Abstract
Background
Genetic susceptibility is a primary factor contributing to etiology of late-onset Alzheimer's disease (LOAD). The exact mechanisms and timeline through which APOE/PICALM influence brain functions and contribute to LOAD remain unidentified. This includes their effects on individuals prior to the development of the disease.
Objective
To investigate the effects of APOE and PICALM risk genes on brain health and function in non-demented individuals. This study aims to differentiate the combined risk effects of both genes from the risk associated solely with APOE, and to examine how PICALM alleles influence the risk linked to APOE.
Methods
APOE/PICALM alleles were assessed to determine the genetic risk of LOAD in 79 healthy, middle-aged participants who underwent electroencephalography (EEG) and functional magnetic resonance imaging (fMRI) recordings. The resting-state signal was analyzed to estimate relative spectral power, complexity (Higuchi's algorithm), and connectivity (coherence in EEG and independent component analysis-based connectivity in fMRI).
Results
The main findings indicated that individuals at risk for LOAD exhibited reduced signal complexity and the so-called “slowing of EEG” which are well-known EEG markers of Alzheimer's disease. Additionally, these individuals showed altered functional connectivity in fMRI (within attention-related areas).
Conclusions
Risk alleles of APOE/PICALM may affect brain integrity and function prior to the clinical onset of the disease.
Introduction
Understanding the early development of late-onset Alzheimer's disease (LOAD) is crucial for its effective diagnosis, prevention, and treatment. The apolipoprotein E gene (APOE) is widely recognized as the predominant genetic factor influencing LOAD. It has three isoforms: ε4, ε3, and ε2. The ε4 increases the risk of developing Alzheimer's disease (AD) by 4–12 times compared to non-carriers.1,2 In contrast, the ε3 isoform appears to have no effect on disease risk, while the ε2 isoform is occasionally associated with a reduced risk. 3 Patients with the homozygous ε4 allele typically exhibit an earlier onset of disease, 4 a phenomenon that is also observed in familial forms of early-onset AD. 5 Genome-wide association studies (GWAS) have identified numerous additional risk genes for AD, including 42 new loci reported in a 2022 study. 6 Among these, the gene encoding phosphatidylinositol binding clathrin assembly protein (PICALM) has been repeatedly identified as a significant risk factor for AD.6–8 The PICALM G allele is more prevalent among AD patients, whereas the A allele is thought to either decrease the risk of AD or have no effect. Furthermore, potential interactions between the APOE and PICALM genes have been observed. 7 Some studies suggest that the combined presence of these genes influence brain atrophy and diminishes cognitive performance in early AD patients. 9 Both genes are also implicated in amyloid pathology, a common pathway in the development of AD. 10 They have never been studied together in a non-demented population.
Electroencephalography (EEG) is one of the most promising tools in search for LOAD diagnostic markers,11,12 as it has high availability, low cost, and non-invasiveness. The most common protocol used in AD patients is the “resting-state” protocol, as it is brief and does not require participants to engage in any specific task. Most studies use eyes-closed condition,13–18 as open eyes resting-state is often characterized by an EEG desynchronization in common bands of interests. A number of changes in spontaneous EEG has been shown in patients with AD, other dementias and mild cognitive impairment (MCI). The most recognized AD hallmark measured with EEG is the so-called “slowing of EEG”,16,17,19–21 i.e., increased amplitude/power of slow waves as delta (∼1–3 Hz)13,15–17 and theta (∼4–7 Hz)13,14,16,17 and decreased amplitude/power of alpha band (∼8–12 Hz).13,15–17 Higher frequency (beta, gamma) is rarely reported to be changed across AD continuum or has no effect.13–17 Signal complexity is another frequently used EEG measure due to the complex and nonlinear dynamics of brain signals. MCI/AD patients have lower signal complexity than healthy controls.20,22,23 Resting-state protocol allows also for studying functional connectivity (FC). This can be done using EEG signal, but a more robust way is to use functional magnetic resonance imaging (fMRI). LOAD tends to be associated with a reduction in functional connectivity in the posterior default mode network (DMN).24–26 This region was shown to be involved in many actions like memory, introspection, mind-wandering, the generation of spontaneous thought, the maintenance of the sense of self, and the integration of information across different cognitive domains. 27 In fMRI studies, functional connectivity is typically measured using either seed-based connectivity (SBC) or independent component analysis (ICA) approaches. Both are meant to detect the correlations in blood-oxygenation-level-dependent (BOLD) signal fluctuations between brain areas which are supposed to reflect similar activity within the same functional network. SBC analysis focuses on the connections of a chosen region of interest (the seed) with all the voxels from the rest of a brain, while ICA separates signals from all of the voxels simultaneously into independent (presumably network-related) components. 28 The ICA approach is a data driven approach. While SBC allows for more specific interrogation of the data, in region-by-region fashion, it suffers from potentially large multiple-comparison problems. By using the ICA method, one can focus on brain networks and their functional alterations. ICA was used extensively in rs-fMRI studies on AD or individuals at risk with different genetic burden.26,29–33
As the LOAD etiology is multifaceted, it is also important to consider the neuropsychological, health related and lifestyle aspect. AD patients are characterized by increased apathy/depression, impaired emotional control, or personality changes 34 and other lifestyle and health factors are linked to the greater dementia risk. 35
There are studies indicating that adjusting lifestyle and beginning interventions early in the disease process can alter (in some cases) the disease's progression.11,36,37 Additionally, modern clinical trials targeting potentially disease-modifying medications focus on the prodromal phases of AD. Thus, understanding the risk-genes influence on health and brain is a key challenge in AD research. In this study we aimed to clarify the combined genetic influence of APOE and PICALM risk alleles on early brain changes in non-demented individuals. We hypothesized that middle-aged individuals carrying APOE risk alleles would exhibit some alterations in brain function (more intense in case of carriers of double, APOE and PICALM, risk) that resemble AD symptoms, i.e., reduced EEG signal complexity, slowing of brainwaves and disrupted functional connectivity as observed through either EEG or fMRI.
Methods
This report is a part of a larger research project, for which the whole dataset was made openly available at OpenNeuro server as “A Polish Electroencephalography, Alzheimer's Risk-genes, Lifestyle and Neuroimaging (PEARL-Neuro) Database” (Accession Number ds004796) and described in detail in a Data Note article. 38 Below we expand methodological information most relevant for the current report.
Participants and genetic screening
The full cohort (N = 200) from PEARL-Neuro Database underwent genetic screening and completed questionnaires on demographics, health, and psychometric assessments. AD risk genes, the APOE (rs429358/rs7412) and PICALM (rs3851179) alleles were determined using the traditional Sanger sequencing protocol, which was outsourced to the certified third-party company. From this cohort, 79 subjects were selected based on their genetic scores to form the experimental groups for neuroimaging study reported here (see Table 2 in a corresponding Data Note 38 for a frequency of detected genotypes and participants.tsv table in PEARL-Neuro dataset for genetic characteristic of each participant). Exclusion and inclusion criteria were thoroughly described and illustrated in diagrams in the data note article (see Figures 1 and 2 in this corresponding Data Note 38 ). The participants in the genetic-based research groups were matched based on age, gender, education, and various health factors, particularly those influencing dementia risk. The groups were constructed based on APOE/PICALM risk: APOE-ε4/PICALM GG non-carriers (referred to as “N”), single-risk carriers (APOE-ε4 carriers without the PICALM risky GG alleles, referred to as “A+P-”), and double-risk carriers (APOE-ε4 carriers with the PICALM risky GG alleles, referred to as “A+P+”; two participants in this group were homozygous for the ε4 allele). Note that carriers of APOE-ε2 alleles (supposedly protective) were not included in a control (N) group, and no separate “A-P+” group was formed, because our study was focused on the accumulated risk and on the APOE (the most recognized AD risk factor) effects.
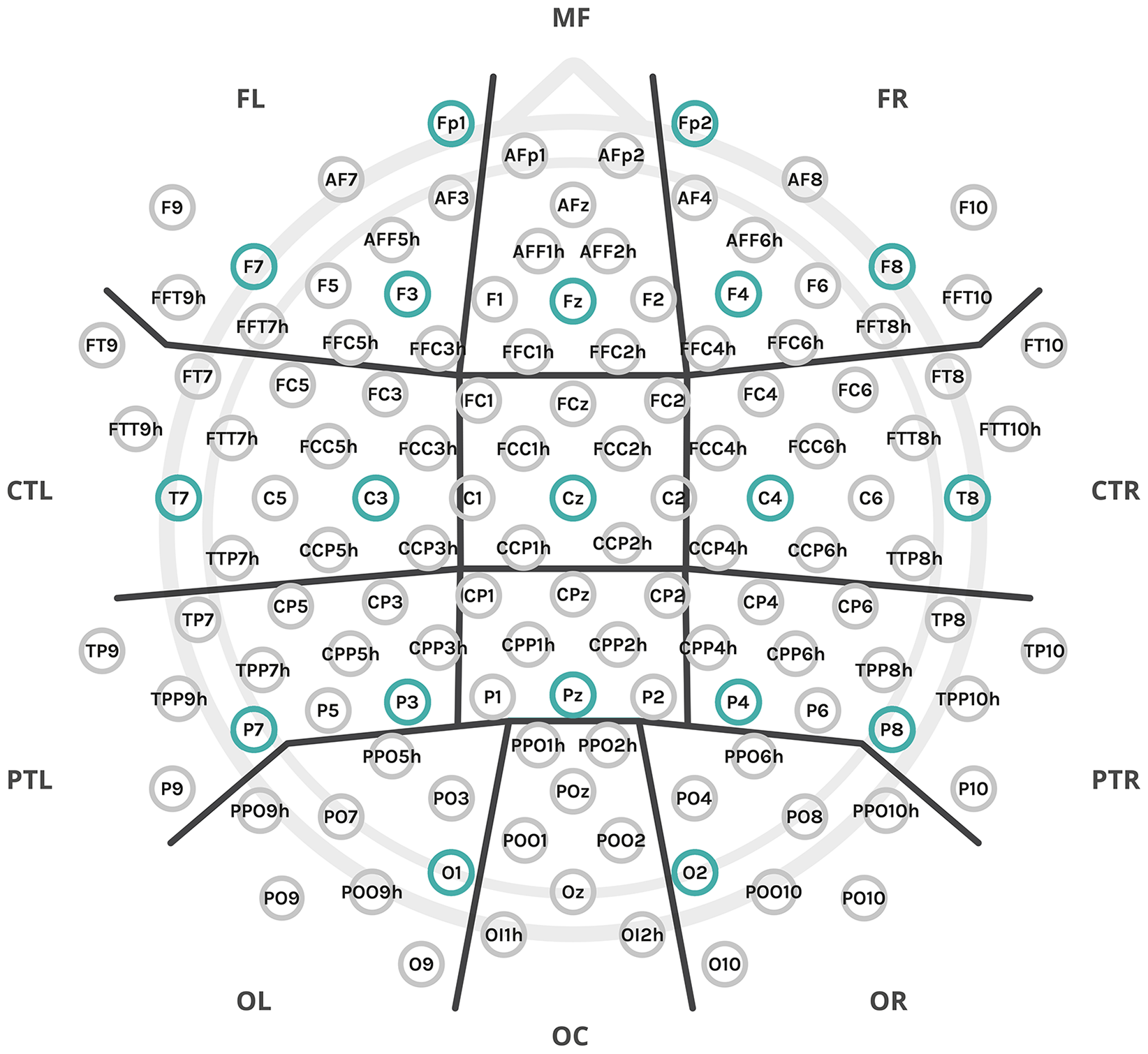
The experiment employed a setup of 128 electrodes, which were organized into anatomical clusters for data analysis, as denoted by the thick black lines: MF: midfrontal; FL: frontal left; FR: frontal right; C: central; CTL: central-temporal left; CTR: central-temporal right; PR: parietal central; PTL: parietal-temporal left; PTR: parietal-temporal right; OC: occipital central; OL: occipital left; OR: occipital right. Additionally, two midline clusters, not marked in the figure, include C – central (comprising FCz, Cz, and neighboring electrodes) and CP – central-parietal (comprising CPz, Pz, and neighboring electrodes). The subset of electrodes from the 10–20 montage used for connectivity analysis was highlighted in a stronger color.
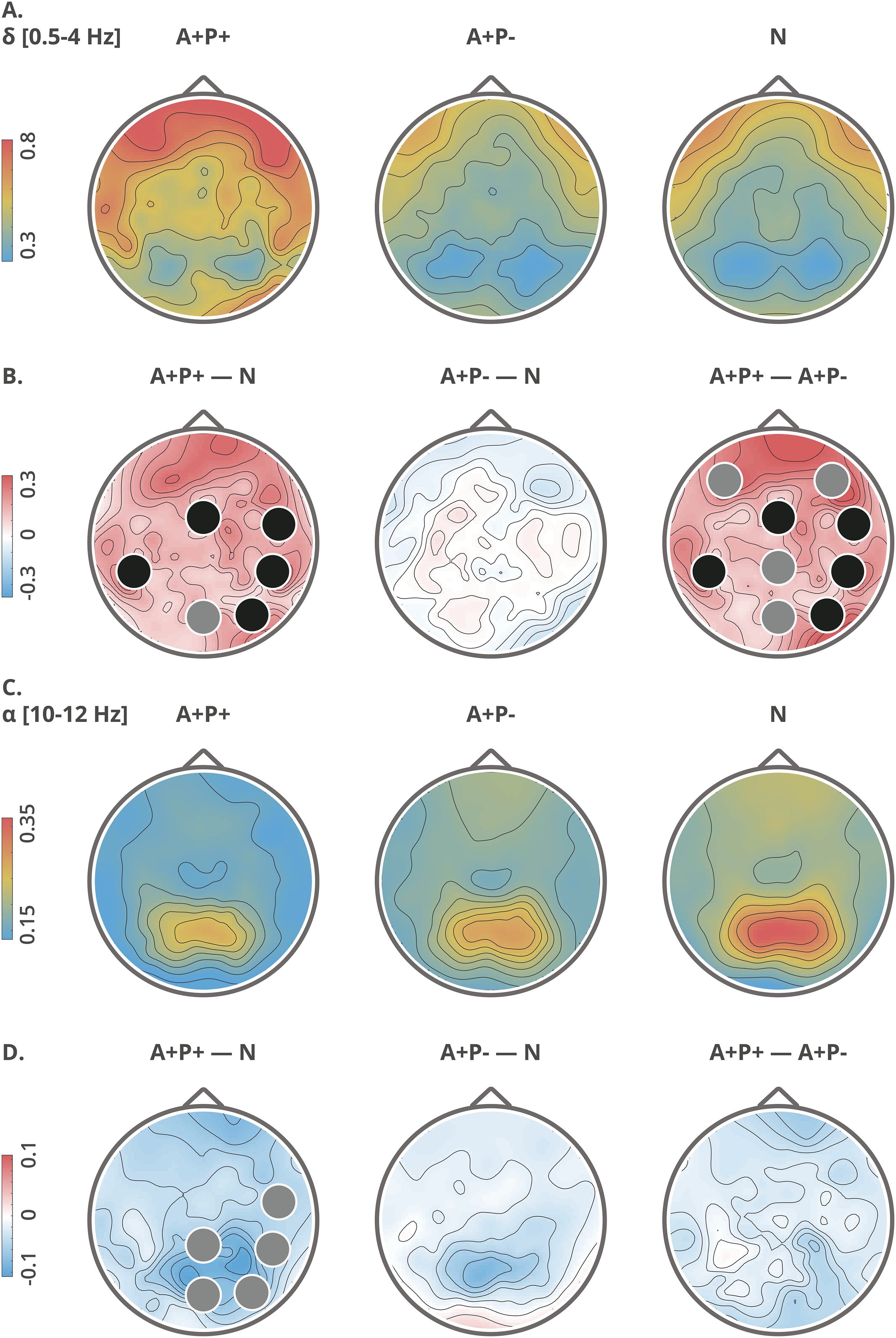
Topographical maps of delta (A, B) and alpha (C, D) relative power: group averages (upper rows: A, C) and group differences (lower rows: B, D). Clusters with significant differences (p < 0.05) between the groups are indicated with black circles, while trend-level differences (p < 0.09) are indicated with gray circles. The topographic maps are computed from high-density data, utilizing all 128 electrodes.
Demographic characteristics, health and psychometric tests assessment: descriptive statistics and group differences.
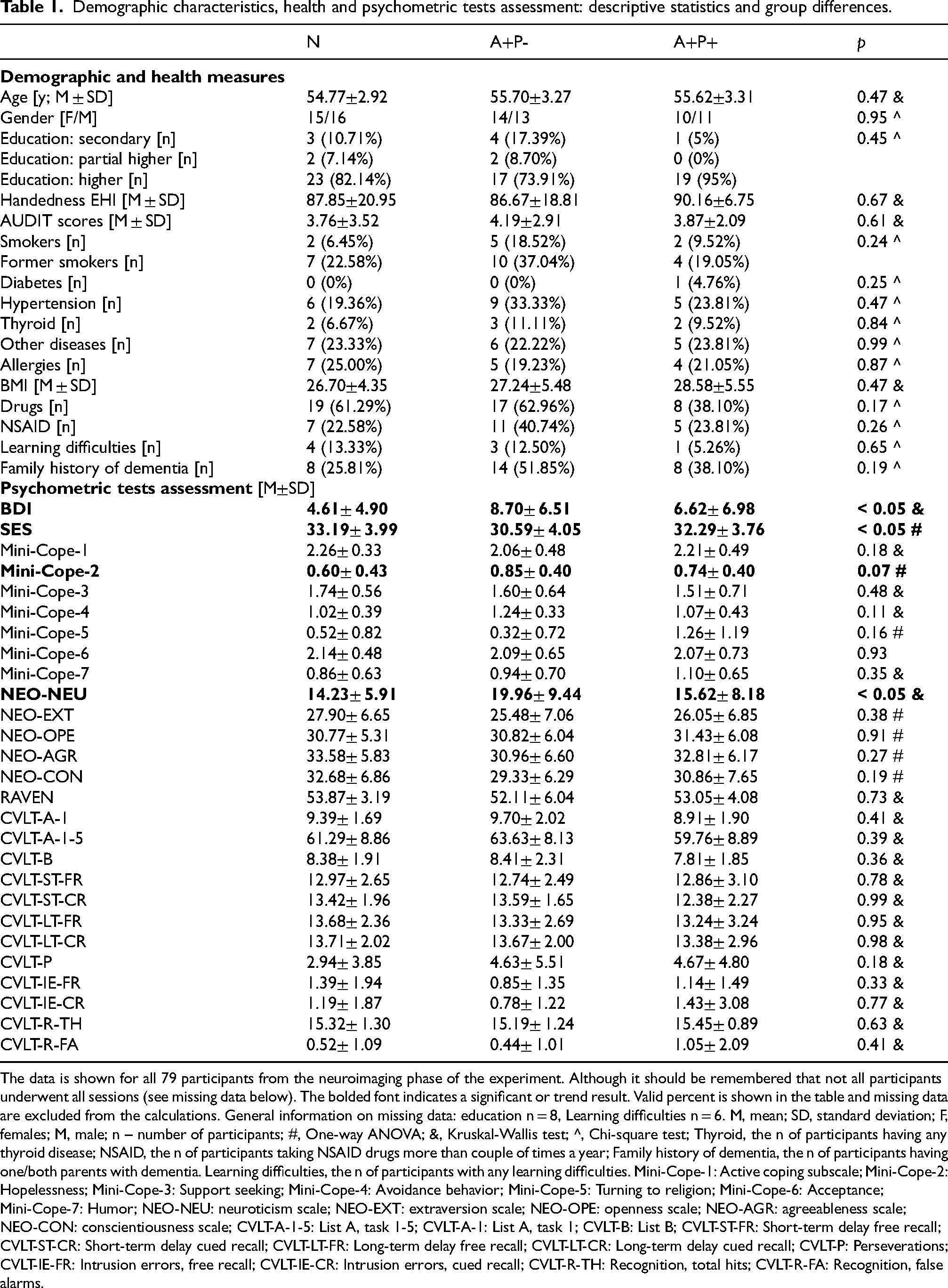
The data is shown for all 79 participants from the neuroimaging phase of the experiment. Although it should be remembered that not all participants underwent all sessions (see missing data below). The bolded font indicates a significant or trend result. Valid percent is shown in the table and missing data are excluded from the calculations. General information on missing data: education n = 8, Learning difficulties n = 6. M, mean; SD, standard deviation; F, females; M, male; n – number of participants; #, One-way ANOVA; &, Kruskal-Wallis test; ^, Chi-square test; Thyroid, the n of participants having any thyroid disease; NSAID, the n of participants taking NSAID drugs more than couple of times a year; Family history of dementia, the n of participants having one/both parents with dementia. Learning difficulties, the n of participants with any learning difficulties. Mini-Cope-1: Active coping subscale; Mini-Cope-2: Hopelessness; Mini-Cope-3: Support seeking; Mini-Cope-4: Avoidance behavior; Mini-Cope-5: Turning to religion; Mini-Cope-6: Acceptance; Mini-Cope-7: Humor; NEO-NEU: neuroticism scale; NEO-EXT: extraversion scale; NEO-OPE: openness scale; NEO-AGR: agreeableness scale; NEO-CON: conscientiousness scale; CVLT-A-1-5: List A, task 1-5; CVLT-A-1: List A, task 1; CVLT-B: List B; CVLT-ST-FR: Short-term delay free recall; CVLT-ST-CR: Short-term delay cued recall; CVLT-LT-FR: Long-term delay free recall; CVLT-LT-CR: Long-term delay cued recall; CVLT-P: Perseverations; CVLT-IE-FR: Intrusion errors, free recall; CVLT-IE-CR: Intrusion errors, cued recall; CVLT-R-TH: Recognition, total hits; CVLT-R-FA: Recognition, false alarms.
Several participants withdrew from the study for reasons such as MRI contraindications, and some data were lost due to technical issues. One participant did not undergo the REST protocol during the EEG session but completed all other protocols on a different day during the MRI/fMRI study. The exact number of participants for each experiment is as follows:
⎯ Health and psychometric tests were completed by all 79 participants (details of missing data in various questionnaires are provided in the Results section in the Data Note article[38]). ⎯ EEG data: N = 78 participants (N group: 31, A+P- group: 27, A+P+ group: 20). ⎯ MRI/fMRI data: N = 69 participants (N group: 27, A+P- group: 24, A+P+ group: 18).
Relative power for delta, theta, alpha-1 and alpha-2 bands: statistics, p-value and post-hoc details regarding group differences.
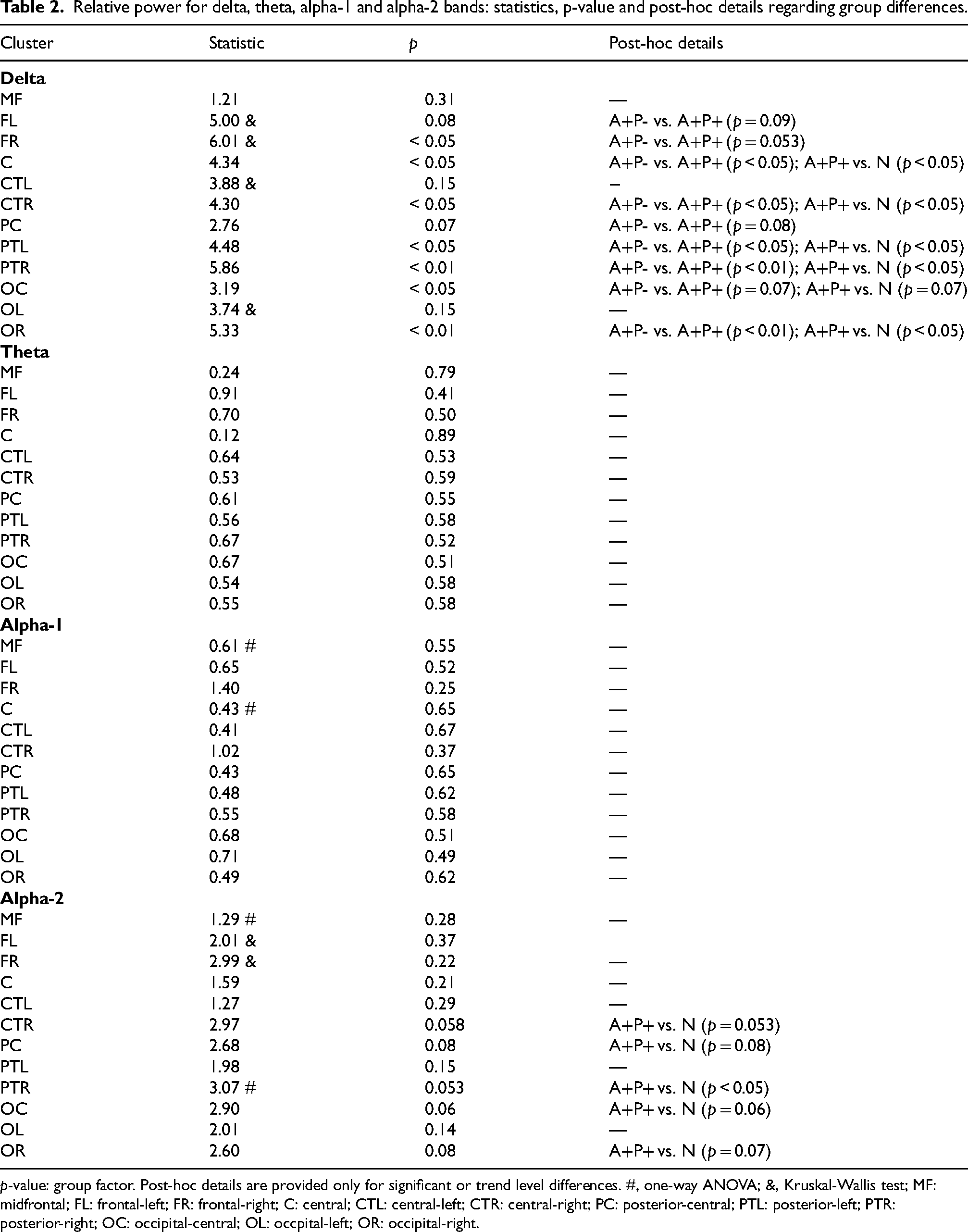
p-value: group factor. Post-hoc details are provided only for significant or trend level differences. #, one-way ANOVA; &, Kruskal-Wallis test; MF: midfrontal; FL: frontal-left; FR: frontal-right; C: central; CTL: central-left; CTR: central-right; PC: posterior-central; PTL: posterior-left; PTR: posterior-right; OC: occipital-central; OL: occpital-left; OR: occipital-right.
The study was approved by the local bioethics committee (Bioethics Committee of the Nicolaus Copernicus University in Toruń functioning at Collegium Medicum in Bydgoszcz, Poland). Written informed consent was provided by all participants and all participants received cash remuneration.
Demographics, health, and psychometric assessment
Participants provided standard demographic information along with details about their health status (e.g., diabetes, hypertension, etc.; all measures are presented in Table 1 in Results section). They were then assessed using a comprehensive battery of psychometric tests to evaluate basic characteristics linked to increased dementia risk or typically found in dementia patients. These tests included:
⎯ Depression/apathy (measured by Beck's Depression Inventory, BDI) ⎯ Self-esteem (measured by Rosenberg's Self-Esteem Scale, SES) ⎯ Stress and stress coping strategies (measured by Mini-Cope Questionnaire) ⎯ Personality (measured by NEO-FFI Personality Inventory) ⎯ Intelligence (measured by Raven's Progressive Matrices – standard/classic version, RPM) ⎯ Memory (measured by California Verbal Learning Test, CVLT)
Additionally, alcohol use was measured by the Alcohol Use Disorders Identification Test (AUDIT; a threshold of ≥ 8 points could indicate unhealthy alcohol usage) and handedness by the Edinburgh Handedness Inventory (EHI).
Data acquisition
EEG session was conducted in the EEG laboratory at the Nencki Institute of Experimental Biology PAS (Poland) and MRI/fMRI session in the Bioimaging Research Center, Institute of Physiology and Pathology of Hearing (Poland). Both EEG and fMRI experiments included an eyes-closed resting-state condition. To ensure participants were rested, EEG sessions were conducted exclusively in the morning and early afternoon. The sessions took place in a comfortable room with dim lighting, where participants sat in a comfortable chair with armrests and faced a monitor. A researcher supervised the study remotely via computer and online LAN camera. Participants were instructed to relax, avoid thinking about anything specific, and remain still. EEG was recorded for 6 min using the extended 10–20 international system for electrode placement (Figure 1), with 128 active electrodes (actiCAP, Brain Products, Munich, Germany) on a Brain Products EEG system. The online reference was set to FCz. At the end of the session, a handheld CapTrak 3D scanner (Brain Products) was used to obtain accurate electrode locations. Impedance was kept as low as possible (average 7.84 ± 3.15 kΩ) through skin rubbing and gel application (Supervisc, extra viscous gel). A low-pass filter was set to 280 Hz, and no high-pass or Notch filters were used during recording. The sampling rate was 1000 Hz.
MRI/fMRI experiments were performed on a 3T Siemens Prisma FIT scanner (Siemens Medical Systems, Erlangen, Germany) equipped with a 64-channel phased-array RF head coil. The acquisition parameters were as follows: multi-band (slice acceleration factor = 8) EPI sequence, repetition time (TR) = 0.8 s, echo time (TE) = 0.038 s, slice thickness = 2 mm, 72 slices, IPAT = 1, FOV = 216 × 216 mm, 52° flip angle, voxel size = 2 × 2 × 2 mm, and acquisition time (TA) = 7:30 for each series (15 min total rs-fMRI acquisition time). Each subject underwent two resting-state EPI sequences, one with anterior-posterior encoding phase and the other with posterior-anterior encoding phase. Both were subjected to subsequent unwarping procedure (see Data preprocessing section). Structural T1-weighted 3D MP-Rage images were acquired with the following parameters: TR = 2400 ms, TI = 1000 ms, TE = 2.74 ms, 8° flip angle, FOV = 256 × 256 mm, voxel size = 0.8 × 0.8 × 0.8 mm, and TA = 6:52 min.
Data preprocessing
The EEG data were preprocessed using the EEGLAB toolbox 39 within MATLAB 2022a. For each participant, standard electrode positions in EEG data files were replaced with individual positions obtained from the CapTrak localizer. The data were downsampled to 250 Hz and filtered within the range of 0.1–40 Hz using standard filter parameters from the toolbox. Additional files were saved with filtering specifically set to 1–40 Hz for subsequent ICA. Channels with excessive noise were removed based on the EEGLAB clean raw data algorithm, which utilizes criteria such as no-signal/flat line, channel correlation, and line noise, as well as through visual inspection. On average, 5.33 channels out of 127 were removed per participant, and the removed channels were interpolated. An average reference was applied, and the initial reference electrode (FCz) was restored and included in the data. The data were segmented into non-overlapping epochs, and those containing excessive artifacts were removed using the ASR algorithm (artifact subspace reconstruction bad burst correction) and further visual inspection. On average, 1.87 epochs per participant were removed. ICA was then applied to detect and separate components with evident physiological artifacts (e.g., eye-blink, muscle, ECG artifacts), resulting in the removal of an average of 4.72 components per participant. The data were visually inspected again, and if necessary, additional cleaning of epochs was performed, with an average of 1.41 additional epochs removed per participant.
Preprocessing of the MRI/fMRI data was performed using SPM12 (Wellcome Trust Centre for Neuroimaging, London, UK) and FSL. 40 Functional data were first realigned, followed by correction of spatial distortions from phase encoding using FSL's topup function. The average field distortion map was generated based on both opposing phase-encoded EPI acquisition series. 40 The structural T1-weighted image was co-registered with the functional images, segmented, and normalized to the common 1-mm isometric MNI space. Transformation parameters obtained from this process were then applied to the functional images after resampling to a 2-mm isometric voxel size. A 6 mm Gaussian kernel (full width half maximum, FWHM) was used for spatial smoothing. Functional data were filtered in the 0.008 to 0.09 Hz band range and denoised using ArtToolbox, as implemented in CONN, 41 with ‘intermediate settings’ (Global-signal z-value < 5; motion < 0.9 mm). On average, 7.2 ± 6.2 volumes (0.6%) were scrubbed per participant. Additionally, the COMPCOR 42 approach was utilized on white matter and cerebrospinal fluid signals to generate nuisance regressors related to physiological artifacts (6 PCA components for each mask).
Data analysis
The analysis of the EEG data was performed using MATLAB. For each participant, Welch's power spectral density estimate was computed at each channel with a 4-s window and 50% overlap (pwelch MATLAB function; spectral resolution 0.25 Hz). The average power for the delta (0.5–4 Hz), theta (4–7 Hz), alpha-1 (7.5–9.5 Hz), and alpha-2 (10–12 Hz) frequency bands was then calculated using the bandpower MATLAB function. Relative average band power was calculated by dividing the band scores by the total power of the signal in the 1–30 Hz range. Global relative band powers (average of all 128 electrodes) and regional relative band powers (average of selected electrodes) were computed. For regional power estimation, electrodes were grouped into 12 anatomical regions of interest (ROIs)/clusters: midfrontal (MF), left and right frontal (FL and FR), left and right central-temporal (CTL and CTR), left and right parietal-temporal (PTL and PTR), and left and right occipital (OL and OR) (Figure 1). To ensure an approximately normal distribution of data for statistical analysis, the results were logit-transformed using the function
The EEG signal complexity was calculated using Higuchi's fractal dimension (HFD) algorithm,44,45 which is renowned for its accurate estimation of signal fractal dimension in electrophysiology data and for its computational efficiency. 46 Among various algorithms, HFD consistently provides the most precise fractal dimension estimations47,48 and has been effective in distinguishing between AD patients and healthy subjects.23,49 The fractal dimension ranges from 1 to 2, with higher values indicating more complex signals. This value depends on the algorithm's tuning parameter, kmax, which can be defined using several approaches. We selected kmax using the plateau criterion, which has proven efficient for EEG data, though it is not always recommended for other data types. 50 This approach, validated for various data types,50,51 identifies the best discrimination between predefined groups. To determine the appropriate kmax for calculating group difference statistics, we first computed the absolute percentage change between average HFD values at consecutive k values to detect the plateau. A threshold of 0.05% was used to find the start of the function minima. The first minimum was at kmax = 36, but subsequent values exceeded the threshold. The second minimum, at kmax = 61, marked the start of the function plateau, where percentage changes remained below 0.05%. Next, we calculated the distance metric (pairwise difference) between the three groups over the plateau values (from kmax = 61 to kmax = 100). The sum of differences for each kmax was then computed. The kmax = 82 corresponded to the largest sum of differences among the groups, indicating the point where the groups were most distinct from each other.
EEG connectivity (coherence) was calculated using built-in Fieldtrip 52 functions with the mtmfft method (which implements multitaper frequency transformation; taper: dpss) on 4-s segments, using the previously described bands of interest. Coherence values between two signals range from 0 to 1, with 1 indicating highly correlated signals. For statistical analysis, we selected a subset of electrodes from the standard 10–20 montage, commonly used in studies on AD patients.53,54 However, the graphical results, including connectograms and matrix representations, are also displayed in the Supplemental Figure 4 for the high-density montage with 128 electrodes.
Functional MRI resting-state analysis was conducted using the CONN toolbox (version 21.a). 41 Group-level ICA was used to identify 21 temporally coherent networks from the combined fMRI data of all subjects. The choice of 21 components is supported by prior research on individuals with various AD-related genetic polymorphisms 30 , and using around 20 components is considered reasonable. 25 The selection of 21 components was further validated through visual inspection to ensure the distinctiveness of networks of interest. For subject-specific dimensionality reduction, a singular value decomposition of the z-score normalized BOLD signal was performed, with 64 components applied separately for each subject. Group-level analyses were conducted using a General Linear Model (GLM). 55 The CONN software automatically assigned neural networks to components using the spatial match to template algorithm, which calculated the correlation between each group-level spatial map and CONN's default networks with varying levels of spatial correlation coefficient. Although groups were matched by age and sex, these variables were included as nuisance regressors due to the significant age- and gender-related variability in rs-fMRI data. 25 Changes in the default mode network (DMN) are a characteristic feature of AD. Therefore, our primary focus was on this network, and we employed a hypothesis-driven approach to analyze the resting-state fMRI data, specifically examining the ICs with the highest correlation coefficient with the DMN.
Statistics
Most statistical analyses were conducted using R 56 with RStudio 57 and custom scripts, with the exception of some neuroimaging statistics computed within specific toolboxes/software. Initially, we assessed the linearity between age and the dependent variables; since no linearity was observed, age was not considered a covariate for statistics performed in R. For quantitative variables, either a two-way ANOVA test (with sex as a fixed factor) or a one-way ANOVA was used after verifying the assumptions of the tests. The normality of residuals was first assessed using the Shapiro-Wilk test and quantile (Q-Q) plots. Additionally, homoscedasticity was tested using Levene's test (criterion: p < 0.05). For nominal variables, the chi-square test was employed. For ordinal variables and when ANOVA assumptions were not met, the non-parametric Kruskal-Wallis test was applied. ANOVAs were supplemented by standard post-hoc tests with Tukey correction for multiple comparisons. If the ANOVA test with Welch's homogeneity correction was used, the Games-Howell post-hoc approach (with Tukey's correction) was applied. For the Kruskal-Wallis test, Dunn's post-hoc tests were used (with Holm correction). Statistical analysis of EEG coherence results was conducted in MATLAB using two-sample t-tests, with coherence calculated within the Fieldtrip toolbox. For t-test coherence results, FDR correction was applied using the Benjamini & Hochberg method 58 with a custom MATLAB function. 59 fMRI data statistics were derived from the SPM12 toolbox. Statistics on fMRI data connectivity were conducted at the cluster level, relying on parametric statistics derived from Gaussian Random Field theory.55,60 The results were subjected to a thresholding approach involving a voxel-level threshold of p < 0.001 for cluster formation and a familywise corrected cluster-size threshold of p-FDR < 0.05. 61 Statistical significance was defined as follows: a p-value ≤ 0.05 was considered significant, and a p-value > 0.05 and ≤ 0.09 was considered a trend. Where possible, all data were presented as mean (M) ± standard deviation (SD). Annotations on the figures are as follows: a p-value ≤ 0.05 is marked with *, < 0.01 with **, and < 0.001 with ***. Trend-level differences are marked with ∼ and the exact p-value.
Results
Characteristics of the participants
The study cohort comprised right-handed, middle-aged adults, with an equal distribution of females and males, ensuring gender balance in each subgroup (Table 1). No significant differences between the groups regarding age, education, gender, handedness, possible alcohol problems, smoking status, health status were found (Table 1). Most respondents were generally healthy and were non-smokers. A+P- and A+P+ groups have more relatives (parents) with dementia than the N group (51.85% and 38.10% versus 25.81%), but there were no statistical differences.
The psychometric assessments indicated minor differences in depression/mood and self-esteem scales (BDI and SES, Table 1), with the A+P- group scoring worse than the N group (BDI: H(2) = 6.37, p < 0.05, post-hoc: A+P- versus N p < 0.05; SES: F(2,76) = 3.17, p < 0.05, post-hoc: A+P- versus N p < 0.05). Additionally, the A+P- group showed increased levels of neuroticism (NEO-NEU: H(2) = 6.04, p < 0.05, post-hoc: A+P- versus N p < 0.05) and a tendency towards higher scores on the hopelessness scale in a stress coping strategies test (MINI-2 F(2,74) = 2.72, p = 0.07, post-hoc: A+P- versus N p = 0.06). Since the full cohort (N = 200) completed psychometric testing and provided data on family history of dementia and demographics, the same comparisons made for the significant findings identified during the neuroimaging phase were repeated for all participants, including the A-P+ group, which was not included in the neuroimaging phase. To maintain consistent group structures based on allele assignment (as in the neuroimaging phase), individuals with the APOE-ε2 allele were excluded, leaving 174 participants. This resulted in unequal group sizes compared to the neuroimaging phase (A + P + group, N = 21; A+P- group, N = 28; N group, N = 76; A-P+ group, N = 49). This extended comparison revealed significant differences in family history of dementia (χ2(3) = 10.03, p < 0.05, N = 174, post-hoc: A+P- versus N p < 0.05), with the A+P- group having more first-degree relatives with dementia than the N group. No significant differences were observed for BDI, SES, and the Mini-Cope-2 scale. Regarding neuroticism, differences were again identified (H(3) = 7.72, p = 0.05), but post-hoc tests considering six comparisons after introducing the A-P+ group revealed no significant differences after adjusting for multiple comparisons. Despite this, the A+P- group consistently showed higher neuroticism scores compared to the N group, with the A-P+ group also exhibiting relatively high scores and the A+P+ group displaying the lowest scores (A+P-: 20.29 ± 9.42, A-P+: 20.41 ± 8.77, A+P+: 15.62 ± 8.18, N: 17.31 ± 8.48).
Power spectral measures
The A+P+ group demonstrated higher delta relative power than the N and/or A+P- groups both globally (F(2,72) = 4.34, p < 0.05, post-hoc: A+P- versus A+ P + p < 0.05, A+P+ versus N p < 0.05) and in certain electrode clusters (Figure 2 and Table 2). There were no global differences in theta (p = 0.60), alpha-1 (p = 0.54), nor alpha-2 (p = 0.11) bands. The A+P+ group exhibited lower relative alpha-2 power in comparison to the N group in specific clusters (Figure 2 and Table 2).
Male participants were consistently characterized by higher theta relative power than females, both globally (F(1,72) = 7.04, p < 0.05) and in certain electrode clusters (Supplemental Table 2). They were also characterized by lower high alpha band power than females, both globally (F(1,72) = 6.95, p < .05) and in certain clusters (Supplementary Table 2). Additionally, there was an interaction between sex and group regarding delta relative power (Supplemental Figure 1), indicating that males in the A+P+ group had higher delta power than males or females in the N or A+P- groups, both globally (F(2,72) = 3.30, p < 0.05, post-hoc: M A+P- versus M A+P+ p < 0.01, M A+P+ versus M N p < 0.05, M A+P+ versus F N p < 0.01, F A+P- versus M A+P+ p < 0.05) and in specific electrode clusters (Supplemental Table 1). A statistically significant interaction was also observed between the group and sex factors on lower alpha relative power, both globally (F(2,72) = 5.03, p < 0.01, post-hoc: F A+P- versus M A+P- p < 0.05, M A+P- versus M N p = 0.09, M A+P- versus F A+P+ p = 0.07) and in specific electrode clusters (Supplemental Figure 1). Female participants from the A+P+ group had lower low alpha relative power than males in the A+P- group, and males from the N group had lower low alpha relative power compared to males in the A+P- group. Additionally, females in the A+P- group exhibited lower low alpha relative power than males in the same group.
Signal complexity
The signal complexity results are depicted in Figure 3, showing the average global HFD plotted against the tuning parameter kmax. The A+P+ group demonstrated significantly lower signal complexity (M ± SD, 1.83 ± 0.04) compared to the N group (M ± SD, 1.85 ± 0.03
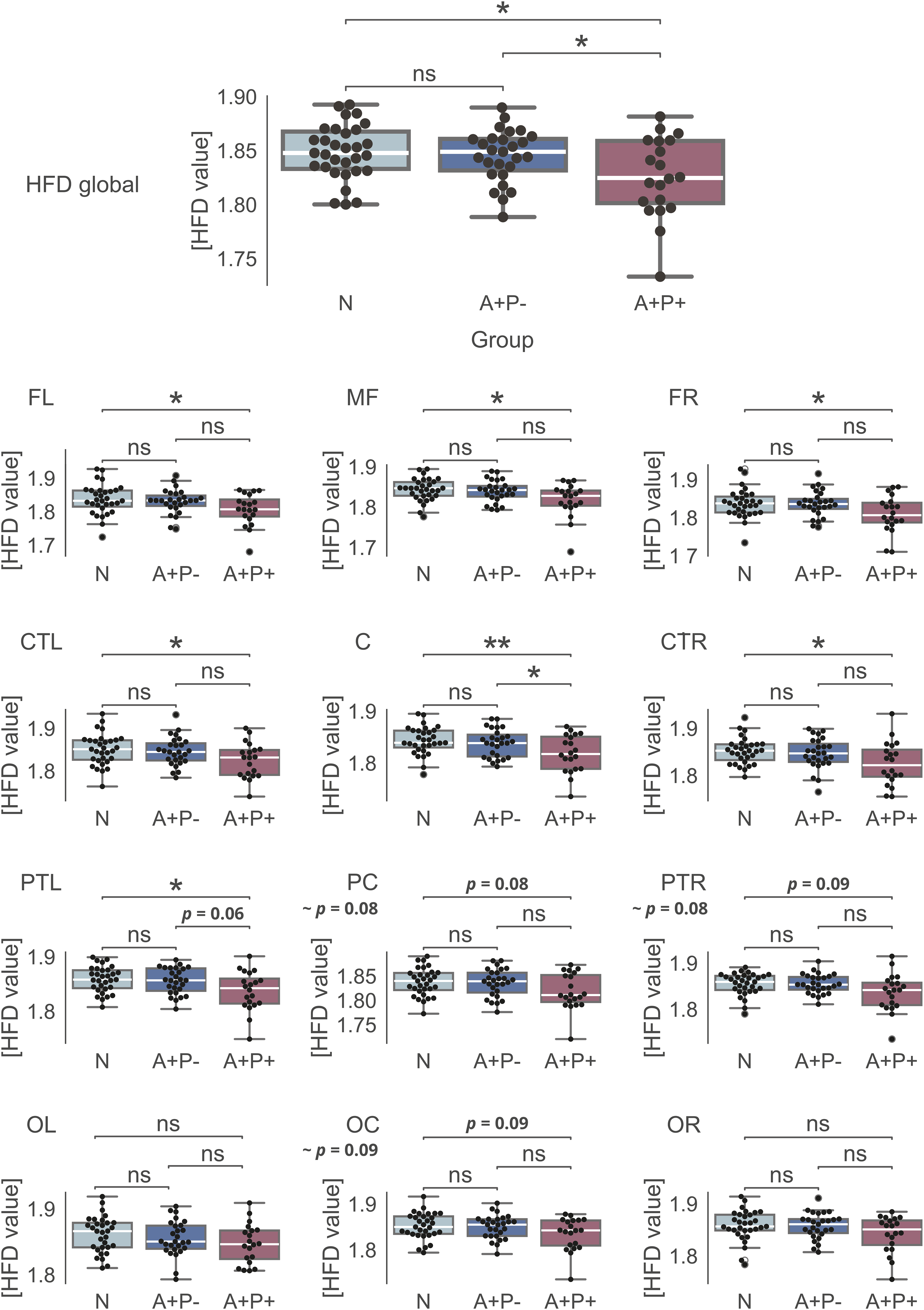
Average Higuchi's fractal dimension. Differences between the groups on kmax = 82.
HFD values. Descriptive statistics and group differences.
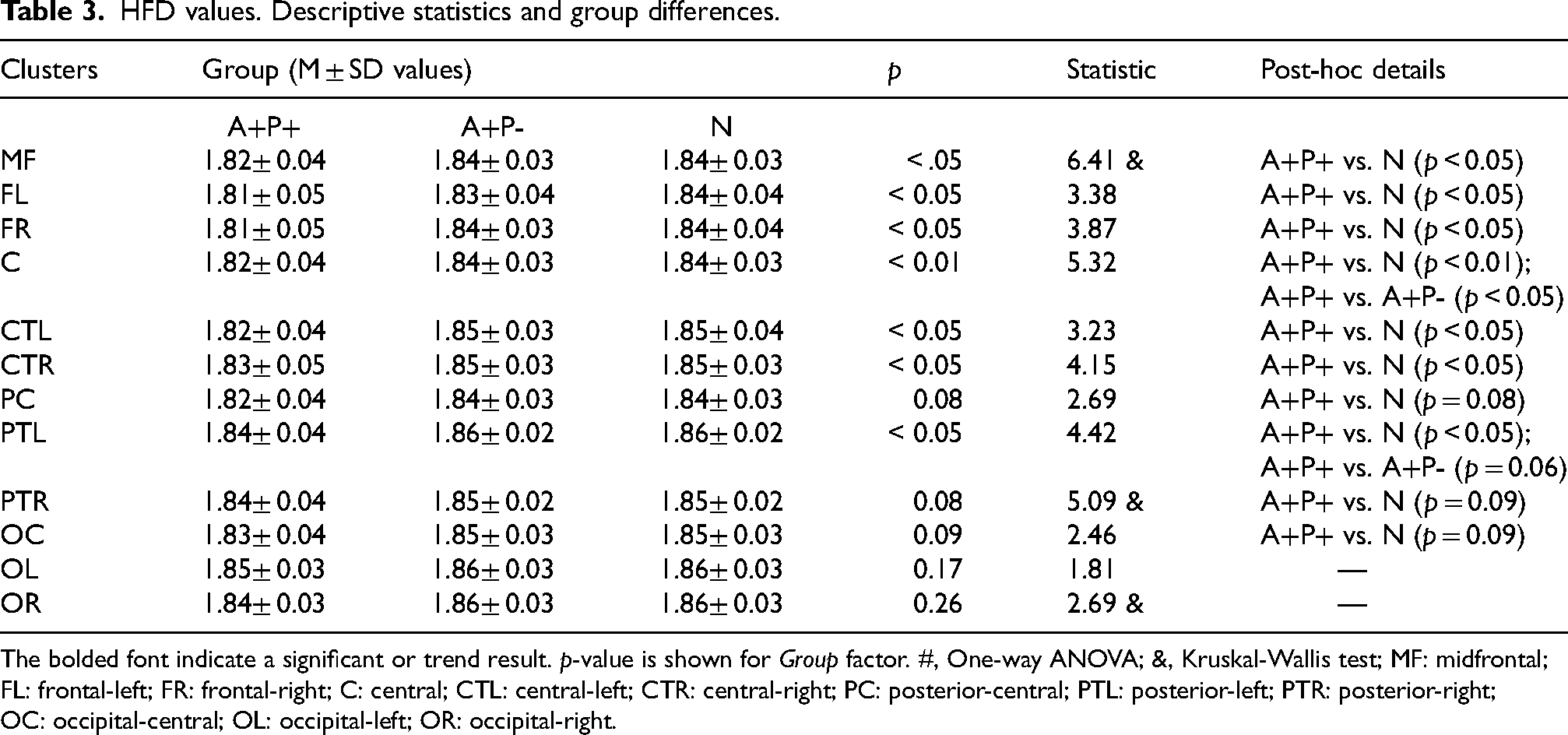
The bolded font indicate a significant or trend result. p-value is shown for Group factor. #, One-way ANOVA; &, Kruskal-Wallis test; MF: midfrontal; FL: frontal-left; FR: frontal-right; C: central; CTL: central-left; CTR: central-right; PC: posterior-central; PTL: posterior-left; PTR: posterior-right; OC: occipital-central; OL: occipital-left; OR: occipital-right.
Moreover, sex had a significant impact on HFD values, with males displaying reduced signal complexity both globally (F(1,72) = 5.72, p < 0.05) and in certain electrode clusters (Supplemental Table 2)—C cluster: F(1,72) = 7.14, p < 0.01, CTR cluster: F(1,72) = 5.70, p < .05; PC cluster: F(1,72) = 4.59, p < .05; PTL: F(1,72) = 8.44, p < .01; OC cluster: F(1,72) = 6.39, p < .05; OL cluster: F(1,72) = 12.14, p < .001. A trend level (p = .053) interaction between sex and group factors was found in FL cluster (Supplementary Table 4). These findings mirrored the main results for the group factor alone, with females/males from the A+P+ group exhibiting the lowest HFD values compared to females/males from the A+P- and N groups (which had the highest values).
Connectivity
The analysis of EEG connectivity, measured by the coherence of frequency components (bands), identified subtle patterns indicating some differences between the groups at the p-uncorrected level. In the delta band, the A+P+ group displayed greater coherence compared to the A+P- and N groups. Furthermore, the A+P- group showed reduced theta coherence relative to the N group. Both the A+P+ (low and high alpha) and A+P- (low alpha) groups exhibited (mostly) decreased coherence in the alpha band compared to the N group (as depicted in Supplemental Figure 3, with uncorrected p-values below the alpha 0.05 level). However, it is crucial to highlight that none of these associations remained significant after applying the false discovery rate (FDR) correction. Additionally, Supplemental Figure 4 shows coherence results using a high-density montage with all 128 electrodes, including connectograms and difference matrix representations for a thorough depiction of global connectivity.
The analysis of fMRI ICA-based connectivity revealed some subtle differences between the studied groups. The correlation of 21 estimated components with known networks is detailed in Supplemental Table 5, showing the best three matching components: IC 11 (r = 0.38), IC 5 (r = 0.34), and IC 20 (r = 0.11). Their representations are shown in Supplemental Figure 5. While IC 5 and IC 11 clearly depict posterior parts of the DMN, IC 20 shows mixed areas, which is explained by only an r = 0.11 correlation coefficient with the DMN. Additionally, IC 20 includes signals from the cerebrospinal fluid (CSF) and ventricular regions (cyan color). As indicated by the correlation coefficients, these components do not perfectly align with traditional DMN areas and also include other, nearby regions. Two DMN related components (IC 11 and IC 5) were selected for further group comparisons.
The A+P- group exhibited significantly lower network strength in a cluster in the right temporo-occipital part of the middle temporal gyrus (toMTG, IC11) compared to the N group (Table 4 and Figure 4). This effect was significant at p-FDR < 0.01. No significant differences were found in IC 5 between the groups.
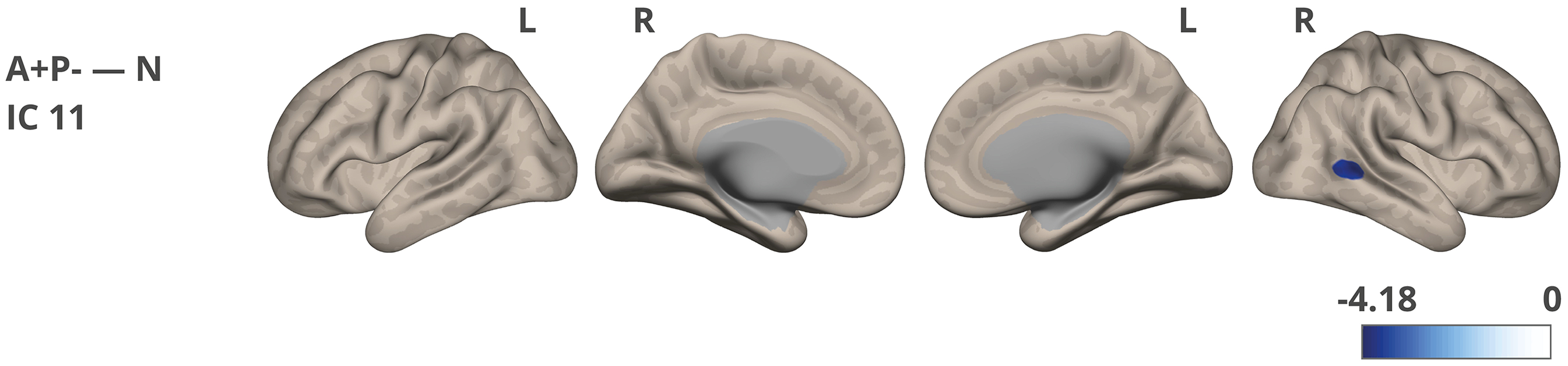
Network strength/connectivity differences were noted between the A+P- and N groups for IC 11. The color corresponds to the direction of the difference and obtained t-value. The images were visualized using the CONN toolbox, with t-values adjusted to fit the cortical structure on a glass ICBM template brain surface display, presented in lateral and medial views.
Spatial cluster (location, size, and anatomical alignment) associated with IC 11, highlighting significant difference between the groups.

r: right; k: cluster size; toMTG: middle temporal gyrus temporo-occipital part.
Key findings summary
EEG “slowing,” a marker of AD, was observed in participants with combined APOE and PICALM risk (A+P+) compared to those without risk (N group). A similar marker, indicating reduced EEG signal complexity, was also present in the A+P+ group compared to the N group. EEG coherence in both the A+P+ and A+P- groups showed a trend of changes consistent with those observed in AD patients reported in the literature. Lower network strength in the posterior (temporo-occipital) region of middle temporal gyrus (toMTG) was identified in the A+P- group compared to the N group during the analysis of independent components related to the DMN.
Discussion
We examined EEG and fMRI resting-state data from healthy middle-aged individuals with various alleles of the AD risk genes APOE and PICALM. The groups were demographically balanced, and we discovered that risk carriers exhibit some AD hallmarks in domains directly (EEG, fMRI) and indirectly (psychometric evaluation) associated with brain functioning.
The entire cohort consisted of well-functioning individuals from major urban areas who were professionally active and had a high level of education. The groups performed equally well on both the intelligence test (RAVEN) and the California Verbal Learning Test (CVLT) which assesses verbal learning and memory. Previously reported CVLT results for healthy but much older APOE carriers showed that they either did not differ from no-risk groups 62 or exhibited a higher frequency of recall intrusion errors. 63 Despite the lack of clear cognitive impairment, the subjects displayed group differences in specific psychological characteristics. Participants with a single risk factor (A+P-) declared worse psychological well-being compared to the no-risk group. They exhibited lower self-esteem, higher scores on the BDI scale (related to depression and lowered mood), heightened levels of neuroticism, and a tendency to use less effective stress coping strategies. It is well established that factors such as depression, lowered mood, and neuroticism act as risk factors for AD and may interact with APOE in provoking the disease.2,63–66 The BDI scores differentiated the groups, although the average scores were within the normal range. Prolonged stress, which can cause these psychological symptoms, is also described as a risk factor. 65 Specifically, midlife stress is associated with the development of dementia in later life. 67 Notably, these differences were significant primarily for the cohort that underwent neuroimaging testing. However, for the entire cohort of 200 individuals recruited for genetic screening, statistical significance was achieved only for neuroticism.
In double risk carriers (A+P+ group), EEG resting state was characterized by two of the most noticeable and sensitive markers of AD: a shift of the power spectrum to lower frequencies (known as “slowing of the EEG”) and decreased signal complexity. EEG “slowing” was evidenced by higher delta and lower alpha relative power, while a lower Higuchi fractal dimension indicated reduced signal complexity. These two measures, complexity and EEG dynamics, are strongly related 68 and correspond to the capacity for information processing (i.e., less complex signal = lower capacity), which changes due to neurodegeneration as well as during natural stages of brain development, maturation, and aging. Additionally, the impact of common genetic variations (e.g., APOE gene) on cognitive/brain functioning increases with aging. 69 EEG abnormalities in normal aging also include changes in spectral content, such as decreased power in delta, theta, and alpha peak frequencies 13 and decline in complexity (significantly affecting central-parietal areas, especially right-shifted clusters). 49 In MCI patients, research has shown a small increase in the power of delta and theta bands in temporal areas. 13 Upper alpha, but not lower alpha power, was distinguishable among controls and AD patients in another study. 15 AD patients with APOE risk have stronger EEG “slowing” than carriers of neutral APOE alleles,4,70 although one study showed opposite results. 71 Power perturbations within the temporal and parietal areas are especially sensitive indicators for distinguishing AD patients from healthy controls. 18 A reduction in complexity in AD patients is clearly noticeable, particularly affecting the temporal-occipital regions. 49 The EEG “slowing” marker is believed to be associated with neuronal loss, axonal pathology, and cholinergic deficits, which affect functional connections in the cortex. 20 Decreased signal complexity is also related to either neuronal loss or neurotransmitter deficiencies, such as acetylcholine. 20 Atrophy in cholinergic neurons may be the primary source of EEG “slowing,” as these neurons are most affected by AD. The cholinergic hypothesis of AD and memory dysfunction in the elderly was proposed over 50 years ago.20,72,73 The cholinergic system regulates various aspects of brain function—cognition, locomotion, attention, sleep, arousal, and sensory processing—by modulating neuronal activity via acetylcholine receptors. Cholinergic drugs tend to reverse EEG “slowing,” supporting this hypothesis.20,74 Anticholinergic drugs (e.g., scopolamine), which block the stimulation of post-synaptic receptors, cause EEG “slowing”.20,74,75 APOE-ε4 positive AD patients are characterized by more severe cholinergic deficits than patients with a neutral APOE genotype. 76
In our study, the groups did not exhibit significant differences in connectivity, as measured by EEG coherence at the FDR-corrected level. However, on the uncorrected level, they showed subtle trends in the same direction as reported among AD patients: decrease in alpha coherence in the at-risk (A+P+ or A+P-) groups compared to the N group, and increased delta coherence in the at-risk A+P+ group compared to the A+P- and N groups. A recent systematic review showed that in 24 out of 34 studies comparing AD patients to healthy controls, AD patients had significantly decreased coherence within the alpha band. 77 The results for coherence in lower frequencies (<7 Hz) were less consistent and less frequently significant. 77 Generally, alpha coherence tends to be decreased in AD patients, 13 while delta and theta coherences tend to be increased compared to matching controls. 13
Using an ICA-based approach, we identified small yet significant differences in fMRI connectivity. This analysis primarily focused on independent components linked to the DMN, which is often impaired in AD patients.25,78 The A+P- group (compared to the N group) exhibited significantly reduced network strength in a cluster encompassing the right temporo-occipital part of the middle temporal gyrus. Although the MTG is not traditionally considered a core component of the DMN, numerous studies demonstrate its involvement within the posterior DMN and discuss DMN role in semantic cognition.79–81 Network changes in APOE risk-carries (without amyloid burden) were previously reported in the literature.32,82 Certain studies have revealed decreased connectivity within the posterior DMN in older APOE-ε4 carriers (70–89 years old) compared to non-carriers, 83 while no effect was observed in middle-aged adults. 33 Our results support the previous report on the effects of the APOE-ε4 risk allele alone and further suggest that the interplay between APOE and PICALM may accelerate the onset of this symptom. Research has consistently indicated decreased connectivity within the posterior DMN in individuals with MCI and AD patients, while recently showing increased connectivity within the frontal parts of the DMN.25,78 The causes of network changes are still unclear; it is unknown whether they are related to amyloid deposition, whether they represent a compensatory mechanism in response to amyloid atrophy and toxicity, or to what extent they are influenced by genetic factors. 25 Atrophy and hypometabolism are known to be partially responsible for observable network changes in amyloid-positive individuals. 84 The concept of network-based functional compensation suggests that alterations in the brain's functional architecture, influenced by genes such as APOE and PICALM, may enable the brain to adapt and compensate for changes or disruptions in specific brain regions or networks. Intuitively, we would expect to observe compensation through increased activity; however, in the case of the DMN, the opposite may occur, 85 with reduced activity potentially being beneficial for task-related engagements.
The study had several limitations. First, a larger sample size would strengthen the findings and would allow for the separation of an additional (A-P+) experiential group in the neuroimaging phase of the study, and for more comprehensive analyses through a multivariate design. Additionally, group differences in psychological traits were not accounted for as covariates/additional factors in the main analysis. Another limitation is the lack of data on well-established AD biomarkers, such as amyloid-β 42/40 or phosphorylated tau, which can be measured in plasma. Due to the current study design, we cannot associate our participant's results with the potential development of the earliest phases of AD. Such conclusions can only be drawn after future follow-up tests within the same cohort.
In summary, our findings reinforce previous research and suggest that APOE and PICALM shape the functional architecture of the resting brain even in the absence of dementia. Future research should prioritize studying non-demented risk carriers throughout their lifespan to understand the impact of these genetic variations on aging and to uncover the biological mechanisms underlying their association with neurodegenerative diseases. Early detection of AD should employ multimodal approaches that consider genetic burden (such as the APOE and PICALM risks in our study), and additionally some or all markers like blood-based biomarkers, MRI/fMRI/EEG abnormalities, cognitive performance, health, lifestyle, demographic factors, and neuropsychological assessments. Together, these markers may help identify individuals at risk of developing dementia in the future, allowing for potential early and successful interventions.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251317489 - Supplemental material for Alzheimer's disease-like features in resting state EEG/fMRI of cognitively intact and healthy middle-aged APOE/PICALM risk carriers
Supplemental material, sj-docx-1-alz-10.1177_13872877251317489 for Alzheimer's disease-like features in resting state EEG/fMRI of cognitively intact and healthy middle-aged APOE/PICALM risk carriers by Patrycja Dzianok, Jakub Wojciechowski, Tomasz Wolak and Ewa Kublik in Journal of Alzheimer's Disease
Footnotes
Acknowledgments
We thank participants for their valuable time and willingness to participate in the study. We appreciate the help of Ingrida Antonova and Olga Stefańska in EEG acquisition. In preparing this manuscript, we utilized OpenAI's language model ChatGPT (4o) to assist in English language correction of selected paragraphs.
Author contributions
Patrycja Dzianok (Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Software; Visualization; Writing – original draft); Jakub Wojciechowski (Formal analysis; Software; Writing – review & editing); Tomasz Wolak (Supervision; Writing – review & editing); Ewa Kublik (Conceptualization; Funding acquisition; Methodology; Supervision; Writing – review & editing).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Polish National Science Centre (NCN) grants no. 2018/31/N/HS6/03551 and 2016/20/W/NZ4/00354. The funding body has not participated at any stage in study design, data collection, analysis, or interpretation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data (PEARL-Neuro Dataset) supporting the findings of this study are openly available in OpenNeuro repository at https://doi.org/10.18112/openneuro.ds004796.v1.0.8 and described in the corresponding data note article. 38
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
