Abstract
Background
Research on the influence of heart failure on mortality after Alzheimer's disease diagnosis is limited.
Objective
To evaluate the association between comorbid heart failure and mortality following Alzheimer's disease diagnosis, particularly considering sex differences.
Methods
We analyzed administrative claims data from Japan, involving 32,363 individuals (11,064 men and 21,299 women) aged 75 or older newly diagnosed with Alzheimer's disease, with 7% having comorbid heart failure. Cox proportional hazard models and population attributable fractions (PAFs) were used to evaluate the association between comorbid heart failure and mortality within one year following Alzheimer's disease diagnosis.
Results
Individuals with Alzheimer's disease and heart failure had a multivariate-adjusted hazard ratio of 1.51 (95% confidence interval [CI], 1.32–1.73) for mortality during the one-year follow-up period compared to those with Alzheimer's disease and without heart failure. Subgroup analysis by sex revealed a higher mortality hazard ratio in women of 1.63 (95% CI, 1.36–1.95) than that in men of 1.39 (95% CI, 1.13–1.71). Further age and sex subgroup analysis indicated that women across all age brackets—75–79, 80–84, and ≥ 85 years—had higher mortality hazard ratios. The PAF for heart failure increased with age in both sexes, with women having higher PAFs than men, and the sex difference in PAF being most pronounced in the 75–79 age category (men: 1.4%, women: 4.0%).
Conclusions
Hazard ratios and PAFs for mortality associated with comorbid heart failure in newly diagnosed Alzheimer's disease are higher in women than in men, which persists across all age subgroups.
Introduction
The aging of society is progressing rapidly throughout the world. The number of people aged 80 years or older is expected to triple between 2020 and 2050, reaching 426 million. 1 With aging, there is an increase in geriatric conditions associated with mortality and hospitalization, such as dementia and heart failure.2–4 The prevalence of cognitive impairment among heart failure patients is approximately 40%, according to a systematic review. 5 Alzheimer's disease, a primary cause of dementia, impairs cognitive functions and the ability to perform daily activities. 6 Alzheimer's disease can affect the management of heart failure in several ways, particularly in the areas of medical adherence, caregiving, and symptom monitoring. 7 Therefore, the co-occurrence of these conditions poses unique challenges in an increasingly aging global society.
Despite the increasing prevalence of comorbidity between Alzheimer's disease and heart failure, notable research gaps remain in our understanding of the association between heart failure and mortality among individuals with Alzheimer's disease. Older adults with heart failure and cognitive impairment have increased risks of mortality. 8 Alzheimer's disease is more prevalent in women, yet men diagnosed with Alzheimer's disease have been observed to have a higher mortality risk. 9 These sex differences in Alzheimer's disease prevalence and mortality might further influence the association between comorbid heart failure and outcomes. By contrast, in older patients with heart failure and preserved ejection fraction, men had higher risk of all-cause mortality. 10 These findings suggest the possibility of effect modification by sex in the relationship between Alzheimer's disease, heart failure, and mortality. In addition, a recent study in the United States demonstrated that older adults with both heart failure and dementia faced significantly higher risks of hospitalization within one year and mortality within two years compared to those with heart failure alone. 11 Given the increasing prevalence of these comorbidities in our aging society,1,5 investigating the short-term mortality risk, particularly within one year, is crucial. This focus on short-term outcomes is essential for understanding the clinical course of these co-existing conditions and may contribute to more effective clinical management strategies. However, one-year mortality among older adults with comorbid heart failure following a diagnosis of Alzheimer's disease is still not clearly understood.
This study aims to address these research gaps by analyzing the variation in mortality rates during the one-year follow-up period among individuals aged 75 and older who have been newly diagnosed with Alzheimer's disease, with a particular focus on the association with comorbid heart failure. Using administrative claims data from Japan, we stratify our analysis by sex and age to explore demographic differences and calculate the PAF to assess the public health implications of heart failure comorbidity in this population. The findings of this study may inform the development of targeted interventions and healthcare policies for older adults with newly diagnosed Alzheimer's disease and comorbid heart failure.
Methods
Data source
This study utilized health insurance claims data provided by DeSC Healthcare Inc. (Tokyo, Japan), encompassing the period from April 2014 to August 2021. This comprehensive dataset contains anonymized, healthcare-related information derived from insurance claims, including diagnostic, prescription, and procedure codes. The dataset enables tracking of patient information across all medical institutions in Japan. However, this is contingent upon the individual's continuous enrollment in the same health insurance society. Comprehensive details regarding this health insurance claims dataset are provided elsewhere. 12
Within this dataset, our analysis specifically included members of the Medical care system for the elderly in the latter-stage (MCSEL), which predominantly serves individuals aged 75 and above across Japan. The MCSEL is a public health insurance system in Japan designed to provide medical coverage for individuals aged 75 and older, as well as those between 65 and 74 years old who are certified as being disabled by the Wide Area Union. Upon reaching the age of 75, individuals are automatically enrolled in the MCSEL, regardless of their employment status, and their previous health insurance coverage (e.g., National Health Insurance, Employees’ Health Insurance, or Mutual Aid Associations) is transferred to the MCSEL. This system ensures that all older adults in Japan have access to healthcare services, irrespective of their employment history or pre-existing health conditions, except those receiving public assistance.
The MCSEL provides a highly representative sample of the Japanese population aged 75 and older. By focusing on this population, we ensured a well-defined cohort with reliable and representative data, enhancing the validity and generalizability of our findings for the Japanese population aged 75 and older.
Study population
The study identified individuals experiencing their first episode of Alzheimer's disease, as determined by specific International Classification of Diseases, 10th revision (ICD-10) codes. We first identified dementia patients based on the ICD-10 codes specified by the Global Burden of Disease Study 13 : F00, F01, F02, F03, G20, G30, and G31. Among these, we specifically defined Alzheimer's disease cases as those with the codes for “Dementia in Alzheimer's disease” (F00) or “Alzheimer's disease” (G30). We focused on those aged 75 years or older at the onset of the recorded data period, who were newly diagnosed with dementia. This approach ensured a minimum look-back period of 365 days from the data period's start to confirm the newness of the diagnosis. We excluded individuals for whom outcome information (i.e., all-cause mortality) was unavailable from their insurers. Additionally, we excluded those who were hospitalized at the time of their dementia diagnosis, as their patient backgrounds and disease progression might differ considerably from those diagnosed in an outpatient setting. Inpatients may represent more advanced dementia cases, often admitted due to severe cognitive decline or behavioral symptoms, while outpatients are likely to be in early to moderate stages with milder impairments. By focusing on outpatients, we aimed to evaluate a more representative sample of the general population with dementia. This approach enhances the applicability of our findings to the broader community-dwelling population with dementia and avoids potential confounding due to the inherent differences in disease severity between inpatients and outpatients. Finally, among those diagnosed with dementia, individuals with Alzheimer's disease were included for analysis. The definition of dementia is described in Supplemental Table 1.
Exposure and outcome measures
Exposure to heart failure was defined using ICD-10 codes of I50, specifically when these codes were listed as the principal diagnosis. The primary outcome measure was all-cause mortality following an Alzheimer's disease diagnosis, with mortality determined through the process of qualification loss within MCSEL. In MCSEL, the procedure for qualification loss is initiated upon an insured individual's death, thereby ascertaining the cause of qualification loss, such as death.
Variables
The study collected demographic data (age and sex) and medical history. Medical history included variables like the modified Charlson Comorbidity Index (excluding the contribution of heart failure), 14 and previous hospitalizations within 365 days preceding the dementia diagnosis. The analysis also considered the use of dementia medications within the first 30 days following the dementia diagnosis. These medications encompassed anti-dementia and antipsychotic drugs as described in Supplemental Table 2. We identified specific etiologies and comorbidities associated with heart failure. Etiologies included valvular disease, cardiomyopathy, atrial fibrillation/flutter, coronary artery disease, and pulmonary hypertension. Comorbidities assessed were hypertension, venous thromboembolism, chronic obstructive pulmonary disease (COPD), peripheral artery disease, and chronic kidney disease as described in Supplemental Table 3.
Statistical analysis
We compared participants’ characteristics based on the presence or absence of heart failure exposure. Categorical variables were expressed as numbers and percentages, and continuous variables as means with standard deviations or medians with interquartile ranges. Using Cox proportional hazards models, we estimated the hazard ratio (HR) of all-cause mortality during the one-year follow-up period associated with heart failure in individuals newly diagnosed with Alzheimer's disease, adjusting for factors such as age, sex, the modified Charlson Comorbidity Index, and previous hospitalizations in the multivariable model. To explore potential effect modification by sex, we conducted subgroup analyses stratified by sex. Additionally, we formally tested for interaction by including an interaction term between sex and heart failure in our Cox models and reported the p-value for this interaction. Further subgroup analyses were conducted by age groups (75–79, 80–84, ≥ 85 years). PAFs for mortality due to heart failure exposure were calculated to estimate the public health implications of heart failure exposure in the population, both overall and stratified by sex and age group. In our analysis, the PAFs for mortality were calculated using the following equation: PAF = Pd (HR−1)/HR. Pd is the proportion of participants with heart failure among those who died. 15 HR is the multivariable-adjusted HR of mortality. All statistical analyses were carried out in R version 4.3.1.
Ethical considerations
Because of the anonymous nature of the data, the requirement for informed consent was waived. As our study utilized “existing anonymously processed information,” it is exempt from the “Ethical Guidelines for Medical and Biological Research Involving Human Subjects” as per Japanese regulations (https://www.mext.go.jp/lifescience/bioethics/files/pdf/n2373_01.pdf, only available in Japanese). Despite this exemption, study approval was obtained from the institutional review board of the Fujita Health University (No. HM23-239).
Results
Among the 32,363 individuals with Alzheimer's disease enrolled in our observational study, 2161 (6.7%) were identified as having heart failure. The selection process is detailed in Figure 1. Table 1 outlines the baseline characteristics of participants, categorized by the presence or absence of heart failure and stratified by sex. Participants with heart failure were found to be older on average, with ages of 83.6 years for men and 84.7 years for women, compared with 81.4 and 82.5 years, respectively, for those without heart failure. In the heart failure group, the proportion of participants increased with age, with the highest percentage in the 85 and above category. Among those with heart failure, 42% of men and 50% of women were aged 85 years or older. The proportion of individuals with a history of hospitalization within the last year was higher in the heart failure group compared to the non-heart failure group. Additionally, the heart failure group had higher Modified Charlson Comorbidity Index scores than the non-heart failure group. Variations in the use of anti-dementia and antipsychotic medications within 30 days following diagnosis were noted. Heart failure patients showed lower usage of anti-dementia drugs and higher usage of antipsychotic drugs compared to those without heart failure. Regarding sex differences in etiology and comorbidity in the heart failure group, the proportions of cardiomyopathy, atrial fibrillation/flutter, and coronary artery disease were higher in men than in women. For comorbidities, the proportions of COPD, peripheral artery disease, and chronic kidney disease were higher in men compared to women.
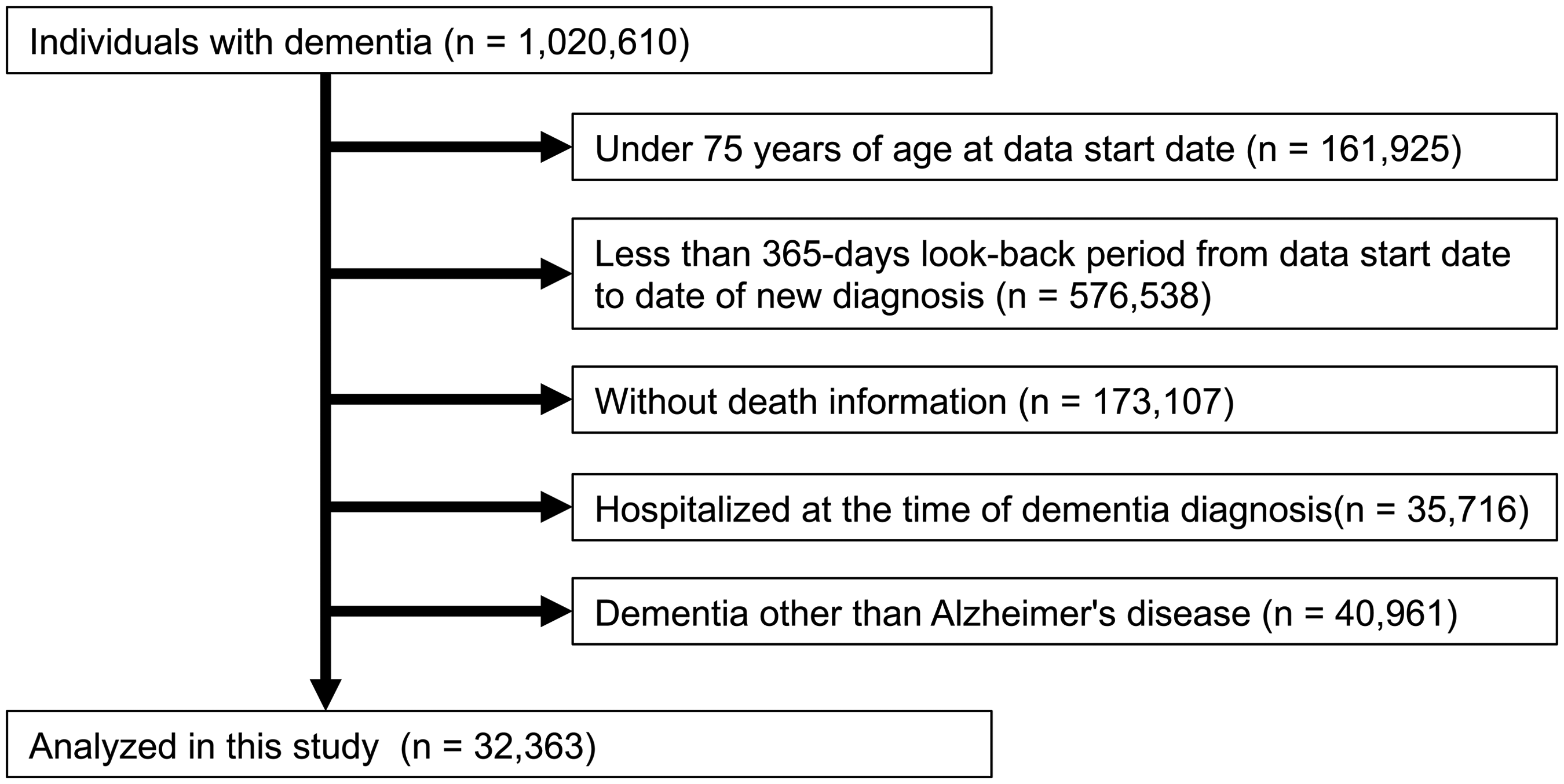
Flow chart of participants selection.
Baseline characteristics of participants with and without heart failure.
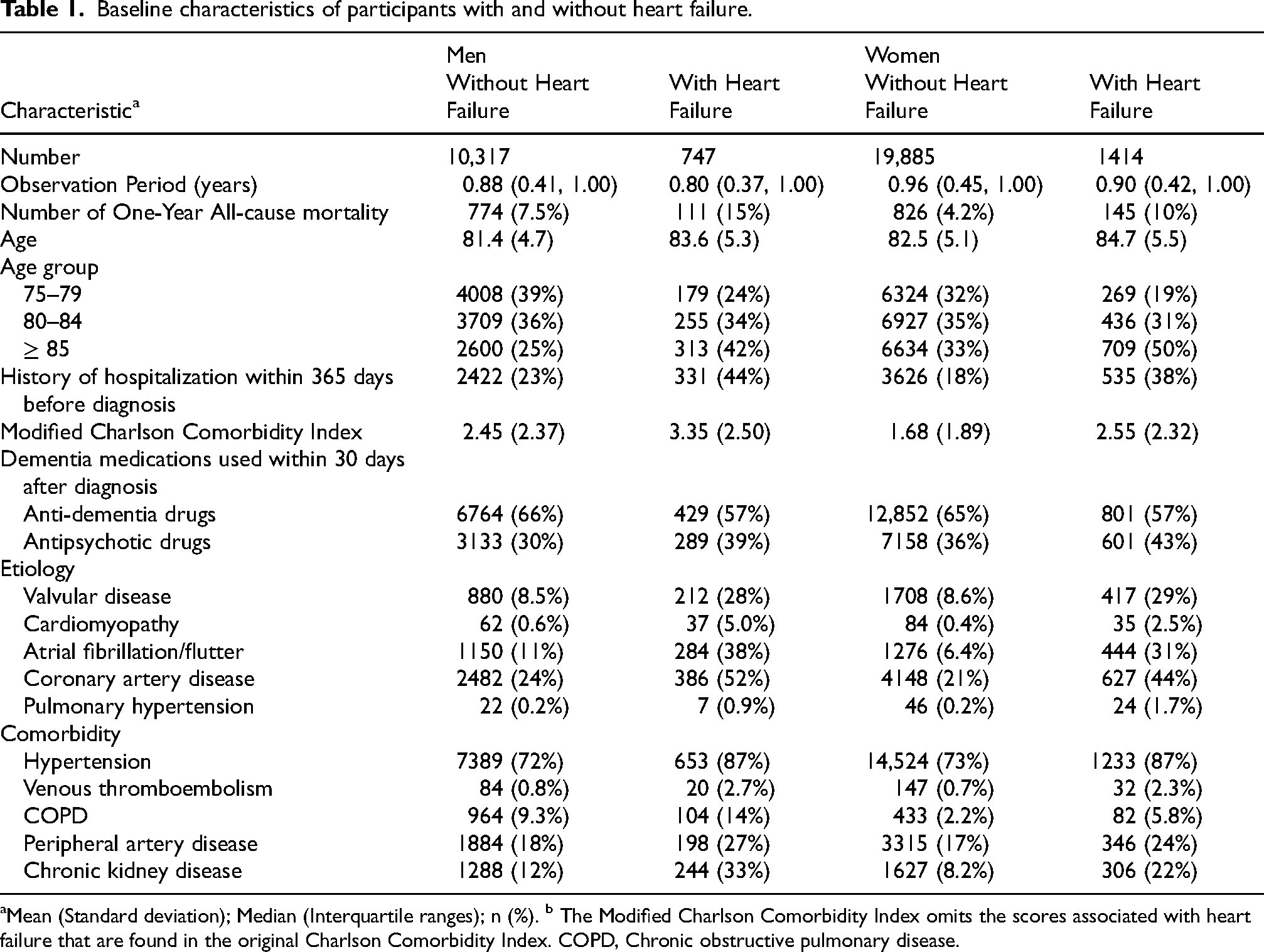
Mean (Standard deviation); Median (Interquartile ranges); n (%). b The Modified Charlson Comorbidity Index omits the scores associated with heart failure that are found in the original Charlson Comorbidity Index. COPD, Chronic obstructive pulmonary disease.
During the one-year follow-up period, the all-cause mortality rate among individuals with Alzheimer's disease and heart failure was higher compared to those without heart failure, with a rate of 16.9 per 100 person-years in the heart failure group versus 7.3 in the non-heart failure group. Censoring occurred for the following reasons: diagnosis of heart failure after the start of observation (n = 731), relocation to another Wide Area Union (n = 184), transition to public assistance (n = 164), other reasons (n = 4), and the end of the available data period (n = 14,190). The crude HR for all-cause mortality in the heart failure group was 2.30, which adjusted to 1.51 after controlling for age, sex, the modified Charlson Comorbidity Index, and hospitalizations within the last year. When analyzed by sex, the mortality rate for the male heart failure group was 21.7 per 100 person-years with a multivariate-adjusted HR of 1.39, while the mortality rate for the female heart failure group was 10.6 per 100 person-years with a multivariate-adjusted HR of 1.63. These findings indicate that while men have a higher mortality rate than women among individuals with Alzheimer's disease and heart failure, the mortality HR for comorbid heart failure was higher in women than in men (p for interaction of sex = 0.06). Further stratification by age and sex revealed that the increased mortality rate associated with heart failure persisted across all age groups for both men and women, with higher HRs among women compared to men. These results are summarized in Table 2, which presents the mortality rates and HRs for the heart failure and non-heart failure groups, as well as the sex-specific and age-stratified analyses.
One-year mortality rates and hazard ratios for all-cause mortality by heart failure status among individuals with Alzheimer's disease.
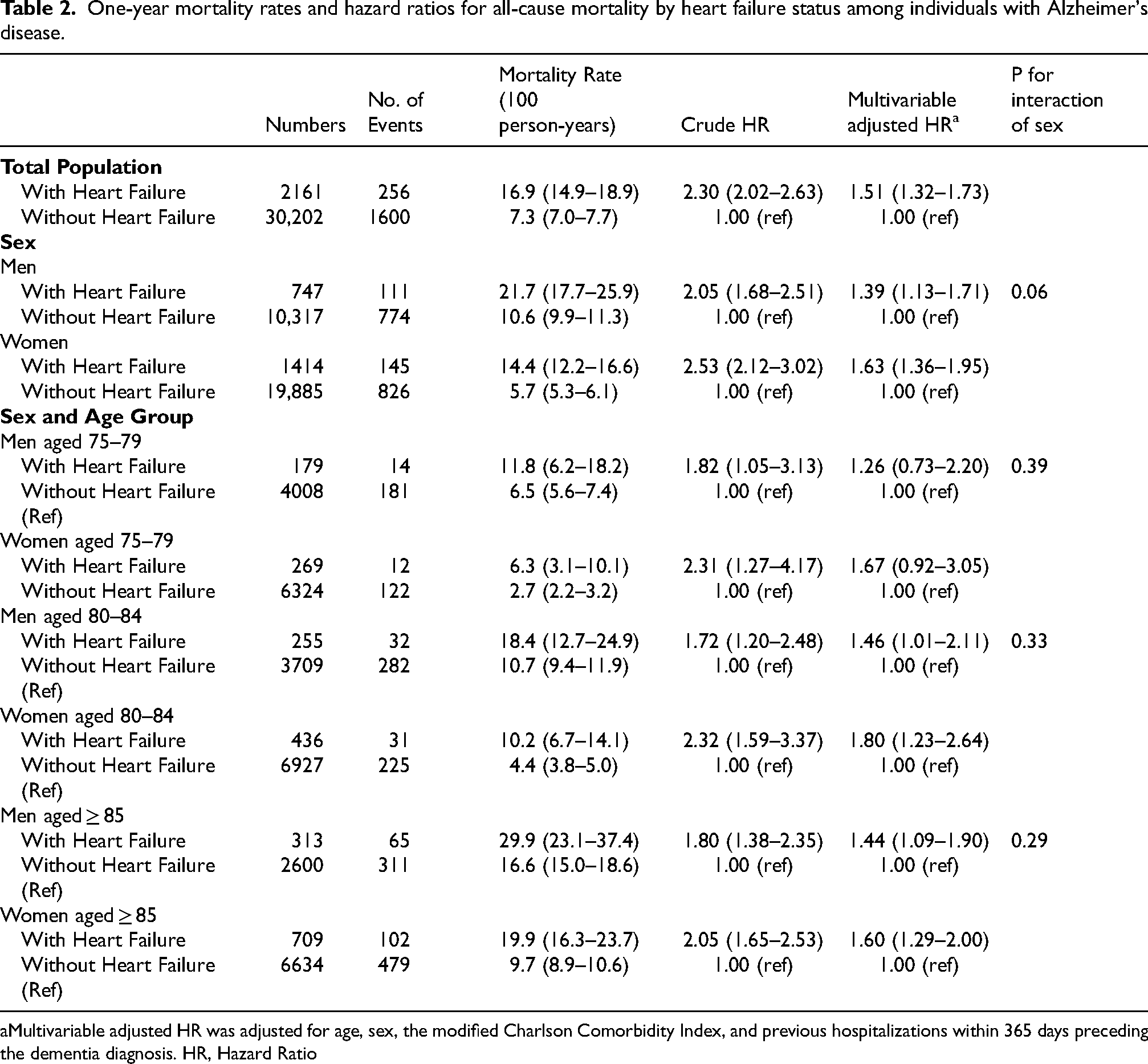
Multivariable adjusted HR was adjusted for age, sex, the modified Charlson Comorbidity Index, and previous hospitalizations within 365 days preceding the dementia diagnosis. HR, Hazard Ratio
The PAF for heart failure contributing to mortality in individuals with Alzheimer's disease was found to be highest among those aged 85 and above, with 5.3% for men and 6.2% for women. Notably, the PAF was consistently higher in women across all age groups, with the largest sex difference observed in the 75–79 age category. These PAF results are visually represented in a heatmap in Figure 2.
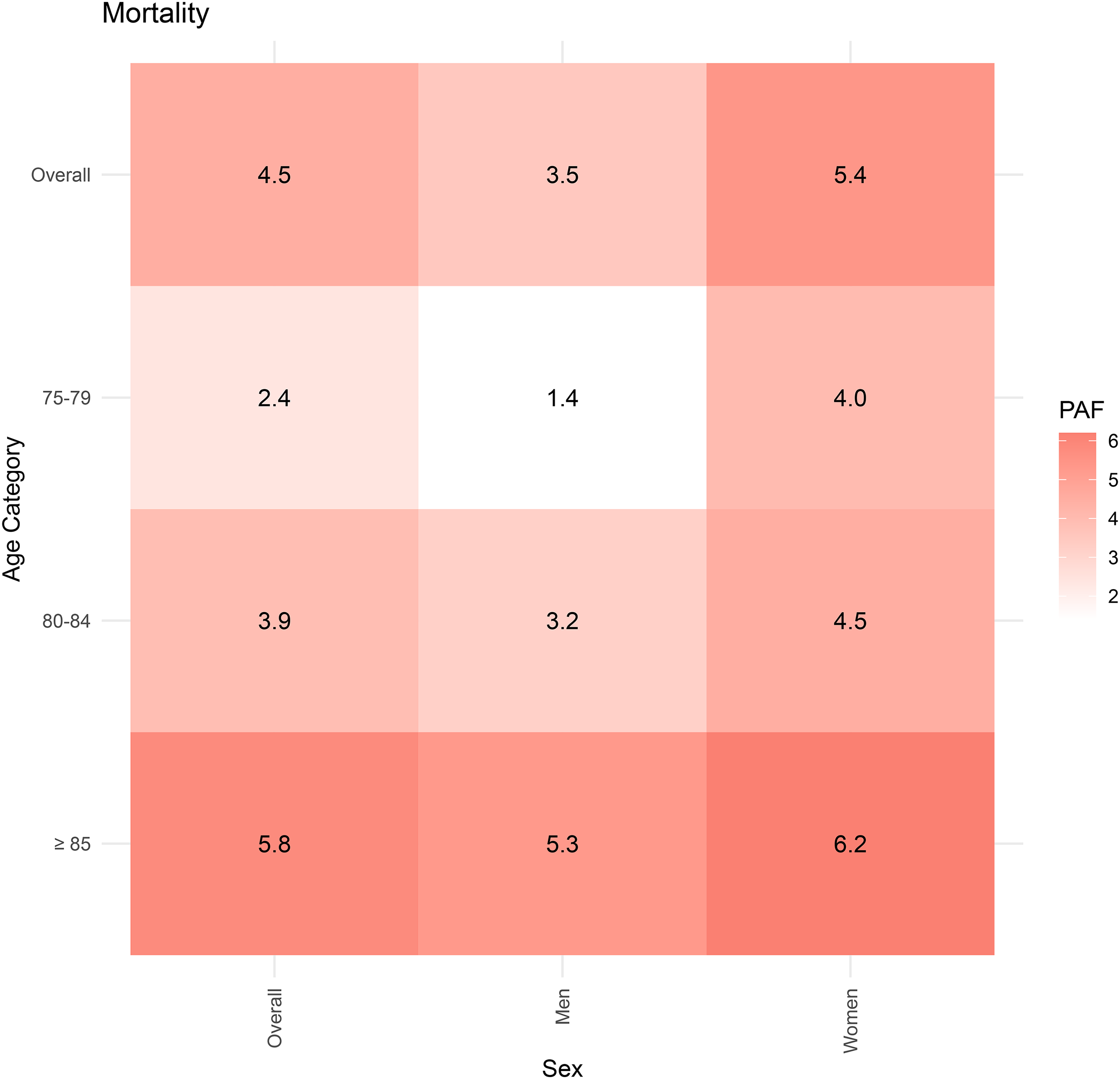
Population attributable fractions due to heart failure for mortality among participants with Alzheimer's disease.
Discussion
Our study reveals two critical findings regarding the association between comorbid heart failure and mortality rates among older individuals with newly diagnosed Alzheimer's disease. Firstly, we observed that individuals with both Alzheimer's disease and heart failure face a significantly higher mortality rate compared to those with Alzheimer's disease alone. This difference in mortality is more pronounced across sexes; while men with heart failure generally exhibit higher mortality rates, the mortality HR associated with heart failure is more pronounced in women. Secondly, the PAFs show a consistent pattern where women, across all age brackets, bear a higher burden of mortality due to heart failure, with the most marked difference appearing in the 75–79 age group.
Our study demonstrated that mortality rates among older individuals with comorbid Alzheimer's disease and heart failure were significantly higher compared to those diagnosed with Alzheimer's disease alone. This observation is supported by existing literature, which identifies cognitive impairment in heart failure patients as a significant risk factor for increased mortality, with a pooled hazard ratio of 1.64 (95% CI, 1.42–1.88). 16 Importantly, our research addresses a gap in the existing research by examining sex differences in mortality HRs, revealing a higher mortality HR associated with heart failure in women compared to men among individuals with Alzheimer's disease. Our analysis of the interaction between sex and heart failure yielded a borderline significant result (p for interaction of sex = 0.06), highlighting a possible effect that should not be overlooked, even though it does not meet the conventional threshold for statistical significance. This finding of a potentially higher impact of heart failure on mortality in women with Alzheimer's disease stands in contrast to previous research. Earlier studies have indicated that men with Alzheimer's disease exhibit a higher mortality risk compared to women. 9 Furthermore, in the broader context of heart failure, research has shown that older women with heart failure and preserved ejection fraction 10 or those hospitalized with heart failure in Japan 17 demonstrate a lower risk of all-cause events compared to men. While the co-occurrence of Alzheimer's disease and heart failure is a known risk factor for mortality, our study highlights the higher mortality HRs in women compared to men, underscoring the need for sex-specific strategies in managing this comorbidity.
The observed PAFs for mortality among older individuals with comorbid Alzheimer's disease and heart failure were higher for women across all age groups examined in this study. These findings are particularly relevant when considered within the context of the average life expectancy in Japan, reported as 81.64 years for men and 87.74 years for women in 2020, 18 and the earlier age of heart failure diagnosis in men compared to women, as reported in England with 74.8 years for men and 79.6 years for women. 19 In men, PAF showed an increasing trend with advancing age, particularly at older ages than life expectancy or age of heart failure diagnosis. In contrast, women demonstrated an increase in PAFs even in age groups younger than their average life expectancy and the average age at heart failure diagnosis. These findings highlight the need for targeted interventions and policies to address the higher mortality burden among women with comorbid Alzheimer's disease and heart failure, especially in younger age groups, and further research into the factors driving these observed disparities.
Multiple factors likely influence the underlying mechanisms of our findings. First, Alzheimer's disease complicates the management of heart failure through challenges in medication management, caregiving, and symptom monitoring. Individuals with Alzheimer's disease face difficulties in managing their complex medication schedules, which is crucial for heart failure treatment. 20 Second, the caregiving burden for Alzheimer's patients is notably higher than for other chronic diseases. 21 This heightened burden may impede effective heart failure management by caregivers. Lastly, Alzheimer's disease complicates symptom monitoring, potentially causing delays in treatment adjustments and worsening heart failure conditions. 22 Our study could not directly assess the severity of heart failure or the three potential mechanisms mentioned above due to the nature of health insurance claims data, but it is plausible that inadequate treatment management and care due to the challenges associated with Alzheimer's disease may have contributed to the increased mortality rates observed.
In examining the sex differences in mortality HRs among older individuals with comorbid Alzheimer's disease and heart failure across various older age groups, the potential role of caregiving in compensating for the reduced self-care abilities typical of dementia warrants attention. Heart failure requires careful post-discharge disease management due to the high risk of readmission from exacerbations. In Japan, only 7.3% of patients attend outpatient cardiac rehabilitation, 23 emphasizing the importance of heart failure care provided by cohabiting family members. A registry study of heart failure patients in Japan found that care services utilization reduced the risk of heart failure readmission within one year after discharge, 24 highlighting the need for heart failure patients diagnosed with dementia to introduce and coordinate care services. The Comprehensive Survey of Living Conditions in Japan reveals that women, constituting 65.0% of caregivers at home, primarily provide caregiving. 25 This statistic suggests a possible disparity in the support systems available to men and women, with men potentially receiving more consistent caregiving support, an essential aspect of heart failure management. This potential gender imbalance in caregiving aligns with trends reported in international literature. For instance, male caregivers of individuals with dementia in Finland experienced lower burden than female caregivers, despite care recipients having more severe disease. 26 A meta-analysis of gender differences in caregiver found that female caregivers, compared to male caregivers, provided more caregiving hours, assisted with more caregiving tasks, and reported higher levels of caregiver burden. 27 Additionally, research in Finland found that male caregivers of Alzheimer's patients were associated with poorer nutrient intake in both patients and caregivers. 28 Regarding our findings on PAFs, we observed a reduction in sex differences among those aged 85 and above. This observation might imply a decrease in caregiving capacity due to the caregivers’ advancing age, which could influence the quality of care received by male patients. While our data do not directly demonstrate a decline in caregiving quality, these findings highlight the need for sex-specific management strategies that consider the caregiving context and cater to the support needs of both patients and their caregivers.
One strength of our study is the highly comprehensive data for individuals aged 75 and older. The data captures nearly all residents aged 75 and older (except those receiving public assistance) in the regions where data were collected, enhancing the generalizability of our findings.
However, our study has several potential limitations. First, using administrative claims data introduces potential biases due to variations in coding accuracy. The identification of Alzheimer's disease and heart failure based on ICD-10 codes depends on the precision of clinical diagnosis and coding for insurance claims. While we used codes commonly employed in research, validation studies in the Japanese context are limited. For heart failure, a Japanese validation study reported moderate sensitivity (84.7%) but lower positive predictive value (57.0%) for ICD-10 code-based identification. 29 Although we attempted to improve specificity by focusing on primary diagnoses, the inherent limitations of claims data mean that some degree of misclassification likely remains. Future research needs to conduct comprehensive validation studies of ICD-10 codes for these conditions in Japanese claims data to further enhance the reliability of such database studies. Second, the observational nature of our research cannot confirm causality between heart failure and increased mortality in individuals with Alzheimer's disease, with the possibility of unmeasured variables like lifestyle, caregiving, and socioeconomic status influencing outcomes. Third, as we defined heart failure patients as those with heart failure as the principal diagnosis, our study population might consist of individuals with relatively severe heart failure. A systematic review found that the median prevalence of heart failure in older populations was 11.8% (range 4.7–13.3%). 30 Considering this, our study's heart failure prevalence of 6.7% might suggest an underrepresentation of the broader heart failure population, potentially limiting the generalizability of our findings. Lastly, the absence of detailed clinical data on the severity of Alzheimer's disease and heart failure restricts our understanding of the observed associations, which is crucial for developing targeted management strategies.
In conclusion, our study underscores the substantial association between comorbid heart failure and increased mortality rates within one year of Alzheimer's disease diagnosis in older individuals, with women experiencing higher HRs and PAFs across all age groups. These findings highlight the critical need for early intervention and sex-specific management strategies to improve outcomes for older individuals with newly diagnosed Alzheimer's disease and comorbid heart failure.
Supplemental Material
sj-docx-1-alz-10.1177_13872877241305813 - Supplemental material for Sex-specific association of comorbid heart failure on mortality after Alzheimer's disease diagnosis in older adults aged 75 years and above: A health insurance claims data analysis in Japan
Supplemental material, sj-docx-1-alz-10.1177_13872877241305813 for Sex-specific association of comorbid heart failure on mortality after Alzheimer's disease diagnosis in older adults aged 75 years and above: A health insurance claims data analysis in Japan by Masaaki Matsunaga, Shinichi Tanihara, Yupeng He, Hiroshi Yatsuya and Atsuhiko Ota in Journal of Alzheimer's Disease
Footnotes
Acknowledgments
The database was provided by DeSC Healthcare Inc. under their academic research support program. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. We thank all staff at DeSC Healthcare Inc. ChatGPT, version 4.0, a language model developed by OpenAI (![]() ), was used for language refinement of this manuscript.
), was used for language refinement of this manuscript.
Author contributions
Masaaki Matsunaga (Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Resources; Software; Validation; Visualization; Writing – original draft; Writing – review & editing); Shinichi Tanihara (Validation; Writing – review & editing); Yupeng He (Validation; Writing – review & editing); Hiroshi Yatsuya (Validation; Writing – review & editing); Atsuhiko Ota (Validation; Writing – review & editing).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data sets of the current study are not publicly available due to restrictions on the use of the data provided from DeSC Healthcare Inc. under the license for the current study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
