Abstract
Background
Adherence is critical in patients with Alzheimer's disease (AD) in order to achieve optimal benefit from therapy. However, patient compliance with the treatment remains a challenge.
Objective
To evaluate, in a real-world clinical setting, caregiver preference and treatment compliance with twice-weekly versus daily transdermal rivastigmine patch in mild-to-moderate AD.
Methods
92 patients who had been treated with daily rivastigmine patch for at least six months prior to switching to twice-weekly patch were evaluated. The change in therapeutic regimen was decided by the treating physician in accordance with standard practice. Caregivers’ satisfaction with daily rivastigmine patch was assessed at study entry. Caregiver's preference and satisfaction with twice-weekly patch as well as patient compliance were evaluated at weeks 12 and 24 using the Alzheimer's Disease Caregiver Preference Questionnaire.
Results
A significantly higher proportion of caregivers expressed a preference for the twice-weekly patch over the daily patch (p < 0.001), and this preference was found to be associated with ease of use (p < 0.001), ease of following the schedule (p < 0.001), and ease of compliance (p < 0.001). Furthermore, caregivers were more satisfied with the twice-weekly patch (p < 0.0001). At 24 weeks, patient compliance was significantly better with the twice-weekly patch than with the daily patch (p = 0.002). Caregiver burden significantly improved at the end of the treatment (p = 0.003). No serious adverse events were reported.
Conclusions
The twice-weekly rivastigmine patch offers a convenient and straightforward dosing regimen for caregivers, with potential to enhance adherence with treatment in AD patients without causing serious adverse events.
Keywords
Introduction
Dealing with dementia is one of the greatest challenges in medicine today. Of all of them, Alzheimer's disease (AD) is the most frequent, accounting for nearly 70% of the total of cases. 1
Significant advancements have been made in the development and approval of disease-modifying therapies in AD. Two anti-amyloid monoclonal antibodies, aducanumab and lecanemab, that demonstrate efficacy in slowing the cognitive decline associated with AD have been approved, and a third, donanemab, is currently under review.2–4 The cholinesterase inhibitors (rivastigmine, galantamine, and donepezil) have been shown to be an effective treatment for mild to moderate AD manifestations.5,6 They can help to slow down the progression of symptoms or delay institutionalization of the patient.6,7
Adherence is critical in patients with AD in order to achieve optimal benefit from therapy and improve or maintain quality of life.8,9 However, patient compliance with the treatment remains a challenge. Indeed, lack of adherence to drug treatment is a major problem in older people with often multiple and chronic diseases, as is the case with AD. 9 Memory problems and other cognitive deficits associated with dementia make patients with AD especially susceptible to therapeutic noncompliance. Medication adherence in these patients is also affected by several other factors that are common in elderly people, such as physical impairments, polymedication and an increased risk of drug interactions and sensitivity to adverse effects.10–12
As the dementia progresses, patients require increasing levels of care and most people with AD need help managing their medication. 13 In this sense, caregivers, especially family members, are often responsible for administering and managing medications and play a key role in the care and treatment compliance of patients with AD.9,14 In fact, medication management contributes to the burden associated with caring for patients with AD.9,14
The caregiveŕs responsibility is often a cause for concern, especially when side effects associated with medications occur or there are concurrent illnesses involving other drug administration schedules. In addition, having time to administer treatment can also be a source of stress for caregivers who have other commitments, such as work. 15 Thus, the choice of treatments among the available options is likely to be influenced by the preference of the caregiver, who will tend to choose easier to use modes of drug administration. This could decrease caregiver stress, favoring patient health and medication adherence. 9 Understanding caregivers’ treatment preferences and the reasons for those preferences can help maximize the effectiveness of the intervention.
Rivastigmine is an acetylcholinesterase and butyrylcholinesterase inhibitor approved for the symptomatic treatment of mild to moderate AD and is available to be administered orally or as transdermal patches. Its efficacy and safety have been demonstrated in several randomized controlled trials.16–19
In the 24-week, randomized, double-blind, placebo controlled IDEAL trial, which investigated once-daily rivastigmine patches versus twice-daily capsules in patients with moderate AD, more than 70% of caregivers expressed a preference for the patch over the capsules.20,21 The patch was preferred to capsules in terms of ease of use and adherence to the administration schedule. Caregivers reported greater satisfaction with the patch overall and less interference with daily life compared to capsules. Study results have showed that treatment with the daily rivastigmine transdermal patch may improve adherence and compliance compared to oral administration, which could lead to increased treatment benefits for patients.21,22
In addition to once-daily rivastigmine patch, a twice-weekly patch has recently been developed. 23 The innovative drug delivery system is designed to deliver the drug continuously over an extended period of up to four days, allowing twice-weekly administration with the patch being replaced after 4 days/3 days. This allows for two fixed days of patch change per week.
Consequently, the twice-weekly patch requires a less frequent application, which will provide a better guarantee of receiving rivastigmine at a consistent and regular interval. Since patient and caregiver satisfaction is essential for good treatment adherence, 9 the use of twice-weekly rivastigmine patch could be associated with a need for less planning and logistics of patients and caregivers and thus less burden on the caregiver. It may offer tolerability, convenience and therapeutic advantages for this patient population and could be associated with increased treatment compliance compared to once-daily rivastigmine patch.
The primary objective of this study was to evaluate, in a real-world clinical setting, caregiver preference and satisfaction with twice-weekly versus daily transdermal rivastigmine patch. Additionally, the study aimed to evaluate caregiver assessment of patient compliance with treatment with twice-weekly versus daily rivastigmine patch in patients with mild-to-moderate AD. As a secondary objective, we aimed to assess caregiver burden and examine the patientś overall clinical global impression of change and cognitive performance. The data presented in this study are derived from routine clinical practice
Methods
Study design
This was a 24-week, multicenter, prospective, noninterventional study conducted with AD patients who have been switched to twice-weekly transdermal rivastigmine patch from daily rivastigmine patch by their treating physician. Patients were recruited from eight sites that included one hospital, four neurology clinics and three research centers in different cities in Spain between June 2023 and December 2023. The observational period for each participant was 24 weeks. Data were collected at three time points: study entry (baseline), week 12 (±2 weeks), and week 24 (±2 weeks; end of the study).
The study was conducted in accordance with the definition of non-interventional trials. 24 Accordingly, the study drugs were prescribed in compliance with the marketing authorization, and the treatment decision was made as part of the patients’ routine medical care and prior to study participation. Patient assignment to therapy was decided within current practice and medical indication and was clearly separated from the decision to include the patient in the study. No diagnostic procedures or follow-up were performed in addition to routine and usual care. Patient treatment, visits, and evaluations were independent of this study and were at the discretion of the treating physician following local standard medical practice and local prescribing information.
The study protocol was approved by the Málaga Research Ethics Committee. A written informed consent was signed by patients who were able to give or their legal representative prior to their inclusion. Informed consent was also requested from caregivers who participated in the study. The study followed the ethical standards adopted by the Declaration of Helsinki in its latest version (Fortaleza, Brasil, 2013) and was conducted in accordance with the standards of Good Clinical Practice, as described in the Tripartite Harmonized Standards of the International Conference on Harmonization for Good Clinical Practice 1996. This study is registered in the Spanish Clinical Trials Registry (REEC) under the number 0067-2024-OBS.
Study participants
Eligible participants were patients with a diagnosis of AD according to the benchmarks set forth by the National Institute on Ageing Alzheimer's Association workgroups. 25 To be included in the study AD patients had to be at stage 3 to 5 of the Reisberg Global Deterioration Scale (GDS), 26 indicating mild to moderate dementia. In addition, eligible participants were patients who had been treated with daily rivastigmine patch for a minimum period of six months prior to switching to twice weekly rivastigmine patch.
An additional criterion for inclusion in the study was that the patient is cared for by a family caregiver. In this sense, family caregivers are relatives who provide a broad range of assistance to the person suffering from dementia including, among other tasks, the administration of medications needed by the patient. They must have sufficient knowledge of the patient, usually a first-degree relative or partner living in the same patient's home.
Assessments
The primary objectives were to assess caregiver preference and satisfaction for twice-weekly rivastigmine transdermal patch and caregiver assessment of patient compliance with the treatment from baseline to week 24. For this purpose, we used the Alzheimer's Disease Caregiver Preference Questionnaire (ADCPQ). The ADCPQ was developed to assess aspects of AD treatment influential in caregiver preferences, including treatment expectations, expected preferences, treatment evaluation and satisfaction and overall treatment satisfaction.19,21,27
Caregivers’ satisfaction with daily rivastigmine patch was assessed at study entry. Caregivers’ satisfaction with twice-weekly rivastigmine patch was assessed at weeks 12 and 24. In both cases, satisfaction was evaluated using a Likert scale ranging from 1–5, where 1 represents the worst result (e.g. very dissatisfied) and 5 the best (e.g. very satisfied).
Caregiverś preference for twice-weekly rivastigmine patch or daily rivastigmine patch was evaluated at weeks 12 and 24. The ADCPQ required caregivers to respond to, “Which of the following treatments (daily patch or twice weekly patch) would you prefer to give in the future to manage Alzheimer's disease?”. Also caregivers were asked to indicate the reasons for treatment preference: ease of use (e.g. easy to open, apply or peel off), ease of following the schedule (e.g. the number of time per day) or ease of compliance with treatment.
In addition to assessing caregiveŕs satisfaction and preference, patient´s compliance with both the daily rivastigmine patch at study entry and the twice-weekly patch at weeks 12 and 24 was also assessed. In both cases compliance with treatment was evaluated using a Likert scale ranging from 1–5, where 1 represents the worst result (e.g. never took the medication as prescribed) and 5 the best (e.g. always took the medication as prescribed).
The secondary objective was to assess the caregiver burden using the Caregiver Burden Interview 28 at study entry and at the end of the study. The Caregiver Burden Interview (CBI) is a 22-item self-report questionnaire, which measures caregivers’ subjective burden of caregiving, in terms of health, finance, and social/interpersonal relationships. Participants’ responses were made on a five-point scale (0 = never to 4 = nearly always). The total score can range from 0 to 88.
Additional assessments included the Mini-Mental State Examination (MMSE) 29 at study entry and week 24 to evaluate cognitive functioning and the Clinical Global Impression of Improvement scale (CGI-I)30,31 at week 24 to assess the improvement perceived by the clinician and the caregiver. The CGI-I assesses improvement due to therapeutic interventions. It ranges from 1 (very much improved) to 7 (very much worse).
Safety evaluations included the collection of all treatment-emergent adverse events (TEAEs). The investigator conducted a visual inspection of the skin at the site of application at each visit to assess for any signs of irritation. The caregiver also provided feedback on any observed skin irritation.
Statistical analysis
To estimate the sample size, we based on the IDEAL study 21 which assessed caregiver preference for rivastigmine patch over capsules for treatment of AD. Our analysis indicates that a sample of 81 subjects would be sufficient to detect a preference rate for twice-weekly rivastigmine patch of 70%, assuming a 95% confidence level and a 10% margin of error. This sample size would also allow detection of a statistically significant difference of at least 0.5 points (10%) in the mean score of caregivers’ overall satisfaction with twice-weekly rivastigmine patches relative to daily rivastigmine patches. To do this calculation we have assumed a relative variability of 22%. In both cases we have set a power of 80% and a significance level of 5%.
For the analysis, we included all caregivers who in addition to the baseline completed the ADCPQ at week 12 and/or 24 and were caregivers of patients who received at least one dose of twice-weekly rivastigmine patch.
Sociodemographic data were recorded at baseline and analyzed by means of number of observations, mean and standard deviation for quantitative variables and absolute and relative frequencies for categorical variables.
Direct caregiver preference for daily or twice-weekly rivastigmine patches was compared by examining percentages using tests of binomial proportions. A 95% confidence interval was calculated around proportions of caregivers showing preferences for daily or twice weekly patches. Quantitative differences in caregiver satisfaction comparing daily or twice-weekly patches were analyzed using paired Student's t-test for means. These analyses were replicated using a nonparametric test, the Wilcoxon signed-rank test, to confirm the results obtained with parametric paired t-tests. To assess significant differences in patient compliance depending on the use of daily or twice-weekly patches Student's t-test for means were used. These analyses were repeated with a nonparametric test, the Wilcoxon signed-rank test, to confirm the results obtained with parametric paired t-tests.
In addition to the analyses described above, subgroup analyses were conducted to determine whether caregiver characteristics (sex, disease severity, relationship to patient or caregiveŕs place of residence) influenced overall preference for twice-weekly patch versus daily patch.
Statistical analyses were performed using JASP statistical package version 0.18.03 (JASP, University of Amsterdam, Amsterdam, The Netherlands). All analyses were performed applying two-sided tests and the significance level was set at p < 0.05.
Results
Patients and caregivers population
Patient and caregiver demographics and clinical characteristics at baseline are shown in Table 1. A total of 92 patients met the inclusion criteria and their data were available for analysis (47 female, 45 male). Patients had a mean age of 77.8 years (range 60-93) and mean years of education of 9.73 (range 3–20). With the exception of one patient who resided in a nursing home, all others were living at home.
Baseline patient and caregiver demographic and clinical characteristics.

Values are mean ± SD (Standard Deviation) or number (%).
MMSE: Mini-Mental State Examination; GDS, Global Deterioration Scale; CBI: Caregiver Burden Interview.
Following the completion of the second assessment at week 12, one participant withdrew from the study due to a diagnosis of cancer, while another participant died due to heart failure unrelated to the treatment. Most patients had comorbid medical conditions at baseline, the most common being hypertension (57.6% of patients), dyslipidemia (35.9%), and diabetes mellitus (18.5%). All these patients were receiving concomitant medications for their pathologies that were continued during the study period.
Caregivers had a mean age of 60 years (range 34–89) and mean years of education of 11.55 (range 3–20). A greater proportion of caregivers were female (71%) and approximately two-thirds (64.1%) were living with the patient. Caregivers were predominantly spouses (52%) or children (46%) of the patient. All patients and caregivers were Caucasian.
Caregiver preference
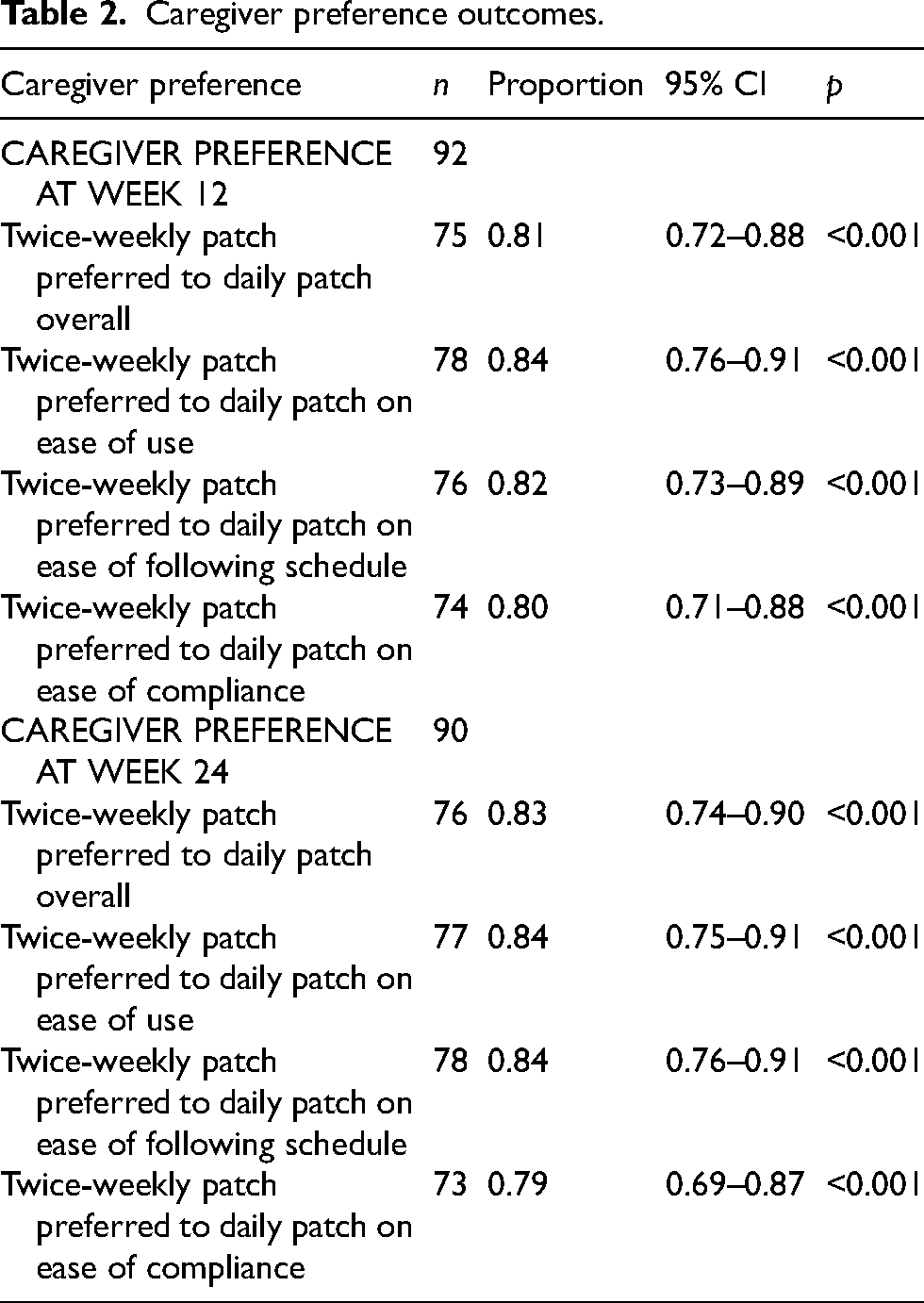
At week 12, 81% of caregivers expressed a direct preference for the twice-weekly patch to the daily patch overall for AD treatment administration (p < 0.001). Similarly, more caregivers preferred the twice-weekly patch to the daily patch on ease to use, ease following the schedule, and ease of compliance with treatment (all p < 0.001). At 24 weeks, 83% of caregivers expressed a direct preference for the twice-weekly patch to the daily patch overall (p < 0.001). Likewise, more caregivers preferred the twice-weekly patch to the daily patch on ease to use, ease following the schedule, and ease of adherence with treatment (all p < 0.001) (Table 2 and Figure 1).
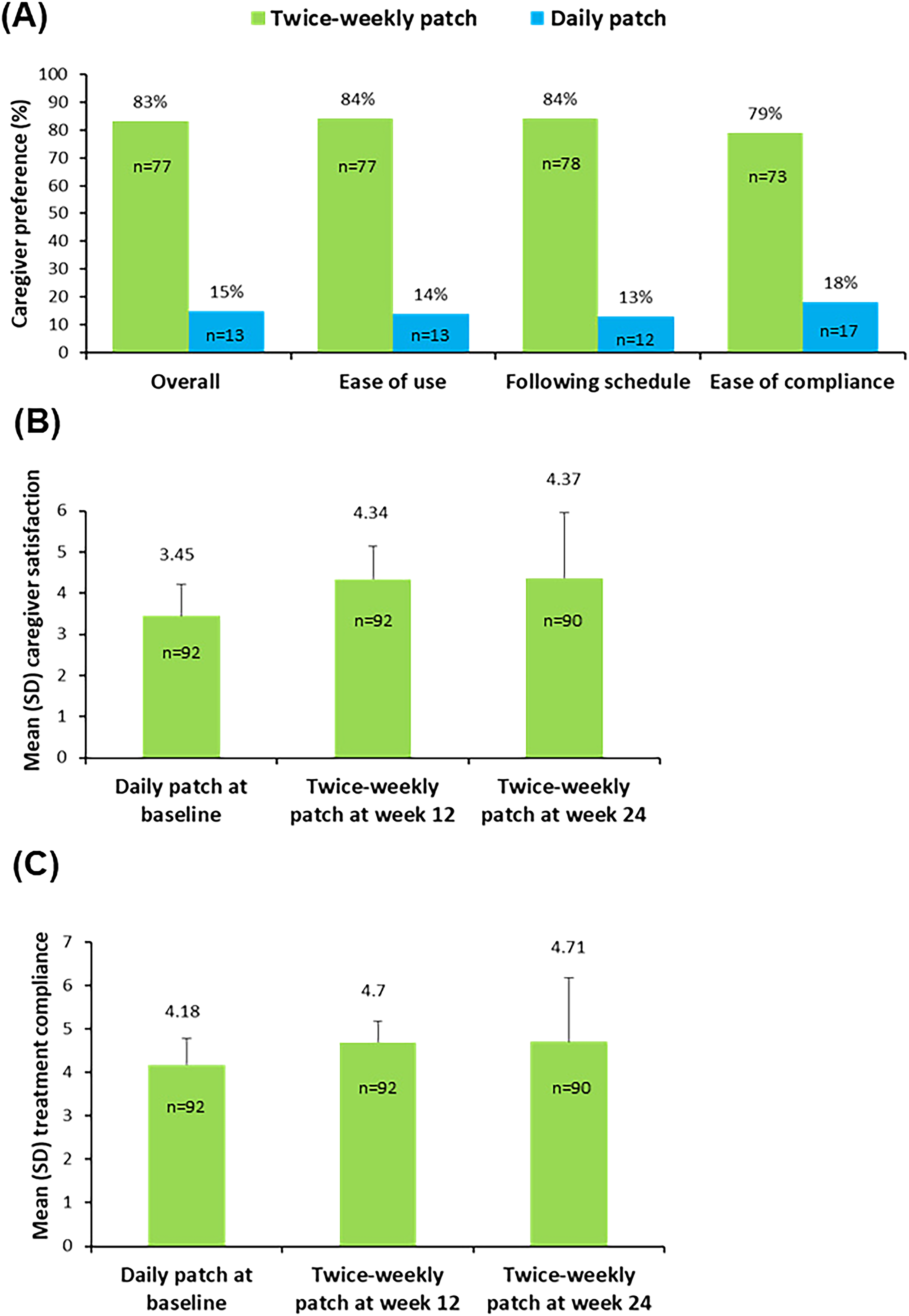
Caregiver preference and satisfaction and compliance with treatment. (A) Caregiver preference and reasons provided for preference for twice-weekly or daily rivastigmine patch at 24 weeks (n = 90). For all outcomes, p < 0.001, twice-weekly patch versus daily patch. (B) Caregiver satisfaction for twice-weekly or daily rivastigmine patch. For all outcomes, p < 0.0001, twice-weekly patch versus daily patch. (C) Compliance with the treatment. p < 0.0001, twice-weekly patch versus daily rivastigmine patch at week 12; p = 0.002 twice-weekly patch versus daily rivastigmine patch at week 24.
Caregiver preference outcomes.
Caregiver satisfaction
At 12 weeks, caregivers indicated greater satisfaction with the twice-weekly patch versus the daily patch (mean score 4.34 ± 0.80 versus 3.45 ± 0.76, mean difference 0.89, t = -8.11, p < 0.0001). At 24 weeks, caregivers also indicated greater satisfaction with the twice-weekly patch versus the daily patch (mean score 4.37 ± 1.58 versus 3.45 ± 0.76, mean difference 0.92, t = -5.10, p < 0.0001) (Figure 1). Similar results were obtained using the Wilcoxon signed-rank test and the paired t-test analyses. Seventy-seven (83.7%) caregivers at week 12 and 79 (85.9%) caregivers at week 24 reported that were satisfied or very satisfied with the twice-weekly patch versus 48 (52.1%) of patients taking the daily patch.
Treatment compliance
At week 12, patient compliance was significantly higher with the twice-weekly patch than with the daily patch (mean score 4.70 ± 0.48 versus 4.18 ± 0.61, mean difference 0.52, t = -6.54, p < 0.0001). At 24 weeks, patient compliance was also significantly higher with the twice-weekly patch than with the daily patch (mean score 4.71 ± 1.47 versus 4.18 ± 0.61, mean difference 0.53, t = -3.22, p = 0.002) (Figure 1). Similar results were obtained using the Wilcoxon signed-rank test and the paired t-test analyses. Sixty-five (70.7%) caregivers at week 12 and 87 (94.6%) caregivers at week 24 reported that the patient always took the twice-weekly patch as prescribed versus 27 (29.3%) of patients taking the daily patch.
Caregiver burden
A statistically significant improvement in the caregiver burden, as measured by the CBI, was seen from baseline to week 24 (mean score 40.64 ± 15.24 versus 37.68 ± 14.64, mean difference 2.96, t = 3.04, p = 0.003) (Figure 2).
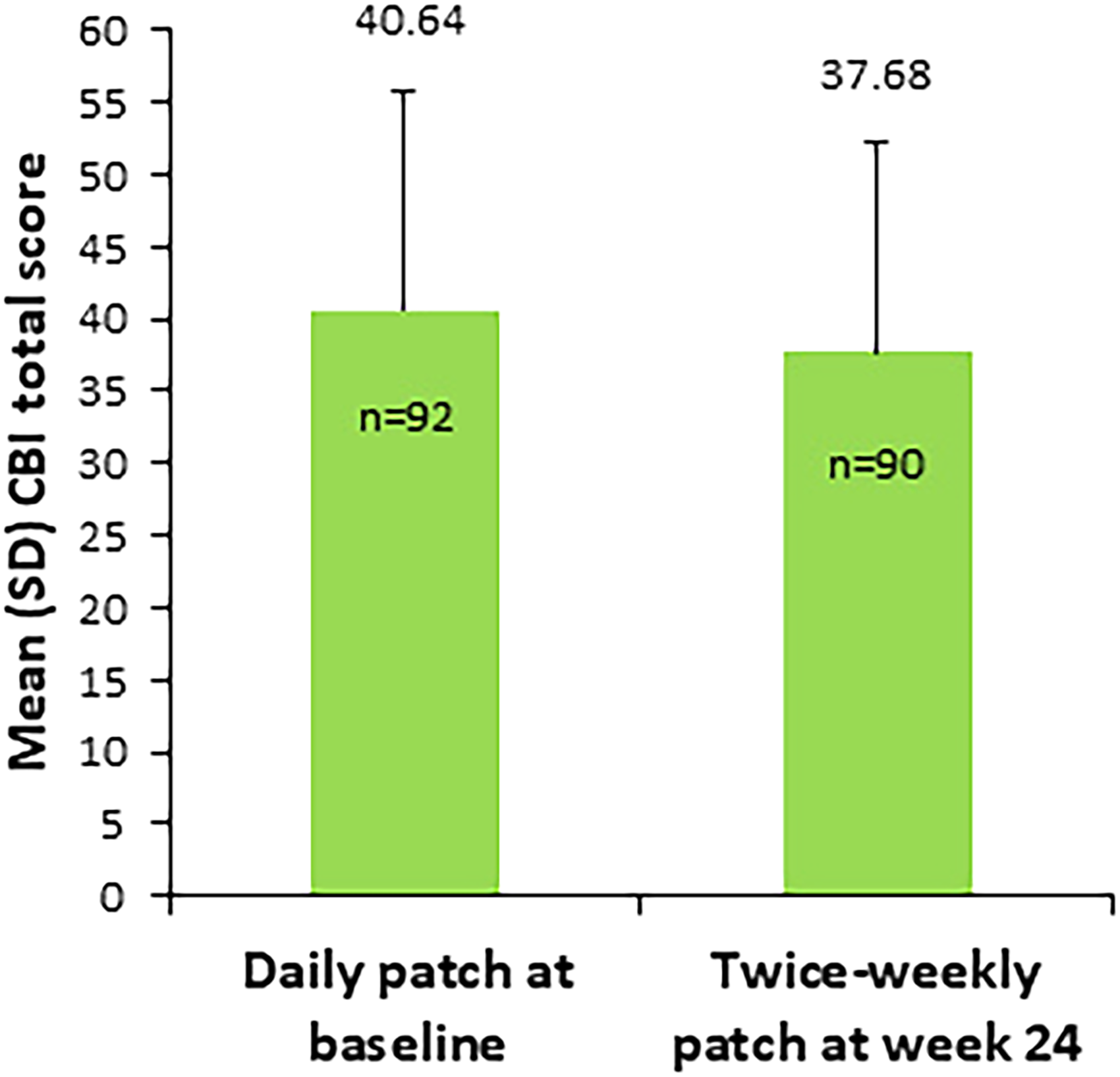
Caregiver burden total score from baseline to week 24. CBI: Caregiver Burden Interview; Values are mean (Standard Deviation). p = 0.003 versus baseline.
Additional assessments
There were no statistically significant differences in the MMSE score from baseline to week 24 (20.39 ± 4.73 versus 19.53 ± 7.19, p = 0.453). At the end of the study, the mean cliniciańs perceived improvement compared to the initiation of treatment, as measured by the CGI-I, was 3.10 ± 2.21 which corresponds approximately to the minimally improved condition on the CGI-I score. In the same line, the mean CGI-I score reported by caregivers at the end of the study was 3.33 ± 2.16 (Figure 3).
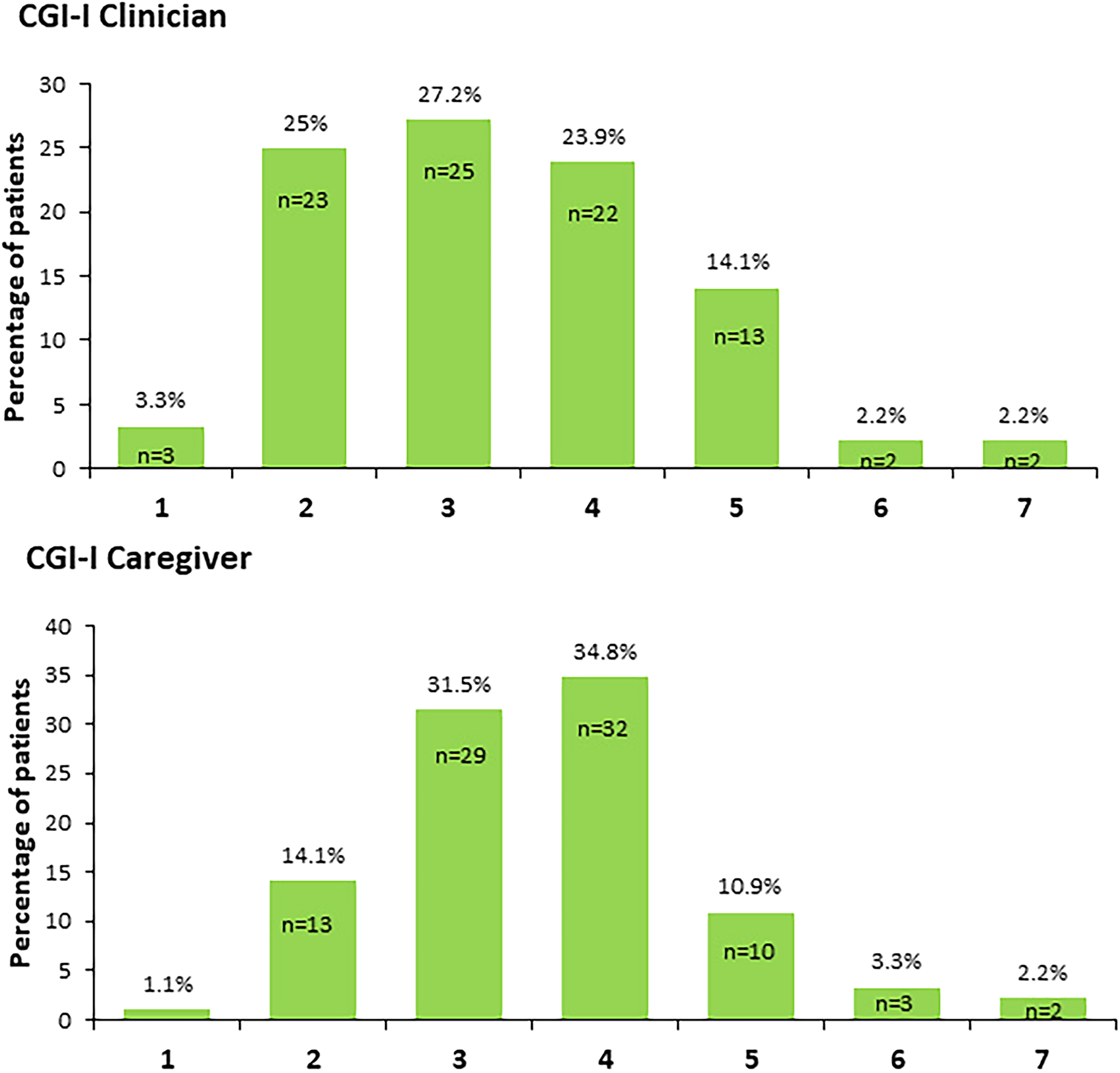
Individual items of the Clinical Global Impression–Improvement (CGI-I) Scale at the end of the study. 1, Very much improved; 2, Much improved; 3, Minimally improved; 4, No change; 5, Minimally worse; 6, Much worse; 7, Very much worse.
Subgroup analysis
At week 24, preference for the twice-weekly patch was consistent across all subgroups: caregiver sex (p = 0.132), disease severity (p = 0.230), relationship to caregiver (p = 0.389) and caregiveŕs place of residence (p = 0.165). The results in the subgroups were similar to those observed in the overall study population, indicating that preference does not seem to be affected by caregiver characteristics.
Safety analysis
Rivastigmine twice-weekly patch was well tolerated and no unexpected TEAEs were reported (Table 3). During the 24-week treatment period, 28 TEAEs were reported by 16 patients (17.39%). The most commonly reported TEAEs during treatment were pruritus (7.6%) and skin irritation (6.5%). Most TEAEs were transient, were mild or moderate, and resolved spontaneously. No deaths or serious TEAEs occurred during the study. Skin tolerability was good, and most patients (93.5%) did not experience skin irritation. Adverse events did not lead to treatment interruptions in any patient.
Summary of adverse events reported (n = 92).
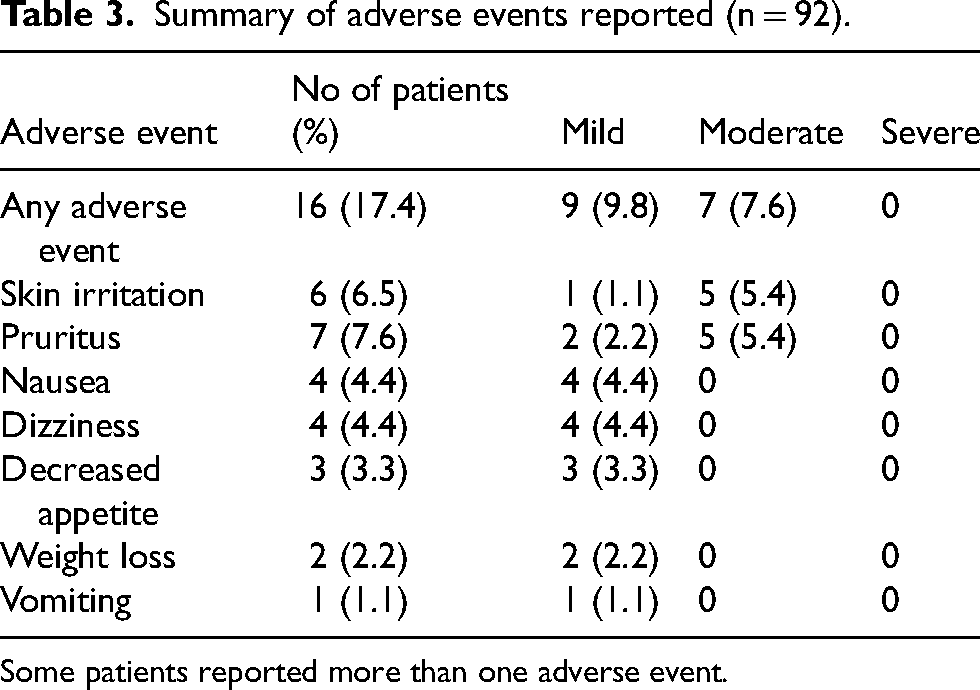
Some patients reported more than one adverse event.
Discussion
To the best of our knowledge, this is the first study to evaluate, in real-world clinical practice, caregiver preference and satisfaction with twice-weekly versus daily transdermal rivastigmine patch and caregiver assessment of patient compliance with treatment in patients with mild-to-moderate AD.
In our study, a significantly greater proportion of caregivers participating in the study expressed a preference for the twice-weekly patch over the daily patch in patients who were exposed to both modes of administration, and this preference was found to be associated with ease of use, ease of following the schedule, and ease of treatment compliance. At 24 weeks, patient compliance with treatment was also significantly better with the twice-weekly patch than with the daily patch. Furthermore, caregivers expressed greater satisfaction with the twice-weekly patch, noting that it had a minimal impact on their daily lives. Caregiver burden significantly improved at the end of the treatment.
It is possible that some participants’ initial preferences for the twice-weekly patch may have been influenced by a sense of novelty associated with the introduction of a novel treatment. Nevertheless, by week 24, this factor should have dissipated, and its impact on the results should be minimal.
Lack of adherence to drug treatment is a major problem in AD patients. 9 The presence of cognitive deficits, physical impairments, polymedication, and sensitivity to adverse effects renders AD patients particularly susceptible to therapeutic noncompliance with most patients needing help from their caregivers for medication management.10–12 In this context, caregivers, particularly family members, are mainly responsible for medication management, playing a key role in the treatment compliance of patients with AD. Indeed, medication management contributes to the considerable burden associated with caring for patients with AD. 32 It is therefore likely that the choice of treatments among the available options will be influenced by the preference of the caregiver, who will tend to choose easier-to-use modes of drug administration. 13 Thus, the simplification of treatment regimens may assist in reducing the burden on caregivers, which may subsequently result in enhanced medication adherence and a positive impact on the patient's condition. 9
Caregivers’ preference for treatment of AD and compliance has been previously assessed in various other studies. The IDEAL trial, the first to demonstrate the efficacy of the rivastigmine transdermal patch, also showed that 72% of caregivers preferred the transdermal monotherapy over the oral capsules. 21 The results of the RECAP study showed that transdermal monotherapy is a preferred treatment option for both caregivers and physicians, and that patients are more likely to comply with this treatment in daily practice. 33 Similarly, the EMBRACE study, which evaluated the real-world efficacy and tolerability of the rivastigmine patch, demonstrated a high level of caregiver preference (88.2%) for the daily patch over oral medication in patients with mild to moderate AD. 34 In addition, findings from an observational study in patients with mild-to-moderate AD showed that the transdermal formulation of rivastigmine can reduce the burden on caregivers in daily practice. 35
The findings of the present study indicate that the twice-weekly rivastigmine patch may offer significant advantages to caregivers in terms of ease of medication administration and management of treatment schedules. Patients using twice-weekly patches exhibited a higher level of compliance compared to those using daily patches. In addition, patients using daily patches tended to miss medication more often than those using twice-weekly patches. Therefore, it may be suggested that both the presence of more satisfied caregivers and a preference for the twice-weekly patch may result in better treatment compliance in patients with mild-to-moderate AD.
The most frequently identified reasons for medication noncompliance are forgetfulness, avoidance of adverse effects and complex dosing regimen. 9 Due to the challenges associated with memory in patients with AD, ensuring that patients take their medication as prescribed and at the appropriate time and dosage can be particularly difficult. 9 Our findings suggest that twice-weekly rivastigmine patches represent a more streamlined and straightforward approach to medication management, improving treatment adherence in forgetful patients. In our study, almost all caregivers (94.6%) informed that the patient always took the twice-weekly rivastigmine patch as prescribed.
In addition to forgetfulness, older individuals may choose to discontinue medication to avoid the associated adverse effects. 9 Although with the use of rivastigmine patches skin irritation can occur, its incidence is relatively low and rarely results in the patch being withdrawn (2.4% in the rivastigmine patch group in the IDEAL study). 19 Similarly, a 6-month open-label study showed a favorable therapeutic response and safety profile of the rivastigmine patch, with only 6.8% of withdrawals due to local skin reactions. 36 In the present study, rivastigmine twice-weekly patch was shown to be well tolerated in elderly patients with AD. No patients discontinued treatment due to adverse events. Most adverse events reported were of mild to moderated intensity and only 6.5% of patients experienced mild to moderate skin irritation.
Finally, the frequency of medication dosing has been shown to have a direct impact on good treatment compliance. The dosing frequency has an inverse relationship with de compliance rates, 37 so therapies administered in a simpler form, such as twice-weekly patches, are easier to adhere with. In this sense, in our study the use of twice-weekly patches contributed to simplify the drug regimen.
Over 6 months of treatment with rivastigmine patch, approximately 50% of patients experienced overall clinical improvement based on the CGI-I score (i.e. CGI-I score ≤3). A total of 51 (55.5%) clinicians considered the patient´s condition as very much improved to minimally improved. In the same line, 43 (46.7%) caregivers reporting the patient´s condition as very much improved to minimally improved. These overall clinical improvement data together with the strong caregiver preference and the greater satisfaction with the twice-weekly rivastigmine patch, suggest that a twice-weekly patch may have the potential to enhance overall patient outcomes of treatment.
Caregiver burden associated with treatment not only has a major impact on personal satisfaction, but also on adherence to treatment. 22 In our study, caregivers reported greater satisfaction and less interference with daily life using the twice-weekly patch. These findings showed that caregiver burden exhibited a significant reduction at the end of the treatment period. One potential explanation for this outcome is that the administration of the twice-weekly rivastigmine patch may have enhanced the sense of self-sufficiency and empowerment among caregivers. Thus, our data suggest that by simplifying the treatment regimen and daily routine, the twice-weekly patches have the potential to reduce the caregiver burden associated with administering medication for an individual with AD, which may in turn lead to enhanced treatment compliance and have a favorable impact on the patient´s condition.32,38
The strengths of this study were that it was conducted under “real-world” medical practice conditions and that all patients were exposed to both forms of therapy, allowing all caregivers to offer a comparison of satisfaction, preference, and compliance. The main limitation of this study is that it was an observational, open-label study, which inherently carries the potential for bias. Although this study was multicenter, is advisable to exercise caution in generalizing these results.
Conclusions
In conclusion, the results of this study show that the use of the twice-weekly rivastigmine patch is an easy-to-use and friendly dosing regimen for the caregiver. Its use has the potential to help to achieve higher benefits of the treatment and may provide a novel and valuable strategy to enhance compliance with treatment in AD therapy. In addition, the twice-weekly rivastigmine patch was well tolerated and no unexpected or serious adverse events were reported. Further prospective studies will be required to substantiate these promising observational findings.
Footnotes
Acknowledgments
We wish to thank patients and caregivers for their participation in the study.
ORCID iDs
Author contributions
José María García-Alberca (Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Supervision; Validation; Writing—original draft; Writing—review & editing); Paz de la Guia (Investigation; Resources; Software; Supervision; Writing—original draft; Writing—review & editing); Esther Gris (Investigation; Methodology; Resources; Software; Supervision; Writing—original draft; Writing—review & editing); Silvia Mendoza (Investigation; Methodology; Software; Supervision; Writing—original draft; Writing—review & editing); Maria Lopez de la Rica (Investigation; Methodology; Software; Supervision; Writing—original draft; Writing—review & editing); Jose Antonio Lopez -Trigo (Investigation; Methodology; Supervision; Writing—original draft; Writing—review & editing); Rosa Lopez-Mongil (Investigation; Methodology; Supervision; Writing—original draft; Writing—review & editing); Teresa Garcia-Lopez (Formal analysis; Investigation; Supervision; Writing—original draft; Writing—review & editing); Raquel López-Garcia (Formal analysis; Investigation; Methodology; Writing—original draft; Writing—review & editing); Teresa Rodriguez-del Rey (Investigation; Methodology; Supervision; Writing—original draft; Writing—review & editing); Javier Gay-Puente (Investigation; Methodology; Supervision; Writing—original draft; Writing—review & editing); Jesus Garcia-Castro (Investigation; Methodology; Supervision; Writing—original draft; Writing—review & editing); Federico Casales (Investigation; Methodology; Supervision; Writing—original draft; Writing—review & editing); Xavier Morato (Investigation; Methodology; Supervision; Writing—original draft; Writing—review & editing); Merce Boada (Investigation; Methodology; Supervision; Writing—original draft; Writing—review & editing); Gemma Gonzalez-Velasco (Investigation; Methodology; Supervision; Writing—original draft; Writing—review & editing); Jose Manuel Marin-Carmona (Investigation; Methodology; Supervision; Writing—original draft; Writing—review & editing); Nora Ines Paez (Investigation; Methodology; Supervision; Writing—original draft; Writing—review & editing); Maria Leon (Investigation; Methodology; Supervision; Writing—original draft; Writing—review & editing); Rosario Carrillejo (Investigation; Writing—original draft); Francisca Rius (Formal analysis; Writing—original draft; Writing—review & editing); Miguel Angel Barbancho (Formal analysis; Investigation; Methodology; Writing—original draft; Writing—review & editing); Jose Pablo Lara (Data curation; Formal analysis; Investigation; Methodology; Validation; Writing—original draft; Writing—review & editing); Encarnacion Blanco-Reina (Data curation; Formal analysis; Investigation; Methodology; Validation; Writing—original draft; Writing—review & editing).
Funding
Research reported in this publication was supported in part by Esteve and Luye. The funders of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
