Abstract
This article takes as a case study a set of disagreements in the early 2000s about randomised controlled trial (RCT) evidence for a newly developed drug in the field of intensive care medicine. The interpretation of RCT findings – and in particular, the application of these findings to clinical practice – were contested among research-active intensive care doctors, despite their shared professional and epistemic values. I examine the arguments about scientific interpretation and application to clinical practice advanced by two readily identifiable groups. The analysis documents how four particular aspects of scientific knowledge were perceived and portrayed differently by the two groups, and notes how each group was associated with different kinds of routine work practices and external networks. My argument is that these differences give rise to distinctive hermeneutic frames and orientations towards the scientific results and disparities in their consequential judgements regarding the legitimate use of the newly developed drug, and I extend Stones and Turner’s concept situationally specific habitus to make the link between context and hermeneutic frames and orientations. The analysis has implications for furthering our understanding of how the clinical meaning attributed to scientific evidence is affected by the context of reception of results, even where epistemic and professional values are shared.
Keywords
Introduction
The claim is made on behalf of randomised controlled trials (RCTs) that they provide the highest level of objective evidence for evaluating the efficacy and safety of medical interventions (e.g. GRADE Working Group, 2004), and therefore, by implication, the application of RCT evidence to clinical practice should be straightforward. However, previous social scientific research has subjected these kinds of claims to detailed empirical and theoretical scrutiny (see, e.g. collections edited by Wahlberg and McGoey (2007), Will and Moreira (2010) and Brives et al. (2016)). Researchers have problematised the presumed epistemological foundations of RCTs, such as the meanings of placebo (Lakoff, 2007), efficacy (Dumit and Sanabira, 2022) and the plausibility of mechanisms of pharmacological effect (Wahlberg, 2008). Furthermore, researchers have drawn attention to the contingencies of geographical contexts (Dumit and Sanabria, 2022), patient selection (Lakoff, 2007), regulative regimes (Abraham, 2007) and dissemination practices (Wahlberg, 2008; Will, 2010). Underlying these findings is the inherent tension between research, clinical and scientific perspectives (Brives, 2013; Will, 2007).
My primary aim in this article is to build on this social scientific research, in particular research on the context of dissemination, by a focus on the reception of RCT findings by clinicians – that is, their interpretation and application to clinical practice. The analysis is founded on the notion that scientific results such as those reported in RCTs are a product which must be interpreted in order to be applied to clinical practice, and the main contribution of the article is in showing how aspects of RCT results are perceived and portrayed differently according to different kinds of routine work practices and external networks. The article also has two subsidiary contributions. First, noting that much productive work in the social science of RCTs has been on psychiatric or chronic conditions, the case which I analyse is an RCT for a novel intervention for an acute biomedical emergency condition (sepsis). Second, I provide further support for the long-standing social scientific awareness of the distinction – and tensions – between science and medicine.
The analysis is of a case where there were significant disagreements about the meaning of evidence from an RCT in the field of intensive care medicine. The disagreements reveal that despite a shared overall frame of meaning (in this case, provided by the professional field of intensive care medicine and research), there are significant differences in the frames by which scientifically sophisticated clinicians evaluate, interpret and apply scientific results. The central contribution of the article is in positing a precise psychosocial mechanism, situationally-specific habitus (Stones and Turner, 2020: 193–195), for the development and consolidation of these different frames for the interpretation of scientific results.
The case
The disagreements examined in this article concerned the findings of a randomised, controlled trial (RCT) for an innovative treatment for sepsis. 1 Sepsis is defined in the medical literature as life-threatening organ dysfunction caused by a dysregulated host response to infection (Singer et al., 2016), and is regarded as a medical emergency (Evans et al., 2021). It is a heterogeneous and serious syndrome that is costly to treat, and which can have a high mortality rate even following intensive care. The trial at the centre of the disagreements was for a newly developed drug, Drotrecogin alfa (activated) [DAA]. 2 The efficacy and safety of DAA was tested in a large clinical trial with the results published in the New England Journal of Medicine (NEJM) showing a relative risk reduction in 28-day mortality of 19.4% (Bernard et al., 2001). Prima facie, this is a substantial risk reduction; if replicated in clinical practice it could reduce the proportion of deaths by almost one-fifth, although a risk of serious bleeding was also associated with the drug.
While this relative risk reduction was widely regarded as very encouraging, medicines regulators (the Food and Drug Administration in the USA and the European Medicines Agency) noted the safety concern of potential for serious bleeding. Both regulators imposed conditions for approval (for example, safety monitoring on an annual, rather than five-yearly, basis; and mandating a confirmatory trial in lower risk patients). Subsequently, a trial in low-risk patients detected no beneficial effect (Abraham et al., 2005), although the drug remained approved for use in high-risk patents. However, practising critical care doctors expressed scepticism over the drug’s effectiveness (Hopley and van Schalkwyk, 2006) and there was demonstrable ambivalence over its use in clinical practice (Rowan et al., 2008). Scientific debates at conferences and the medical literature continued during the period 2002 to 2008. These debates and disagreements polarised into a ‘bitter controversy’ (Mancebo and Antonelli, 2008) which only subsided once a new trial was announced. This trial took place in 2008–2011 and the results were in striking contrast to the first one: DAA ‘did not significantly reduce mortality’ (Ranieri et al., 2012), and the drug was withdrawn. The result of this subsequent clinical trial, and the degree of controversy which precipitated it, are not directly connected with the argument of this paper, which is concerned, rather, with the form of scientific discussion, debate and disagreement prior to this second trial.
Overview of argument
The analysis focuses on the specific scientific arguments presented by two readily identifiable groups of intensive care doctors during the period following the publication of the first trial. For analytical purposes the two groups are labelled Pragmatist and Purist, as delineated in Table 1 and elaborated in the analysis.
Overview of analysis: institutional positioning, external networks and the perspectives on science for two sets of actors in the DAA clinical trial controversy.
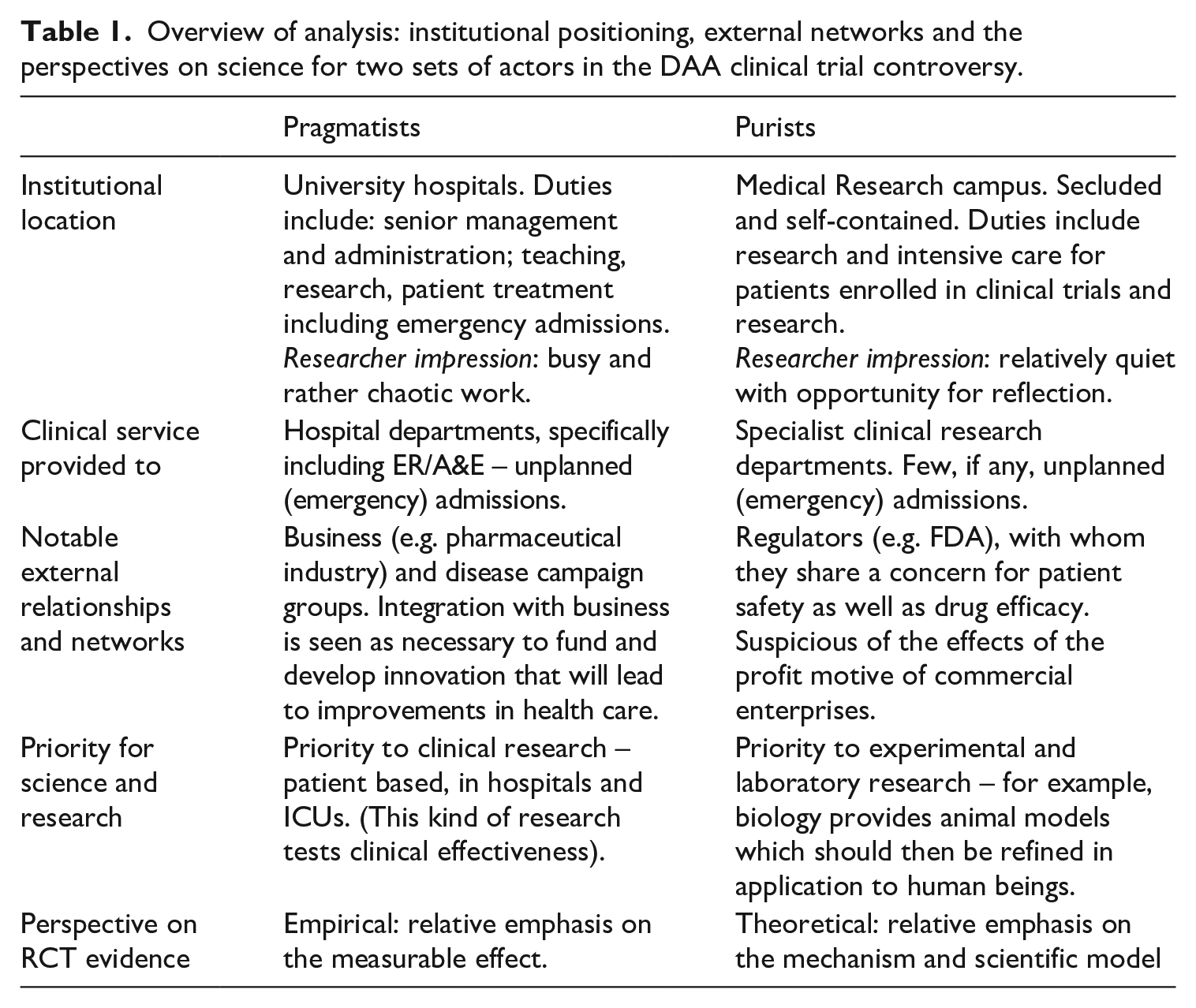
Each group consisted of widely published and internationally known intensive care doctors. The analysis examines closely the contextual positioning and hermeneutic frames of the protagonists, discussing their contexts in terms of routine work practices and external collaborative networks. This leads to an analysis of how, from within their respective frames, particular aspects of scientific knowledge are perceived differently:
Regarding experimental practice: different opinions about the importance of a change to the original protocol for the trial;
In terms of a general model of science: whether scientific evidence needs to be replicated;
In terms of the specific scientific explanation: whether there needs to be an established plausible mechanism by which the new drug treatment produces its effects;
In terms of the clinical application: the implications of the risk-safety profile of the drug.
The argument of this article is that the differing contexts of the protagonists give rise to distinctive hermeneutic frames, and that this difference is the basis of the schisms between the two camps in their respective orientations towards the scientific results. The paper proceeds by explaining the Analytical and theoretical approach, followed by the Methods and sources of data collection. Turning to the analysis itself, I first explore the Contexts for the two camps, specifically noting routine work practices and external networks. In the following two sections, I firstly elaborate the Purist perceptions of the science and then the Pragmatist perceptions of the science, each with respect to the four aspects of scientific knowledge mentioned above. In the Discussion I infer the connection between these differing perspectives on science and the kinds of scientific research which were valorised by each group, before considering wider implications in the Conclusion.
Analytical approach: Strong structuration theory
Strong structuration theory (SST; Stones, 2005) is used to explore the relations between the internal life of actors (phenomenological and hermeneutic) and their external contexts. SST assumes external constraints and resources are internalised, thus emphasising the power of positioning within a contextual field to influence the hermeneutic frames of individuals and groups. The key aim in the analysis of these disagreements is to illuminate the conflicting viewpoints of key participants in the debates by drawing out the linkages between the differences in the hermeneutic framing of the science and the specifics of their positional situation.
Since SST posits the internalisation of external constraints and resources (‘internal structures’) it has developed a sophisticated understanding and vocabulary for different kinds of internal structures. In the original formulation of SST there were two aspects to these ‘internal structures’ that can be separated out. The first is the way in which the external forces of socialisation (including, in this instance, socialisation into the professional values of the general field of intensive care medicine and research), slowly but surely shape certain enduring dispositions within actors, who tend to internalise and conform to many of their various codes and behaviours. Following Bourdieu, these general dispositions may be termed habitus (Bourdieu, 2000: 138; Bourdieu and Wacquant, 1992; Stones, 2005). While habitus may be thought of as forms of capital accumulated over the long term, it is important also to recognise that it can be conceived as a multilayered concept (Wacquant, 2016: 68). Thus the protagonists in the controversy could be assumed to share a professional habitus associated with training, education and socialisation in scientific research and intensive care medicine, layered on top of other layers of habitus such as general education, social class and cultural background.
SST’s second category of internal structure, originally termed conjuncturally-specific, was formulated to refer to specific knowledge necessary for responding to issues in the immediate structural terrain. An example of this, for the current case, would be the decision to publish a clinical review, commentary or letter: this would require specific knowledge of which journals are the best target for which kind of writing.
In addition to these two categories of internal structure, the empirical features of this case reveal how general and transposable dimensions of habitus that are associated with a given field can be differentiated from another sub-set of durable dispositions, which have a more specific anchoring in engagements with particular contexts, settings and processes within the relevant field. That these are durable, rather than conjuncturally-specific, gives motivation to draw on and develop the concept of situationally-specific habitus (Stones and Turner, 2020: 193–195).
Situationally-specific habitus
The key concept of this paper is based on the observation that as a consequence of their different contexts (to be elaborated in the next section), each of these groups were involved in distinguishable sets of customary work practices and external networks. The sets of attendant practices, orientations and dispositions undertaken by one group were subtly but significantly different to those assumed by the other, and the relatively enduring tasks, obligations, duties and expectations are interwoven with particular roles in specific contexts and settings.
The dispositions connected with the different settings (work practices and external networks) are less transposable than, say, professional habitus, but typically more enduring than the resources and constraints featuring in the many unique and transient elements of unfolding in situ events and processes. Following Wacquant’s (2016) characterisation of habitus as multi-layered, and Stones and Turner’s (2020) introduction of the term, the concept situationally-specific habitus is intended to capture this aspect of the ‘internalisation of the external’ which plays a key role in explaining the difference of orientation of the two medical groupings to the RCT results. 3
The analysis which follows, then, identifies two different forms of situationally-specific habitus embedded in the two medical groupings. The different contexts for work and relational networks of the members of these two main groups are regarded as constitutive of their respective frames of meaning, and provide an explanation for the development of different frames for interpreting and applying scientific results.
Methods and sources of data collection
The primary methods and sources of data collection used for this study were analyses of published articles in medical journals and interviews with key individuals who were involved in the scientific production and subsequent debates. Key journal articles were regarded as providing clear, carefully crafted representations of the perspectives on the scientific issues. These were combined with interview data and observational insights garnered during data collection. Relevant individuals who had been prominent protagonists in the debates were identified through the author’s and funder’s awareness of key participants in the field of intensive care research. The individuals were contacted and invited to participate once university ethics approval had been obtained.
The main period of fieldwork (interviewing) took place when the interviewees were anticipating the results of the second clinical trial. Interviews took place at international conferences and at the workplaces of individual doctors (hospitals and research centres in the USA and Europe). The location of interviews facilitated observations and reflections on the nature of professional duties and obligations in different kinds of workplace setting, enabling the identification of key dimensions of the different frames of meaning. The interview and documentary data presented is selected to illustrate particular aspects of the science pertinent to the schism between the two groups.
Contexts for the two camps: Work and network
I delineate key features of the two groups in Table 1 to provide an overview of the analysis which follows. In this section I describe in more detail the contexts for each group, and in the following two sections, I analyse the perception of the science for each group. I label one group as Pragmatists, and the other as Purists – terms which I use to indicate their general perspective on RCTs, particularly how RCT evidence pertains to clinical practice.
Context (work and networks) for the ‘Pragmatists’
Many of the doctors whom I describe as ‘Pragmatists’ were leading members of national and international intensive care societies and several had been directly involved in the original clinical trial (Bernard et al., 2001). They were nearly all professors of Intensive Care Medicine as well as being practising clinicians, very active in clinical research and generally with strong networks to other clinician-researchers, relationships with industry (device manufacturers and pharmaceutical companies) and disease advocacy and research organisations. One respondent described organising conferences: We organise a [conference]. . . I go to the industry and I try to get support. . . [from] various companies. . . that’s the world we live in right? (Respondent M)
In other words, this respondent saw gaining commercial sponsors for meetings as part and parcel of organising conferences and other networking opportunities. These networks tended to be of like-minded individuals who generally had a shared interest in clinical research programmes rather than laboratory research, and in raising awareness of the problem of sepsis which they encountered in hospital care. The need for commercial involvement is firmly embedded within this respondent’s perception of the ‘world we live in’, that is, the contextual field from his point of view, and the networking operated in the reverse direction too. In marketing terms, the individuals with a pragmatist orientation were ‘Key Opinion Leaders’, that is, individuals feted by the manufacturer of DAA because of their national and international influence.
So, the duties and obligations of networks and clinical trials were interwoven with their routine clinical work, provided the situational context in which they formed their pragmatic conceptions of the need to solve the clinical problem of sepsis, a condition they regularly encountered in intensive care. Furthermore, these individuals often had overlapping additional roles (e.g. in hospital or departmental management) and were required to switch between their various responsibilities at short notice. For example, during interviews there were frequent interruptions and at least one rearrangement at short notice, including splitting an interview in two, or suggesting a telephone interview when in an airport taxi. The general sense of busy-ness, both within their institution and in the extensive networks outside it, contrasted with the ‘Purists’, as I now discuss.
Context (work and networks) for the ‘Purists’
The most prominent individuals displaying a purist orientation were the authors of several notable publications, who felt that some of the scientific evidence may have been misrepresented. The major figures on this side of the debates were senior investigators at the Medical Research Institute (MRI– a pseudonym), a government funded medical research centre.
In contrast to the Pragmatists, the Purists were precluded from involvement with industry by a formal institutional requirement of the MRI, designed to ensure that their scientists remain immune from both inappropriate influence and financial conflicts of interest. This coheres with what we can think of as purist professional values: assiduously maintaining a space for professional autonomy by resisting what they perceived as the inappropriate intrusion of commodification. In this way, specific institutional and professional values and commitments reinforce one another. This is expressed in the Purists’ straightforward scepticism towards the profit motive of business and to the implications of too close a relationship between industry and the medical profession: Pharmaceutical companies are beholden to their shareholders. . .. Industry and doctors have been scrutinised heavily in the last ten years. . . and there is undue influence [e.g. on State Medical Boards]. (Respondent G).
While the Purists were resistant to involvement with industry, they were relationally connected to the regulators, for example serving on advisory panels to the Food and Drug Administration. Thus, for the Purists, there were regular and routinely embedded duties and obligations beyond clinical practice that supported regulation and required a focus on safety.
A fieldwork observation was that an air of quiet autonomy seemed to mark their place of work, in contrast to the expansive busy-ness of the Pragmatists. The MRI itself felt more like a quiet university or college campus than a busy hospital: Before the interview we walked through the [MRI] campus and [Respondent G] pointed out the separate Departments. They each run clinical trials and admit patients for their clinical trials. The critical care service is independent, not tied to any particular Department but in fact serves them. There are no emergency admissions to the [MRI]. (Research diary)
In addition, it is noteworthy that the preferred forms of scientific research for this group was animal research – that is, translating animal models of disease for use in humans.
There are, then, several implications to draw out. First, the experience and intellectual interest of the ‘Purists’ in sepsis was expressed in terms of animal models and experiments rather than clinical experience or research. Second, it seems to be not only the prestige of the MRI that gave weight to their side of the debate, nor simply the connections with the regulators which steered them towards a particular point of view. It is also, in part, that there seemed to be more time and space to think and reflect at some length and in detail, and perhaps in a more considered way, about the meaning and implications of the trial results.
Having presented the contrasting situations (in particular, routine work practices and external relationships and networks), in the next two sections I consider the perception of the RCT of the two groups according to four particular aspects of scientific knowledge: the conduct of the original trial; reproducibility of results; the quality of knowledge of the condition (and the drug’s mechanism of action); and the risk-safety profile of the drug. In doing so I follow the chronology of the debate and start with the viewpoints of the Purists, who presented their objections following the original NEJM publication of the positive trial results. This is followed by an account of the Pragmatists’ views, which was articulated in response to the Purists’ objections.
Purist perceptions of the science
Purist perception of the implications of a change to the trial protocol
About halfway through the trial several changes in the protocol were made, including changes in the exclusion criteria, a new placebo and a new master lot of cells to make DAA. That there was a change to the original trial protocol was portrayed by people associated with this viewpoint as problematic (MacKenzie, 2005; Warren et al., 2002), summarised as follows: The FDA analysis indicated that the efficacy of the drug after the amendment of the protocol differed substantially from its efficacy before the changes were made. Whereas. . . [DAA] did not improve survival in the phase before amendment of the protocol. . .. it was very effective in the phase after the protocol was amended. . . It is not possible to tell from the available data whether the striking improvement in efficacy resulted from the enrolment of a somewhat different population of patients, an undetected change in the drug itself, chance alone, or some combination of all three. (Warren et al., 2002: 1027)
Warren and colleagues are careful to say that it is not that the drug is ineffective – indeed, in the second half of the trial, following the change to the protocol, it seems to be
Purist perception of the reproducibility of results
One doctor from the purist camp expressed his point of view on issues of replication in the following no-nonsense manner: The data looked like the drug was effective. . ..it was potentially a good drug. . . [but] another trial would lay doubts to rest (Respondent G).
He explained his reasoning as follows: [Clinical practice should] not [be based on] a single trial. Reproducibility is the essence of science. . .. The NEJM article [was being] taken as gospel. No-one [was saying] “we need another trial”. (Respondent G).
This concern about reproducibility was dealt with at length in an article in the journal Intensive Care Medicine, where the authors described six different therapies for sepsis over four decades. For each of the therapies, the first trial showed a positive effect that was not reproduced in a subsequent trial.
The history of [sepsis therapies] argues that two beneficial RCTs are necessary with at least one confirmatory trial for the regulatory approval of any new drug. . . [the pattern] of inconsistent findings between trials serves to remind us of the limits of the single RCT. Namely, while the RCT design minimises selection bias within a trial, it is still only a single experiment. (Sweeney et al., 2008: 1956).
The argument here indicates fidelity to the idea that an RCT is a single experiment, so there is a need for a confirmatory trial.
Purist perception of the quality of knowledge regarding the condition itself and the mechanism of the drug
The third area where the Purists took issue with the scientific findings was with respect to the putative mechanism of the drug. The primary article raising these issues was published in the NEJM. Noting how this article begins is analytically instructive: A new hypothesis with implications for the treatment of sepsis has recently been tested – the hypothesis that part of the pathophysiology of sepsis is caused by unrestricted or inappropriate coagulation in the microcirculation. (Warren et al., 2002: 1027)
That is to say, the trial itself and its findings are portrayed as testing a hypothesis about the pathophysiology of sepsis. The subtlety is that this starting point is not a question about the efficacy of the drug – it is a logically prior theoretical question about the nature of the condition. This pushes the debate towards the domain of the ontology of the condition, and towards epistemological models that can try and capture this, rather than being in the first instance a clinical issue about how to treat the condition. For the Purists, then, what should work in clinical practice is dependent on the adequacy of the model with respect to the condition and the suggested mechanism.
Purist perception of the risk-safety profile of the drug
The Purists wanted to explore the potential issue with serious bleeding. In a 2007 editorial in the journal Intensive Care Medicine several authors discussed more trials and clinical use surveys which noted serious bleeding in patients where DAA had been administered. Their overall argument was that the critical care community should reconsider whether the bleeding risks associated with this agent outweigh its purported benefit. (Eichacker and Natanson, 2007: 396).
A related issue, given this potential toxicity, was the possibility of over-use in clinical practice: . . .. During controlled trials that are designed primarily to show the efficacy of an agent, exclusion criteria are frequently applied that minimise the occurrence of adverse effects. During clinical use of an agent, such exclusion criteria may not be applied as rigorously and the incidence of adverse events would be expected to increase. (Eichacker and Natanson, 2007: 398–399).
The doctors with a purist orientation acknowledged ‘it was potentially a good drug’, but: There were clear risks and concerns about clinical use – once approved, the use will be extended. (Respondent G).
All this can be summarised in the notion of ‘primum non nocere’ (‘first, do no harm’). This principle of medical ethics was a common refrain (MacKenzie, 2005; Sweeney et al., 2008).
Pragmatist perceptions of the science
The doctors with what I am calling a pragmatist orientation revealed contrasting viewpoints on each of these aspects of the scientific findings.
Pragmatist perception of the implications of a change to the trial protocol
Several respondents interviewed were generally persuaded that the trial had been well-conducted, despite the change in protocol.
I am someone who thinks the science was more than adequate in that first trial. . . There were a couple of things that were done, the protocol was changed in the middle of the study but that is done in. . . many clinical trials, that is not out of the ordinary; and they switched the batches. . . [but] they had good data which showed that the agent was the same [effect] throughout the trial. . . (Respondent E). Some people say [there’s a problem] because there was an amendment to the protocol during the study but that is something that is quite common in drug development so I don't think there is anything unique there. (Respondent M).
That is, a change to the protocol should not be a cause for concern as it is a ‘common occurrence’ in clinical trials. The pragmatist orientation considers the difference between phases of the trial as more or less inconsequential.
Pragmatist perception of the reproducibility of results
The basic response of the Pragmatists to the purist claim about reproducibility being the essence of science was that the observed size of the drug’s effect was overwhelming. Those of a pragmatist orientation were impressed with 19% relative risk reduction.
When the project ended I was incredibly optimistic. I had been doing these kinds of studies for 30 years and to have a positive trial, mortality difference which was important clinically and highly statistically significant. . . [was] so powerful.. [I] was elated. . . (Respondent D). It felt revolutionary. (Respondent E).
Terms like ‘elated’ and ‘revolutionary’ show the excitement that, from this point of view, there was (at last) a powerfully effective treatment for sepsis.
It was not only the size of the effect that was felt to be persuasive, but also the statistical significance. One respondent argued explicitly that the size of the statistical significance was such that only one trial was needed to provide the necessary evidence. From his point of view, the statistical significance of the DAA trial, at .006, is ‘fairly close to two trials at .05’ (the actual statistical significance of which would be .0025). Thus, this empirical finding alone is said to be close to having had two trials: There is a sort of qualitative sense that a second trial feels somehow different. [But] statistically the level of certainty of one trial with a highly significant finding may not be that different. (Respondent C).
The view of Respondent C is that a second trial, seen as essential by the Purists, might ‘feel different’, but there are reasons to doubt whether it is necessary from a strictly statistical point of view. The difference in the perspectives of the two groups on this issue is striking.
Pragmatist perception of the quality of knowledge regarding the condition itself and the mechanism of the drug
Whereas the Purists were seeking an adequate model for the condition (and by implication, a clear biological mechanism for the drug’s effect), the Pragmatists, at least for the purposes of clinical practice, were happy to remain relatively agnostic about the condition and the precise mechanism. That is, while they were interested in the potential mechanism(s) of the drug from the point of view of scientific inquiry, they placed greater trust in the statistical evidence of the size of the relative risk reduction so that for the purposes of clinical practice, they were happy to remain ignorant about DAA’s precise mechanism. Somewhat tongue-in-cheek, one respondent commented I think if most doctors are honest they realise that most of the drugs they use, we don’t really fully understand the mechanisms [laughs]. (Respondent D).
That said, putting the humour to one side, the desire for an explanation for how a drug works – the mechanism – is a natural part of clinical practice. The original claim for the drug was that it produced an anti-coagulation effect. A number of respondents were unsatisfied with this explanation.
So my recollection is that the trial catches everyone by surprise. No one really knows what the mechanism of action is. No one has been thinking about the coagulation at all, and it’s a little hard to understand why [that would work]. (Respondent C).
It is interesting to note the difference from the Purists here. Whereas the Purists’ formal description of the trial was of testing the hypothesis of ‘inappropriate coagulation’, it is here stated that ‘no one has been thinking about coagulation at all’. But this itself does not render the trial results invalid. On the contrary, given the impressive mortality difference and statistical significance, it seems probable that a number of diverse potential mechanisms could be important.
You know the biology. . . is clear that this molecule is effective, that it works in a variety of important ways. . . (Respondent B). I remember discussing [at a conference] that it just didn’t seem plausible that an anti-coagulant was going to give us this improvement in outcome. . . I thought there was more to it and I also thought it was quite plausible that it did have quite diverse immunological and inflammatory actions which could indeed explain it. (Respondent L).
That is, a number of possible mechanisms were thought to be plausible; indeed, perhaps the drug worked in more than one way. This signals, in part, those elements of the scientific frame of meaning of the Pragmatists that are shared with the Purists: as scientific researchers (and, indeed, as practising doctors) both groups want questions about the mechanisms at work to be answered.
However, despite sharing in principle a desire for a plausible mechanism, the overall frame of meaning, and hence the behaviour, of the Pragmatists differs from that of the Purists in the following key respect. While the Purists focus on the putative mechanism of coagulation as a hypothesis, the Pragmatists are more persuaded by the size of the relative risk reduction, and consequently place their focus on this. According to this logic, the size of the mortality difference obviates the immediate need for a definitive mechanism. They are happy to use the drug for the time being, while in the meantime scientific studies can continue to ‘backfill’ a clearer conception of the mechanism – that is, establish the mechanism in laboratory studies, in a post-approval phase.
Pragmatist perception of the risk-safety profile of the drug
In contrast to the Purist reaction to the risk-safety profile of the drug, the Pragmatists were less concerned: [DAA]. . . has associated with it a number of safety questions. Of course if you have a biologically active compound that is genuinely biologically active, as [DAA] is, then of course it is going to have the negative as well as the positive effects. You know it’s only drugs that don't do anything that have no downside. . . (Respondent B).
The point is, then, that only a biologically inactive substance would have absolutely no negative effect. The argument put forward here is that with suitable care and monitoring, the drug can be used. They also softened the danger of over-use (a problem identified by the Purists), since they felt that those who use the drug in practice will gain experience and they will be using it within its licence.
If you understand how to use and can you clearly say it works, well it is difficult in. . . small numbers but the only thing to be said is that an experienced clinician who has a lot of experience with these patients you do occasionally see dramatic improvement that seems difficult to explain or highly coincidental if it were not this [. . .]. But you certainly don't see anything to suggest that this is a bad thing to be doing, if you use it within it original license and you observe strictly the safety criteria and you understand that. (Respondent B)
Here we can see a pragmatic orientation encompassing both interpretation of scientific studies and clinical practice. Faced with intensive care patients with the condition of sepsis, the drug DAA can be considered – with care – as a tool at an ‘experienced clinician’s’ disposal.
Discussion
In Strong Structuration Theory terminology, the trial evidence from the RCT was conjuncturally-specific information, perceived from within the frame of meaning of the more embedded phenomenologies of habitus. The immediate but enduring contexts and locations of the two groups produced different situationally-specific forms of habitus, which translated into divergent frames of interpretation of the science. The general, shared aspects of the medical frames of meaning of the two groups were challenged by the divergent frames for interpretation of the trial results, mediated by situationally-specific habitus. These fissures became subtly apparent in the situational (re-) framing of professional and ethical values with respect to science and medicine. Differences emerged, for example, in orientations to patient safety and the balance of risk and benefit in conditions with high mortality, the consideration of whether a clinical trial is a single experiment testing a hypothesis, whether there needs to be precise established mechanism for a drug’s effect, and whether it matters that the protocol is changed. These kinds of issues were interwoven in different ways within the frames of each of the two groups.
The delineation of Pragmatist and Purist as I have identified in this case study has resonance with extant literature in studies of clinical research and innovation. For example, Podolsky’s (2014) study of the development of antibiotics noted medical participants’ ‘explicit and self-conscious stances as either “enthusiasts” or “sceptics”’ (p. 14). For the two groups in this case study, the most fundamental difference is in the overall perspective on the RCT as a scientific method and how it is supposed to connect with clinical practice. There was considerable interpretive flexibility on RCT results, the flexibility being expressed in different registers of scientific discourse. The pragmatic attitude extended to being more comfortable with what they saw as a minor change to the protocol ‘mid-experiment’, emphasising the empirical evidence (size of the effect) and potential efficacy of the drug and perceiving a need for flexibility in clinical practice. The purist attitude saw the trial as a single experiment and so were uncomfortable with a change to the protocol; they emphasised the theoretical model and mechanism(s) of the drug’s effect, safety concerns and saw dangers of flexibility in clinical practice. These registers are in turn related to their overall attitude towards involvement with commercial enterprises at this time.
The Pragmatists’ orientation to the RCT results supports a greater readiness to countenance commercial interests and be involved with business, justifying this involvement pragmatically as a means of facilitating technological and pharmaceutical innovation and development. Their hermeneutic frame gives rise to an experiential orientation to the acquisition of knowledge and more weight is given to the practical good that might come from these arrangements. Within their frame this seems to be construed as pragmatic virtue: the importance to their medical practice of actually getting something done, which is perceived to necessitate collaborating with industry and with campaign groups.
Their immediate contextual field has moulded their situationally specific habitus, through the duties and obligations attached to their interconnecting everyday roles in treating patients, organising medical conferences and participating in clinical trials. Presented with the clinical problem of patients in intensive care with sepsis, pharmaceutical companies and other businesses are seen as powerful potential allies in finding a solution. The Pragmatists’ interconnection with this particular pharmaceutical company is an inherent consequence of the nature of their clinical work. Once established, the joint networks of institutionally positioned individuals reinforce the emphasis on shared problems requiring a solution, which in turn precipitates further collaboration.
Having said this, it should be noted that while Pragmatists were prepared to countenance involvement with commercial enterprises, this did not amount to an uncritical acceptance of business practices. For example, Pragmatists were critical in interviews of post-approval marketing activity for the drug, one example of which was a marketing campaign which portrayed the low ‘real-life use’ of DAA (Rowan et al., 2008) as a case of unethical cost-driven health care rationing. However, overall, from within their own, partial, viewpoint, the Pragmatists see themselves as doing all they can to solve the clinical problem of sepsis.
The Pragmatists’ valorisation of certain scientific approaches is associated with this situationally-specific habitus. They prioritise and participate in clinical research, that is, patient based studies, in hospitals and in intensive care units. Given this context for research, they can be said to be concerned with drug effectiveness rather than efficacy (cf. Moreira and Will, 2010), and this connects with an empiricist interpretation of RCT results: an emphasis on the measurable effect of the drug under experimentation rather than the precise explanation, mechanism or model.
On the other hand, the Purists’ orientation to the RCT results exhibits a deep suspicion towards commercial enterprises in general (and the pharmaceutical industry in particular). The scientific priority for these individuals is the safety of the drug and they conceived of the pharmaceutical industry, with its strong profit motive, as having an incentive to not put safety first. In part, the Purists’ circumspection may be aided by their being less often faced with unplanned, emergency admissions. In addition, the autonomous character of the MRI meant that the Purists could realistically aspire to greater, more scrupulous, objectivity and so felt more able to adopt a deontological purism in defending a professional space from what they saw as the encroachment of inappropriate commercial values. A formal institutional requirement against involvement with business can be seen as fostering and supporting their determination to make explicit the risks and safety concerns of the drug, for example. For those with a purist orientation, furthermore, their duties and obligations beyond clinical practice were connected with drug regulation, thus reinforcing a routine focus on safety. This, in turn, informed their focus on the quality of knowledge about the condition itself, in terms of a model and the intimately related issue of mechanisms, as core elements of what is important in terms of scientific integrity. Thus their situationally-specific habitus is moulded and reinforced through the duties and obligations attached to their everyday roles in the clinical and regulatory services they provide.
The Purists’ valorisation of certain scientific approaches is associated with their situationally-specific habitus. They tend to prioritise laboratory-based research, that is, studies which may derive from animal experiments, where the animal models of disease can subsequently be refined in application to human beings. Given this context for research, they can be said to be concerned with drug efficacy and safety, and this connects with their understanding of RCT results, where there is a relative emphasis on the trial as an experiment, and the scientific model and mechanism for the drug’s biological effect, rather than its empirical measurement.
Conclusion
As described in the introduction to this paper, a corpus of social scientific research has sought to scrutinise the claims made on behalf of RCTs. In this article I have added to this corpus by looking in detail at the context of the reception of RCT evidence. I examined a disagreement about RCT evidence in intensive care which took place among a well-defined set of medical experts with largely shared epistemic and professional values. By carefully observing the contexts, networks of collaborators and normal scientific work of two prominent groups, I have proposed that these contexts inform their scientific interpretations via the concept of situationally-specific habitus. That is, certain commitments, norms and values are reinforced according to the routine work practices and cultures, and this leads to differing hermeneutic frames.
I am not suggesting that for either group that their context determines the interpretation of the science: there will always be a role for active agency (Stones, 2005: 100–109). Furthermore, general and professional dispositions (habitus), acquired through professional socialisation, education and training will be an important factor in the perception and interpretation of scientific evidence. So, noting again active agency, individuals may well select places of work according to their general dispositions; the institutional and other contexts can then reinforce certain pre-existing general dispositions and counteract others. In this way different individuals with similar education, training and professional values end up with distinctive hermeneutic frames for the interpretation of biomedical evidence.
It may be that the specificity of this case study precludes straightforward generalisation to other medical specialties. In particular, intensive care is a relatively new specialty, developed in the era of evidence based medicine, and so scientific acumen is part of the specialty’s self-understanding. It is also technologically advanced, highly invasive and with a broad knowledge base (an amalgam of acute forms of general medicine and holistic anaesthesia). The specificity of this case is also highlighted in the condition on which it was based, sepsis, which is a heterogeneous condition with no single causative factor. These features will need to be considered in relation to the potential for generalising these findings to other medical specialities.
Having acknowledged the specificity of the case, I do want to suggest three wider implications. First, there is an implication for the community of practitioners (Lave and Wenger, 1991: 98ff). As Podolsky mentions the perennial character of the tension between enthusiasm and scepticism, it is worth noting that both have something to offer – indeed, the tension can, ultimately, be a productive one. This point can be elaborated in terms of the professional values enacted by the two groups (cf. Weber, 2008[1919]: 198ff). On the one hand there is a pragmatic virtue in taking responsibility to try to solve the problem of sepsis and in identifying collaborators to do so. On the other hand there is a purist virtue in maintaining a conviction about resisting business and commercial encroachment into professional space. It would appear to be incumbent for the community of practitioners (e.g. as represented in this case by intensive care societies and conferences) to provide space for the practical implications of these ethical positions to be debated and worked through. Medical journals and editors, too, have their role to play in facilitating such a space (Will, 2010).
A second implication concerns the relation between clinical practice and biomedicine. At the heart of this analysis is a distinction between science and medicine: clinicians have a professional purpose and practical epistemology which draws on scientific evidence, yet in itself must go beyond it. There has long been recognition of the tensions, difficulties and differences between reasoning within science and reasoning within medicine (e.g. Fleck, 1986[1927]; Gordon, 1988). But what makes medicine interesting and important for science is that its practitioners, even those who do not produce science, are self-consciously scientifically trained and consumers of scientific findings. Ethnographic research has demonstrated the complexities and difficulties in making clinical trials work in research practice (Will and Moreira, 2010). There is a need for further ethnographic studies to examine the routine practices whereby scientific and other knowledge is brought to bear on clinical practice.
This leads, more specifically, to a third and final implication. By developing and using the concept of situationally-specific habitus, this case study has investigated a local debate within one medical specialism and elaborated precise relationships between structural contexts and interpretations of a particular RCT result. There may be potential for generalisation to other specialities where there are distinct influential groups and approaches to the application of RCT results to clinical practice. Social science research on RCTs has examined the local construction of epistemological considerations such as efficacy, plausibility and placebo (Dumit and Sanabira, 2022; Lakoff, 2007; Wahlberg, 2008), and there are doubtless many factors which can contribute to the variations in these constructions. From the contribution of this article, one factor among these which might explain systematic differences in interpretive frames of meaning is situationally-specific habitus, which is associated with the routine work, contexts and collaborative networks of participants.
Footnotes
Acknowledgements
I am grateful for the contribution of several institutions and individuals in the production of this article. While not funded by a specified grant, research expenses (travel, accommodation and interview transcription) were provided by the Intensive Care National Audit & Research Centre. I also thank the University of Essex for a period of research study leave and Western Sydney University for research hospitality. Earlier versions of the article were jointly conceptualised and written with Professor Rob Stones (Western Sydney University), but protracted Long Covid led to his reluctant withdrawal from the project. I would like to thank Rob for his invaluable contributions to earlier versions of the paper and for urging me to continue to develop and rework the project alone. I would also like to thank Professor Kathy Rowan (Intensive Care National Audit & Research Centre) for challenging and productive conversations. Finally, I thank the editor and anonymous reviewers of Health for their constructive critique which helped improve the argument.
Correction (December 2022):
Article updated to add a new reference “Stones R and Turner B (2020) Successful societies: Decision-making and the quality of attentiveness. British Journal of Sociology 71(1): 183–199.” and its citation “Stones and Turner, 2020”.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for data collection and interview transcription provided by the Intensive Care National Audit and Research Centre.
