Abstract
To facilitate multisensory processing, the brain binds multisensory information when presented within a certain maximum time lag (temporal binding window). In addition, and in audiovisual perception specifically, the brain adapts rapidly to asynchronies within a single trial and shifts the point of subjective simultaneity. Both processes, temporal binding and rapid recalibration, have been found to be altered in individuals with an autism spectrum disorder diagnosis. Here, we used a large adult sample (autism spectrum disorder: n = 75, no autism spectrum disorder: n = 85) to replicate these earlier findings. In this study, audiovisual stimuli were presented in a random order across a range of stimulus onset asynchronies, and participants indicated whether they were perceived simultaneously. Based on the synchrony distribution, their individual temporal binding window and point of subjective simultaneity were calculated. Contrary to previous findings, we found that the temporal binding window was not significantly different between both groups. Rapid recalibration was observed for both groups but did not differ significantly between groups. Evidence of an age effect was found which might explain discrepancies to previous studies. In addition, neither temporal binding window nor rapid recalibration was correlated with self-reported autistic symptoms or sensory sensitivity.
Lay abstract
It has been known for a long time that individuals diagnosed with autism spectrum disorder perceive the world differently. In this study, we investigated how people with or without autism perceive visual and auditory information. We know that an auditory and a visual stimulus do not have to be perfectly synchronous for us to perceive them as synchronous: first, when the two are within a certain time window (temporal binding window), the brain will tell us that they are synchronous. Second, the brain can also adapt quickly to audiovisual asynchronies (rapid recalibration). Although previous studies have shown that people with autism spectrum disorder have different temporal binding windows, and less rapid recalibration, we did not find these differences in our study. However, we did find that both processes develop over age, and since previous studies tested only young people (children, adolescents, and young adults), and we tested adults from 18 to 55 years, this might explain the different findings. In the end, there might be quite a complex story, where people with and without autism spectrum disorder perceive the world differently, even dependent on how old they are.
Sensory processing has been proposed to be an underlying mechanism for behavioral characteristics observed in individuals diagnosed with autism spectrum disorder (ASD). More specifically, temporal processing has been hypothesized to be central, leading to differences notably in multisensory processing (Baum et al., 2015; Brock et al., 2002; Stevenson et al., 2014, 2016). To understand how variation in multisensory processing can lead to different (non-) social behavior, it is crucial to investigate how multisensory processing differs between autistic and non-autistic individuals. 1
One of the most important processing abilities of the brain’s sensory system is its ability to integrate information from multiple modalities. This interaction usually occurs when multisensory information is presented in close temporal proximity (see, for example, Alais & Burr, 2004; MacDonald & McGurk, 1978; Vroomen & de Gelder, 2000). For instance, in a noisy environment, we can understand a speaker better when we observe the speaker’s lip-movements (Sumby & Pollack, 1954), and in a densely cluttered environment, we can easily find a visual target when its color change coincides with a spatially uninformative auditory signal (Van der Burg et al., 2008, 2010, 2011). Such multisensory interactions are optimal when the information is presented synchronously (e.g. Van der Burg et al., 2008, 2010, 2015a). However, from the perspective of the observer, perfect synchronicity is unlikely. For instance, the perceived synchrony depends on the distance between the observer and the multisensory object, as auditory signals travel slower through the air than visual signals. Furthermore, auditory information reaches the brain 30–50 ms faster than visual information (Fujisaki et al., 2004). Other factors, such as spatial allocation of attention (i.e. prior-entry; Spence et al., 2001) and stimulus intensity (Los & Van der Burg, 2013), also have an impact on how fast we process information from different sensory modalities and thus affect our percept of multisensory synchrony.
The nervous system has developed several strategies to perceptually accommodate for audiovisual asynchronies, and some of these strategies have been found to differ between individuals with and without ASD. Two of these strategies, the temporal binding window (TBW) and rapid temporal recalibration, are the topic of this study. The first strategy is that although perfect temporal alignment (i.e. synchrony) is optimal for observing audiovisual interactions, perfect temporal alignment is not a prerequisite for observing multisensory interactions. Indeed, events from different sensory modalities may interact with each other when both signals are presented within the so-called TBW (Slutsky & Recanzone, 2001; Van der Burg et al., 2008, 2010, 2014). This window reflects the maximum multisensory asynchrony under which binding of multisensory information occurs, leading to a synchronous perception. The likelihood of perceiving multisensory signals as synchronous decreases with increasing temporal delay between the events.
Brock et al. (2002) suggested that variation in temporal binding might explain some characteristics of autism. Namely, the focus on global features and context information observed in individuals without ASD is a result of an increased temporal binding; more information is combined into a coherent percept (see also Frith, 1989). However, studies investigating temporal binding in individuals with ASD reported inconsistent results: an increased TBW has been found in multiple studies (Feldman et al., 2019; Foss-Feig et al., 2010; Kwakye et al., 2011; Woynaroski et al., 2013), but others reported exactly the opposite: a narrower TBW in individuals with ASD (e.g. Kawakami, Uono, Otsuka, Yoshimura, et al., 2020). Similarly, a correlation of a narrow TBW with higher autistic traits in the general population has been observed (e.g. Kawakami, Uono, Otsuka, Zhao, et al., 2020), while another study found no correlation between TBW and autistic traits at all (Zhou et al., 2019). If variation in the TBW is indeed a central mechanism for observable differences in multisensory perception, different stimuli or experimental setups should not be able to explain these differences. However, the size of the study sample is crucial to be able to determine variations within and between groups (e.g. Ioannidis, 2005).
The TBW is not the only mechanism to compensate for audiovisual asynchronies. A second strategy is the ability to adapt to audiovisual asynchronies over time (Fujisaki et al., 2004; Vroomen et al., 2004), for example, when watching a video where the auditory and visual part is not aligned perfectly. Most people adapt to this mismatch after a short time, in a process called temporal recalibration. Experimentally, it has been shown that this recalibration can occur extremely rapidly, on the basis of a single asynchronous audiovisual event (Van der Burg et al., 2013). In the Van der Burg et al. study, participants perceived an audiovisual pair across a range of stimulus onset asynchronies (SOAs), and participants were asked to judge whether these events were presented simultaneously or not. Typically, such a task results in a synchrony distribution (i.e. a bell-shaped function). The peak of this function corresponds to the point of subjective simultaneity (PSS), that is, the audiovisual asynchrony at which the audiovisual pair is perceived simultaneously. The width of the function corresponds to the TBW. Van der Burg and colleagues (2013) illustrated that the PSS on a given trial t depends on the modality order on the previous trial (t – 1). More specifically, the PSS was shifted toward the leading modality on the previous trial. That is, the whole synchrony distribution shifted toward the leading sense on the previous trial. In other words, when participants were presented with an auditory-leading, visual-lagging stimulus pair at t – 1, they were more likely to judge a stimulus pair of the same order as synchronous at t. Thus, the participants adapted to the asynchrony within seconds and perceived simultaneity in the following trial.
This so-called rapid temporal recalibration has been shown to be robust across a range of different audiovisual stimuli (e.g. Simon et al., 2017; Van der Burg et al., 2013, 2015b; Van der Burg & Goodbourn, 2015). Furthermore, Van der Burg and colleagues (2013) showed that the PSS did not only depend on the modality order on the previous trial (t – 1) but also on the modality order two trials back (t –2; see also Alais et al., 2015; Roseboom, 2019; Van der Burg & Goodbourn, 2015), although the latter effect was significantly reduced compared to the effect on trial t – 1. This shows that participants were able to integrate information to inform their current perception from not only one but even two trials back. Whether this finding holds in autistic populations, however, has to our knowledge not been investigated yet. In addition, it has not been investigated in any population whether the rapid recalibration effect reaches even further than two trials back. It is to be expected that the rapid recalibration effect will decrease further over time. This decrease in itself might provide further information for differences in ASD: if the decrease is similar to the one observed in individuals without ASD, it would indicate that previous experience in general gets weighted less in individuals with ASD. However, if the rate of decrease is increased in individuals with ASD, it would indicate that the recalibration effect might be particularly short lived. This would provide an interesting perspective for theories, such as the E/I imbalance, as for theories proposing a general weaker influence of experiences on perception (e.g. Pellicano & Burr, 2012; Van de Cruys et al., 2014).
Several studies investigated rapid temporal recalibration in individuals with and without ASD. Individuals with ASD showed a reduced (Noel et al., 2017), or even entirely absent rapid temporal recalibration (Turi et al., 2016) compared to individuals without ASD. This finding indicates that individuals with ASD did not (or to a lesser extend) recalibrate, but continued to perceive asynchronies accurately. However, Zhou and colleagues (2019) did not find a correlation between autistic traits and rapid recalibration in the general population. One issue with previous studies is that samples are potentially biased: first, participants with ASD are mostly male (e.g. Noel et al., 2017: 92% male participants with ASD, Turi et al., 2016: 75% male participants with ASD). Second, many autism studies focus on children and adolescents; even studies with adults usually concentrate on young adults (e.g. Noel et al., 2017: age range 7–17 years, Turi et al., 2016: 17–34 years). Finally, samples are often rather small (e.g. Noel et al., 2017: 26 participants with ASD, Turi et al., 2016: 16 participants with ASD). Taken together, the rather young, male, and small samples represent a selection of the autistic community. This study therefore aimed at including a larger and more diverse sample in terms of gender and age. Both were additionally explored whether they are significant factors for multisensory processing. Past studies have shown evidence that multisensory processing changes over age (e.g. Noel et al., 2016 for rapid recalibration specifically).
In this study, the goal is to investigate the TBW and rapid temporal recalibration to audiovisual asynchronies and its relation to an ASD diagnosis, autistic symptoms, gender, and age. In the experiment, participants were presented an audiovisual pair across a range of stimulus onset asynchronies (SOAs) and were asked to judge whether these events were synchronous or not (see also Van der Burg et al., 2013). An example of the paradigm is illustrated in Figure 1.
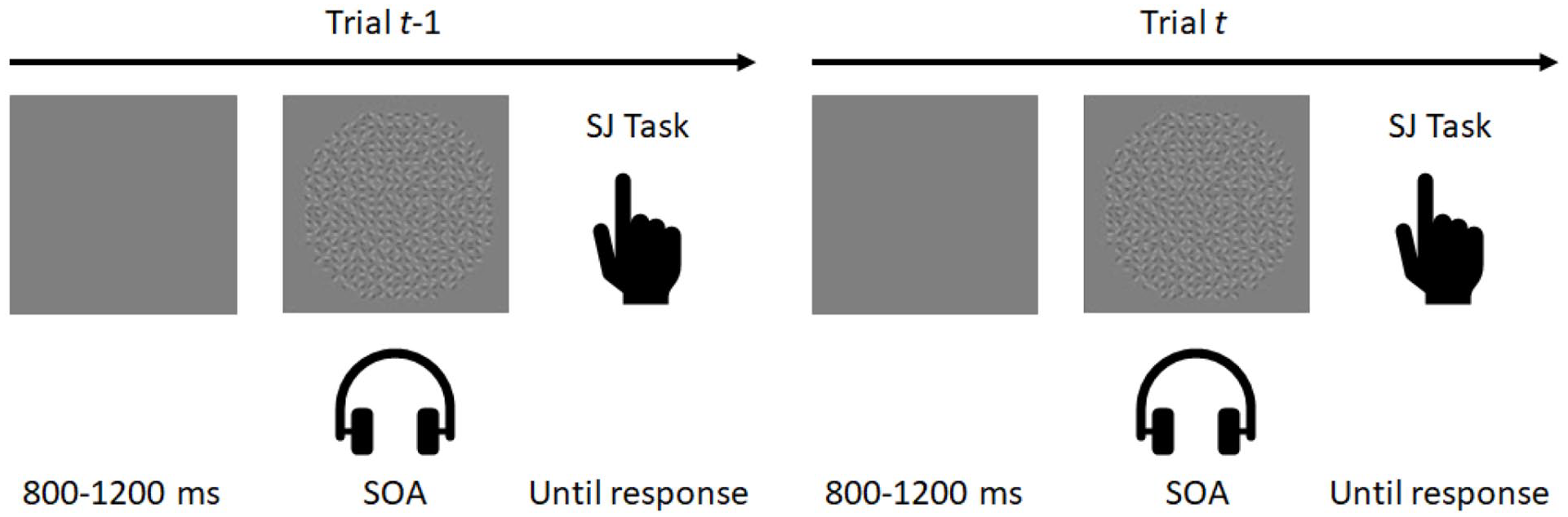
Paradigm used in this study. Participants perceived an audiovisual pair across a range of stimulus onset asynchronies (SOAs) and were instructed to judge whether these events were present in synchrony or not (i.e. a synchrony judgment (SJ) task).
Contrary to other studies, we use a large sample of adults with and without an ASD diagnosis to increase statistical power and have a more representative sample. The large sample allowed us to test for age and gender effects on rapid temporal recalibration. In addition, we explore correlations with autism symptoms and sensory perception. Furthermore, we extend previous analyses by investigating the rapid recalibration not only to the previous trial (t – 1) but also to two and three preceding trials (t – 2 and t – 3).
Methods
This study has been preregistered on the Open Science Framework (https://osf.io/2mvsx/?view_only=b5cecb3747534fdd9bded65cbfacc975). We deviate from our proposed analyses when investigating Group, Gender, and Trial History effects for the rapid recalibration effect (see below). Instead of conducting one analysis of variance (ANOVA) per Trial History condition, we added it as a main factor and performed one ANOVA instead.
Participants
One hundred participants with ASD (47 male, 53 female, mean age: 43.6 years, ranging from 19 to 55 years) and 92 participants without ASD (40 male, 52 female, mean age: 32.5 years, ranging from 18 to 56 years) were recruited from the Netherlands Autism Register (NAR, https://www.nederlandsautismeregister.nl/english/) and from psychology students of the Vrije Universiteit Amsterdam. ASD was diagnosed prior to the recruitment according to Diagnostic and Statistical Manual of Mental Disorders (4th ed., text rev.; DSM-IV-TR or 5th ed.; DSM-5) criteria by psychiatrists/psychologists who worked independently from the authors, and who were unaware of the goals and outcomes of this study. The diagnostic process included anamneses, proxy reports, and psychiatric and neuropsychological examinations. None of the adults in the no-ASD group reported an ASD diagnosis and none of the participants was a firsthand relative of someone with an ASD diagnosis. Inclusion criterion for both groups was age ranging between 18 and 56 years. Participants were screened for hearing and vision problems. All participants were naive to the purpose of the experiment and received course credits or 25 euro for their participation. Informed consent was obtained prior to the experiment. This research was approved by the medical ethical committee of the Vrije Universiteit Amsterdam.
Questionnaire measures
The Autism-Spectrum Quotient (AQ)-Short measures autistic symptoms on the two higher-order factors—Social Behavior, and Numbers and Patterns—while the former factor consists of the lower-order factors Social Skills, Routine, Attention switching, and Imagination (Hoekstra et al., 2011). The Sensory Perception Quotient (SPQ)-Short measures sensory sensitivity on the factors vision, hearing, touch, taste, and smell (Tavassoli et al., 2014; Weiland et al., 2020).
Behavioral experiment
Procedure
Participants filled in online questionnaires (AQ-Short and SPQ-Short) and demographical data as part of their membership of the NAR. The experiment was part of a large project and took place at the Vrije Universiteit Amsterdam. Participants received information about the experiment prior by email.
Apparatus and stimuli
The experiment was programmed and presented using OpenSesame (Mathôt et al., 2012). Participants performed the task in a soundproof booth. The stimuli were shown on a 22-inch Samsung monitor (refresh rate 120 Hz, 1680 by 1050 pixels resolution) and participants sat at a distance of ~80 cm from the monitor. Auditory stimuli were presented using Harman Kardon HK195 speakers located to either side of the monitor, and responses were made using a standard QWERTY keyboard. The visual stimulus was a combination of multiple Gabor patches arranged into a circle (see Figure 1, stimuli adapted from Snijders et al., 2013) presented on gray background (luminance 300 cd/m²). It was presented on a Samsung monitor, model CM22WS, with a refresh rate of 120 Hz. The monitor was alternating between the stimulus screen and a black screen in order to induce electroencephalogram (EEG)-related measures that are independent from the behavioral task. The auditory stimulus was a 40-Hz amplitude modulated white noise burst (65 dB), similar to the one used in Wilson et al. (2007). The auditory and visual stimuli were presented for 500 ms. The experiment was part of a larger project and electroencephalography (EEG) was recorded while participants performed the task. The other tasks and EEG will be reported elsewhere.
Design and procedure
A trial started with the presentation of a gray screen for a random interval between 800 and 1200 ms. Subsequently, the audiovisual pair was presented across a range of stimulus onset asynchronies (SOAs; −400, −200, −50, 50, 200, or 400 ms). Here, negative values indicate that audition leads vision, and positive values indicate that vision leads. Participants were asked to make an unspeeded response by either pressing the left, or right control key if they perceived the audiovisual pair asynchronous or synchronous, respectively. The next trial was initiated after participants made a response.
Prior to the experiment, participants practiced a couple of trials to get familiar with the task. Subsequently, participants performed 384 experimental trials in four blocks. Participants were allowed to take breaks between blocks.
Analyses
Figure 2 illustrates the calculation of the two main measures: TBW and ΔPSS. Both are derived from Gaussian functions fitted to individual participants’ synchrony distributions with the parameters PSS (i.e. mean; initial value 0 ms, constrained between −401 and 401 ms), TBW (i.e. the standard deviation of the distribution; initial value 250 ms, constrained between 50 and 400 ms), and amplitude (initial value 0.9, constrained between 0 and 1). The shift in PSS (ΔPSS) depending on the modality order in the previous trial(s), two Gaussians were fitted per participants, depending on the modality order in previous trial(s) (see also Van der Burg et al., 2013).
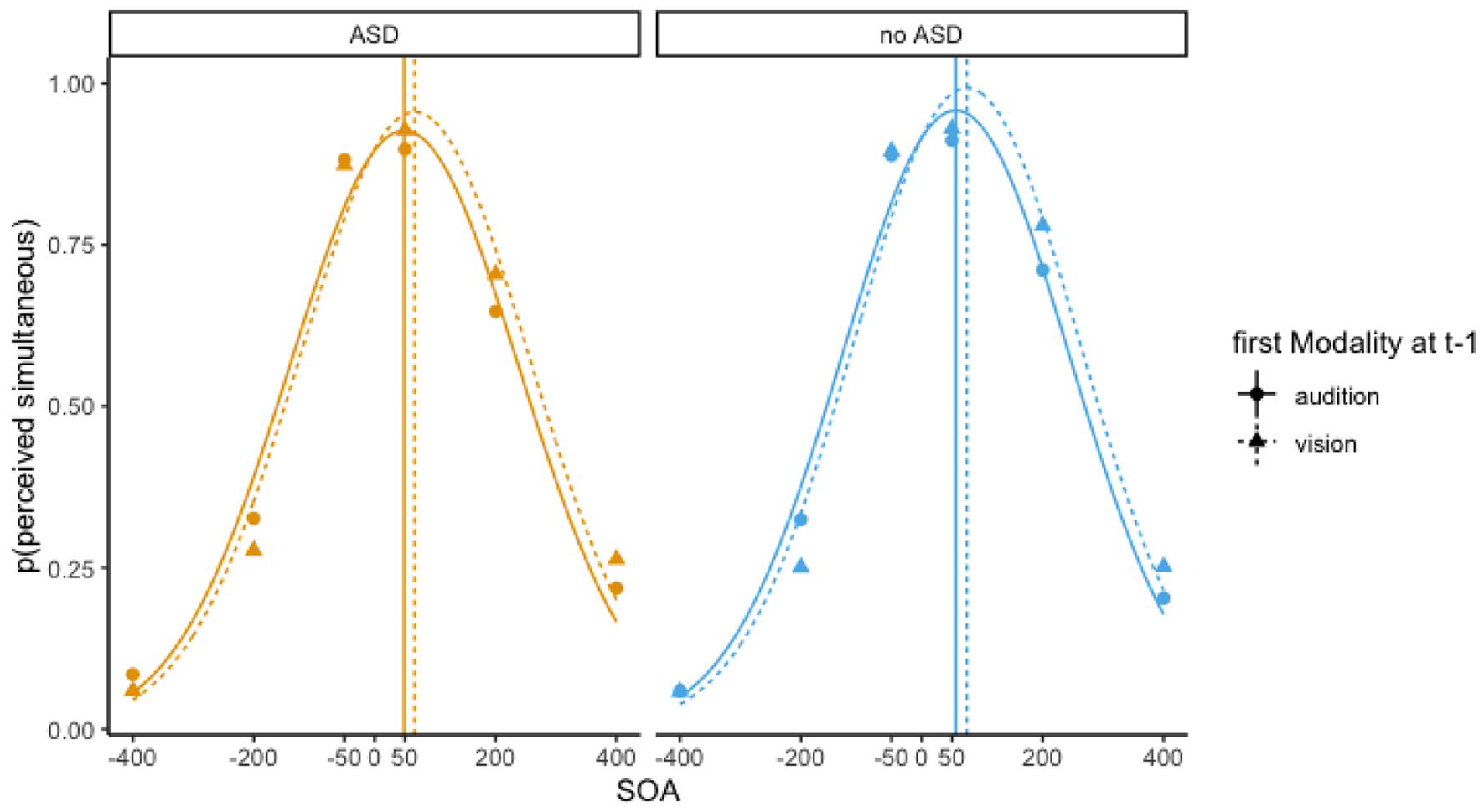
Average performances per group. Pictured are probabilities of a simultaneous response as a function of SOA. Negative SOA values indicate that auditory stimulus was leading; positive SOA values indicate that visual stimulus was leading. Data are split based on the modality order in the previous trial t – 1: circles indicate that in the previous trial audition was leading; triangles indicate that in the previous trial vision was leading. Through those datapoints, two Gaussian functions are drawn. Vertical lines indicate the mean, or the point of subjective simultaneity (PSS). The PSS shifts based on the previous modality order; this reflects the rapid recalibration effect (ΔPSS).
Community involvement
Individuals with ASD were not directly involved in the design of the study but gave feedback. The NAR survey is inspired by needs and interests expressed by stakeholders from the autism community, which included the focus on sensory sensitivity. Each year, the NAR invites stakeholders (autistic adults and parents of children with varying abilities) to exchange ideas on relevant research topics and dissemination of findings.
Results
The first three trials from each block were excluded from further analyses since we were interested in history effects up to three trials back. Trials in which participants responded slower than 3000 ms (measured from the offset of the stimulus pair) were excluded from further analyses, which resulted in the exclusion of 2.8% of trials in the ASD group (mean 10.6 trials, SD = 13.1) and 1.2% of trials in non-ASD group (mean 4.77 trials, SD = 5.55). An unpaired Welch’s t-test showed that significantly more trials were excluded from the ASD group, t(105) = 3.73, p < 0.001.
Furthermore, data from four participants (all with ASD) were excluded as the estimated overall TBW was larger than 400 ms (i.e. the maximum SOA), and nine participants (two with ASD) were excluded since they reported synchrony in 75% of the trials where the SOA was 400 ms. This leaves 75 participants with and 85 without ASD. For gender-related analyses, two more participants were excluded (one with ASD) for statistical reasons since they indicated their gender as neither male nor female (i.e. gender was “other”).
The goodness of fit of the fitted Gaussian functions was measured using residual sum of squares (RSS), for the t – 1 condition and averaged over the visual first and auditory first condition. A Welch two-sample t-test revealed that functions fitted significantly better in the ASD group (M = 0.03, SD = 0.02) compared to the non-ASD group, M = 0.04, SD = 0.03; t(157.84) = −2.99, p = 0.003. RSS values ranged from 0.002 to 0.122 in the ASD group, and from 0.003 to 0.109 in the non-ASD group.
TBW
We conducted an ANOVA on the TBW with Group (ASD vs non-ASD) and Gender (male vs female) as between subject variables (see Figure 3), and Age as a covariate. The ANOVA yielded no significant main effects of either Group or Gender, F(1, 154) = 0.001, p = 0.976, and F(1, 154) = 0.195, p = 0.659, respectively. The two-way interactions also failed to reach significance, F < 1, indicating the TBW was not affected by Group or Gender. Also, Age was not significant, F(1, 154) = 1.124, p = 0.291. A Bayesian ANOVA to investigate whether evidence for an absence of Group and Gender effects confirmed that the null model was the most likely one. The full model (with factors Group and Gender, and their interaction) merely yielded a Bayes factor of 0.010 which is strong evidence for the null hypothesis (see Lee & Wagenmakers, 2014; van Doorn et al., 2021).
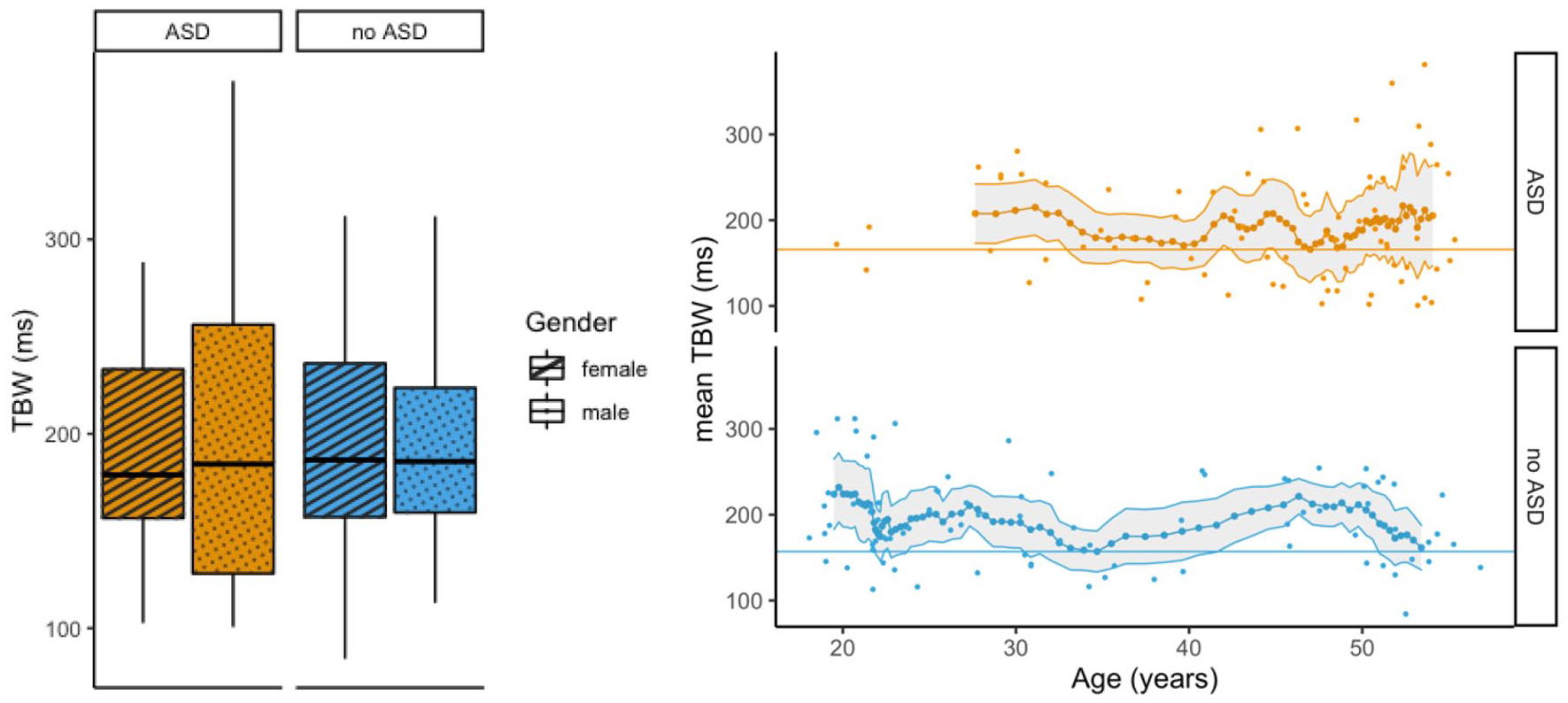
TBW results. The left figure shows the TBW for both Group (ASD and no ASD) and Gender (female and male). Boxplots represent five summary statistics: the median, 25th and 75th percentiles (lower and upper hinges), and minimum and maximum values (lower and upper whisker). The right panel indicates the TBW as a function of age. Small dots indicate individual participants’ datapoints. Larger dots connected by a line indicate group averages per binned data set. The shaded area around the line represents a 95% confidence interval. The horizontal lines signify the baselines (i.e. the minimum TBW magnitude) per group.
Age effects
Figure 3 illustrates age effects for the TBW in both groups. Since groups differed significantly in terms of age (see Table 1), both groups were investigated separately, using a sliding window approach (see Noel et al., 2016): data were sorted by group from youngest to oldest participant. Then, a sliding window of 11 participants was moved one participant at a time and determined binned data sets. There were 64 binned data sets in the ASD group, and 74 binned data sets in the non-ASD group. As a baseline for further testing, the binned data set with the minimum average TBW was chosen, respectively, for the ASD and non-ASD group (ASD group: minimum TBW = 165.9 ms, at 47.0 years; non-ASD group: minimum TBW = 157.1 ms at 34.7 years). Then, each binned data set was tested whether the TBW was significantly larger than the baseline, using one-sided t-tests. Correction for multiple comparisons was done using the false discovery rate (FDR) method.
Demographic information about participants.
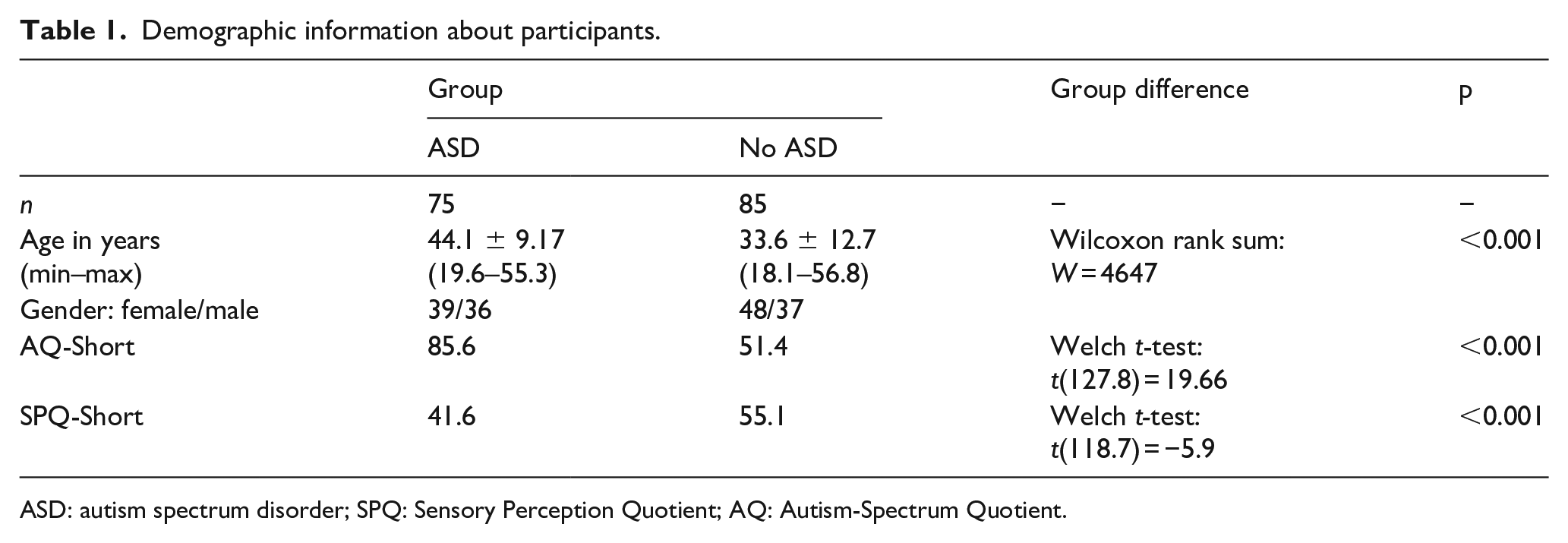
ASD: autism spectrum disorder; SPQ: Sensory Perception Quotient; AQ: Autism-Spectrum Quotient.
In the ASD group, none of the bins were significantly larger than the baseline, indicating that the TBW did not vary with age (all FDR-corrected ps > 0.242). In the non-ASD group, 34.7% of binned data sets yielded a significant result after false discovery rate (FDR) correction. Namely, the youngest age groups, from ca. 19 to 21 years and 24 to 30 years, and older age groups between ca. 42 to 50 years had larger TBWs compared to the baseline (see Figure 3).
Rapid temporal recalibration (ΔPSS)
Figure 4 illustrates the rapid recalibration effect (difference of points of subjective simultaneity, ΔPSS = PSS vision leads on the previous trial − PSS audition leads on the previous trial) as a function of Trial History (t – 1, t – 2, and t – 3). In this study, a positive ΔPSS reflects a rapid temporal recalibration effect. Based on a mixed two-way ANOVA on the mean ΔPSS with Trial History (t – 1, t – 2, and t – 3) as within subject variable, Group and Gender as between-subject variables, and Age as a covariate. The ANOVA yielded no main effects of Group, F(1, 155) = 0.69, p = 0.408, and Gender, F(1, 155) = 1.00, p = 0.318, and also the covariate Age did not reach significance, F(1, 155) = 0.53, p = 0.467. The Trial History main effect was significant, F(2, 312) = 10.87, p < 0.001. We conducted three separate one-tailed t-tests to test whether the ΔPSS (i.e. rapid recalibration) was significantly larger than zero for each trial history condition. All t-tests were significant—t – 1 rapid recalibration effect was 18.1 ms: t(159) = 8.33, p < 0.001; t – 2 rapid recalibration effect was 7.4 ms: t(159) = 4.15, p < 0.001; and t – 3 rapid recalibration effect was 6.5 ms: t(159) = 3.60, p < 0.001. Finally, we investigated whether ΔPSS differed between Trial History conditions, using paired, one-sided t-tests. Results showed that while ΔPSS was greater in the t – 1 compared to t – 2 condition, t(159) = 3.97, p < 0.001, there was no significant difference between the t – 2 and t – 3 condition, t(159) = 0.34, p = 0.366. Finally, the two-way interaction between Group and Trial History did not reach significance, F(2, 312) = 0.54, p = 0.586, suggesting that the rapid recalibration effect over several trials back was not depending on the group.
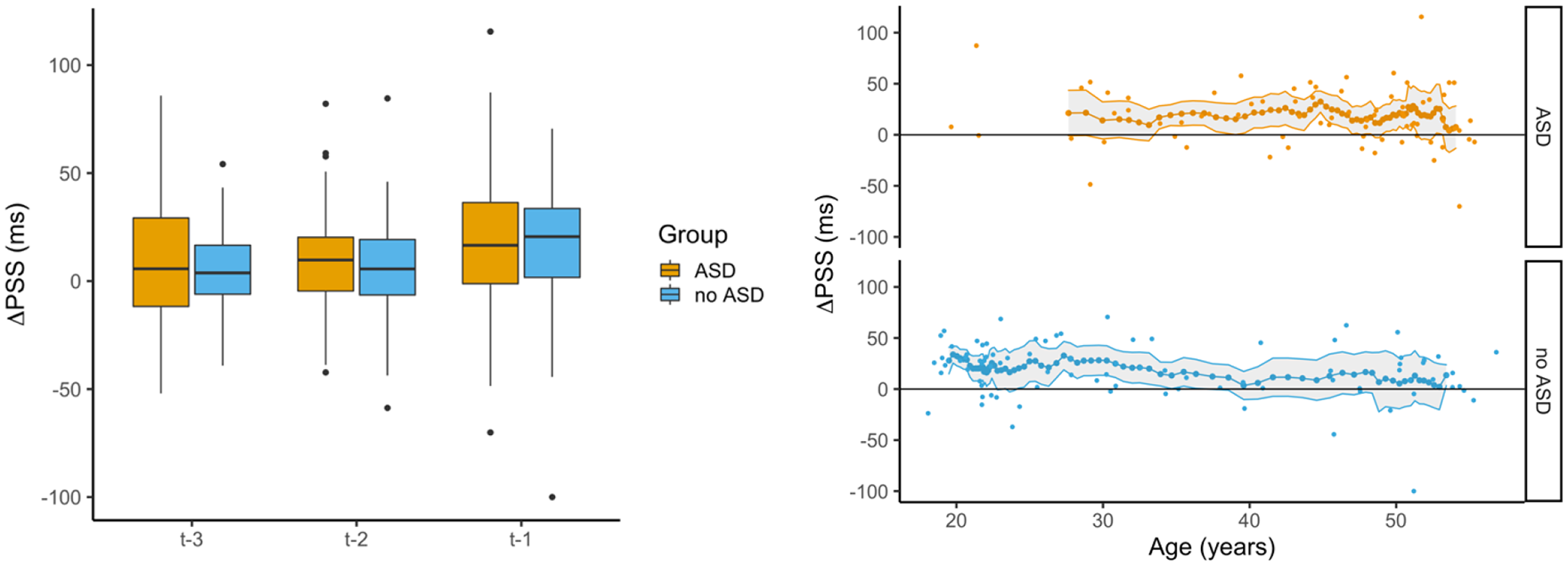
Rapid recalibration (ΔPSS) results. The left figure shows rapid temporal recalibration (ΔPSS) as a function of Trial History (t – 3, t – 2, and t – 1) for each Group (ASD and no ASD). Boxplots represent five summary statistics: the median, 25th and 75th percentiles (lower and upper hinges), and minimum and maximum values within 1.5 times the interquartile range between 25th and 75th percentile (lower and upper whisker). Values beyond that range are represented as black dots. The right figure shows rapid temporal recalibration as a function of age for each Group (ASD and no ASD). Small dots represent individual participants’ datapoints, the larger dotes connected by a line represent averages per bin. The shaded area around the line represents 95% confidence interval.
A Bayesian analysis was added to confirm the absence of Group and Gender effects. Indeed, an ANOVA with Group, Gender, and their interaction yielded a Bayes factor of 0.012.
In addition, we investigated the correlation between ΔPSS at t – 1 and the TBW in both groups (see also Van der Burg et al., 2013). In the ASD group, the two measures seemed to be uncorrelated (Pearson’s r = 0.17, p = 0.144). However, in the non-ASD group, the correlation was significant (r = 0.246, p = 0.023) albeit it considerably smaller than previously reported (r = 0.69 in Van der Burg et al., 2013; r = 0.59 in Harvey et al., 2014; r = 0.40 in Simon et al., 2017).
Age effects
Age effects in ΔPSS were investigated for t – 1, similarly but not identically to age effects in TBW: the same sliding window approach was used; however, with binned data sets, it was tested whether ΔPSS was significantly greater than zero, using one-tailed t-tests (n = 64 bins for ASD group, n = 74 bins for non-ASD group). After FDR correction for multiple comparisons, in the ASD group, 76.6% of the binned data sets had a ΔPSS significantly larger than zero, whereas in the non-ASD group, 64.9% of tests remained significant (see Figure 4 for development over age). In the non-ASD group, only young age groups (until ca. 38 years) showed a ΔPSS significantly larger than zero. In the ASD group, age groups in the mid-range (from ca. 35 to 50 years) showed consistently significant ΔPSS.
Questionnaire correlations
Relationships between both TBW and ΔPSS on trial t-1 with the SPQ-Short (total, vision, and audition scores) and the AQ-Short total and subscale scores were investigated (see Table 2). Data were missing for some participants.
Correlations of the behavioral variables TBW and ΔPSS with the questionnaire subscales of the SPQ-Short and AQ-Short.
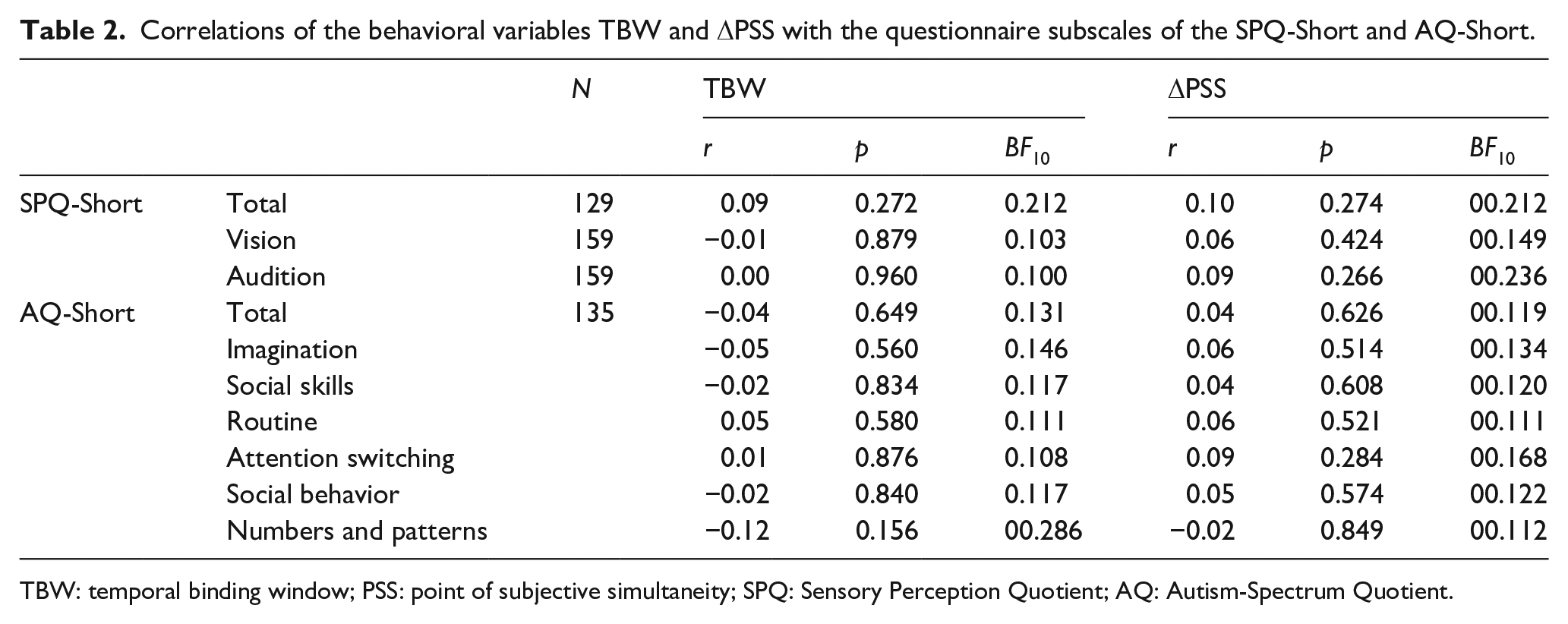
TBW: temporal binding window; PSS: point of subjective simultaneity; SPQ: Sensory Perception Quotient; AQ: Autism-Spectrum Quotient.
None of the correlations were significant even without correction for multiple testing (all ps ⩾ 0.156, and all rs ⩽ 0.12), and Bayes factors were consistently low (all BF10 < 0.29, see Table 2).
Discussion
This study investigated the TBW and ΔPSS in individuals with and without an ASD diagnosis. Regarding the TBW, no group or gender differences were observed. In fact, Bayesian analysis confirmed the notion that the TBW was not significantly different between the ASD and non-ASD group. However, the TBW did vary as a function of age, but only for those participants without an ASD diagnosis. The PSS on a given trial was depending on the modality order on the previous trial (i.e. rapid temporal recalibration; see also Van der Burg et al., 2013) but also depending on the modality order two and even three trials back. Importantly, rapid recalibration was observed for individuals with and without an autism diagnosis. In contrast to previous studies (Noel et al., 2017; Turi et al., 2016), we could not find any evidence for group differences. However, group differences might have been masked by age effects: in the non-ASD group, a significant rapid recalibration was only observed in the younger age groups (up to 38 years), whereas in the ASD group only the middle age groups (ca. 35–50 years) showed a significant rapid recalibration effect. Finally, neither autistic symptoms (AQ-Short) or sensory processing (SPQ-Short) were correlated with either TBW or the rapid recalibration effect.
The absence of group differences in the TBW is uncommon; however, we focused on adults whereas most studies reporting differences examined behavior in children (e.g. Foss-Feig et al., 2010; Kwakye et al., 2011). In line with our findings, another study also reported the absence of a significant group difference when investigating the TBW in adults (Kawakami, Uono, Otsuka, Yoshimura, et al., 2020). A recent meta-analysis reported that audiovisual processing differences between groups with and without ASD reduce with age (Feldman et al., 2018). In this study, we did not compare groups in the development over age but investigated groups separately, since the two groups were not matched for age (see Table 1). However, as we did not find changes over age in the ASD group, this might be a counter argument for a delayed development of multisensory processing in ASD (e.g. Feldman et al., 2018). Instead, we found changes over time in the non-ASD group. These changes seemed to follow a U-curve, where the youngest and the oldest participants showed a larger TBW. Similarly, Noel and colleagues (2016) reported that younger participants (non-ASD) showed a larger TBW, although they did not find an increase in older participants. However, Zhou et al. (2019) did not find differences in TBW between their adolescent compared with their young adult sample. Given the inconsistencies and the cross-sectional nature of these results, it is difficult to speculate about underlying mechanisms responsible for age-related changes. However, it highlights how some findings seem to be difficult to generalize, even over simple demographic characteristics as age.
The absence of group differences in rapid temporal recalibration effects was surprising, as other studies reported significant group differences (Noel et al., 2017; Turi et al., 2016). Still, we found evidence for a significant rapid recalibration effect for both groups tested. The first study reporting group differences observed a rapid recalibration effect for the non-ASD group and no effect whatsoever for the ASD group (Turi et al., 2016). Noel et al. (2017) did find evidence for rapid recalibration in their participants with and without ASD, but the recalibration effect was largely reduced in the first group compared with the last group. Since the sample size in this study was comparatively large (ca. three times more participants with ASD than Noel et al., 2017, and ca. 4.5 times more than Turi et al., 2016), it substantially reduced the likelihood that our results are due to a lack of statistical power. Moreover, the Bayesian analysis supported the null finding. One possible explanation for these contrary findings is that these studies investigated rapid recalibration with younger individuals (Turi et al., 2016: 17-34 years, Noel et al., 2017: 7-17 years). These studies might have missed the effect that older participants without ASD do not seem to have a (strong) rapid recalibration effect (see also Noel et al., 2016), and that older participants with ASD do seem to have a rapid recalibration effect. Past studies mostly investigated young adults (without ASD) for whom rapid recalibration effects might be particularly large (e.g. Noel et al., 2016, 2017; Turi et al., 2016; Zhou et al., 2019). Another possible explanation lies in the ASD group: when investigating age effects, we found that young individuals with ASD did not show a significant rapid recalibration effect, however, individuals from ca. 35 to 50 years did. Previous studies have tested populations younger than this age range, so it is possible that rapid recalibration develops later in the life of diagnosed individuals (Feldman et al., 2018). The present ASD group also has a larger percentage of women. This gender distribution, however, is unlikely to explain the different outcomes of our study, since the rapid recalibration effect was not depending on the participants’ gender.
In addition, autistic symptoms and sensory processing were not significantly correlated with either measure of audiovisual processing in this study while the questionnaires used distinguish well between individuals with and without ASD (e.g. Hoekstra et al., 2011; Weiland et al., 2020). This absence of correlation is in line with some previous research: Zhou and colleagues (2019) did not find significant correlations between AQ scores and either rapid recalibration or TBW. Similarly, another study reported no significant correlation between AQ scores and TBW (Kawakami, Uono, Otsuka, Zhao, et al., 2020). Other previous research, however, did find correlations with questionnaires: Turi and colleagues (2016) report medium correlations in both their ASD and non-ASD group, between an autism questionnaire (SRS-2) and rapid recalibration. It is not clear why they did find a correlation where others did not, but it might be plausible that this is due to their small sample size.
Finally, it is possible that the lack of correlations and group differences in this study is part of another phenomenon that has been observed: effect sizes in autism research have been decreasing over the past decades (Rødgaard et al., 2019). This trend seems to be unique to autism research; is not related to increasing sample sizes; and has been found in social, cognitive, and neural measures. It has been suggested that this decrease is the result of the change in definition and criteria for ASD which has led to an increased heterogeneity, and therefore noise in samples (Mottron & Bzdok, 2020). While this certainly raises important questions for the way ASD is defined and diagnosed, we feel it remains necessary to report findings on individuals that are currently diagnosed with ASD.
This study casts doubt on previous finding of group differences in either TBW or rapid recalibration. It showed that gender is unlikely to play a role; however, most measures did change with age (except TBW in the ASD group). This opens new research questions, for example, to properly map out development over age with extended samples and to investigate whether these developments have implications for everyday life. In addition, it questions the validity of psychophysical measurements as indicators for subjectively experienced stimulus intensity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NWO ZonMW Top grant (No. 2017/02015/ZONMW).
