Abstract
The oxytocin system may be different in autistic people, which could explain some of the deficits in social behavior and cognition associated with autism spectrum disorder. However, studies comparing oxytocin levels in autistic and neurotypical individuals have shown conflicting results and a 2016 meta-analysis on seven studies concluded that there was no significant difference. Here, we greatly expanded the sample of studies to 31, warranting a reassessment of this finding. We searched Web of Science with MEDLINE®, SciELO Citation Index, and BIOSIS Citation Index for articles that measured oxytocin in plasma/serum (k = 26 studies), saliva (4), or cerebrospinal fluid (1) in autistic individuals (total n = 1233 participants) compared to neurotypical individuals (n = 1304). We found that oxytocin levels were significantly lower in autistic people (Cohen’s d = −0.36, 95% confidence interval = [−0.61, −0.10], p = 0.007), with no evidence for publication bias. This overall effect was driven entirely by differences among children (k = 25, d = −0.44, 95% confidence interval = [−0.72, −0.16], p = 0.002) but not adults (k = 6, d = 0.03, 95% confidence interval = [−0.55, 0.61], p = 0.92). These results support further research into the use of oxytocin to treat social deficits in children.
Lay abstract
Oxytocin is a hormone that mediates interpersonal relationships through enhancing social recognition, social memory, and reducing stress. It is released centrally into the cerebrospinal fluid, as well as peripherally into the blood, where it can easily be measured. Some studies indicate that the oxytocin system with its social implications might be different in people with autism spectrum disorder. With summarizing evidence of 31 studies, this meta-analysis suggests that children with autism spectrum disorder have lower blood oxytocin levels compared to neurotypical individuals. This might not be the case for adults with autism spectrum disorder, where we could not find a difference. Our findings motivate further exploration of the oxytocin system in children with autism spectrum disorder. This could lead to therapeutic options in treating autism spectrum disorder in childhood.
Introduction
Autism spectrum disorder (henceforth ASD) is a neuro-developmental condition characterized by two broad symptom clusters, namely, (1) deficits in social interaction and social communication as well as (2) restricted or narrow interests and repetitive behaviors (American Psychiatric Association, 2013). The social deficits specifically include difficulties in social cognition, emotion-recognition and mentalizing, maintaining eye contact and processing gaze information as well as initiating and fostering social affiliations. These traits are all thought to be mediated by oxytocin (OT).
OT and ASD
OT is a neuropeptide produced in the hypothalamus and released from axonal projections to other parts of the central nervous system and from the posterior pituitary into peripheral circulation (Meyer-Lindenberg et al., 2011). OT is well-known for its role in parturition, breastfeeding, and parent-infant bonding, but has also been shown to mediate broader social relationships (Donaldson & Young, 2008). Fundamentally, OT enhances social recognition, social memory, and reward through modulation of dopaminergic pathways, as well as by reducing anxiety and stress by dampening amygdala activity and the hypothalamic–pituitary–adrenal axis (Bethlehem et al., 2014; Hurlemann & Grinevich, 2018; Piva & Chang, 2018; Uvnäs-Moberg, 1998).
Based on these reported social effects of OT, it is thought to be implicated in the social deficits of ASD. Indeed, several components of the OT system—such as the genes for OT itself, for its receptor, and for cluster of differentiation 38 (a transmembrane protein that regulates OT release)—have been associated with ASD (Feldman et al., 2016). Variation in these genes could result in differences in the binding affinity between OT and its receptor, the distribution of OT receptors, as well as circulating OT levels. While examining receptor distributions and binding affinity is not typically possible in humans (but see Freeman et al., 2018), several reviews have argued that autistic people have lower levels of circulating OT (Cochran et al., 2013; Crespi, 2016). Such a difference in OT levels would bolster efforts to use OT administration for alleviating social deficits in ASD patients (Peled-Avron et al., 2020; Phaik Ooi et al., 2017; Wang et al., 2019). Thus, establishing whether OT levels differ in ASD is an important step toward developing therapeutic uses. In the following, we briefly review previous studies comparing OT levels between autistic and neurotypical individuals.
Previous studies comparing OT levels in autistic versus neurotypical people
Modahl et al. (1998) was the first study to find lower OT plasma levels in autistic boys. The same researchers also found differences in the actual OT peptide forms, suggesting broader differences in OT metabolism (Green et al., 2001). Al-Ayadhi (2005) and El-masry et al. (2010) confirmed lower OT plasma levels in autistic children. Later, the hypothesis of lower OT levels in autistic children was challenged. Miller et al. (2013) did not find lower OT plasma levels, nor did (Jacobson et al., 2014; Parker et al., 2014; Taurines et al., 2014). The latter study did, however, show a correlation between OT plasma levels as well as polymorphisms in the OT receptor gene with theory of mind and social communication performance not only in autistic children but also in their siblings and unrelated neurotypical children, indicating differences in the OT system not to be uniquely associated with ASD. Hence, OT may be associated with socio-cognitive function in a continuous fashion that is not always captured by a categorical comparison of children diagnosed with ASD to neurotypical ones.
Much fewer studies were conducted in autistic adults. Both Althaus et al. (2016) and Jansen et al. (2006) found higher plasma OT levels in autistic adults compared to neurotypical ones, the former including only males. Andari et al. (2010) were the only authors to find lower OT levels in autistic adults, including mostly males with Asperger’s syndrome and high-functioning autism. Munesue et al. (2010) and Procyshyn et al. (2020) found no difference in plasma and saliva OT levels in autistic adults, with the latter measuring women only. The same was true for Aita et al. (2019) regarding plasma OT levels in a sample of hospitalized adults with severe intellectual disabilities.
In an attempt to capture some of the continuous variation in socio-cognitive function, 19 of the papers reviewed in the present meta-analysis correlated OT levels with ASD symptoms, quantified by at least 15 different scores or diagnostic instruments. Some studies did find OT levels to be inversely correlated with ASD symptom scores (Alabdali et al., 2014; Taurines et al., 2014), ASD severity (Kobylinska et al., 2019), repetitive behavior (Aita et al., 2019; Miller et al., 2013; Yang et al., 2015), impairment in verbal communication (Zhang et al., 2016), or attention to detail (Husarova et al., 2013). Counterintuitively, other studies found positive correlations of OT levels and ASD severity or symptoms (Husarova et al., 2016; Jacobson et al., 2014; Tomova et al., 2015). Autistic individuals with higher OT levels were found to be more socially and developmentally impaired (Modahl et al., 1998) or to have difficulties in “Adaptation to change” (Yang et al., 2017). Meanwhile, El-masry et al. (2010) could not find a correlation between OT levels and ASD symptom severity. The same was true for Oztan, Jackson, et al. (2018), but they found a lower expression of the receptors for OT and arginine vasopressin (AVP) in autistic people as measured in blood by neuropeptide mRNA levels, suggesting that OT receptor quantity or distribution is also involved in ASD. Here, we focus solely on the overall difference in OT levels between autistic and neurotypical people, acknowledging that there could be more subtle, continuous associations between OT levels and socio-cognitive performance.
While the focus of the present meta-analysis is on differences in baseline OT levels, several studies measured changes in OT levels in the context of behavioral experiments. Feldman et al. (2014) showed that the lower baseline OT levels of autistic children normalized during parent–child interaction, that is, reached the same levels as those of neurotypical children, before returning to relatively lower baseline levels. Fujisawa et al. (2014) found an association between salivary OT levels and attention to pointed-at objects, but only in neurotypical children and not in autistic ones. Corbett et al. (2016) administered a low dose of hydrocortisone to children and measured a subsequent increase in OT plasma levels, proposed to reflect OT’s stress-buffering role—but only in neurotypical children and not in autistic ones. Mariscal et al. (2019) found autistic children to have a decreased contagious yawning response, but only those with low plasma OT levels. In sum, these studies indicate that not only baseline OT levels but also OT reactivity to different contexts may differ between autistic and neurotypical individuals.
Given these varied findings, it is important to know whether there are robust differences in OT levels between autistic and neurotypical individuals, and what factors might moderate such differences. To date, only one formal meta-analysis has addressed whether baseline OT levels differ in autistic and neurotypical people (Rutigliano et al., 2016). These authors found suggestive evidence for such a difference (plasma: k = 5, Hedges’ g = −0.58, 95% confidence interval (CI) [−1.44, 0.29], p = 0.193; saliva: k = 2, g = −0.35, 95% CI [−0.70, −0.01], p = 0.046). However, due to small sample size, methodological heterogeneity, and a Bonferroni correction applied to all meta-analyses in their article (which included several other psychiatric conditions), it was concluded that there was no significant difference in OT levels. A recent systematic review by Wilczynski et al. (2019) compared OT plasma levels (k = 10) and AVP plasma levels (k = 5) but did not provide a formal meta-analysis. They deemed it impossible to draw unequivocal conclusions due to, among others, heterogeneous methodologies, demographic differences among groups, or generally low quality of publications. They also excluded 17 of 29 articles from their final analysis because they did not meet their inclusion criteria of a “clearly and comprehensively presented” and “good methodology.” However, we could not identify which studies were excluded or what the exact criteria were.
Here, we greatly expanded the sample size compared to our predecessors (k = 31 studies), partly thanks to much recent research. We accounted for methodological heterogeneity using moderator analysis and followed the Newcastle–Ottawa Scale for assessing study quality in meta-analyses (Wells et al., 2000). Our goal was to test whether the current state of the field supported an overall difference in OT levels between autistic and neurotypical people, and what methodological or demographic factors might influence the magnitude of this difference.
Methods
Search and inclusion
A systematic search was conducted to find relevant articles. First, Web of Science was searched until 1 April 2020 (see Supplementary Materials for the search code). Articles were screened by title or abstract. If eligibility could not be evaluated, articles were read in full text. Second, reference lists of the retrieved articles were screened for additional articles that met the inclusion criteria. The search process is summarized in Figure 1.
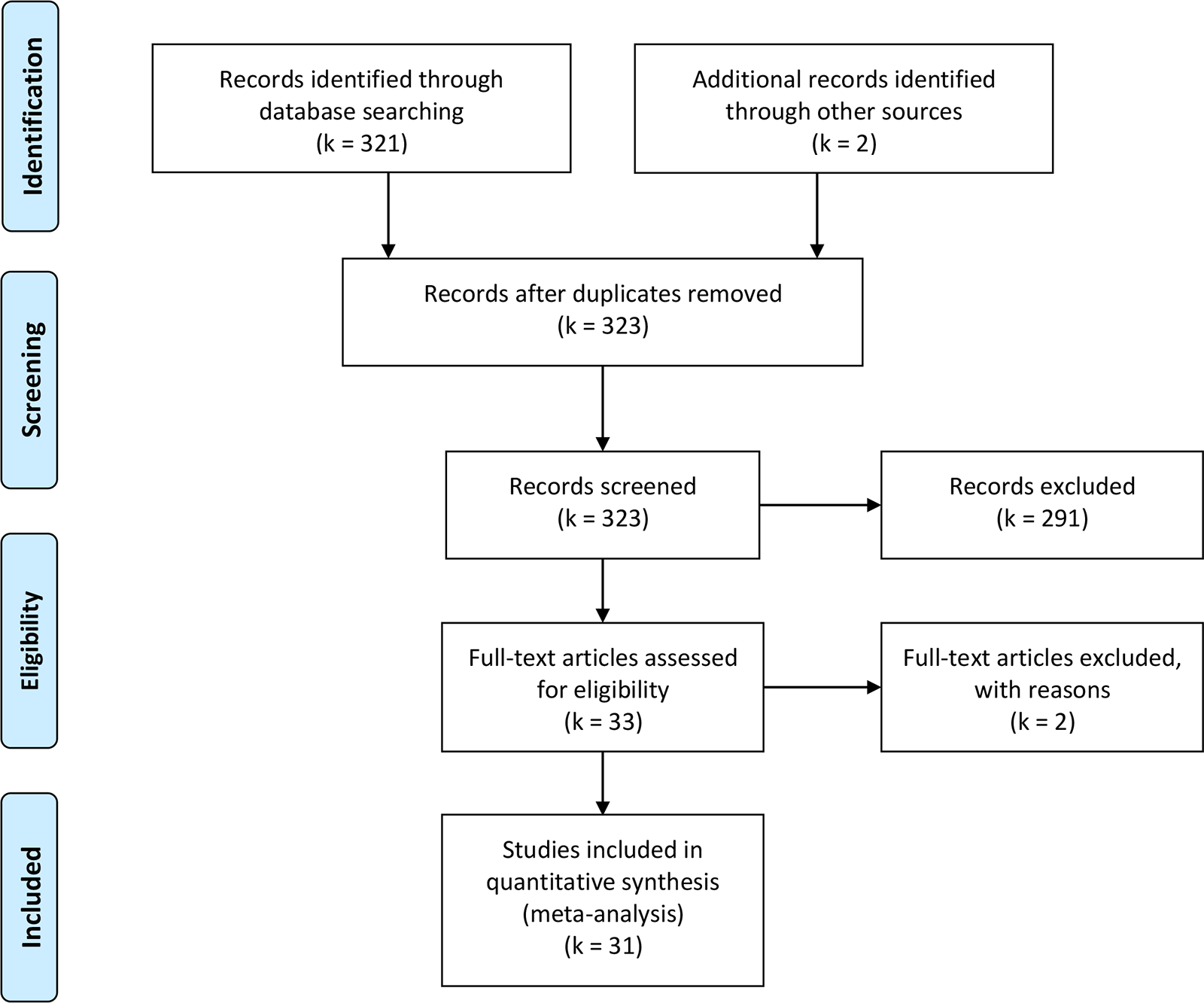
PRISMA workflow summarizing our search process and final sample.
Eligibility for the meta-analysis was met if articles: (1) were original articles written in English; (2) included participants with a diagnosis of ASD according to the Diagnostic and Statistical Manual of Mental Disorders (American Psychiatric Association, 2013) or the International Statistical Classification of Diseases and Related Health Problems (World Health Organization, 1993) or validated diagnostic instruments; (3) included a comparison group; and (4) reported mean levels or statistical differences of OT measurements between groups.
Articles were excluded if they: (1) reported an overlapping data set with another retrieved article. In this case, the article with the larger data set was included. In some cases, we suspected overlap but could not confirm it; we therefore report results with and without these studies. (2) Failed to report enough information by neither reporting mean OT levels nor statistical indicators of differences between groups. In those cases, we tried to contact the authors to obtain missing numbers. Hence, Green et al. (2001) had to be excluded because of an overlapping dataset with Modahl et al. (1998). Husarova et al. (2013); Husarova et al. (2016); Lakatosova et al. (2015); Ostatnikova et al. (2016) and Tomova et al. (2015) were all written by a largely identical research team and recruited their participants at the same institution. Because we could neither confirm nor rule out overlap of data and did not receive an answer from these researchers, we conducted the meta-analysis including all articles as well as excluding all but the one with the largest sample size (Lakatosova et al., 2015). We received additional information from Jansen et al. (2006) and Zhang et al. (2016), allowing us to include these studies. Sufficient information could not be obtained from Corbett et al. (2011).
Study quality
The Newcastle–Ottawa Scale for assessing the quality of non-randomized studies in meta-analysis (Wells et al., 2000) includes eight criteria, of which five are applicable here: case definition, representativeness of the cases, selection of controls, definition of controls, and comparability. We found virtually no variation in study quality across these domains, except “selection of controls,” where most studies included community samples but two included hospital samples (Aita et al., 2019; Oztan, Garner, et al., 2018). Thus, there were no obvious differences in recruiting, diagnosis, or sample composition that could impact study quality (Wells et al., 2000) and would therefore need to be accounted for in a meta-analysis.
Data extraction and meta-analysis
We extracted mean OT levels and their standard error (SE) or deviation (SD) for ASD and comparison groups as well as the sample sizes of each group to calculate Cohen’s d and its variance as a standardized measure of mean difference. If raw mean values and variances were not reported and the authors did not respond to emails soliciting this information, we extracted statistical indicators of differences between groups (t-test, F-test, p-value, or Mann–Whitney U Z) and calculated Cohen’s d from these values. The latter procedure was followed in Feldman et al. (2014); Jacobson et al. (2014); Ostatnikova et al. (2016); Oztan, Garner, et al. (2018); Oztan, Jackson, et al. (2018); Parker et al. (2014); Tomova et al. (2015); Yang et al. (2015, 2017). Table 1 lists Cohen’s d and its variance for all studies as well as further details on diagnosis, number of participants, their age and sexes, specimen type, and assay method. Specific data on socioeconomic status and educational attainment levels were typically not recorded in these studies, with the single exception of Feldman et al. (2014, who reported all parents of autistic children to have completed high school). We calculated an overall weighted effect size using random effects meta-analysis. We also assessed several moderators using meta-regression, including whether the subjects were children or adults (mean age >18), the difference in sex composition between patient and comparison groups (since males generally have lower OT levels (Carter, 2007)), as well as the assay type (enzyme-linked immunosorbent assay (ELISA) or radioimmunoassay (RIA)) and specimen type (blood, cerebrospinal fluid (CSF), and saliva) used. We tested for publication bias and missing studies using Egger’s regression test (Egger et al., 1997) as well as the trim-and-fill method (Duval & Tweedie, 2000). These tests are based on the idea that all studies should cluster symmetrically around the overall effect size with a degree of spread given by the precision of the study (see Supplementary Figure S1); at lower precision, that is, smaller sample sizes, effect sizes are scattered more widely, and studies with large effect sizes in the expected direction may be overrepresented, leading to detectable asymmetry. Finally, we tested for influential outliers using various diagnostics combined in the “influence” function. All analyses were conducted using the metafor package in R 3.5.2 (Viechtbauer, 2010). Community stakeholders were not involved in the preparation of this meta-analysis.
Articles included in the meta-analysis.
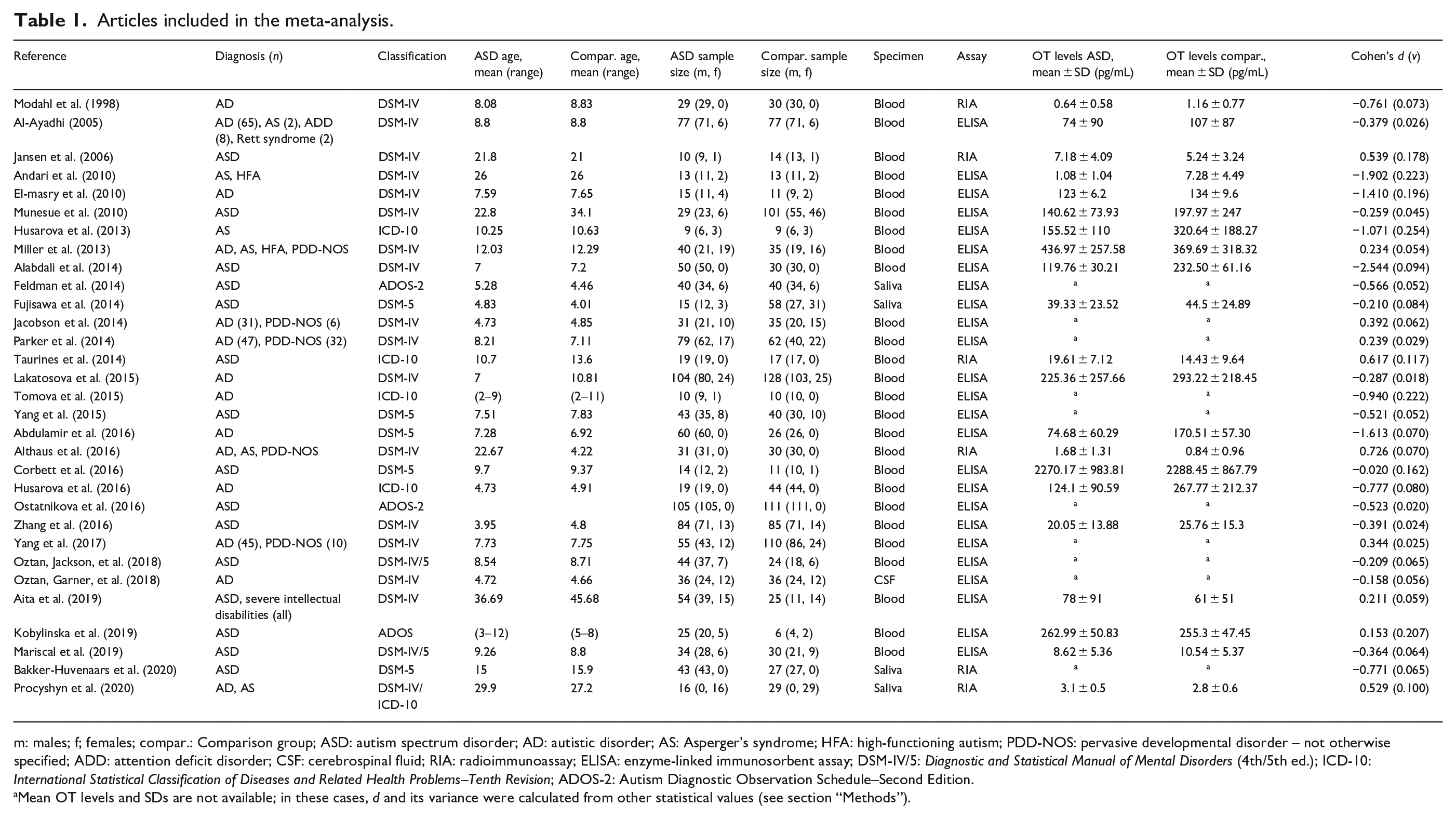
m: males; f; females; compar.: Comparison group; ASD: autism spectrum disorder; AD: autistic disorder; AS: Asperger’s syndrome; HFA: high-functioning autism; PDD-NOS: pervasive developmental disorder – not otherwise specified; ADD: attention deficit disorder; CSF: cerebrospinal fluid; RIA: radioimmunoassay; ELISA: enzyme-linked immunosorbent assay; DSM-IV/5: Diagnostic and Statistical Manual of Mental Disorders (4th/5th ed.); ICD-10: International Statistical Classification of Diseases and Related Health Problems–Tenth Revision; ADOS-2: Autism Diagnostic Observation Schedule–Second Edition.
Mean OT levels and SDs are not available; in these cases, d and its variance were calculated from other statistical values (see section “Methods”).
Results
Thirty-one articles were selected for meta-analysis (see Figure 1 and Table 1). Overall, we found that OT levels were significantly lower in autistic people compared to neurotypical individuals (Cohen’s d = −0.36, 95% CI = [−0.61, −0.10], k = 31, p = 0.007, Figure 2). There was no evidence for publication bias as Egger’s test was not significant (p = 0.17) and the trim-and-fill method did not detect any missing studies (see Supplementary Figure S1). There was one influential outlier study (Alabdali et al., 2014), the removal of which slightly reduced effect size (d = −0.28, 95% CI = [−0.49, −0.06], k = 30, p = 0.01). Excluding the four studies with potential data overlap still yielded a significant effect (d = −0.30, 95% CI = [−0.58, −0.01], k = 27, p = 0.04). Since there was significant heterogeneity in effect sizes among studies (Q = 193.81, degree of freedom (df) = 30, p < 0.0001), we examined the influence of potential moderators.
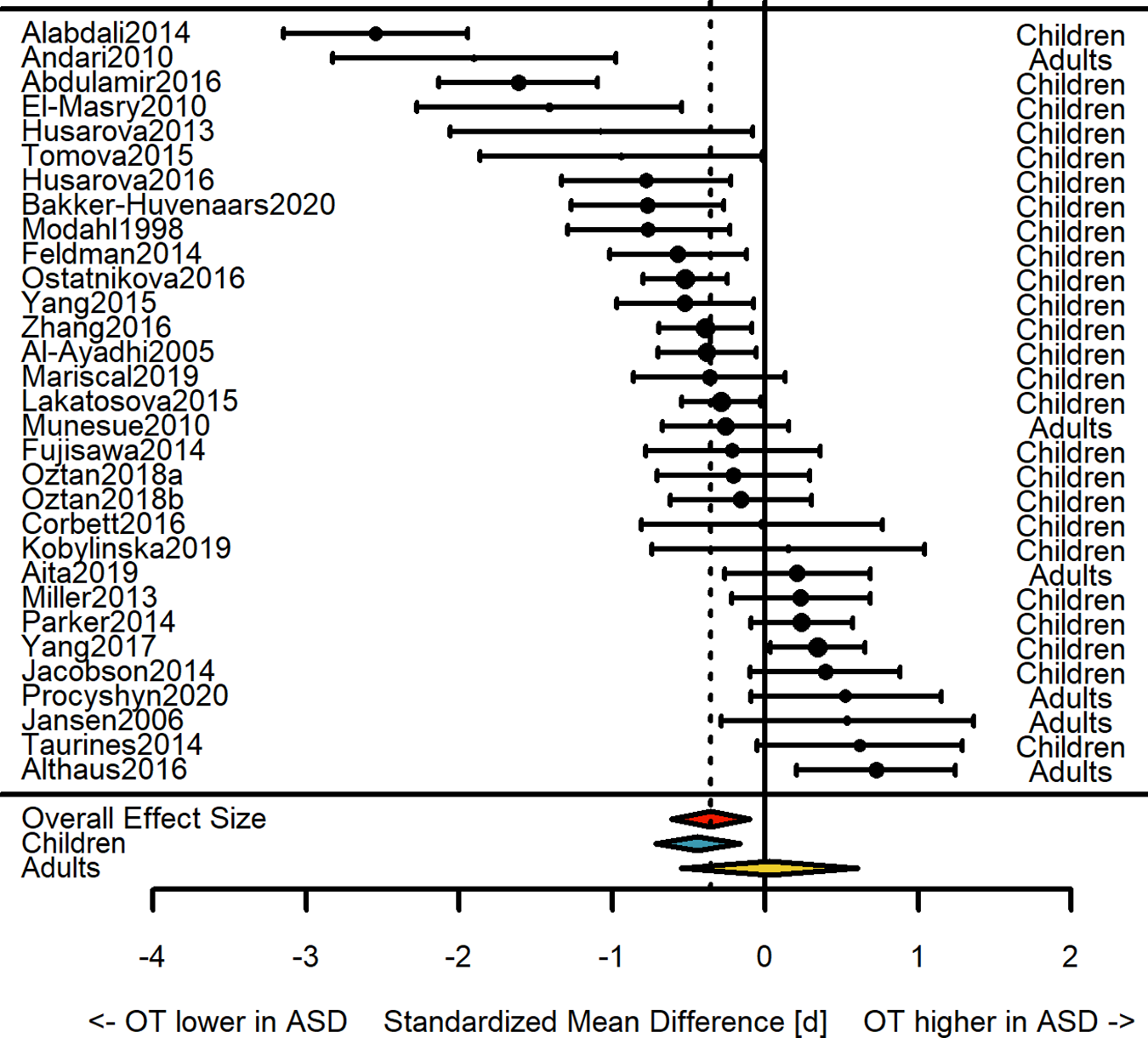
Forest plot showing effect sizes (Cohen’s d) from individual studies and the overall weighted effect size (with 95% CI).
Meta-regressions showed that significant differences in OT levels between ASD and comparison groups were only found in children (k = 25, d = −0.44, 95% CI = [−0.72, −0.16], p = 0.002) but not adults (k = 6, d = 0.03, 95% CI = [−0.55, 0.61], p = 0.92; test for difference: QM = 9.72, df = 2, p = 0.008), and only in studies using ELISA (k = 25, d = −0.46, 95% CI = [−0.74, −0.19], p = 0.001) but not RIA (k = 6, d = 0.12, 95% CI = [−0.45, 0.70], p = 0.68; QM = 11.01, df = 2, p = 0.004). Furthermore, there was a trend toward different effect sizes according to specimen type (QM = 7.04, df = 3, p = 0.07), with a significant effect only detectable in blood samples (k = 26, d = −0.38, 95% CI = [−0.67, −0.09], p = 0.01) but not saliva (k = 4, d = −0.27, 95% CI = [−1.00, 0.47], p = 0.47) or CSF (k = 1, d = −0.16, 95% CI = [−1.60, 1.28], p = 0.83); given the small sample sizes of the latter two and the fact that all overall effect sizes are negative, this difference seems negligible. Finally, the overall effect size was not driven by a greater proportion of males in the ASD group; indeed, when correcting for differences in the proportion of males in the ASD and comparison groups, the overall effect size becomes slightly stronger (d = −0.43, 95% CI = [−0.70, −0.15], p = 0.002), although this moderator was not significant (QM = 1.87, df = 1, p = 0.17). In all meta-regressions, significant residual heterogeneity remained.
Discussion
We found strong evidence that OT levels differed between autistic children (but not adults) and neurotypical individuals. Our results support and strengthen the suggestive difference found in an earlier meta-analysis on a much smaller sample of studies (Rutigliano et al., 2016).
Our finding of lower OT levels in autistic children points to an involvement of the OT system in the development or manifestation of ASD. The substantial heterogeneity of the results, even after accounting for moderators, may be partly due to the inclusion of a wide range of ASD phenotypes (see Table 1). Furthermore, in at least 19 articles, OT levels were correlated with ASD symptom severity, with the majority reaching significance. Together with studies relating OT levels to socio-cognitive functions in neurotypical individuals as well as in siblings of autistic children (Parker et al., 2014), our finding is consistent with OT levels mediating a continuous range of socio-cognitive function, at the extreme of which are autistic people (Crespi, 2016).
In contrast to the strong evidence of lower OT levels in autistic children, OT levels in autistic adults were virtually indistinguishable from those of neurotypical adults. Although only a few studies included adults (k = 6), this suggests that OT levels in autistic people might normalize as they grow older. Consistent with this possibility, longitudinal studies point to improvement in symptoms and adaptive functioning toward adulthood (Magiati et al., 2014; Seltzer et al., 2004), including improved communication, perhaps mediated by elevated OT levels. Such a normalization of OT levels could explain why intranasal administration of OT in autistic adults often yields small or no effects on social function or repetitive behaviors (Peled-Avron et al., 2020; Phaik Ooi et al., 2017; Wang et al., 2019).
Instead of OT levels being unusually low in autistic children and later “catching up” in adulthood, it is also possible that OT levels in neurotypical children are unusually high and later decline to match those of autistic people by adulthood. The latter scenario would be analogous to OT receptor densities in the ventral pallidum, which are the highest in neurotypical children and most different from autistic individuals at younger ages, before converging on the lower level of autistic people by adulthood (Freeman et al., 2018). In this view, the OT system may be responsible for priming the brain for social interactions during a critical period in childhood, and one would expect a lack of such priming to have effects lasting into adulthood, as seen in a majority of autistic adults continuing to show social deficits (Magiati et al., 2014). In general, relatively little is known about the developmental trajectory of OT levels, and detailed longitudinal studies tracking OT levels, socio-cognitive skills, and other ASD-related symptoms would be needed to better understand how OT levels mediate ASD and social cognition more broadly across the life course.
In sum, our results warrant further investigation of the connections between the OT system and social deficits in ASD and of the potential therapeutic use of OT to alleviate these deficits, especially in children. This study cannot address causality, that is, whether social deficits cause low OT levels or vice versa; future work on the genetics of the OT system or longitudinal studies of ASD symptom severity and OT levels may help establish such causal connections.
Supplemental Material
sj-docx-1-aut-10.1177_13623613211034375 – Supplemental material for Oxytocin levels tend to be lower in autistic children: A meta-analysis of 31 studies
Supplemental material, sj-docx-1-aut-10.1177_13623613211034375 for Oxytocin levels tend to be lower in autistic children: A meta-analysis of 31 studies by Simon John and Adrian V Jaeggi in Autism
Supplemental Material
sj-docx-2-aut-10.1177_13623613211034375 – Supplemental material for Oxytocin levels tend to be lower in autistic children: A meta-analysis of 31 studies
Supplemental material, sj-docx-2-aut-10.1177_13623613211034375 for Oxytocin levels tend to be lower in autistic children: A meta-analysis of 31 studies by Simon John and Adrian V Jaeggi in Autism
Footnotes
Acknowledgements
The authors thank Dr Lucres Jansen and her research team from the VU Medical Center in Amsterdam, and Professor Rong Zhang and Professor Hongfeng Zhang and their research team from the Neuroscience Research Institute in Beijing for providing additional data.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
