Abstract
Atypical sensory processing is prevalent across neurodevelopmental conditions and a key diagnostic criterion of autism spectrum disorder. It may have cascading effects on the development of adaptive functions. However, its unique contribution to adaptive functioning and the genetic/environmental influences on this link are unclear. In a clinically enriched twin sample (n = 289, 60 diagnosed with autism spectrum disorder), we investigated the associations between the quadrants of the Adult/Adolescent Sensory Profile (low registration, sensory sensitivity, sensation seeking, and sensation avoiding) and adaptive functioning. Associations were modeled across the cohort accounting for the effects of clinical diagnosis, IQ, sex and age, and within-twin pairs, additionally implicitly adjusting for familial factors. Furthermore, we explored interaction effects between atypical sensory processing and autism spectrum disorder diagnosis. Sensory sensitivity and sensation avoiding were associated with reduced adaptive functioning across individuals, but not within-twin pairs. An interaction effect was found between sensation seeking and autism spectrum disorder diagnosis, showing a negative association between sensation seeking and adaptive functioning only in individuals diagnosed with autism spectrum disorder. The results suggest that atypical sensory processing is associated with reduced adaptive functioning and that familial factors influence this link. In addition, sensation seeking behaviors might interfere with adaptive functioning specifically in individuals with autism spectrum disorder.
Lay abstract
Individuals diagnosed with autism tend to process sensory information differently than individuals without autism, resulting for instance in increased sensitivity to sounds or smells. This leads to challenges in everyday life and may restrict the individual’s daily functioning. How direct this link is, however, is currently unclear. We investigated this question in 289 twins of whom 60 were diagnosed with autism and further 61 were diagnosed with other neurodevelopmental disorders. We looked at the association between unusual sensory processing and adaptive skills, both across individuals and within-twin pairs, testing whether individuals with higher levels of atypical sensory processing showed reduced adaptive skills compared to their twins. Since twins share 50%–100% of their genes and part of their environment (e.g. family background), associations within-twin pairs are free from effects of these familial factors. We found that an increased sensitivity to, as well as the avoiding of, sensory input (hyper-responsiveness) was linked to reduced adaptive skills across individuals—but not within-twin pairs. We also found an association between the degree to which individuals seek for sensory input (sensation seeking) and reduced adaptive skills, but only in individuals diagnosed with autism. The results suggest that sensory hyper-responsiveness has negative effects on individuals’ general ability to function, but that this link is influenced by familial factors and hence not direct. In addition, sensation seeking behaviors might have a negative impact on adaptive skills specifically in autistic individuals.
Keywords
Introduction
Alongside impairments in social communication and interaction, atypical sensory processing is considered a defining diagnostic criterion of autism spectrum disorder (ASD) within the domain of restricted and repetitive behaviors, according to the Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5; American Psychiatric Association, 2013, pp. 50–59). Specifically, the DSM-5 specifies hyper- or hypo-responsiveness to sensory input or “unusual interest in sensory aspects of the environment” as relevant sub-criteria to meet an ASD diagnosis.
Atypical sensory processing is considered one of the earliest signs of ASD (Ben-Sasson et al., 2007), observed as early as at 6 months of age in individuals who are later diagnosed with ASD (Germani et al., 2014). Furthermore, many individuals with ASD perceive sensory challenges as the true core of their condition (Chamak et al., 2008) and as a research priority (Pellicano et al., 2014). While also common in other neurodevelopmental and psychiatric disorders, such as attention-deficit hyperactivity disorder (ADHD) (Bijlenga et al., 2017; Ghanizadeh, 2011) and anxiety disorders (Liss et al., 2005; Serafini et al., 2017), sensory processing atypicalities are especially common (45–93%) in children and adults with ASD (Baranek et al., 2006; Leekam et al., 2007; McCormick et al., 2016) - at a level otherwise only found in children with Fragile X syndrome (without ASD) or deaf-blindness (Rogers et al., 2003; Rogers & Ozonoff, 2005). A recent meta-analysis of 55 studies found a high incidence of hypo-responsiveness in ASD compared to typically developing (TD) individuals and increased hyper-responsiveness even compared to clinical comparison groups (Ben-Sasson et al., 2019). It should however be noted that the patterns of sensory features in ASD are heterogeneous (Crane et al., 2009; Uljarević et al., 2017) and a typical ASD profile of atypical sensory processing has yet to be defined (Leekam et al., 2007; Rogers & Ozonoff, 2005; Schaaf & Miller, 2005). Results regarding sensation seeking (i.e. performing activities to increase sensory input) have the greatest variability among studies and the results are moderated by cognitive ability, study design, and age (Ben-Sasson et al., 2019). Adding to the phenotypic complexity in ASD, both hypo- and hyper-responsiveness often co-occur in the same individual (Baranek et al., 2006; Ben-Sasson et al., 2007; Mayer, 2017).
Sensory features in ASD are included in the restricted and repetitive behavior domain of autistic symptoms and consequently correlate with higher symptom levels in this domain (Lidstone et al., 2014; Wigham et al., 2015). However, they also correlate with other ASD and comorbid symptoms, including social communication problems (Glod et al., 2015; Hilton et al., 2010) and anxiety (Green et al., 2012; Uljarević et al., 2016). Furthermore, there is increasing evidence of sensory features being associated with challenges in general adaptive functioning in ASD (Dellapiazza et al., 2018; Williams et al., 2018). Adaptive functioning is formed as the interplay between an individual’s physical and mental functions and the features of the environment (Bölte et al., 2019), including an individual’s capacity to function in everyday life, such as getting dressed, using public transport, being able to converse and understand others, and take part in group or community activities. The level of adaptive functioning largely determines an individual’s degree of independence, educational and vocational achievements, and participation in society as a whole and is thus of paramount relevance for mental health and well-being (De Bildt et al., 2005; Farley et al., 2009; Paul et al., 2004). Many individuals with ASD show pronounced impairments in adaptive functioning (Chatham et al., 2018; Harrison & Oakland, 2008; Kanne et al., 2011; Nevill et al., 2017), leading to a poorer prognosis compared to the general population in the areas of independence, employment, and relationships (Cederlund et al., 2008; Henninger & Taylor, 2013; Shattuck et al., 2012) and higher rates of depression and anxiety (Kraper et al., 2017).
Impairments in adaptive functioning are not fully accounted for by cognitive ability (Charman et al., 2011; Kanne et al., 2011; Tillmann et al., 2019), suggesting that in ASD, additional factors contribute to adaptive functioning challenges. The presence and level of atypical sensory processing has been suggested to be relevant for adaptive functioning, in general and especially for individuals with ASD (Ashburner et al., 2008; Baker et al., 2008; Dellapiazza et al., 2018; Glod et al., 2015; Jasmin et al., 2009; Liss et al., 2006; Pfeiffer et al., 2005). For instance, atypical sensory processing is associated with difficulties in mealtime situations (Zobel-Lachiusa et al., 2015), community participation (Hochhauser & Engel-Yeger, 2010; Pfeiffer et al., 2005), academic achievement (Ashburner et al., 2008; Ismael et al., 2018), communicative skills (Dellapiazza et al., 2018; Glod et al., 2015; Watson et al., 2011), and social awareness in individuals with ASD (Glod et al., 2015). Sensation avoiding, in particular, is negatively associated with self-care skills such as eating and getting dressed in children with ASD (Jasmin et al., 2009).
Interestingly, sensation seeking behaviors, which are associated with positive mood, higher education, and reduced depressive symptoms in TD individuals (Ben-Avi et al., 2012; Engel-Yeger, & Dunn, 2011; Machingura et al., 2019), are in contrast linked to a broad range of behavioral problems, including difficulties in adjusting one's behavior according to social rules (Baker et al., 2008) and academic underachievement in children with ASD diagnosis (Ashburner et al., 2008).
The association between atypical sensory processing and adaptive functioning was also supported by a recent large-scale study including 417 children, adolescents, and adults with ASD (Tillmann et al., 2019). However, this association dropped below the significance level when a range of independent variables was included in the same model, where only older age, lower cognitive ability, and higher social communication symptoms predicted lower adaptive functioning. However, the latter study did not differentiate between different types of sensory processing atypicalities.
Taken together, atypical sensory processing seems to be negatively associated with adaptive functioning, especially in individuals diagnosed with ASD. However, more controlled research designs are required to decipher the impact of sensory processing alterations on adaptive functioning relative to other factors such as comorbid conditions, cognitive ability, sex, or age. In addition, different domains of atypical sensory processing should be assessed separately. Finally, little is known regarding the impact of genetics and environment on the association between sensory processing and adaptive functioning.
Twin and family studies have the power to differentiate genetic and environmental contributions and to implicitly control for a large number of confounding factors, but are sparse in this context. The few existing studies revealed that sensory hyper-responsiveness and its association with autistic traits seems to be strongly influenced by genetics in the general population (Taylor et al., 2018). Furthermore, co-twins of autistic individuals share a similar pattern of altered sensory processing even if they are not diagnosed with ASD themselves (De la Marche et al., 2012), as do parents of children with ASD (Donaldson et al., 2017; Glod et al., 2017; Uljarević et al., 2014) and sensory processing alterations seem to be stronger in families with several family members with ASD diagnosis (Donaldson et al., 2017). In a recent twin study on a sample largely overlapping with the current study’s, we found that genetics influenced the association between sensory sensitivity and autistic traits, while other domains of altered sensory processing assessed with the Adult/Adolescent Sensory Profile (AASP; low registration and sensation avoiding) persisted even within monozygotic (MZ) twins and were hence influenced by non-shared environment (Neufeld et al., 2021).
The few family studies about adaptive functioning in ASD have yielded mixed results, indicating either that adaptive functioning is strongly determined by familial factors (Goin-Kochel et al., 2008; MacLean et al., 1999; Szatmari et al., 1996) or the opposite, that is, non-shared factors (Le Couteur et al., 1996; Spiker et al., 1994). One twin study indicated a strong genetic impact on the association between ASD and adaptive functioning (Goin-Kochel et al., 2008). To the best of our knowledge, no twin study focused on the association between atypical sensory processing and adaptive functioning in individuals with and without ASD or other neurodevelopmental disorders (NDDs).
In this study, we addressed the gaps in the literature by investigating (a) the specific relationships between the four quadrants of atypical sensory processing assessed with the AASP (low registration, sensory sensitivity, sensation seeking, and sensation avoiding) and adaptive functioning while adjusting for ASD diagnosis and other neurodevelopmental diagnoses, age, sex, and cognitive ability. Furthermore, we (b) investigated this link for the first time within-twin pairs where familial factors are implicitly controlled for. These comprise all shared environmental factors and in MZ twins even 100% of the genetics. Associations observed across the cohort but lost within pairs can be assumed to be influenced by familial factors, while associations remaining significant within MZ pairs can be assumed to be driven by non-shared environmental factors. Finally, we (c) explored the possibility that the relationship between atypical sensory processing and adaptive functioning might be different in individuals with and without ASD.
We hypothesized that atypical sensory processing would predict a reduction in adaptive functioning beyond the effects of diagnoses, cognitive ability, sex, and age. We expected that genetics influence these relationships and that associations would therefore be attenuated within-twin pairs.
Methods
Participants
After applying exclusion criteria (see below for detail), a total of 289 individuals (143 twin pairs and one triplet, aged 11–33 years) were included, dividing into 84 MZ (MZ; 49% female, mean age = 18.9 ± 6.0 years) and 59 dizygotic (DZ; 41% female; mean age = 16.4 ± 5.2 years) twin pairs, and one set of triplets. Zygosity was determined on a panel of 48 single nucleotide polymorphisms (Hannelius et al., 2007). In a few cases (21 pairs), where DNA results had not yet been analyzed, a 4-item zygosity questionnaire was used instead. In total, 21% of the sample were diagnosed with ASD (n = 60), 21% had other neurodevelopmental disorders but no ASD (NDD, n = 61, primarily ADHD or specific learning disorders), and the remaining 58% had no NDD diagnosis (No NDD, n = 168, please see Table 1). Twelve twin pairs were concordant for ASD diagnosis (both individuals fulfilling diagnostic criteria) and 35 twin pairs and one triplet were discordant for ASD (only one twin fulfilling diagnostic criteria). All participants and/or their caregivers gave informed consent to take part in the study in accordance with the Declaration of Helsinki.
Sample characteristics.
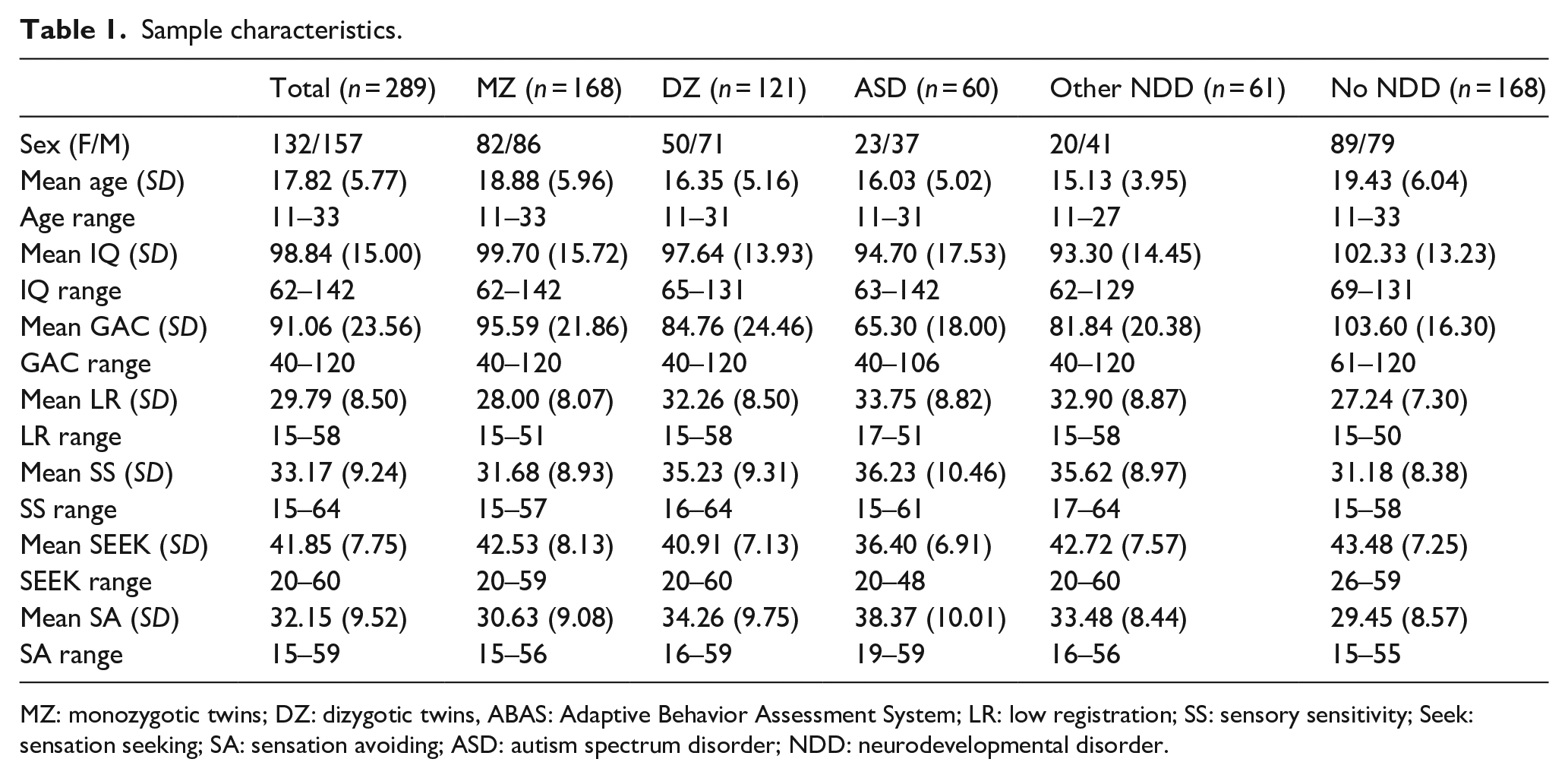
MZ: monozygotic twins; DZ: dizygotic twins, ABAS: Adaptive Behavior Assessment System; LR: low registration; SS: sensory sensitivity; Seek: sensation seeking; SA: sensation avoiding; ASD: autism spectrum disorder; NDD: neurodevelopmental disorder.
The twin sample was recruited within the Roots of Autism and ADHD Twin Study in Sweden (RATSS, Bölte et al., 2014). RATSS recruits twin pairs and triplets, primarily from the population-based Child and Adolescent Twin Study in Sweden (CATSS) (Anckarsäter et al., 2011) which includes all twins born in Sweden since 1994. In RATSS, twins from CATSS are primarily recruited if at least one of the twins scores within the higher range on the autistic or ADHD domain of the “Autism-Tics, ADHD and other Comorbidities inventory” (A-TAC) (Hansson et al., 2005). However, a proportion of likely TD twins (where both twins score low on the A-TAC) is also included. General inclusion criteria for this study were complete data for both twins on all study variables, that is, AASP, adaptive functioning (Adaptive Behavior Assessment System second version (ABAS-II)), and general cognitive ability (Wechsler Adult Intelligence Scale-IV (WAIS-IV)/Wechsler Intelligence Scale for Children-IV (WISC-IV)) and being of the same sex.
From the original sample of 420 assessed individuals (207 twin pairs and 2 sets of triplets), 100 individuals were excluded due to data missing for them or their co-twin on any of the relevant variables. These included 24 twin pairs who were not eligible to complete the AASP because they were below 11 years of age. Furthermore, we excluded 31 individuals for the following reasons: 10 individuals (five twin pairs) due to missing zygosity information, 9 opposite sex twin pairs (= 18 individuals), 1 male individual from a triplet of otherwise female individuals, and 1 twin pair from a family of which 2 pairs (of different ages) had been assessed.
One same sex triplet discordant for ASD with complete data for all individuals was included, since the statistical model at hand can handle clusters of different sizes, enabling within-triplet comparisons as well.
Socioeconomic background
95.6% of the participants’ parents reported in which out of six categories their households total monthly income before tax in Swedish crowns falls (10 Swedish crowns = approximately 1.1 US dollars). These categories were coded as 0 = below 20,000 (6.9%), 2 = 20,000–40,000 (21.8%), 3 = 40,000–60,000 (35.0%), 4 = 60,000–80,000 (20.8%), and 5 = above 80,000 (11.1%). Please see the Supplementary material for income data split for participants with and without NDDs.
Ethnicity was assessed after the initial data collection and 57% of the RATSS participants’ parents replied to the question. Of these, 95% reported to have a European ethnic background.
Measures
Diagnostic assessment for ASD and other NDDs
All diagnoses were best clinical estimate diagnoses according to DSM-5, made by a group of experienced clinicians during a 3-day visit. These were supported by a comprehensive assessment of the participants’ medical history, taking clinical records and standardized diagnostic instruments into account. The gold standard diagnostic tools utilized for the assessment of ASD included Swedish versions of the “Autism Diagnostic Interview–Revised” (ADI-R; Rutter et al., 2003) and the “Autism Diagnostic Observation Schedule” (ADOS; Gotham et al., 2007; Lord et al., 2000), which have good to excellent psychometric properties (Zander et al., 2016, 2017). Other NDDs such as ADHD, tic disorders, specific learning disorders and intellectual disability (ID), as well as psychiatric disorders such as depression and anxiety were determined based on a multitude of information sources. These included the “Kiddie Schedule for Affective Disorders and Schizophrenia” for children (K-SADS; Kaufman et al., 1997), the “Diagnostic Interview for ADHD in adults” (DIVA; Kooij & Francken, 2010), and the “Structured Clinical Interview for DSM-IV” (SCID, axis I; First et al., 1996) for the assessment of ADHD, tic disorders, and psychiatric disorders. Specific learning disorders were determined through history taking, using parent interview. The diagnosis of ID (intellectual disability) was based on IQ, ABAS-II, and the overall clinical consensus assessment made by psychologists meeting the twins and parents.
Sensory processing
The Sensory Profile (Brown & Dunn, 2002) is one of the most commonly used tools to assess atypical sensory processing in individuals with ASD (Brown et al., 2001). It relies on Dunn’s (1997) model of sensory processing, which divides sensory processing along the axes of neurological threshold and self-regulation strategies and covers five sensory modalities: hearing, sight, touch, smell, and taste and in addition activity level and movement. The model generates four quadrants of sensory processing, with two modes representing hypo-responsiveness to stimuli: low registration and sensation seeking, and the other two representing hyper-responsiveness to stimuli: sensory sensitivity and sensation avoiding. Low registration may manifest in not experiencing temperature shifts, slow reactions, passivity, and apparent indifference to pain. Sensory sensitivity implies the contrary: quick, strong, often prolonged registration of sensory input with following adverse reactions. Sensation seeking and sensation avoiding represent active self-regulation strategies to increase or decrease sensory input, respectively. Children engaging in sensation seeking behaviors may make different sounds, fidget or move constantly in an effort to meet their sensory needs (Dunn, 1997).
Here, we used the Adult/Adolescent version of the Sensory Profile (AASP, Brown & Dunn, 2002) which is a self-report questionnaire, typically used from 11 years and consisting of 60 items that assess the frequency of various sensory experiences. Each item is rated on a 5-point Likert-type scale ranging from 1 (“Almost never”) to 5 (“Almost always”), with higher total raw scores on sensory quadrants indicating more atypical sensory processing. Participants and their twins were excluded if more than three items were missing on any of the AASP quadrants (= less than 80% data per quadrant), while up to three missing items per quadrant were imputed with the quadrant mean. The Swedish version has been linguistically and culturally adapted to Swedish conditions and validated in 500 TD individuals (Brown & Dunn, 2014).
Adaptive functioning
Parental ratings on the Swedish child and adolescent version of the ABAS-II was used, which is a scale developed for assessing the adaptive skills of individuals with disabilities (Harrison & Oakland, 2008). Each of the 232 items is a description of an adaptive behavior, spanning from basic abilities such as being able to go to the toilet alone to more advanced abilities such as being able to read a map. There are four response options: “Not Applicable” (0 points), “Never/almost never” (1 point), “Sometimes” (2 points), and “Always/almost always” (3 points), with a maximum total raw score of 696 points. The raw score (sum of points) for each sub-scale is converted to scaled scores which in turn is converted to age-normed index scores. This overall index score, General Adaptive Composite (GAC), represents the individual’s overall level of functioning, ranging between 0 and 120 in this study. GAC builds on three domains: the conceptual, social, and practical domains. The conceptual domain represents the ability to express oneself in speech and writing, applying knowledge in everyday situations and following directions and daily routines. The social domain represents the individual’s ability to independently engage in leisure activities as well as interact with others and maintain relationships. The practical domain focuses on an individuals’ ability to take care of their own health, safety, and hygiene and to manage practical challenges at home or in public such as preparing food or using public transport. Since the Swedish version of ABAS-II does not yet hold age norms for individuals above 21 years, individuals older than 21 (n = 74, 37 twin pairs) were coded based on the norms for 17–21 year olds. Hence, we included age as a covariate in order to control for possible age effects. In addition, we re-run the analyses while excluding these 74 individuals, in order to verify that they did not drive any effects, and reported the outcomes in the supplement. All three ABAS domains have shown high internal consistencies: conceptual index Cronbach’s α = 0.94, social index α = 0.91, and practical index α = 0.95 (ABAS-II) (Harrison & Oakland, 2008). The validity of ABAS-II has been tested and confirmed in a Swedish context (Fors & Tideman, 2007).
Cognitive ability
Depending on the age of participants, general intellectual abilities were assessed with the WAIS-IV in individuals ⩾17 years (Wechsler, 2010) and in individuals younger than 17 years with the WISC-IV (Wechsler, 2007). From both measures, the General Ability Index (GAI) as an estimate of full-scale IQ was calculated. The GAI was used instead of a full-scale IQ because including working memory and processing speed might have led to a disproportionally low IQ estimate in individuals with NDDs, who often have severe difficulties in these areas (Styck et al., 2019; Theiling & Petermann, 2016; Wechsler, 2007, 2010).
Statistical analysis
Sample characterization
Before conducting the main analyses, we characterized the three diagnostic groups: participants diagnosed with ASD (ASD, n = 60), participants without ASD diagnoses who had one or more other NDD diagnoses (Other NDD, n = 61), and participants without any NDD diagnosis (No NDD, n = 168). Twins not fulfilling diagnostic criteria for any NDD diagnosis were included in the No NDD group, regardless of whether or not they had other mental health conditions such as affective disorders.
For all variables of interest, group means were compared using two-sample t-tests and Cohen’s d effect sizes were calculated with conventions of very small (d < 0.2), small (d = 0.2), medium (d = 0.5), and large (d ⩾ 0.8). Group mean comparisons neglected the twin relationships.
The main analyses were conducted using the generalized estimating equations (GEE) framework with doubly robust standard errors (drgee package in R), which does not make any distributional assumptions (Zetterqvist & Sjölander, 2015). The different quadrants of AASP correlated with each other to varying degrees on our sample (r = 0.6–0.7 between sensory sensitivity, low registration, and sensation avoiding while sensation seeking was uncorrelated with the other domains: r = −0.1 to 0.1). They were examined separately, with one model for each domain as predictor of general adaptive functioning (ABAS GAC score). To compensate for multiple comparisons (four separate models), the Bonferroni-corrected p-value threshold was set to p = 0.0125 (at α-level = 0.05).
Across the sample
First, linear regressions were conducted across the sample adjusted for the non-independence of twins (cluster-robust standard errors). ASD diagnosis, other NDD diagnoses, IQ, sex, and age were included as covariates. Where the ABAS GAC score was significantly associated with an AASP quadrant, post hoc analyses were conducted in order to explore whether the three ABAS sub-domains (cognitive, social, and practical skills) were differentially associated with sensory processing atypicality.
In an additional step, we added the interaction between AASP quadrant and ASD diagnostic status in order to investigate whether any of the AASP quadrants were differentially associated with adaptive functioning in individuals with versus without ASD diagnosis and assessed this relationship separately in the diagnostic groups (ASD, Other NDD, and No NDD) if the interaction term was significant.
Within-twin pairs
In addition, the same associations were tested within-twin pairs, using conditional linear regressions. In this analysis, age and sex were implicitly controlled for. Hence, only ASD, other NDD diagnoses, and IQ were included as covariates. Furthermore, all shared environmental factors and either 100% (in MZ pairs) or on average 50% (in DZ pairs) of the genetics were implicitly controlled for. Where any within-pair associations were significant, we further tested them within the sub-sample of 84 MZ twin pairs (n = 168) in order to be able to draw conclusions regarding whether they were influenced by non-shared environmental factors rather than genetics (as non-shared environment is the only component not shared by MZ twins).
Community involvement
There is no explicit community involvement in this study.
Results
Group differences
Means, standard deviations, and ranges per measure and group can be found in Table 1, and outcomes from group comparisons are summarized in Table 2. The ASD group and the Other NDD group differed from the No NDD group in most variables and large effect size differences between all groups were found for adaptive functioning (d range from 0.83 to 2.25). Both clinical groups had lower IQ, lower mean age, and lower overall adaptive functioning compared to the No NDD group (p-values < 0.001) and higher scores in three of the AASP quadrants, low registration, sensory sensitivity, and sensation avoiding (p-values of group comparisons < 0.001 or < 0.0125). The ASD group had lower mean scores in sensation seeking compared to both other groups (p-values < 0.001) while the Other NDD and the No NDD group did not differ from each other in this quadrant (p = 0.50). Furthermore, the ASD group had on average lower overall adaptive functioning than the Other NDD group (p < 0.001) and scored higher in the sensation avoiding quadrant (p < 0.001). The ASD and other NDD groups did not differ in age, IQ, low registration, and sensory sensitivity (p-values > 0.05).
Group comparisons.
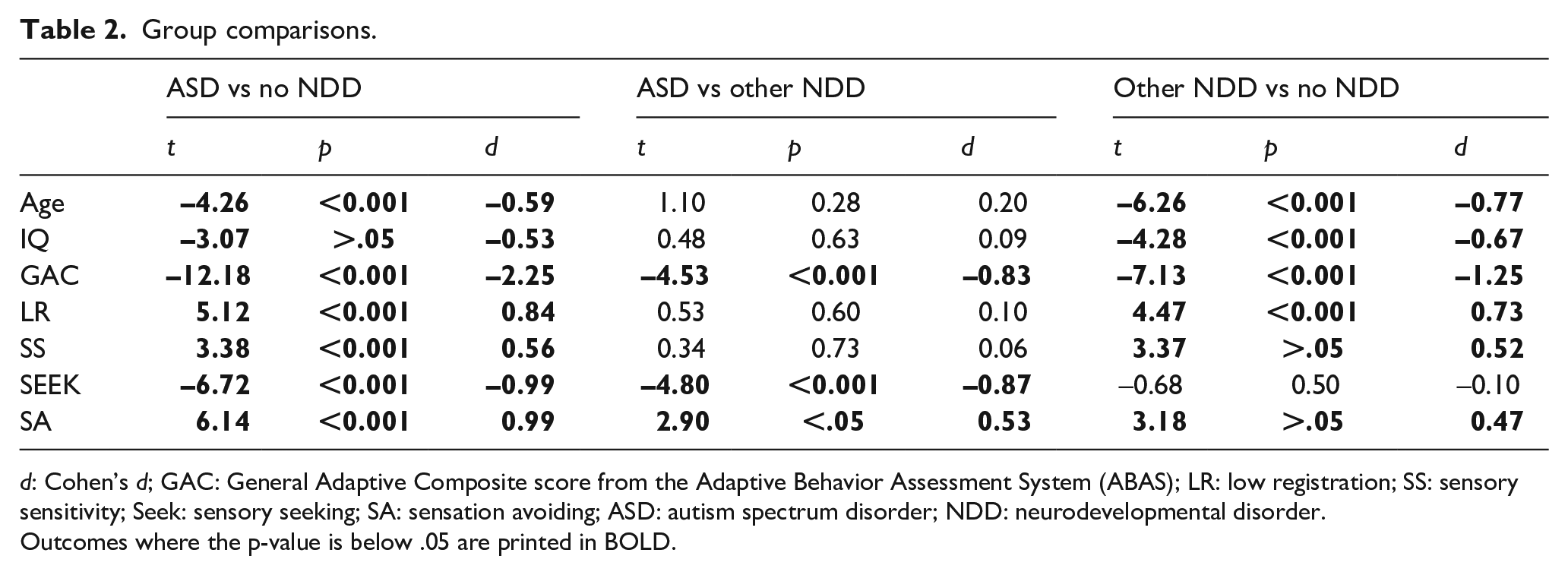
d: Cohen’s d; GAC: General Adaptive Composite score from the Adaptive Behavior Assessment System (ABAS); LR: low registration; SS: sensory sensitivity; Seek: sensory seeking; SA: sensation avoiding; ASD: autism spectrum disorder; NDD: neurodevelopmental disorder. Outcomes where the p-value is below .05 are printed in BOLD.
Across individuals
Scoring higher on low registration, sensory sensitivity, and sensation avoiding was associated with reduced adaptive functioning across individuals (p-values < 0.0125, see Table 3). In all four models, ASD diagnosis was associated with reduced adaptive functioning, while higher IQ and older age were associated with higher adaptive functioning (all p-values < 0.001). Sex and other NDDs did not predict adaptive functioning significantly (all p-values > 0.0125). Sensation seeking was the only AASP quadrant showing an interaction with ASD diagnosis on adaptive functioning (p < 0.0125; see Table 4). When analyzing individuals with and without ASD separately in a follow-up analysis, greater sensation seeking was negatively associated with adaptive functioning in individuals with ASD (b = –0.64, uncorrected p = 0.03). In individuals without ASD (i.e. individuals with or without other NDDs), the estimate was positive instead but not significant (b = 0.22, p = 0.13; see Figure 1).
Association between general adaptive functioning and atypical sensory processing.
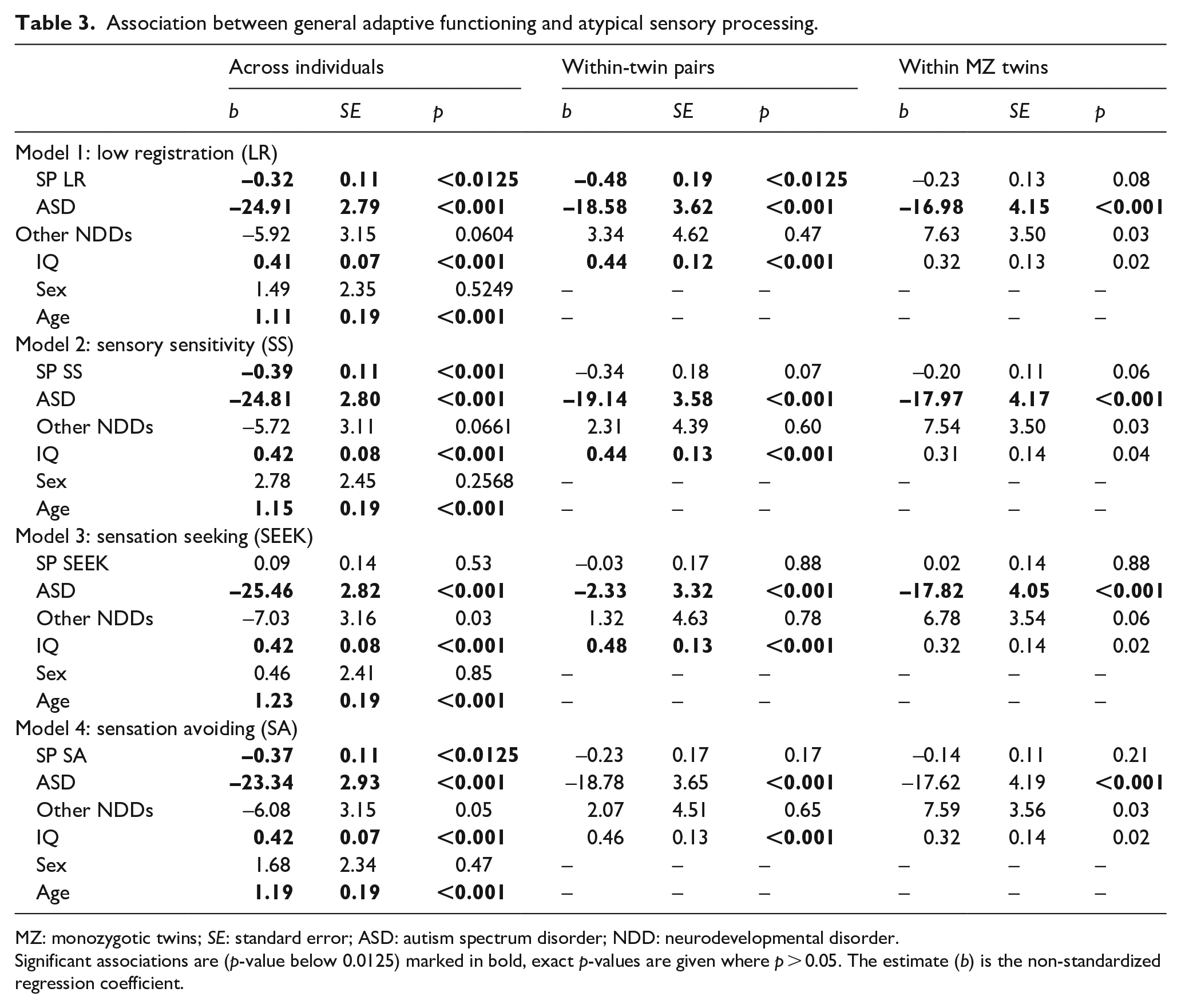
MZ: monozygotic twins; SE: standard error; ASD: autism spectrum disorder; NDD: neurodevelopmental disorder.
Significant associations are (p-value below 0.0125) marked in bold, exact p-values are given where p > 0.05. The estimate (b) is the non-standardized regression coefficient.
Results with interaction between ASD diagnosis and atypical sensory processing.
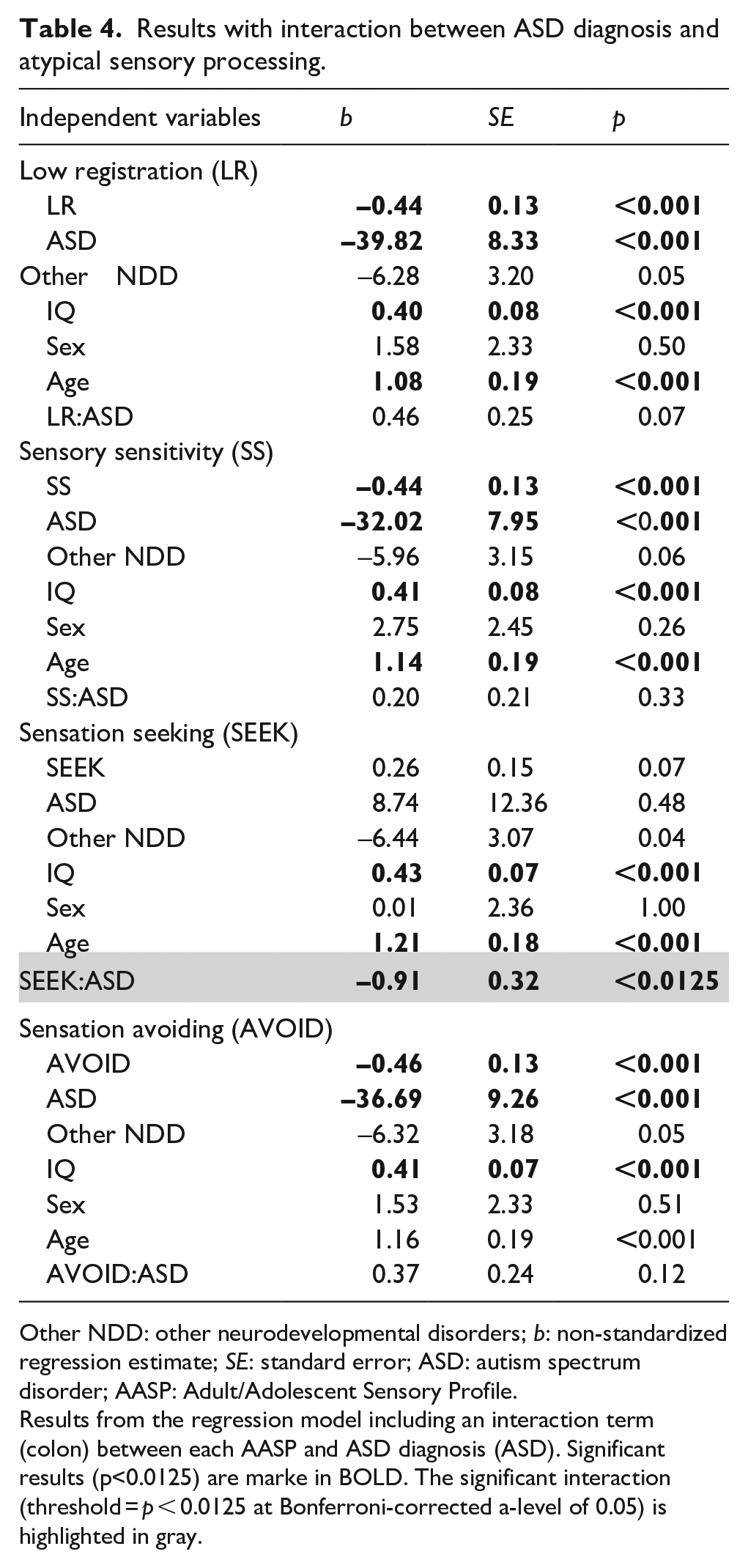
Other NDD: other neurodevelopmental disorders; b: non-standardized regression estimate; SE: standard error; ASD: autism spectrum disorder; AASP: Adult/Adolescent Sensory Profile.
Results from the regression model including an interaction term (colon) between each AASP and ASD diagnosis (ASD). Significant results (p<0.0125) are marke in BOLD. The significant interaction (threshold = p < 0.0125 at Bonferroni-corrected a-level of 0.05) is highlighted in gray.
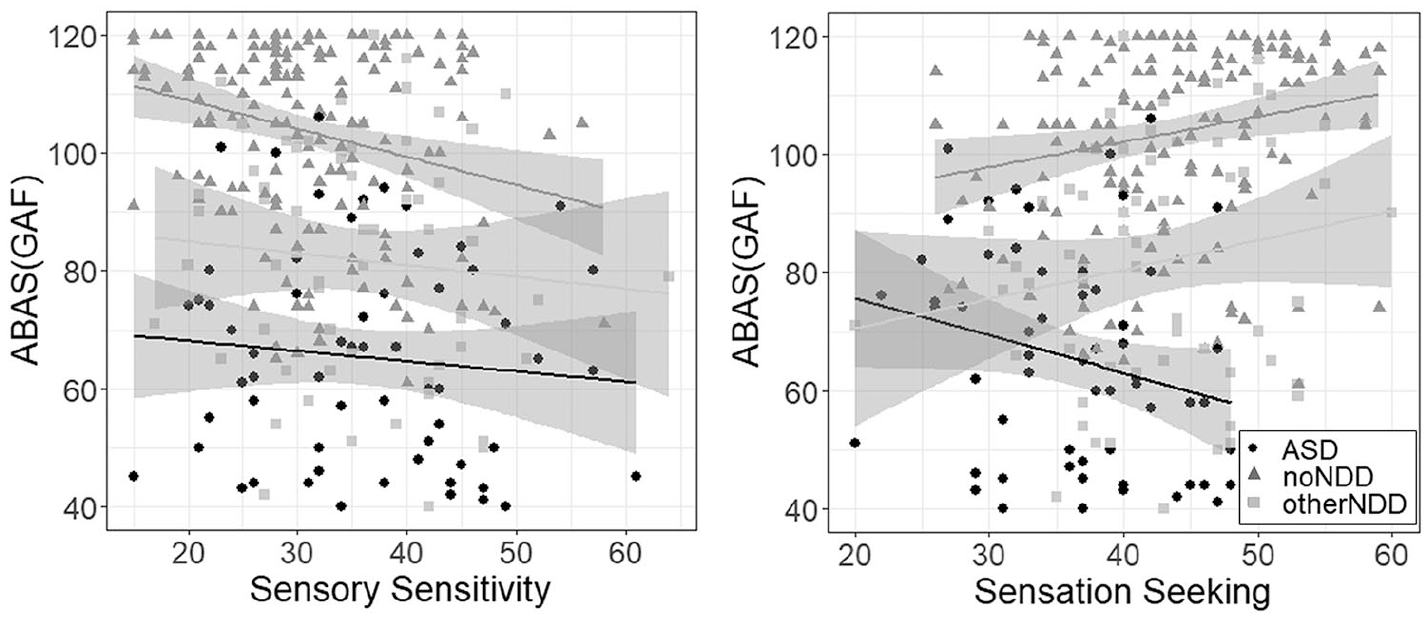
Group-specific associations between SPAA subscales and adaptive functioning. Left: Sensory sensitivity (and similarly also low registration and sensation avoiding) is negatively associated with adaptive functioning within all three groups. Right: In contrast, sensation seeking shows a negative association with adaptive function in participants with ASD diagnosis (black dots) only, while participants with Other NDD diagnosis (light gray squares) or No NDD diagnosis (gray triangles) show a positive relationship.
Furthermore, follow-up analyses revealed that sensory sensitivity was similarly associated with all domains of adaptive functioning (b = −0.29 to −0.36, all p-values < 0.001). In contrast, low registration and sensation avoiding were associated with the social (b = −0.35, p < 0.0125; b = –0.29, p < 0.001) and conceptual domains (b = −0.33, p < 0.0125; b = –0.52, p < 0.001), but their association with the practical domain did not survive the correction (b = 0.25; p = 0.03; b = –0.21, p = 0.06; see Table 5).
Results from follow-up analyses with the three sub-domains of adaptive functioning.
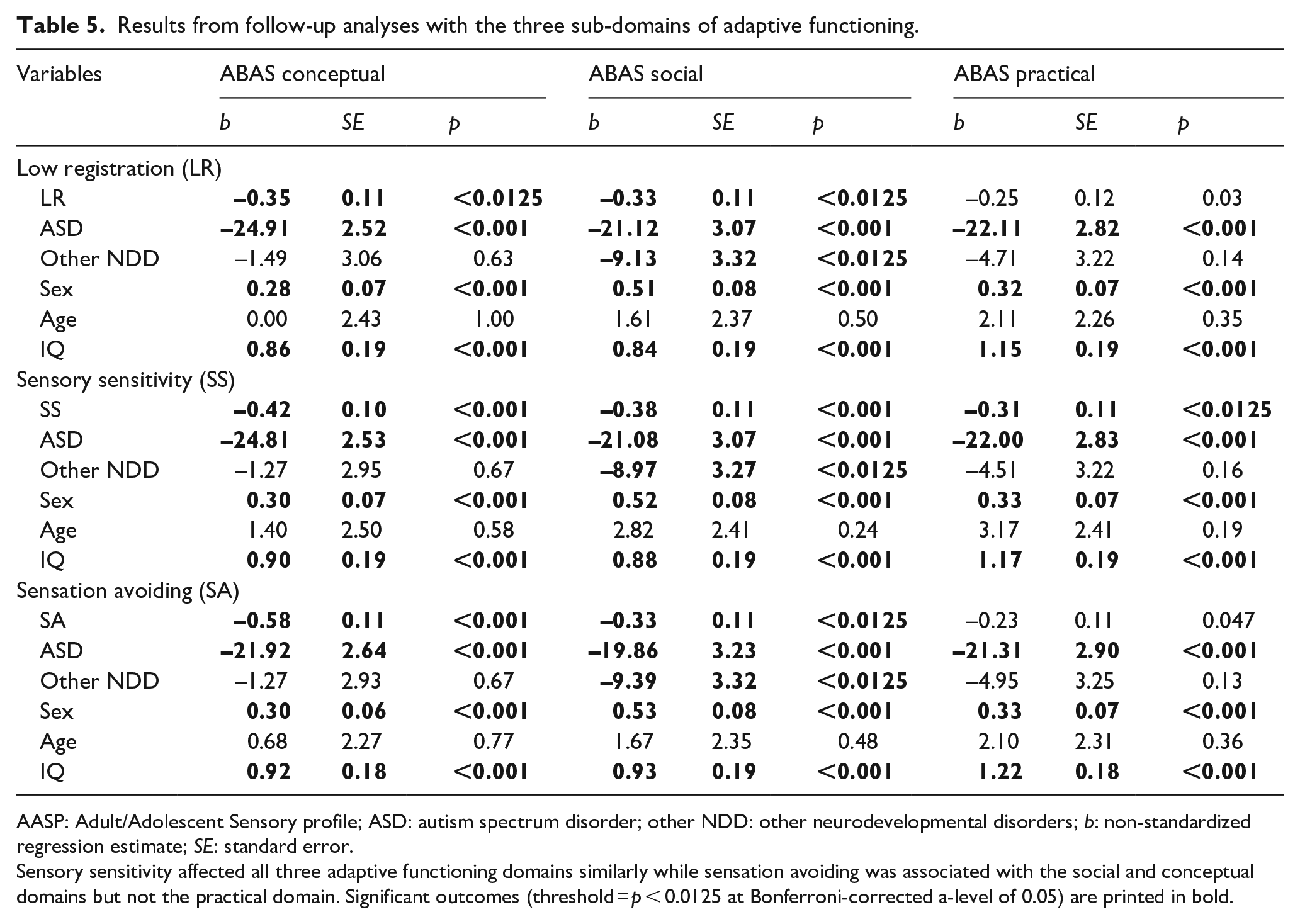
AASP: Adult/Adolescent Sensory profile; ASD: autism spectrum disorder; other NDD: other neurodevelopmental disorders; b: non-standardized regression estimate; SE: standard error.
Sensory sensitivity affected all three adaptive functioning domains similarly while sensation avoiding was associated with the social and conceptual domains but not the practical domain. Significant outcomes (threshold = p < 0.0125 at Bonferroni-corrected a-level of 0.05) are printed in bold.
Within-twin pars
Within-twin pairs, low registration, ASD diagnosis, and IQ were significantly associated with adaptive functioning. More specifically, twins with higher low registration scores or ASD diagnosis scored lower and twins with higher IQ scored higher than their co-twins on the ABAS GAC (p-value for low registration < 0.0125; p-values for ASD diagnosis and IQ < 0.001; see Table 3). We therefore tested these associations also within MZ twin pairs, where they were similar for ASD diagnosis but attenuated below the significance threshold for low registration and IQ. The within-pair associations between the remaining three AASP sub-domains and adaptive functioning exceeded the significance threshold.
Discussion
Although atypical sensory processing is common in individuals with ASD and forms part of the diagnostic criteria, there is limited knowledge about its specific influence on an individual’s functioning in everyday life. This study addressed this question, by investigating the association between four quadrants of atypical sensory processing on adaptive functioning, controlling for ASD and other NDD diagnoses, general cognitive ability (IQ), sex, and age. In addition, we also conducted within-twin pair analyses where all genetic and environmental factors shared by twins are implicitly controlled. Four main findings emerged: first, increased scores of low registration, sensory sensitivity, and sensation avoiding were associated with reduced adaptive functioning in addition to the effects of ASD diagnosis and IQ. However, the effects of ASD diagnosis and IQ were stronger. Second, sensation seeking was differentially associated with adaptive functioning in individuals with and without ASD, predicting reduced adaptive functioning in individuals with ASD only. Finally, within-twin pairs, where all factors shared by twins are implicitly controlled for, the effects of low registration, ASD diagnosis, and IQ remained, while the associations between the sensory processing quadrants related to hyper-reactivity and adaptive functioning were lost. The association with ASD diagnosis was even similar within MZ twins, where genetic influences are adjusted for. The latter results indicate that the effects of ASD diagnosis are influenced by non-shared environmental factors, while familial factors (shared environment and genetics) influence the association between sensory hyper-reactivity and adaptive functioning.
Our findings of ASD diagnosis and IQ predicting adaptive functioning most strongly are consistent with a multitude of studies showing reduced adaptive functioning in individuals with ASD (Chatham et al., 2018; Harrison & Oakland, 2008; Kanne et al., 2011; Nevill et al., 2017) and studies indicating that IQ is one of the factors that is most strongly associated with adaptive functioning in individuals with ASD (Charman et al., 2011; Kanne et al., 2011; Tillmann et al., 2019).
Our main finding that sensory processing atypicalities related to sensory hyper-responsiveness (sensory sensitivity and sensation avoiding) and sensory hypo-responsiveness (low registration) affect adaptive functioning negatively, in addition to the effects of ASD diagnosis and IQ, are in line with previous studies, showing that sensory hyper-and hypo-responsiveness contributes to obstacles in everyday life for individuals with ASD (Ben-Sasson et al., 2019). For sensory hyper-responsiveness, this includes for instance issues with sleep, school activities, mealtime, participation in leisure activities (Ismael et al., 2018), and self-care skills in general (Jasmin et al., 2009). These findings also correspond to a study where individuals diagnosed with autism described the condition from their perspective (Chamak et al., 2008). All of them mentioned alterations in sensory processing, mostly hyper- and hypo-responsiveness, which many perceived as highly challenging and interfering with their daily living. While sensory sensitivity and sensation avoiding are conceptualized as the passive and active expressions of a low neurological threshold for sensory input (Dunn, 1997), they nevertheless represent different nuances of behaviors related to hyper-responsiveness. An individual with high sensory sensitivity might use sensation avoiding in order to deal with over-stimulation or use coping strategies such as self-soothing or distraction. Sensation avoiding might lead to withdrawal from sensory rich social situations and hence reduce an individual’s opportunities to engage and interact with others and develop age-appropriate adaptive functioning skills. Consistent with this view, individuals with ASD in our sample had lower adaptive functioning and higher sensation avoiding than individuals with other NDDs, while these groups did not differ in age or IQ. Furthermore, results from previous subgrouping analyses suggest that those subgroups characterized by the highest severity of sensory atypicalities were also most impaired in adaptive functioning (Ausderau et al., 2016; Tillmann et al., 2020). Our results contradict results of a recent study on a large sample of individuals diagnosed with ASD, where sensory processing alterations (assessed as total score from the caregiver-reported Short Sensory Profile) did not influence adaptive functioning above the effects of IQ, age, and social symptoms (Tillmann et al., 2019). Apart from focusing on individuals diagnosed with ASD alone, this previous study also included younger children (from 6 years of age). Furthermore, they tested ASD trait domains (social-communication symptoms and repetitive behaviors) and other psychiatric symptom domains as continuous measures within the same model with atypical sensory processing. In the same study, sensory processing issues did however predict reduced adaptive functioning when covariates were not included. Most importantly, however, the study did not assess the four quadrants of the Sensory Profile separately, while in our study, only three out of four quadrants predicted reduced adaptive functioning and the fourth, sensation seeking, was differentially associated with adaptive functioning in individuals diagnosed with ASD compared to those without this diagnosis.
This inverse association pattern between sensation seeking and adaptive functioning in individuals diagnosed with ASD compared to those without ASD diagnosis indicates that while sensation seeking behaviors might interfere with adaptive functioning in individuals with ASD, this is not the case for individuals without the condition. Previous research rather suggests that sensation seeking in TD individuals predicts higher functioning, such as higher education and more positive affect (Ben-Avi et al., 2012; Engel-Yeger, & Dunn, 2011; Machingura et al., 2019). Although sensation seeking behaviors might be used by individuals with ASD to regulate under-stimulation and to re-inforce sensory stimuli (Cunningham & Schreibman, 2008; Hebert, 2015), they might also lead to socially unacceptable behaviors. For instance, extensive smelling and touching of objects (as it is given as an example for typically autistic sensation seeking in the DSM-5) might be considered inappropriate in most social contexts. It is possible that TD individuals carry out seeking behaviors that are more adapted to the social context.
Finally, effects of low registration, ASD diagnosis, and IQ remained within-twin pairs in our study. The effect of ASD diagnosis remained even within MZ twins where they are almost solely (except for post-twinning genetic mutations) influenced by non-shared environment (i.e. all non-genetic factors that make twins different from one another). In contrast, the effects of low registration and IQ were lost within MZ twins. The effects of the two quadrants of atypical sensory processing related to sensory hyper-responsiveness were not significant within pairs, in contrast to across the cohort. These results indicate that non-shared environment plays a role in the association between reduced adaptive functioning and ASD diagnosis, while familial factors seem to play a role in the association between hyper-responsiveness and adaptive functioning. Non-shared environmental factors can be everything from differing conditions in utero and birth complications, medical conditions affecting one twin more than the other, to differences in the relationship with the parents or social relationships. Familial factors include both shared environment and genetics. Shared environment comprises for instance socioeconomic background, parental age, parental style, and maternal health conditions during pregnancy. A genetic contribution to the association between atypical sensory processing and adaptive functioning would correspond to findings from twin and family studies showing that atypical sensory processing in ASD is in itself highly genetic (De la Marche et al., 2012; Donaldson et al., 2017; Glod et al., 2017; Taylor et al., 2018; Uljarević et al., 2014).
Implications and future directions
Our results indicate that atypical sensory processing (both hyper-and hypo-responsiveness) contributes to deficits in everyday functioning. Hence, early screening and interventions focusing on sensory issues could help to improve adaptive functioning in people with ASD or other NDDs. Helping individuals find strategies to deal with these symptoms could attenuate their negative impact on their quality of life. Based on our and previous results underlining the importance of familial factors in atypical sensory processing in ASD (especially for the hyper-responsiveness domain), non-diagnosed siblings are likely to share some of the sensory-related difficulties and should hence be included in the assessment of such symptoms and according treatment. Experiencing hyper-responsiveness toward stimuli across diverse contexts may result in the individual withdrawing from environments that are over-stimulating, thereby restricting opportunities for learning age-appropriate adaptive functioning skills. Adjustments of work and school environments to reduce noise, smells, and other sensory inputs would therefore likely be beneficial for a wide range of individuals.
Limitations
Since executive problems such as deficits in self-monitoring, attentional shifting, or inhibition are common in ASD and predict adaptive functioning (Pugliese et al., 2016), future studies should contrast them against sensory issues. Since our study sample was selected specifically for discordance in autistic traits, the results across individuals might not be fully generalizable to the general population. For the same reason, our sample is not suitable for quantitative genetic analyses and likely leads to an over-estimation of non-shared environmental effects for ASD diagnosis. Furthermore, our results might not be generalizable cross-culturally and the impact of sensory issues on adaptive functioning might be more pronounced in societies that offer less support to individuals with ASD than the Swedish society, which allows to some extent for adapted schooling and working conditions as well as domestic assistance. Generally, it needs to be noted that self-report measures of atypical sensory processing have been criticized to focus partly on personal sensory preferences rather than sensory processing per se (Elwin et al., 2013). Objective measures of atypical sensory processing (e.g. sensory threshold tests) and alternative adaptive functioning measures (Henninger & Taylor, 2013) might further nuance the picture of the relationship between atypical sensory processing and adaptive functioning in ASD. Because low IQ might have influenced participants’ ability to complete the AASP, IQ was included as covariate, and in addition, we repeated the analyses restricting the sample to individuals with an IQ score of at least 85 (see Supplementary Table 4). Furthermore, since the Swedish version of the ABAS-II only provides age norms for up to 21 years of age, 74 individuals who were older than 21 were coded according to norms of the age group 17–21 years. Hence, we included age as a covariate and re-run the analyses after excluding these individuals (see Supplementary Table 5). In both these additional analyses, most of the main findings remained largely the same, suggesting that they were not driven by individuals with low cognitive abilities or lacking age-specific norms. However, the age effect, which was significant in the whole sample, was lost in the sample below 22 years and in the sample where the IQ was 85 or higher, IQ was not significant any longer. Furthermore, the within-pair association with low registration was also lost in both these analyses and should hence be interpreted with caution. Finally, our sample likely lacked the power to detect small sized within-pair effects while including several covariates.
Conclusion
In sum, our findings suggest that atypical sensory processing has a negative impact on adaptive outcomes. These effects were weaker than the influence of diagnosis, age, and cognitive ability but remained significant within the same model. Hence, we conclude that atypical sensory processing might contribute to adaptive behavior challenges, while it doesn’t seem to be the most decisive factor. Furthermore, in contrast to the effect of ASD which remained within MZ twins, the effects of sensory hyper-responsiveness on adaptive functioning were lost within twin pairs. This suggests that they were driven by familial factors rather than non-shared environment, indicating an etiologically different relationship to adaptive functioning. More specifically, this might indicate that sensory hyper-reactivity and reduced adaptive functioning share some of their etiological (e.g. genetic) basis, while our findings rather speak against a direct causal relationship. Finally, sensation seeking behaviors might be less compatible with adaptive functioning in individuals with ASD diagnosis than in those without ASD diagnosis.
Supplemental Material
sj-pdf-1-aut-10.1177_13623613211019852 – Supplemental material for The impact of atypical sensory processing on adaptive functioning within and beyond autism: The role of familial factors
Supplemental material, sj-pdf-1-aut-10.1177_13623613211019852 for The impact of atypical sensory processing on adaptive functioning within and beyond autism: The role of familial factors by Janina Neufeld, Lisa Hederos Eriksson, Richard Hammarsten, Karl Lundin Remnélius, Julian Tillmann, Johan Isaksson and Sven Bölte in Autism
Footnotes
Acknowledgements
We would like to thank all participants of the RATSS project as well as our colleagues at the KIND center for their valuable contributions.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare no conflicts of interest with respect to the research. S.B. discloses that he has in the last three years acted as an author, consultant or lecturer for Medicine, Roche, Hogrefe, Kohlhammer and UTB.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Swedish Research Council (no. 2016-01168), “Forskningsrådet för miljö, areella näingar och samhällsbyggande” (FORMAS; no. 259-2012-24), The Swedish Brain Foundation (Hjärnfonden; nos FO2014-0228 and FO2018-0053), Region Stockholm (SLL’s Anslag till forskning, utveckling och utbildning “ALF medicin”; nos 20140134 and 20170016), and Innovative Medical Initiatives (IMI) (no. 115300; EU-AIMS; 2012–2017). We acknowledge The Swedish Twin Registry, which is managed by Karolinska Institutet and receives funding through the Swedish Research Council under the grant no. 2017-00641, for access to data.
Supplemental material
Supplemental material for this article is available online.
Access to additional supplementary material
Those who wish to get access to data, models, or other additional supplementary materials are kindly asked to contact the corresponding author at
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
