Abstract
Atypical learning and memory in early life can promote atypical behaviors in later life. Less relational learning and inflexible retrieval in childhood may enhance restricted and repeated behaviors in patients with autism spectrum disorder. The purpose of this study was to elucidate the mechanisms of atypical memory in children with autism spectrum disorder. We conducted picture–name pair learning and delayed-recognition tests with two groups: one group with high-functioning autism spectrum disorder children (aged 7–16,
Lay abstract
Atypical learning and memory in early life can promote atypical behaviors in later life. Specifically, less relational learning and inflexible retrieval in childhood may enhance restricted and repeated behaviors in patients with autism spectrum disorder. The purpose of this study was to elucidate the mechanisms of atypical memory in children with autism spectrum disorder. We conducted picture–name pair learning and delayed-recognition tests with two groups of youths: one group with high-functioning autism spectrum disorder children (aged 7–16,
Introduction
Reduced cognitive flexibility in autism spectrum disorder (ASD) is characterized by restricted interests and repetitive behaviors (Lopez et al., 2005). Some of the inflexible behaviors in patients with ASD may be linked to their atypical memory. Atypical verbal learning strategies (Bowler et al., 2009), less use of strategy both in learning and retrieval, and impairments in detailed retrieval have been demonstrated in ASD (Gaigg et al., 2015; Wojcik et al., 2018). The greater frequency of traumatic memories in ASD youths may be related to their atypical learning and retrieval which form less organized memories (Rumball, 2019). Over-specificity in perceptual learning (Harris et al., 2015), difficulties in reversal learning (D’Cruz et al., 2013; South et al., 2012), and fewer false memories (Beversdorf et al., 2000; Hillier et al., 2007; Wojcik et al., 2018) suggest atypical, inflexible, and rigid memory in individuals with ASD.
Moreover, Boucher has reported uneven cognitive profiles in ASD, such as simultaneous strength and weakness in the domain of memory (Boucher et al., 2012). For instance, Ring and colleagues (2016) have identified intact item memory and impaired relational memory in adults with ASD. Item memory refers to studying each item in isolation, while relational memory refers to studying associations among a list of items. Although both relational and item-specific encoding enhance correct retrieval, the relational process can develop false memories by combining information across items or events (Huff & Bodner, 2019), which can then be retrieved and reconstructed in different combinations. Distortion is a characteristic of human memory, and these memory errors can be adaptive for memory updating and flexible behaviors (Loftus, 2003; Schacter, 2007; Schacter et al., 2011). By flexibly reconstructing elements of past experiences, we can simulate both likely and unlikely futures and imagine alternative versions of past experiences (Schacter et al., 2015). Conversely, too rigid, robust, and undistorted memory may restrict the modification of thinking and behavior. Children with ASD have exhibited intact source memory but reduced integration of source memory and thinking (Naito et al., 2020). Strong memory for isolated items and weak memory for associated items may relate to the rigid and inflexible memory in ASD.
In typically developing (TD) children, simple binding and associative memory develop earlier, whereas strategic and control processes for relational memory develop later (Shing et al., 2010). Recognition memory for isolated/individual items appears to develop at an early age (4–5 years) (Olson & Newcombe, 2014). The accuracy of 8- to 9-year-olds’ item recognition for visual objects is comparable to that of adults, while fact (knowledge) recall develops linearly between ages 4 and 10 (Riggins, 2014; Rollins & Cloude, 2018). Detailed relational memory develops slowly after age 6 (Lee et al., 2016; Ngo et al., 2019). In addition, both correct and false recognition responses increase with age, and correct recognition is associated with activity in the hippocampus (Paz-Alonso et al., 2008). TD children can use the hippocampal network for successful memory (Ngo et al., 2017; Ofen, 2012; Tang et al., 2018), and hippocampal maturation in the anterior–posterior axis may support successful memory (Demaster & Ghetti, 2013). Encoding-retrieval differentiation in the anterior–posterior axis of the hippocampus has been observed in children (Langnes et al., 2019). As such, altered neural connectivity of the hippocampus may provide information on atypical learning and memory in children with ASD.
Neuroimaging studies have suggested that ASD is associated with altered neural connectivity (Di Martino et al., 2009; Hahamy et al., 2015; Just et al., 2012) and globally weaker resting-state functional connectivity (FC) in children (Yerys et al., 2017). Atypical resting-state FC in ASD children may be associated with their inflexible (i.e. restricted and repetitive) behaviors (Uddin et al., 2013, 2015). Recent studies with healthy adults suggest that resting-state FC can predict memory performance (Dresler et al., 2017; Fjell et al., 2016), and that transfer of learning is related to FC (Gerraty et al., 2014). Altered task-related connectivity in ASD adults may be associated with learning (Schipul et al., 2012; Schipul & Just, 2016). Some studies have suggested that an atypical prefrontal-hippocampus and posterior parietal-hippocampus network may be involved in ASD-related memory deficits (Ben Shalom, 2003; Boucher & Mayes, 2012; Cooper et al., 2017; Solomon et al., 2015). Right–left hippocampal growth differences in healthy children and atypical development in children with ASD have been reported (Reinhardt et al., 2020). However, the neural mechanisms of memory in children with ASD have not yet been well elucidated.
The purpose of this study was to reveal the neural underpinnings of atypical memory in ASD youths that relate to their inflexible behaviors. We used picture–name learning (immediate-recognition) and delayed-recognition tests to measure memory performance in both ASD and TD children. To assess the inflexible characteristics of memory in ASD, we assessed the constancy of memory by classifying delayed-recognition performance based on the immediate-recognition performance for each item. We used successful memory performance (i.e. consecutive correct responses both in immediate and delayed-recognition tests) as the index of memory rigidness, and we also classified gains and distortions in the delayed-recognition test that suggested less memory rigidness. To better understand the characteristics of memory in ASD, we examined the correlations between successful memory performance and resting-state FC in both ASD and TD youths.
Methods
Participants
Participants consisted of 41 ASD (29 boys and 12 girls) and 82 TD (58 boys and 24 girls) Japanese children with full-scale IQ (FSIQ) ≧ 70, aged 7 to 16. Originally, 47 ASD and 92 TD children participated; however, due to head movement in some of the imaging data (deviating from the criteria described in section “Analyses of resting-state FC data”), we only analyzed data from 41 ASD and 82 TD participants. Handedness was determined using the Edinburgh Handedness Inventory. Our assessment of socioeconomic status (SES) consisted of inquiries related to family annual income (seven variables), and the educational qualification of both parents (average of both parents) was measured. Their mean characteristics are represented in Table 1. We confirmed participants’ clinical ASD diagnosis (based on
Mean participant characteristics.
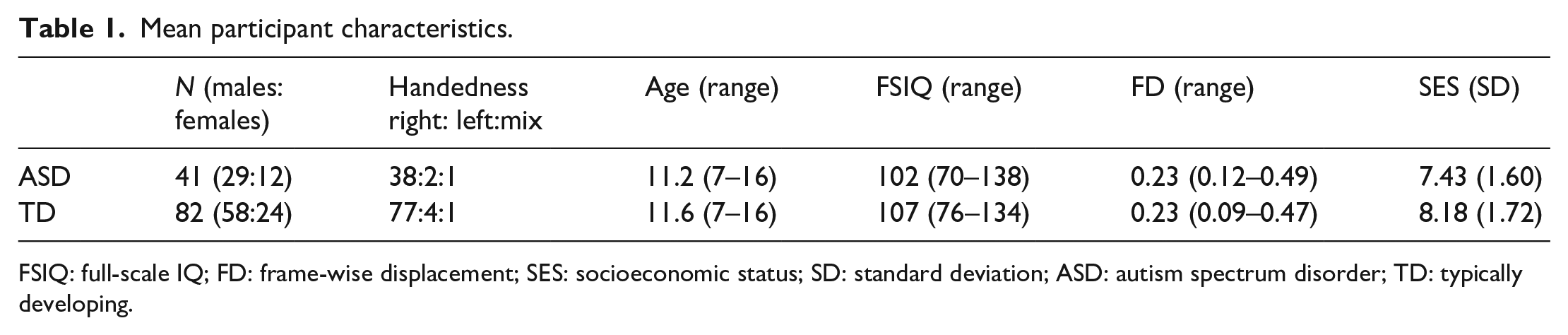
FSIQ: full-scale IQ; FD: frame-wise displacement; SES: socioeconomic status; SD: standard deviation; ASD: autism spectrum disorder; TD: typically developing.
Autistic participants were recruited through (1) authorized non-profit organizations for people with developmental disorders and (2) a newspaper advertisement. TD participants were recruited through a newspaper advertisement. Written informed consent was obtained from each subject’s parent in accordance with the Declaration of Helsinki. This study was approved by the Ethics Committee of Tohoku University.
Memory
We used the Japanese version of the Atlantis picture–name learning test (Maruzen Publishing Co., Ltd, Tokyo, Japan) of the Kaufman Assessment Battery for Children, Second Edition (KABC-II; Kaufman & Kaufman, 2004). This test measures one’s ability to learn new information, and it consists of immediate- and delayed-recognition tests (Figure 1). This test was conducted outside the magnetic resonance imaging (MRI) scanner.
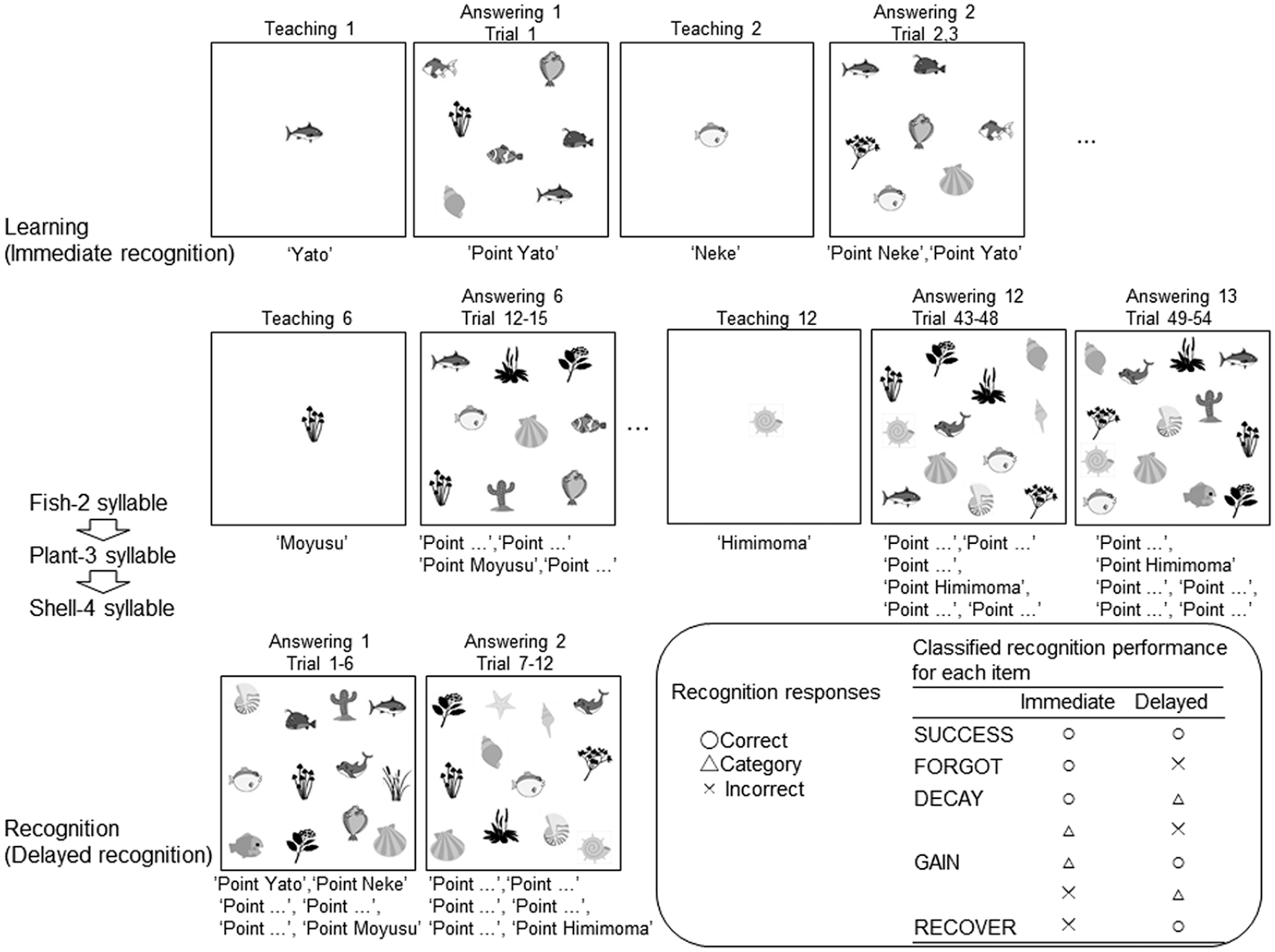
Learning and recognition tests. In the learning test, the experimenter displayed each picture on the teaching card for the participants, instructing corresponding nonsense names. In the following answering card, the experimenter read the nonsense name aloud and instructed participants to point to the correct picture. Relational information of category syllables (fish: 2, plant: 3, and shell: 4) was available. In the recognition test, the experimenter read the nonsense name aloud and instructed participants to point to the correct picture. Recognition responses both in the learning and recognition tests were defined as correct, category correct, and incorrect. The last trial of immediate recognition was used for classified recognition performance (bottom right).
Learning test (immediate-recognition)
Pictures were organized into three categories of fanciful cartoons, including four fish, four plants, and four shells. Fish had two-syllable names, plants had three-syllable names, and shells had four-syllable names. Category-syllable number relation could enhance memory and also induce false recognition within each category. The experimenter showed participants each picture one by one in a fixed order, teaching them corresponding nonsense names. In total, participants were taught 12 picture–name pairs (fish: 2 syllables, fish: 2 syllables, fish: 2 syllables, fish: 2 syllables, plant: 3 syllables, plant: 3 syllables, plant: 3 syllables, plant: 3 syllables, shell: 4 syllables, shell: 4 syllables, shell: 4 syllables, and shell: 4 syllables). Each picture was presented on a teaching card and displayed on an easel (like a picture–card show) for approximately 2 s, after which the teaching card was turned to show an answering card featuring an array of pictures for immediate recognition. The experimenter read the nonsense name aloud and instructed participants to point to the correct picture on the answering card. Twelve teaching cards and 13 answering (pointing) cards were used. Seven to 13 pictures, including no-name pictures (three fish, one plant, and one shell), were presented on the answering cards. Each picture was used repeatedly on the answering cards; 6 to 8 times for fish, 3 to 5 times for plants, and 2 or 3 times for shells. For the first answering card, participants were asked to point to one picture from an array (one trial). For the second card, participants were asked to point to two pictures (two trials). From the 3rd to 13th answering cards, participants were asked to point to two, three, three, four, five, four, five, seven, six, six, and six pictures from the respective arrays. There were 54 learning (immediate recognition) trials in total, of which two involved pointing to a no-name picture. Pointing to the correct picture was defined as correct learning. Pointing to an incorrect picture in the correct picture category (fish/plants/shells) was defined as category learning. For each incorrect answer, the experimenter provided immediate feedback and the correct answer, except in the case of the two no-name pictures and the final (13th) answering card. The maximum possible score for both correct learning and category learning was 54.
A response (i.e. correct learning, category learning, and incorrect learning) in the final learning trial for each picture on the 11th to 13th cards was used to categorize memory performance, in detail, as shown below. A feedback for category learning and incorrect learning was given for seven pictures on the 11th and 12th cards, while no feedback was given for five pictures on the 13th card. For the final learning trials of 12 pictures, a chance level of the correct learning was 0.08 to 0.09 (1/13 to 1/11) and that of category learning was 0.36 to 0.38 (5/13 to 5/11).
Recognition test (delayed-recognition)
During the delay period, participants completed a questionnaire about their relationship with their parents, which consisted of 120 items. Approximately 17 min after the learning test, we conducted a delayed-recognition test using 12 picture–name pairs and a similar procedure as the final (13th) answering card of the learning test. Two answering cards were used featuring a total of 12 pictures, including 11 old pictures (shown in the learning test) and 1 new picture. The order of the 12 picture-pointing prompts was identical to that of the learning test. Pointing to the correct picture was defined as correct recognition. Pointing to an incorrect picture in the correct picture category was defined as category recognition. The maximum possible score for both correct recognition and category recognition was 12. A chance level for correct recognition was 0.08 (1/12) and that for category recognition was 0.33 or 0.5 (4/12 to 6/12).
To assess memory in detail, we classified recognition performance into five categories according to the participant’s response in the final learning trial for each picture: SUCCESS, FORGOT, RECOVERY, DECAY, and GAIN (Figure 1, bottom right). SUCCESS was defined as correct responses in both learning and recognition (i.e. consecutive correct responses both in immediate and delayed-recognition tests), and it reflected the robustness or stability of memory. SUCCESS was the index of interest in this study, as it demonstrates the rigidness of memory in children. FORGOT was defined as correct learning with incorrect recognition. DECAY was defined as correct/category learning with subsequent category/incorrect recognition. GAIN was category/incorrect learning with correct/category recognition. RECOVERY was incorrect learning but correct recognition. The inconsistency between immediate and delayed recognitions suggests lower memory rigidness. Incorrect responses, both in learning and recognition, were not categorized because they left no memory traces. The maximum possible score for each classified recognition performance was 12. Ultimately, we analyzed only the correlations between SUCCESS scores and resting-state FC strength. Due to the small number of responses and lack of variance in the other four indices, they were not used for correlation analyses.
Effects of age on memory
Correlations (Pearson’s correlation coefficients) between age and memory performances were calculated to examine the effects of age on memory in both groups (ASD and TD). Group differences in each memory performance were tested using a Fisher
Image acquisition
All images were acquired using a Philips Intera Achieva 3.0T scanner. For the resting-state functional magnetic resonance imaging (fMRI), 34 trans-axial gradient-echo images (64 × 64 matrix, repetition time (TR) = 2000 ms, echo time (TE) = 30 ms, field of view (FOV) = 24 cm, 3.75 mm slice thickness, voxel size = 3.75 × 3.75 × 3.75 mm3) covering the entire brain were acquired using an echo planar sequence. For this scan, 160 functional volumes (scan duration of 5 min and 20 s) were obtained while subjects were resting (opening their eyes and not moving, not sleeping, and not thinking about anything). Three-dimensional T1-weighted images were collected using a magnetization-prepared rapid gradient-echo (MPRAGE) sequence (240 × 240 matrix, TR = 6.5 ms, TE = 3 ms, inversion time (TI) = 711 ms, FOV = 24 cm, 162 slices, 1.0 mm slice thickness, voxel size = 1.0 × 1.0 × 1.0 mm3, scan duration of 8 min and 3 s).
Analyses of resting-state FC data
The data processing method was generally consistent with that used in our previous study for children with developmental dyslexia (Hashimoto et al., 2020). We performed the MRI data preprocessing and analysis using Statistical Parametric Mapping (SPM12) software (Wellcome Department of Cognitive Neurology, London, UK). Resting-state FC (signal synchrony among remote brain areas) was computed using simple correlations between spontaneous activation levels in multiple brain areas. We did not discard any initial volumes because the MRI scanner automatically discards initial volumes with a non-steady state. Prior to preprocessing, we applied the ArtRepair toolbox implemented in SPM12 to repair spike noise in slices through interpolation from the before and after scans. We used the Data Processing Assistant for Resting-State fMRI (DPARSF, http://rfmri.org/DPARSF) to preprocess the time series volume of each session per participant. This included realignment to the first volume, slice timing correction, T1 image coregistration to fMRI data, segmentation of T1 image with a diffeomorphic anatomical registration through an exponentiated lie (DARTEL) algebraic registration process, normalization to the Montreal Neurological Institute (MNI) space by DARTEL, spatial smoothing (6 mm full-width half-maximum), detrending, and temporal filtering (0.01–0.1 Hz). After spatial smoothing (and before detrending), we used the ArtRepair toolbox to detect and repair bad volumes through interpolation. The criteria for bad volumes were (1) a 1.5% variation in the global signal intensity, and (2) excessive scan-to-scan motion, defined as 0.5 mm frame-wise displacement (FD). In addition, we used the Friston-24 model to regress out nuisance covariates, including six head motion parameters, six head motion parameters one time point before, and the 12 corresponding squared items. The global mean signal was not regressed because global signal regression removes true neuronal signals and can diminish connectivity–behavior relationship (Murphy & Fox, 2017). White matter and cerebrospinal fluid (CSF) signals were regressed out to reduce head motion effects using an anatomical, component-based, noise-correction method (Behzadi et al., 2007; Muschelli et al., 2014) with T1 segment masks and the top 5 principal components. We used lenient exclusion criteria of mean FD > 0.5 mm (Power et al., 2014) to account for the excessive head movement of children. Based on these criteria, we excluded data from 6 of the 47 ASD participants and 9 of the 93 TD participants.
We selected four regions of interest (ROIs) from a resting-state FC study in healthy adults (Wagner et al., 2016). The left anterior hippocampus (MNI coordinates of −28, −12, −20), left posterior hippocampus (−28, −24, −12), right anterior hippocampus (28, −12, −20), and right posterior hippocampus (32, −22, −12)—all with a 6 mm radius sphere—were defined as seed ROIs. The anterior–posterior hippocampal segmentation was consistent with another study examining memory-related activation in a wide age range (6.8–80.8 years) with MNI coordinates of
For the group comparison of correlations between SUCCESS (correct learning and retrieval) and FC, we performed an analysis of variance (ANOVA) with covariates of SUCCESS score, age, sex, FSIQ, and FD. We also performed multiple regression analyses for each (ASD and TD) participant using correlation coefficients of FC and SUCCESS, taking age, sex, FSIQ, and FD as covariates. We applied a statistical threshold for family-wise error (FWE) of
Community involvement
There is no community involved in this study.
Results
Participant characteristics
As shown in Table 1, there were no significant group differences in age (two-sample
Learning and recognition performance
Participants with ASD had similar scores to those with TD in both correct learning and correct recognition, but had slightly lower scores than participants with TD in category learning (Figure 2). We observed no significant group differences in mean number of correct learning (ASD: 41.6, TD: 40.0,
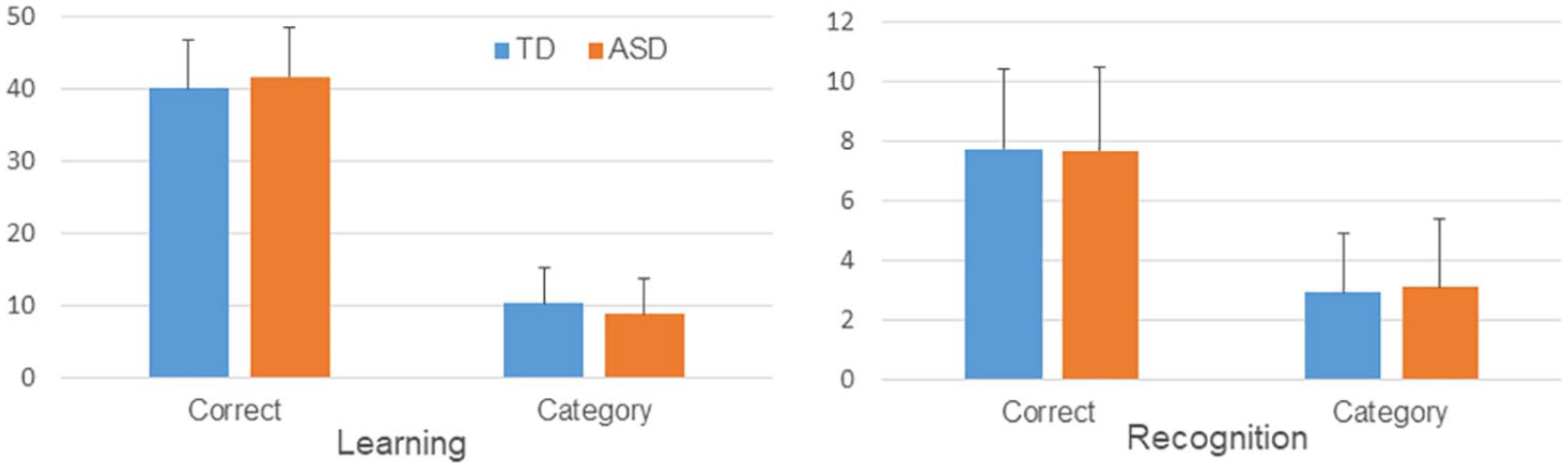
Mean learning and recognition performance of ASD and TD participants. ASD individuals showed marginally fewer category learning responses than did TD individuals. Error bar shows standard error.
Classified memory performance (Figure 3) revealed less GAIN in participants with ASD than those with TD (ASD: 1.76 vs TD: 2.50,
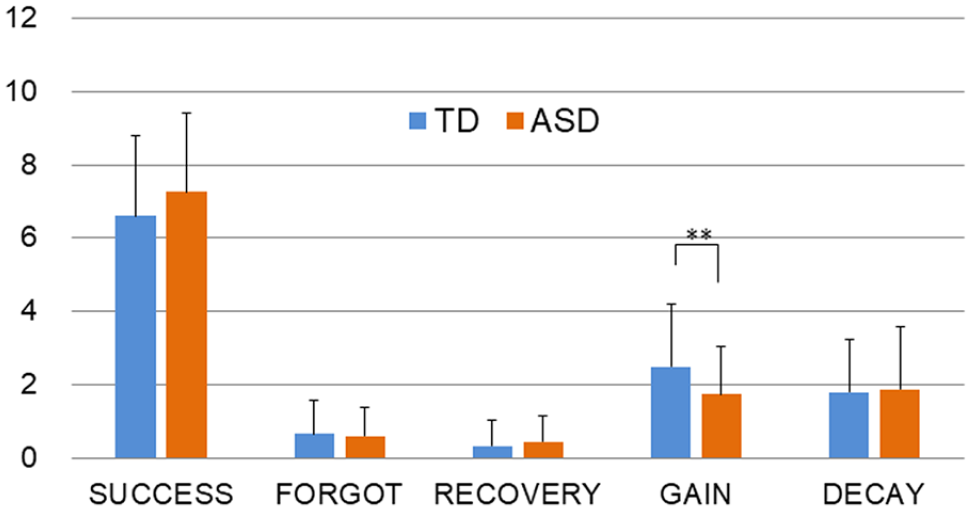
Mean classified recognition scores. Less memory GAIN was observed in ASD than in TD individuals. Error bar shows standard error.
Correlations between age and memory performance are shown in Table 2. Age was positively but weakly correlated (
Correlations (
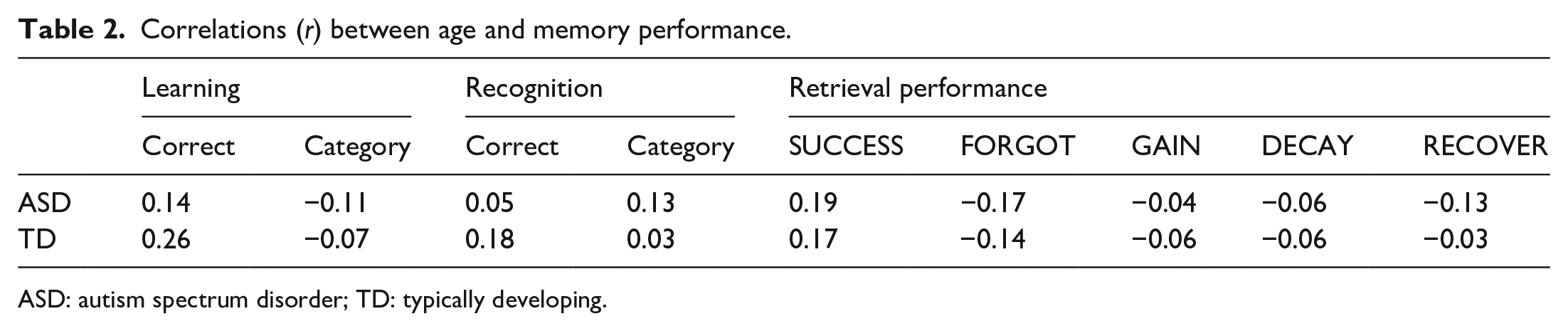
ASD: autism spectrum disorder; TD: typically developing.
Group differences in correlations between resting-state FC and successful retrieval scores
TD children showed greater correlations between resting-state FC and SUCCESS than did ASD children in the left hippocampal networks (Figure 4). Those networks were as follows: the left anterior hippocampus ROI and the left inferior frontal gyrus, the left anterior hippocampus ROI and the left dorsolateral prefrontal cortex, the left anterior hippocampus ROI and posterior cingulate cortex, and the left posterior hippocampus ROI and medial prefrontal cortex (Table 3). ASD children did not show greater resting-state FC than did TD children in any network.
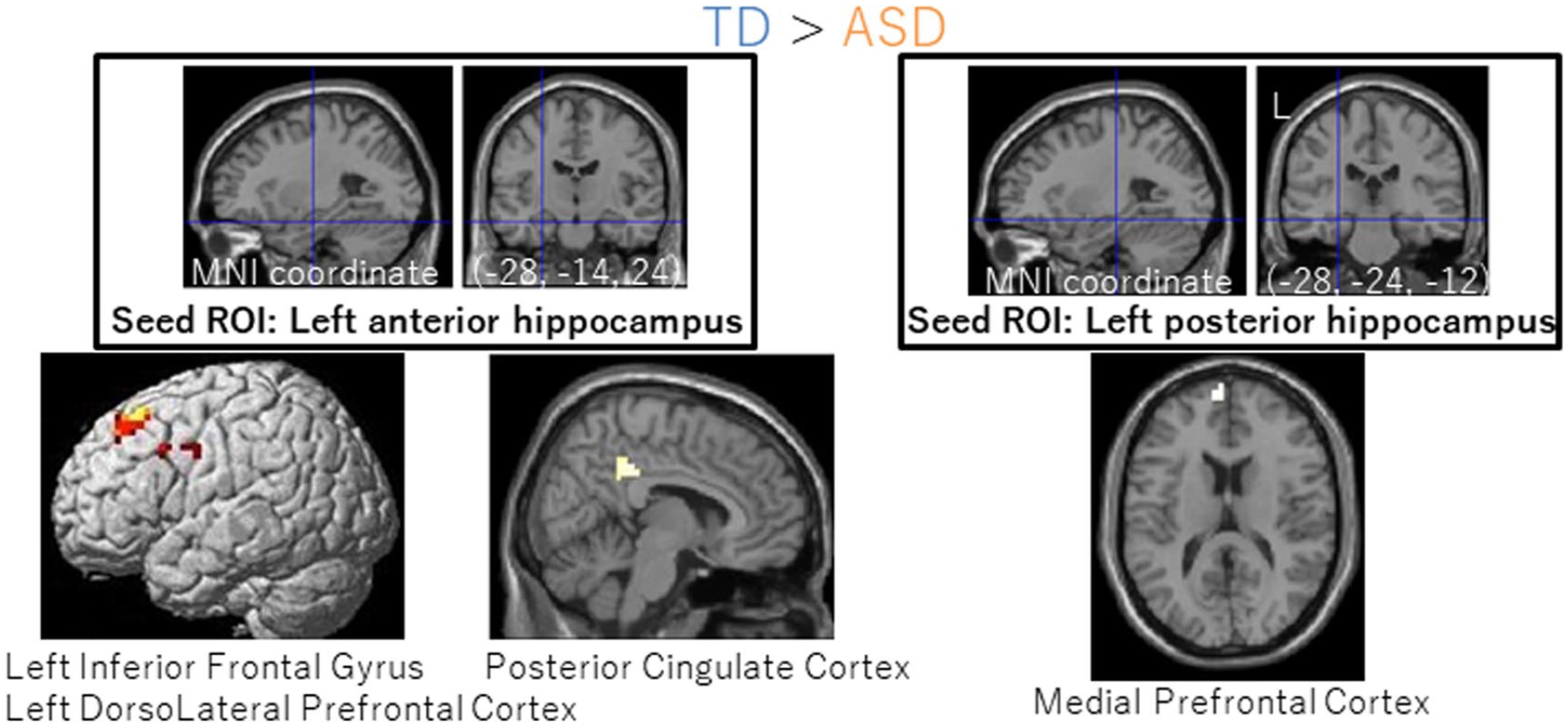
Group differences in correlations between successful memory scores and resting-state functional connectivity. We observed that connectivity was greater in TD than in ASD. This was true between the left anterior hippocampus ROI and the left inferior frontal gyrus/dorsolateral prefrontal cortex (left) and between the left posterior hippocampus ROI and the medial prefrontal cortex (right). Seed ROIs are shown on the top with crosshairs. Clusters survived an FWE-corrected
Hippocampal connectivity showing group differences in correlations with SUCCESS score.
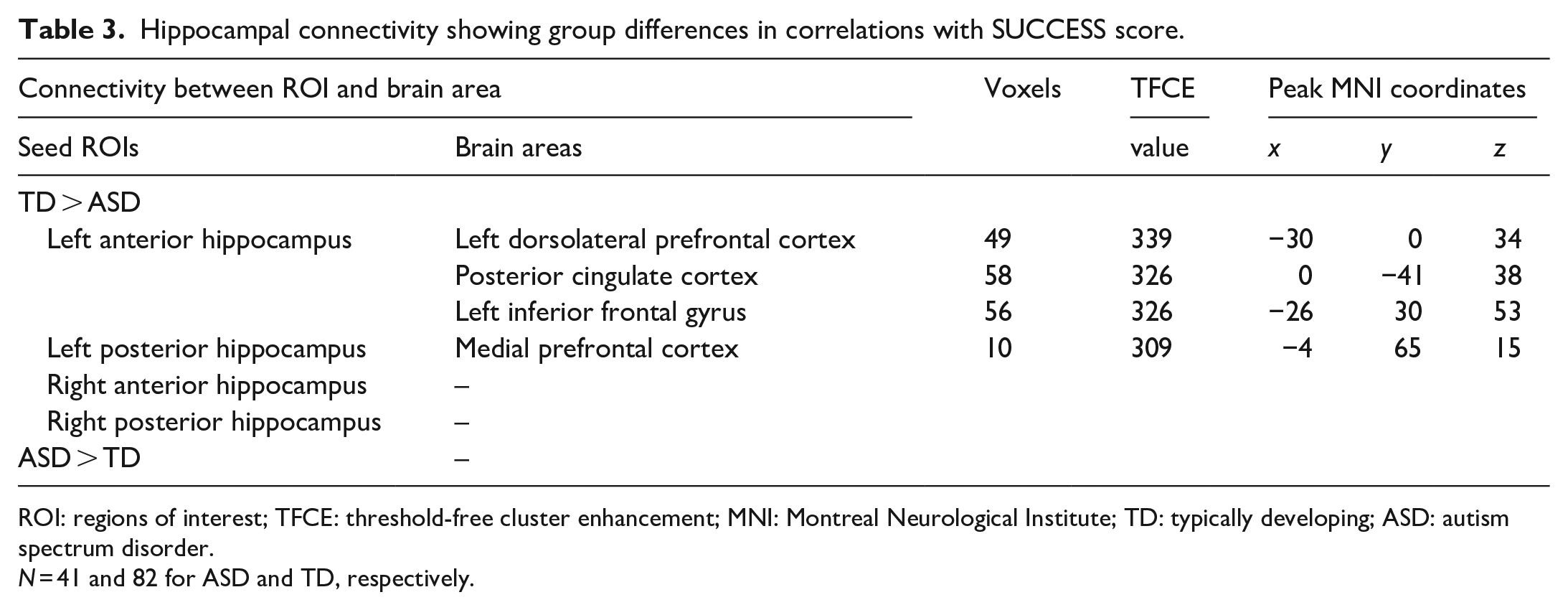
ROI: regions of interest; TFCE: threshold-free cluster enhancement; MNI: Montreal Neurological Institute; TD: typically developing; ASD: autism spectrum disorder.
Correlations between resting-state FC and successful retrieval scores in each group
In TD, the connectivity strength between the right anterior hippocampal ROIs and the left occipitotemporal cortex and between the right anterior hippocampal ROIs and the right dorsolateral prefrontal cortex showed significant positive correlations with successful retrieval scores (Figure 5 and Table 4). Those correlation coefficients (Pearson’s
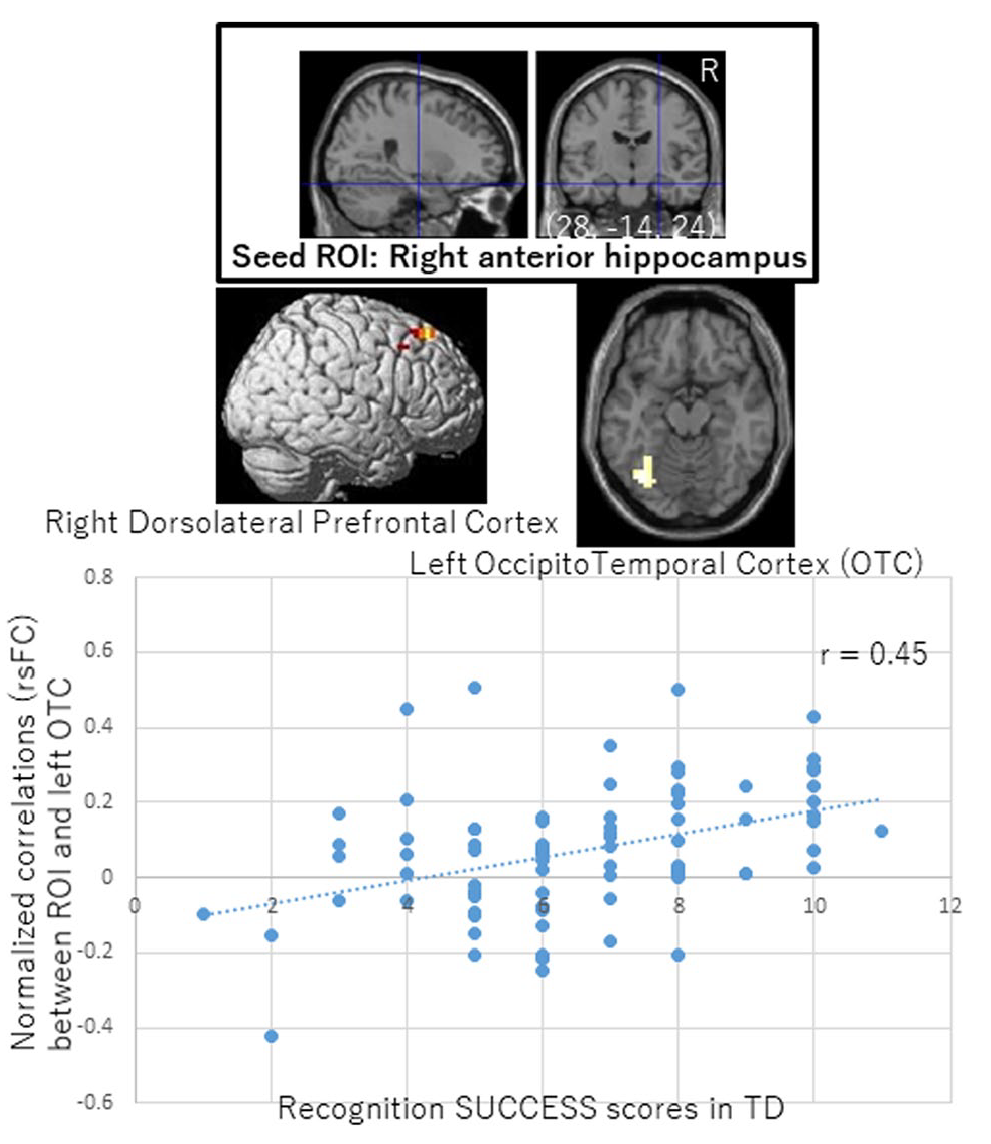
Correlations between successful memory scores and functional connectivity in TD participants. SUCCESS scores in TD were positively associated with resting-state functional connectivity strength between the right anterior hippocampus ROI and the left occipitotemporal cortex/right dorsolateral prefrontal cortex. Seed ROIs are shown at the top with crosshairs. Pearson’s correlation coefficient for the left occipitotemporal cortex is shown. Clusters survived an FWE-corrected
Positive or negative correlations between SUCCESS scores and hippocampal connectivity in each group.
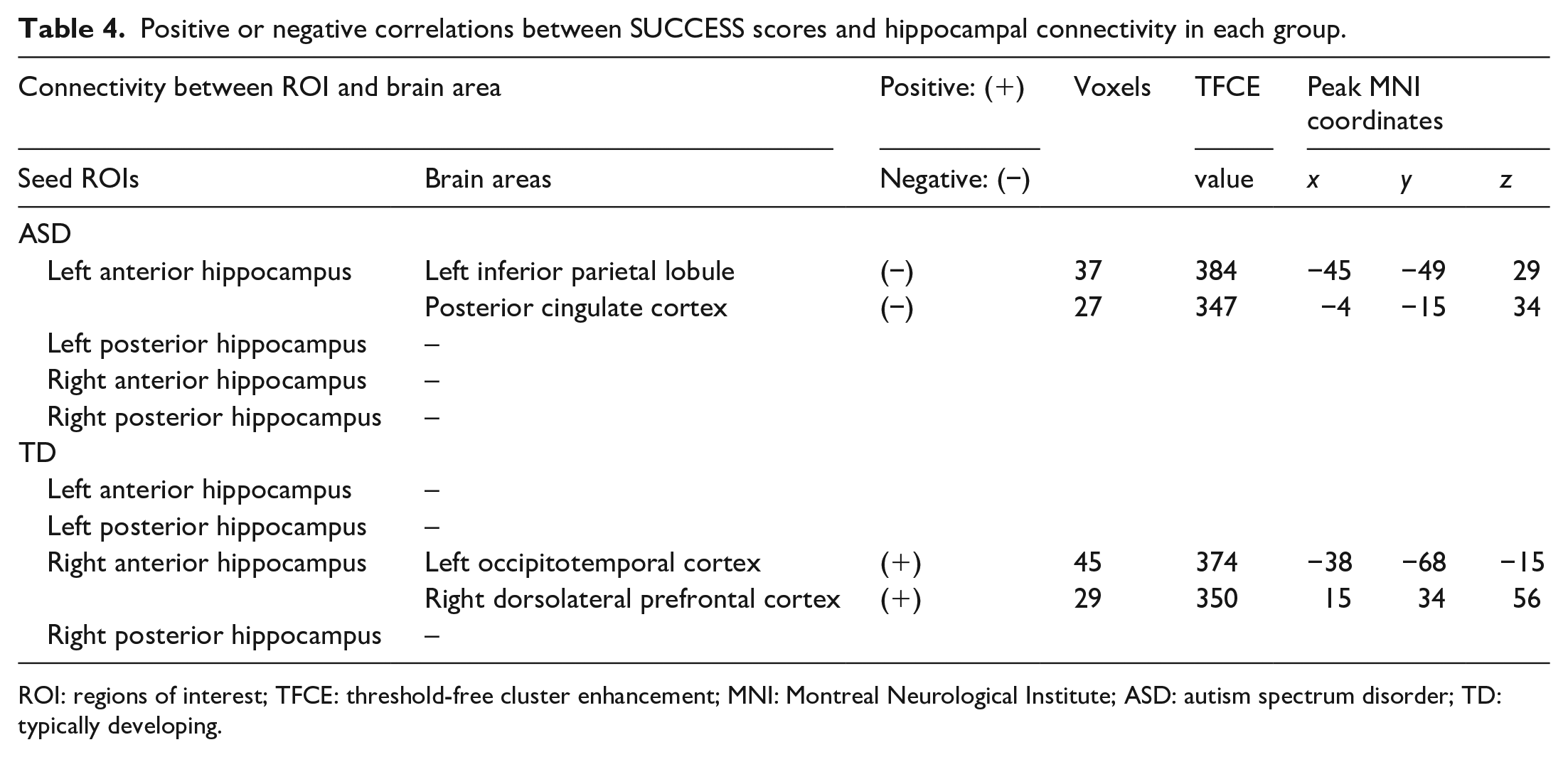
ROI: regions of interest; TFCE: threshold-free cluster enhancement; MNI: Montreal Neurological Institute; ASD: autism spectrum disorder; TD: typically developing.
In ASD, the connectivity strength between the left posterior hippocampus and the left inferior parietal lobule and between the left posterior hippocampus and posterior cingulate cortex showed a significant negative correlation with successful retrieval scores (Figure 6 and Table 4). Those correlation coefficients (Pearson’s
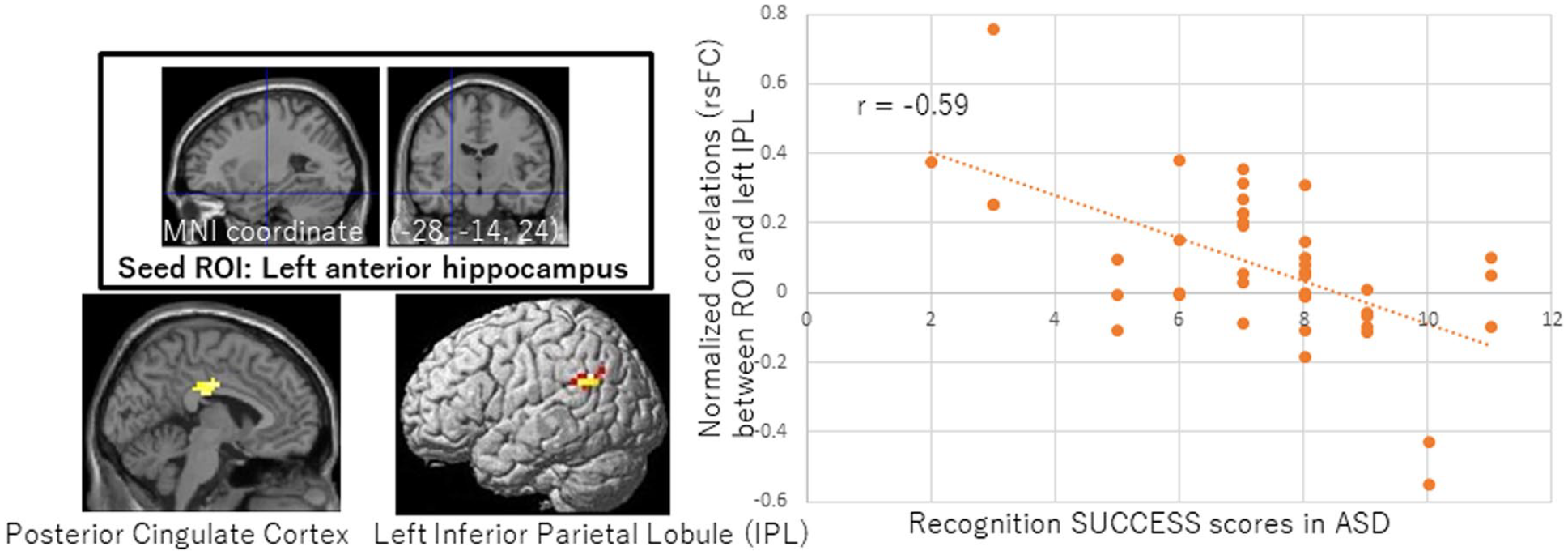
Negative correlations between successful memory scores and functional connectivity in ASD participants. SUCCESS scores in ASD were negatively correlated with neural connectivity strength between the left posterior hippocampus ROI and the left inferior parietal lobule/posterior cingulate cortex. Seed ROIs are shown at the top with crosshairs. Pearson’s correlation coefficient for the left inferior parietal lobule is shown. Clusters survived an FWE-corrected
Discussion
ASD youths exhibited comparable picture–name learning and recognition to TD youths, suggesting intact visual-auditory binding. Notwithstanding their comparable learning and recognition performance, ASD youths showed significantly fewer retrieval gains than their TD peers. Fewer memory gains pointed to an inflexible memory in ASD. Thus, atypical learning in childhood could be a factor linked to atypical cognition and behavior in ASD.
Relational memory might involve positive correlations between successful memory and neural connectivity among TD individuals. Resting-state FC between the anterior hippocampus and higher visual areas after relational learning was related to better retrieval performance (Murty et al., 2017). Likewise, it has been reported that the anterior hippocampal resting-state FC can better predict associative memory (Persson et al., 2018). Among healthy young adults, the right anterior hippocampus and right dorsolateral prefrontal cortex are involved in stable successful retrieval (Hashimoto et al., 2011). In relation to this, the anterior hippocampal network may be associated with the maturation of memory (Demaster & Ghetti, 2013).
In our study, successful memory performance was positively correlated with the right anterior hippocampal network in TD, while it was negatively correlated with the left anterior hippocampal network in ASD. We detected no positive correlations with hippocampal networks in ASD. Our finding that FC was lower in the left hippocampal networks of ASD youths than in those of TD youths was consistent with a previous report identifying globally weak FC in ASD youths (Yerys et al., 2017). In healthy adults, the right hippocampus connects with a distributed network, while the left hippocampus is connected with fronto-limbic areas (Robinson et al., 2016). Right–left asymmetry in hippocampal volume development, both in TD and ASD, has been reported (Reinhardt et al., 2020). The functional lateralization of hippocampus could be involved in memory development in TD children and atypical development in lateralization of hippocampus might be associated with ASD individuals’ problems with social interaction, communication, and inflexible behaviors.
Moreover, the reduced connectivity evident in the left hippocampal networks of ASD individuals suggests the use of local memory systems, which might be associated with non-relational memory. Independent use of the left anterior hippocampus and parietal areas could also explain the successful memory outcomes of our ASD participants. Disengagement of the network for complex and related-information retrieval, including self-related memory (Sheldon et al., 2016), might enhance simple associative (picture–name) memory in ASD youths. Given their lower memory gains in the recognition test, the ASD youths in this study might have applied item-based learning or simple binding rather than memory strategies such as relational learning (Wojcik et al., 2018). The lower age-related improvement in learning and recognition in ASD individuals might point to their reliance on basic, simple, and early-developed memory processes.
We observed significantly fewer memory gains in the ASD group than in the TD group as their learning and retrieval performances were comparable. Although the chance levels of both category learning and recognition were high, the group difference was detected only in terms of gain. Low relational learning and inflexible retrieval in childhood may increase restricted and repeated behaviors in patients with ASD (Gaigg et al., 2015; Lopez et al., 2005; Wojcik et al., 2018). A lower habituation and generalization ability may be associated with patients’ memory characteristics. In the meantime, a strong item memory may compensate for weak relational memory in children with ASD, which can help with the development of a memory performance that is comparable to that of TD children.
Limitations
Our first limitation is that we did not use more stringent exclusion criteria (FD > 0.2 mm) for head movement because this would have further reduced the sample size (Satterthwaite et al., 2012). Nonetheless, our criteria are consistent with previous ASD studies (Falahpour et al., 2016; Gao et al., 2019). Another limitation is that sex differences in both ASD and hippocampal network development may represent a confounding factor in this study (Lai et al., 2017; Riley et al., 2018). High chance levels of both category learning and category recognition could impair the reliability of GAIN and DECAY, although a level much higher than that of chance in both correct immediate- and delayed-recognition tests suggested fewer random responses. Finally, in addition to the SUCCESS score, lower DECAY and GAIN scores (representing fewer memory changes) could indicate rigid memory; however, the small number of responses in this study constrained the assessment of those neural correlates.
Conclusion
Learning and recognition of picture–name pairs were intact in children with ASD, but these individuals showed fewer memory changes than their TD peers. Local memory systems that do not depend to a great degree on hippocampal networks may be involved in rigid memory in ASD, and context-independent and less relational memory processing might be associated with fewer memory gains in these individuals. These atypical memory characteristics in ASD individuals may exaggerate their inflexible behaviors in some situations, or—vice versa—their atypical behaviors may result in rigid and less connected memories. In light of this, our findings may enhance our understanding of memory development in individuals with ASD. The resulting learning strategies and mechanisms may be of interest to educators, parents, and individuals with ASD, who may be able to thereafter capitalize on these insights to improve the effectiveness of special education.
Footnotes
Acknowledgements
We would like to thank Dr Tokio Uchiyama and the Yokohama Psycho-Developmental Clinic for recruiting participants, and Dr Keisuke Wakusawa for fruitful comments on the manuscript.
Author contributions
T.H. conceived of the study. T.H., S.Y., and Y.M. conducted data collection. S.Y. and Y.M. conducted and supervised intelligence quotient testing and ADI-R. T.H. and Y.M. conducted ADOS-2. T.H. performed all analyses and wrote the manuscript. R.K. did supervision and project administration. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
Written informed consent was obtained from each subject in accordance with the Declaration of Helsinki. This study was approved by the Ethics Committee of Tohoku University.
Data policy statement
All data generated and/or analyzed during this study are available from the corresponding author (T.H.) upon reasonable request.
