Abstract
The etiological significance of neurological disorders in autism is debated, but it is clear that they complicate support provision and clinical management, and can have negative impact on outcomes. This systematic review and meta-analysis explored the full range of co-occurring neurological disorders in autism. We estimated the odds of having neurological complications compared to the general population and other neurodevelopmental conditions, as well as the overall prevalence of different neurological disorders. Seventy-nine articles were eligible for the systematic review, including 28 case-control studies, 43 prevalence studies, and 8 cohort studies. Findings were heterogeneous across studies. Overall, autistic individuals were significantly more likely than the general population to exhibit epilepsy, macrocephaly, hydrocephalus, cerebral palsy, migraine/headache, and congenital abnormalities of the nervous system, with prevalence estimates ranging from 1.1% (0%–3.3%; hydrocephalus) to 14.2% (11.3%–17.2%; epilepsy). Epilepsy was also more common in autism than in attention-deficit/hyperactivity disorder (odds ratio [95% confidence interval] = 4.06 [2.81–5.88]). Findings indicate that awareness of neurological disorders and neurological check-ups are indicated in autism to ensure adequate physical health care and support. Prospective studies of neurological disorders in children diagnosed with or at risk of autism might further enhance our understanding of causal pathways.
Lay abstract
Neurological disorders, such as epilepsy and cerebral palsy, have been reported to occur among individuals with autism beyond chance and may have an impact on daily living across the lifespan. Although there has been research investigating neurological disorders in autism, the findings are not always conclusive. Previous summaries of existing studies have not evaluated the full range of neurological disorders. This study aimed to comprehensively explore the neurological problems appearing in autism to provide updated information that is needed for better healthcare and support in this population. We looked at already published studies focusing on risk or frequency of neurological disorders in autism. Our results suggest that individuals with autism are more likely than the general population to have a range of neurological disorders, including epilepsy, macrocephaly, hydrocephalus, cerebral palsy, migraine/headache, and inborn abnormalities of the nervous system. In order to provide individualized healthcare and support of high quality to individuals diagnosed with autism, health care professionals and other support providers need to be attentive to neurological complications. To further improve our understanding about the link between autism and neurological disorders, future research should follow the neurological health of children who are diagnosed with or are at increased likelihood of autism.
Introduction
Autism spectrum conditions (henceforth autism) are heterogeneous in etiology and presentation and are assumed to be associated with altered development and functioning of the nervous system (American Psychiatric Association [APA], 2013). In recent years, there has been increased scientific interest in medical conditions and physical health in autism from clinical, biological, and social perspectives. Compared to the general population, autistic individuals have a higher risk of a range of co-occurring medical disorders and experience premature mortality (Hirvikoski et al., 2016; Kohane et al., 2012). It has been proposed that shared genetic vulnerability and/or underlying biological mechanisms involving multiple systems may contribute to the higher prevalence of somatic complications in autism (Tye et al., 2019; Vorstman et al., 2017). A better understanding of the association between the co-existing medical conditions and autism is therefore needed to inform individualized clinical management and support provision.
Among the several general medical conditions associated with autism, neurological disorders are arguably the most prominent. Neurological disorders encompass a wide range of complications with neurological cause/sequela and have been pinpointed by the World Health Organization (WHO; 2006) as one of the greatest threats to public health. Autistic individuals are prone to neurological complications such as epilepsy and cerebral palsy (Davignon et al., 2018; Schendel et al., 2016), which in turn are linked to a higher degree of impairment (Turk et al., 2009; Viscidi et al., 2013a). Indeed, a longitudinal study has suggested less favorable clinical trajectories in autistic individuals with neurological disorders as compared to those with other medical conditions (Doshi-Velez et al., 2014). Moreover, the clinical approach and healthcare provision can be complicated by the presence of neurological disorders, for instance, owing to the drug-drug interaction and the cognitive and behavioral side effects of medications (Besag, 2018; Depositario-Cabacar & Zelleke, 2010; Frye et al., 2011).
Although an extensive body of epidemiological research has been conducted, the current knowledge of the frequency and specificity of neurological disorders in autism remains limited in several ways. First, the estimates of co-occurrence vary across studies, and study findings are often inconsistent. Second, previous systematic reviews have focused on only a few of the most common neurological disorders, such as epilepsy and macrocephaly, while the full range of neurological disorders has not yet been addressed (Amiet et al., 2008; Sacco et al., 2015; Woolfenden et al., 2012). Third, it is not clear if the prevalence of neurological disorders differs between autism and other neurodevelopmental conditions (NDCs). Fourth, longitudinal studies investigating the association of neurological disorders and autism are scarce. Therefore, an updated and comprehensive systematic review of the current evidence is needed to guide clinical practice and identify gaps in the accumulated body of research. To further understand the association of neurological disorders and autism, we conducted a systematic review and meta-analysis to examine three research questions:
Is there a difference between autism and the general population, or between autism and other NDCs, regarding the prevalence of co-existing specific neurological disorders?
What is the prevalence of specific neurological disorders across the lifespan among individuals with autism?
Is there an association between neurological disorders diagnosed in early childhood and a subsequent diagnosis of autism?
Method
This systematic review was conducted and reported in accordance with the PRISMA statement (Liberati et al., 2009). The protocol was preregistered with PROSPERO (CRD42019120048).
Eligibility criteria
Type of studies
(1) Case-control studies comparing the odds of being affected by a neurological disorder between individuals diagnosed with autism and controls comprising either the general population or individuals with other NDCs. (2) Prevalence studies on neurological disorders in autism. (3) Population-based cohort studies examining the association of specific neurological disorders in early childhood with a subsequent autism diagnosis.
Study population
The population in focus were individuals with a diagnosis of autism as defined by the Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV; autistic disorder, Asperger’s disorder, and pervasive developmental disorder–not otherwise specified) or Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5; autism spectrum disorder), or International Classification of Diseases (10th ed.; ICD-10; F84.0 Childhood autism, F84.1 Atypical autism, F84.5 Asperger syndrome, F84.8 Other pervasive developmental disorders, and F84.9 Pervasive developmental disorder, unspecified) or ninth edition (ICD-9; 299.0 Autistic disorder), ascertained by either clinical assessment or validated instruments. Participants of any age were eligible.
Exposure
A neurological disorder categorized in ICD-10 chapter VI “Diseases of the nervous system” (Table 1). Sleep disorders, listed in both chapter VI (organic) and chapter V (nonorganic), were not included in this review since most of the prevalence studies were unable to differentiate the origins of sleep problems in autism (Cortese et al., 2020), which also involve behavioral and psychiatric aspects (Malow et al., 2012). In addition, the following conditions, which have been reported to occur in autism beyond chance but not listed in chapter VI, were also included: macrocephaly, microcephaly, vertigo, head injury, and congenital malformations of the nervous system.
Chapter VI of ICD-10: Diseases of the nervous system.

Comparator
(1) Case-control studies: controls selected from a sample of typically developing (TD), individuals/general population (non-autism), or a sample with another specific NDC. Controls should be recruited within the same context (community, setting, time) as the cases. (2) Cohort studies: Cohort members without the specific neurological disorder.
Outcome
(1) Case-control/cohort studies: The association of a neurological condition with autism. (2) Prevalence studies: Prevalence of a specific neurological disorder in autism.
Language
Studies published in English.
Publication type
Studies published in peer-reviewed journals from 1990 (the year ICD-10 was endorsed by the World Health Assembly) onward.
Search strategy
Electronic database searches were conducted in the following databases: Embase, Medline, and PsycInfo. The MeSH terms identified for searching Medline were adapted in accordance to corresponding vocabularies in Embase and PsychInfo. Each search concept was also complemented with relevant free-text terms and these were, if appropriate, truncated and/or combined with proximity operators. No language restriction was applied. Databases were searched from inception. The search strategies are available in Appendix S1. The searches were performed by two librarians at the Karolinska Institutet University Library from December 2018 to February 2019.
Study selection and coding
Two reviewers independently screened the titles and abstracts for eligibility according to the criteria and then assessed the potentially relevant publications in full text in the same manner. Disagreements between reviewers were resolved by consensus after discussion. Reference lists of identified review articles were screened for additional studies. A standardized form was used to extract data from the included studies for assessment of study quality and synthesis of the evidence by two independent reviewers. Extracted information included the following: study types, study settings, diagnostic tools, number of participants, demographics characteristics, and information for risk of bias assessment.
Risk of bias assessment
Risk of bias was assessed independently by two reviewers using the Joanna Briggs Institute (JBI) Critical Appraisal tools for case-control studies (Moola et al., 2017), prevalence studies (Munn et al., 2015), and cohort studies (Moola et al., 2017). Methodological quality including the risk of selection bias, misclassification, confounding, and appropriate statistical analysis was appraised. Higher scores indicated less risk of bias. Any discrepancies in the scoring were resolved by discussion and reviewer consensus. Some modifications to the tools were made for this review: (1) For case-control studies, we did not assess item 9 “Was the exposure period of interest long enough to be meaningful?.” This item was not deemed relevant, since neurological disorders do not necessarily precede the diagnosis of autism. (2) For cohort studies, we did not assess item 8 “Was the follow up time reported and sufficient to be long enough for outcomes to occur?” From a development perspective, it was not deemed feasible to specify this. (3) For prevalence studies, we did not assess item 3 “Was the sample size adequate?” To determine this, the estimated prevalence of different neurological disorders in autism must be available. Current estimates are often inconsistent, imprecise, or lacking, which was one of the main reasons for conducting this systematic review.
Planned methods of analysis
The principal summary measures were odds ratio (case-control studies), prevalence rate (studies examining prevalence), and risk ratio (cohort studies). The metafor package (Viechtbauer, 2010) in R version 3.2.4. (RStudio Team, 2015) was used for calculating pooled estimates and 95% confidence intervals (CI). Random-effects model was adopted in data syntheses due to the methodological diversity among the included studies. In the first step, case-control studies were synthesized for each specific neurological condition. If the synthesis of case-control studies showed a significant association, a pooled prevalence estimate (the proportion of the specific neurological disorder in autism) was calculated based on data from both the case-control studies and the prevalence studies and was presented with the caterpillar plots. When synthesizing the prevalence studies, Freeman–Tukey double arcsine transformations were used to stabilize the variance (Freeman & Tukey, 1950). In the back-transformation, the harmonic mean of individual sample sizes was used (Miller, 1978). The consistency for each meta-analysis was measured with the I2 statistic (Higgins & Thompson, 2002). Baujat Plots were used to identify the possible outlying studies that contribute to heterogeneity. The significance of publication bias was tested using Egger’s weighted regression and presented in funnel plots. Subgroup analysis and meta-regression were used to address heterogeneity of outcome measures between studies and to detect possible moderators.
Results
Study selection
A total of 79 studies involving 81 samples were identified for inclusion in the review. The search provided a total of 4963 citations. Checking the references of published systematic reviews and relevant papers identified 19 additional relevant studies. Among the 4982 articles, 4708 were discarded after reviewing the abstracts. Out of the remaining 274 citations examined in full text, 189 did not meet the eligibility criteria, while 6 were duplicates (see Appendix S2 for reasons). See PRISMA flow diagram (Figure 1).
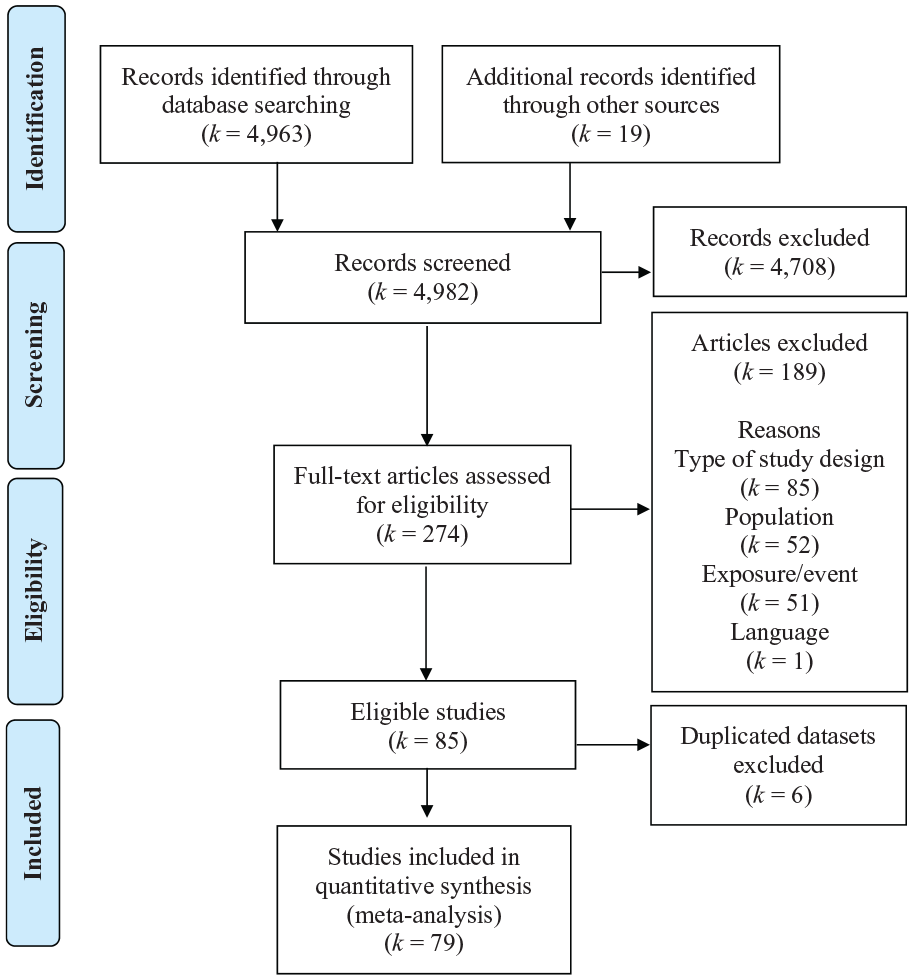
PRISMA flowchart of review process and study selection.
Overview of the included studies
A total of 28 case-control studies investigating 11 different neurological disorders in individuals with and without autism diagnoses were included (see Table S1, articles labeled with “⁎” in the References). Two of the neurological disorders, encephalopathy and muscular dystrophy, were not found among the cases and were consequently not synthesized in the meta-analyses. Regarding the control groups, 26 studies recruited a non-autism or TD population, 5 included an attention-deficit/hyperactivity disorder (ADHD) sample, 4 included a learning disorder (LD) sample, and 1 included an intellectual disability (ID) sample. Forty-three prevalence studies, focusing on 10 different neurological disorders, were eligible for inclusion (see Table S2, articles labeled with “#” in the References). In addition, 8 cohort studies investigating 6 neurological disorders were included for the analysis of neurological disorders preceding a subsequent autism diagnosis (see Table S3, articles labeled with “$” in the References).
The studies were published from January 1992 to December 2018. A rapid increase in publication was noted in the past decade, with 49 (62%) studies published since 2011. Most of the included studies were conducted in Europe (32 studies, 42%) and North America (28 studies, 35%). Thirteen studies (16%) were conducted in Asia, 3 (3%) in Africa, 1 in Australia, and 2 studies recruited samples from different continents (Fortuna et al., 2016; Hussein et al., 2011). About half of the studies (k = 37, 46.8%) used community samples, including national registry data, while the other half used samples enrolled from medical services, including outpatient units and hospitals. The majority of the included articles (46 studies, 58%) focused on children (0 to 18 years), while 25 (32%) had mixed age groups and 8 (10%) focused on adults (>18 years). In terms of the applied standardized diagnostic tools for identifying autism, 20 studies (25%) used either the Autism Diagnostic Interview (Revised; ADI-R; Lord et al., 1994) or the Autism Diagnostic Observation Schedule (ADOS; Lord et al., 2000); 3 (4%) used the Childhood Autism Rating Scale (CARS; Schopler et al., 1988); and 1 used the Autism-Tics, ADHD, and other Comorbidities (A-TAC; 1%; Mårland et al., 2017). Fifty-two studies (66%) relied exclusively on clinical assessments, and 5 studies (6%) only used caregiver report.
Case-control studies
Epilepsy
Study characteristics
A total of 16 case-control studies focused on epilepsy in autism, including a nationwide twin sample (Alabaf et al., 2019) and a sample of multiplex autism pedigrees with sibling controls (Amiet et al., 2013a). The mean male to female ratio of the participants diagnosed with autism was 3.52 (range: 1.87–4.49) across studies. The study populations were recruited from both clinical settings (k = 4, 25%) and communities (included registry data; k = 12, 75%).
Results of individual studies
Out of the 16 case-control studies, 13 measured “epilepsy” as the exposure, while 3 measured “seizures.” Three studies used telephone interviews or questionnaires to identify epilepsy/seizures, and the others obtained the information of epilepsy from medical records. All suggested significantly higher rates of epilepsy in autism than in non-autism controls (range of crude odds ratio 3.26–24.21, Figure 2).
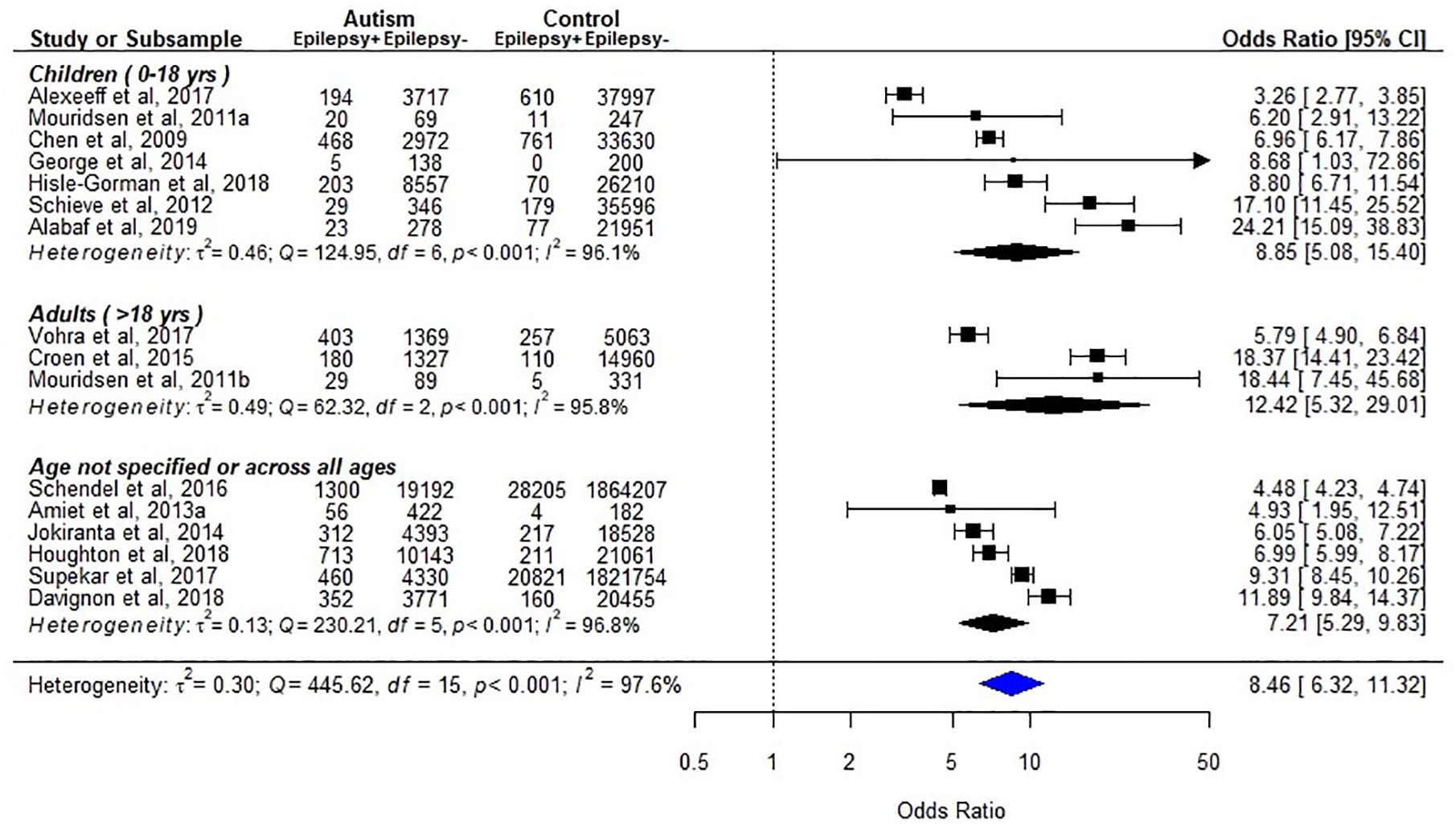
Epilepsy in individuals with autism, as compared to typically developing/general population (k = 16).
Synthesis
Sixteen studies with a total of 65,860 individuals with autism diagnoses and 3,974,070 non-autism controls were included in a meta-analysis (Figure 2). It is important to note that epilepsy and seizures are distinct medical terms and should not be used interchangeably. In addition to the primary analyzes where we collapsed all 16 studies, we therefore conducted subgroup analyses separately for these conditions. Significant heterogeneity across studies was found (p < 0.001; I2 = 97.6%), even in the subgroup analysis by different age groups (children: p < 0.001; I2 = 96.1%, and adults: p < 0.001; I2 = 95.8%). Eight studies did not perform individual matching, limiting confidence of the estimates. However, all studies reported higher prevalence of epilepsy in autism (pooled odds ratio (95% CI): children 8.85 (5.08–15.40); adults 12.42 (5.32–29.01); overall 8.46 (6.32–11.32)). In addition, studies measuring epilepsy and seizures showed similar estimates in the subgroup analyzes (pooled odds ratio (95% CI): epilepsy 8.14 (5.88–11.27); seizures 11.77 (6.62–20.93); Figure S1A). The Baujat plot shows two possible outlying studies (Figure S1B), but the pooled estimate did not change after excluding those two articles. The results of Egger’s test indicated no evidence of publication bias, but some estimates lying on the top of the funnel plot, but outside the triangular region, suggest selection variance across studies (Figure S1C).
Macrocephaly
Study characteristics
Ten case-control studies (including a twin sample (Froehlich et al., 2013) and one study with TD sibling controls (Lainhart et al., 2006)) investigated macrocephaly. A non-autism comparison group was used in eight of the case-control studies, while the two remaining studies compared autism with ADHD and LD, respectively. A majority of studies recruited only children (k = 9), while one study recruited both children and adults.
Results of individual studies
Seven of the 8 studies with non-autism controls identified macrocephaly by reviewing the medical records or by clinical assessments, while the last by ICD codes from a national register database (Timonen-Soivio et al., 2015). The results were inconsistent across studies, with 5 studies reporting no significant association and 3 studies suggested higher prevalence rate of macrocephaly in autism (Figure 3).
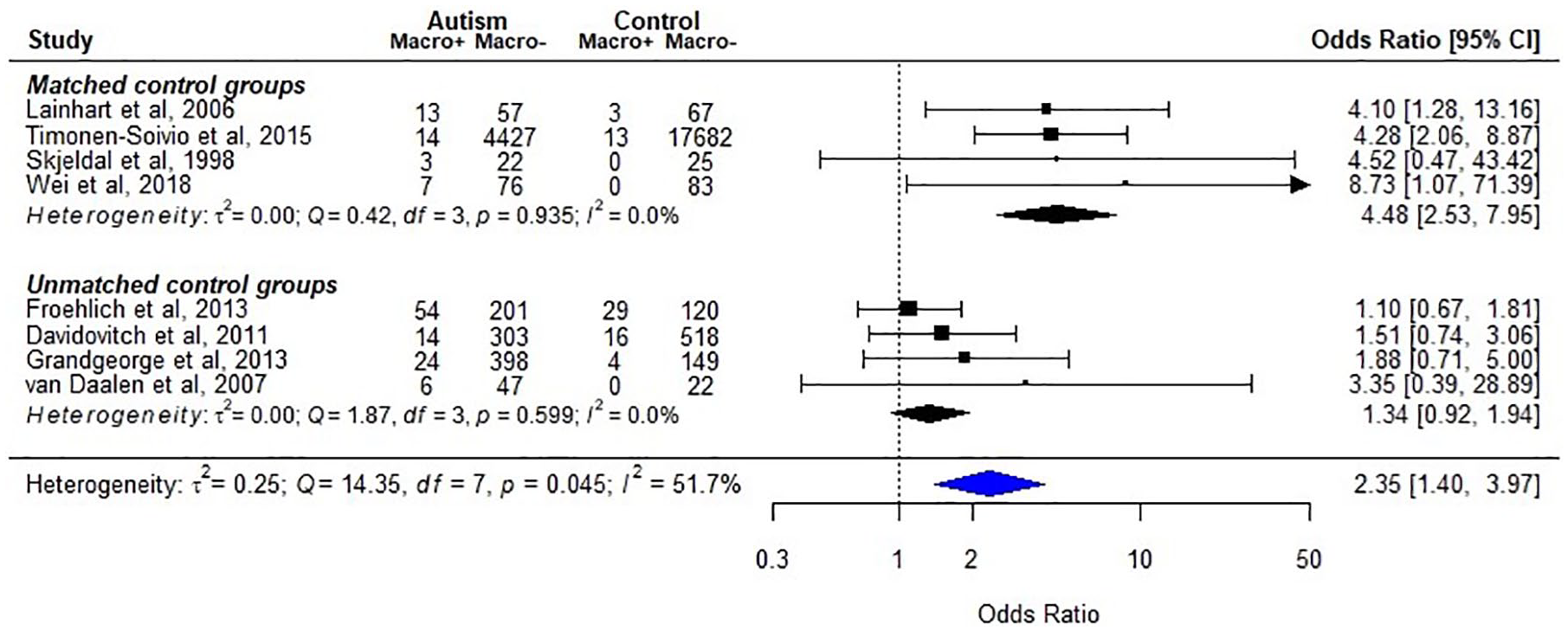
Macrocephaly in individuals with autism, as compared to typically developing/general population (k = 8).
Synthesis
Eight studies including a total of 5666 individuals with autism diagnoses and 18,731 non-autism controls were included in a meta-analysis. Due to the heterogeneity of findings (p = 0.045; I2 = 51.7%), we conducted subgroup analysis based on the matching of the control groups (Figure 3). The synthesized results indicated that the studies with age- and gender-matched controls found a significant association, while the unmatched studies did not (pooled odds ratio (95% CI): matched: 4.48 (2.53–7.95); unmatched: 1.34 (0.92–1.94); overall: 2.35 (1.40–3.97)).
Microcephaly
Study characteristics
Seven case-control studies (one employed sibling controls (Lainhart et al., 2006)) on microcephaly were included, with six of them using non-autism comparison groups and one comparing autism with ADHD only. Five studies recruited only children, while the other two also recruited adults. The number of participants with autism ranged from 25 to 4441 (Figure S2).
Results of individual studies
One study identified individuals with microcephaly by the ICD-9 code in the register dataset (Timonen-Soivio et al., 2015), and the other six by medical records or clinical assessments. No study suggested a significant difference between cases and controls.
Synthesis
The synthesized results of the six studies with non-autism control groups included a total of 5358 individuals with autism and 18,560 non-autism controls (Figure S2). No evidence of heterogeneity was found in the meta-analysis (I2 = 0%), which showed no difference in the risk of microcephaly between autism and controls (pooled odds ratio (95% CI) = 1.08 (0.61–1.93)).
Cerebral palsy
Study characteristics
Seven case-control studies (including three using the same dataset but different cohorts; Alexeeff et al., 2017; Croen et al., 2015; Davignon et al., 2018) focused on cerebral palsy (CP) in autism. A majority of studies recruited only children participants (k = 3), while two had mixed age groups, and the final two recruited only adult participants. Sample sizes varied across studies, with two studies including less than 200 autistic participants and five studies more than 1000 (Figure 4).
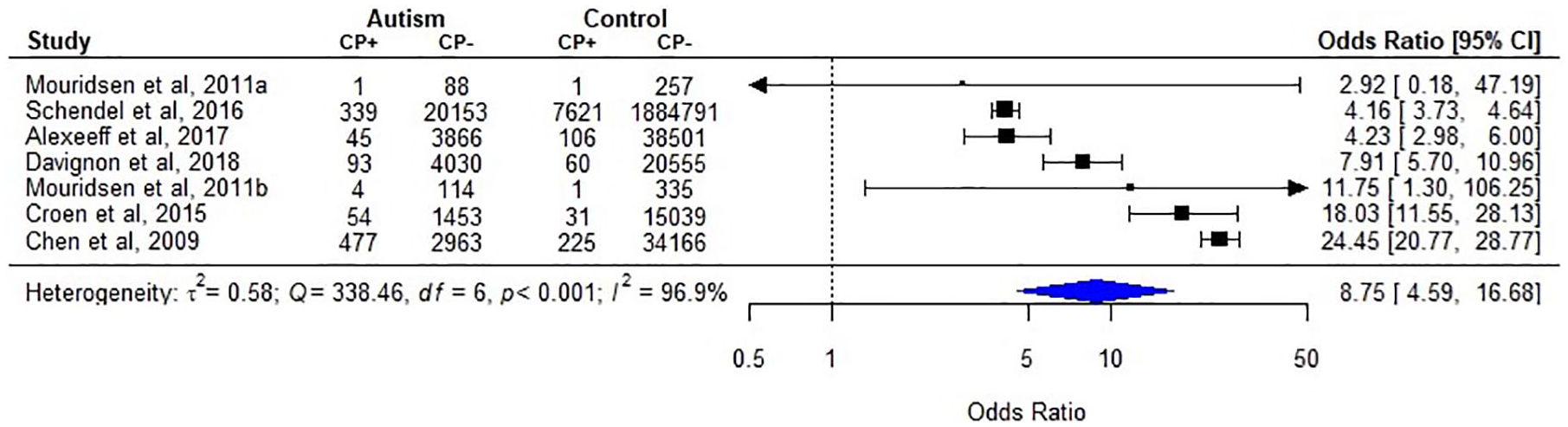
Cerebral palsy in individuals with autism, as compared to typically developing/general population (k = 7).
Results of individual studies
All the case-control studies used ICD code to identify CP. Six of them indicated higher prevalence rate of CP in autism than in non-autism controls (range of crude odds ratio 4.16–24.45), while 1 found no significant difference (Figure 4).
Synthesis
A meta-analysis including all 7 studies was conducted. The synthesis included 33,680 individuals with autism and 2,001,689 non-autism comparators and indicated a significant association of CP and autism (Figure 4). However, our confidence in the pooled estimates was limited due to heterogeneity of results (pooled odds ratio (95% CI) = 8.75 (4.59–16.68); heterogeneity: p < 0.001; I2 = 96.9%).
Migraine/headache
Study characteristics
Nine case-control studies (one used a twin sample; Alabaf et al., 2019) investigated migraine/headache in individuals with autism diagnosis. Five of these studies focused on children, three focused on adults, and one recruited adolescents and young adults. A majority of the studies (k = 7) had more than 300 autism participants.
Results of individual studies
Four of the studies measured “headache” as the exposure, two measured “migraine,” two measured “headache/migraine,” and one measured “headache” and “migraine” separately. Most studies identified migraine/headache by medical records or ICD codes, while one study used telephone interview and one used parent report. Three studies of children reported higher levels of migraine/headache symptoms in autism compared to controls, while others did not find a significant difference (Figure 5).
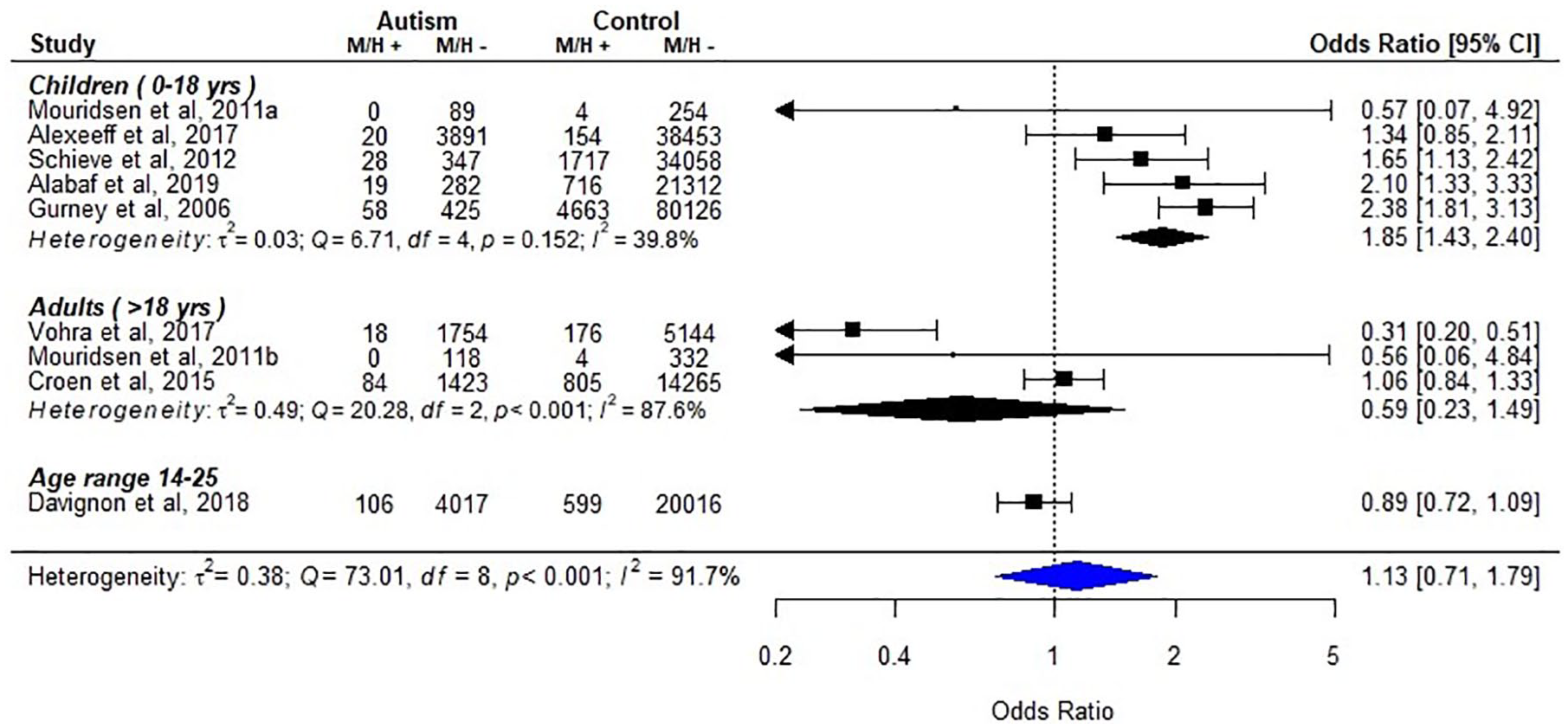
Migraine/headache in individuals with autism, as compared to typically developing/general population (k = 9).
Synthesis
In the primary analyses, we grouped all studies in one overall category of headaches. Due to the heterogeneity of the definitions used in the included studies, which could include both symptoms and official clinical diagnosis, we also conducted subgroup analyses with headache and migraine separately. Nine studies with a total of 12,679 individuals with autism diagnoses and 222,798 non-autism controls were included in a meta-analysis. Due to heterogeneity (p < 0.001; I2 = 91.7%), we conducted subgroup analysis by different age groups (Figure 5). For children, the synthesized results indicated that the odds of migraine/headache in autism were 1.85 times higher than for those without autism diagnosis (pooled odds ratio (95% CI): children 1.85 (1.43–2.40); adults 0.59 (0.23–1.49); overall 1.13 (0.71–1.79)). However, significant heterogeneity was still found among studies with adult participants (p < 0.001; I2 = 87.6%). Among the three studies of adults with autism, two investigated “headache” (Mouridsen et al., 2011b; Vohra et al., 2017) and one investigated “headache” and “migraine” separately (Croen et al., 2015). When analyzed separately, neither headaches nor migraine showed a significant association with autism (Figure S3).
Other neurological disorders
Study characteristics
Figure 6 presents studies comparing the prevalence of other neurological disorders in autism and non-autism populations, including congenital nerve system abnormality (a general term used in those studies without specification, or those abnormalities other than hydrocephalus, macrocephaly, and microcephaly), demyelinating diseases, hydrocephalus, and brain or head injury. Four of these studies focused on children, one on adults, and the other four recruited participants across all ages. A majority of these studies recruited more than 3000 participants with autism diagnoses (k = 6), while three studies recruited less than 1000.
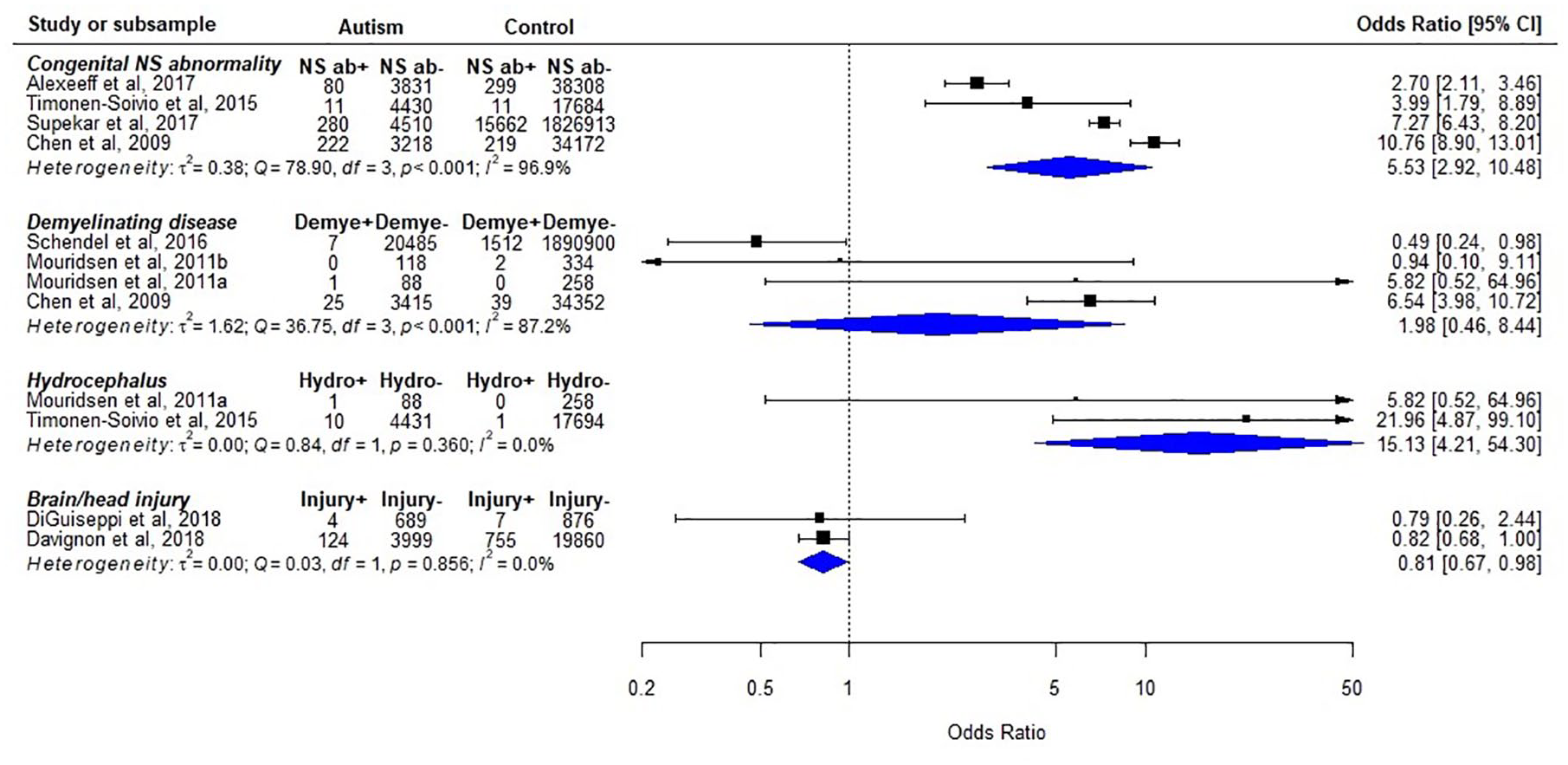
Other neurological disorders in individuals with autism, as compared to typically developing/general population.
Results of individual studies
All studies identified neurological disorders by the ICD diagnostic codes, except one that used parent report to identify traumatic brain injury (DiGuiseppi et al., 2018). For congenital nerve system abnormality, all the four studies suggested a higher prevalence in autism compared to controls (range of crude odds ratio: 2.70–10.76). However, the results for the remaining neurological disorders were inconsistent (Figure 6).
Synthesis
Meta-analyses were carried out for congenital nerve system abnormality and demyelinating diseases (Figure 6). There was significant heterogeneity between studies (p < 0.001; I2 = 96.9% and p < 0.001; I2 = 87.2%, respectively). Nevertheless, all studies investigating congenital nerve system abnormality reported significant higher odds for autism (pooled odds ratio (95% CI): congenital nerve system abnormality 5.53 (2.92–10.48); demyelinating diseases 1.98 (0.46–8.44)).
Comparison between autism and other NDCs
Case-control studies comparing the prevalence of neurological disorders between autism and ADHD are shown in Figure 7. All the four studies focusing on epilepsy reported higher odds in autism, while significant heterogeneity across studies was detected in the meta-analysis (pooled odds ratio (95% CI) = 4.06 (2.81–5.88); heterogeneity: p < 0.001; I2 = 86.3%). Studies which evaluated the risk of neurological disorders in individuals with autism and LD revealed inconsistent findings (Figure S4). There was also one study comparing neurological disorders in autism and ID (Schieve et al., 2012). This study found that children with autism had lower prevalence of headache/migraine than those with ID (odds ratio (95% CI) = 0.54 (0.31–0.92) p = 0.025), but reported no significant difference regarding epilepsy.
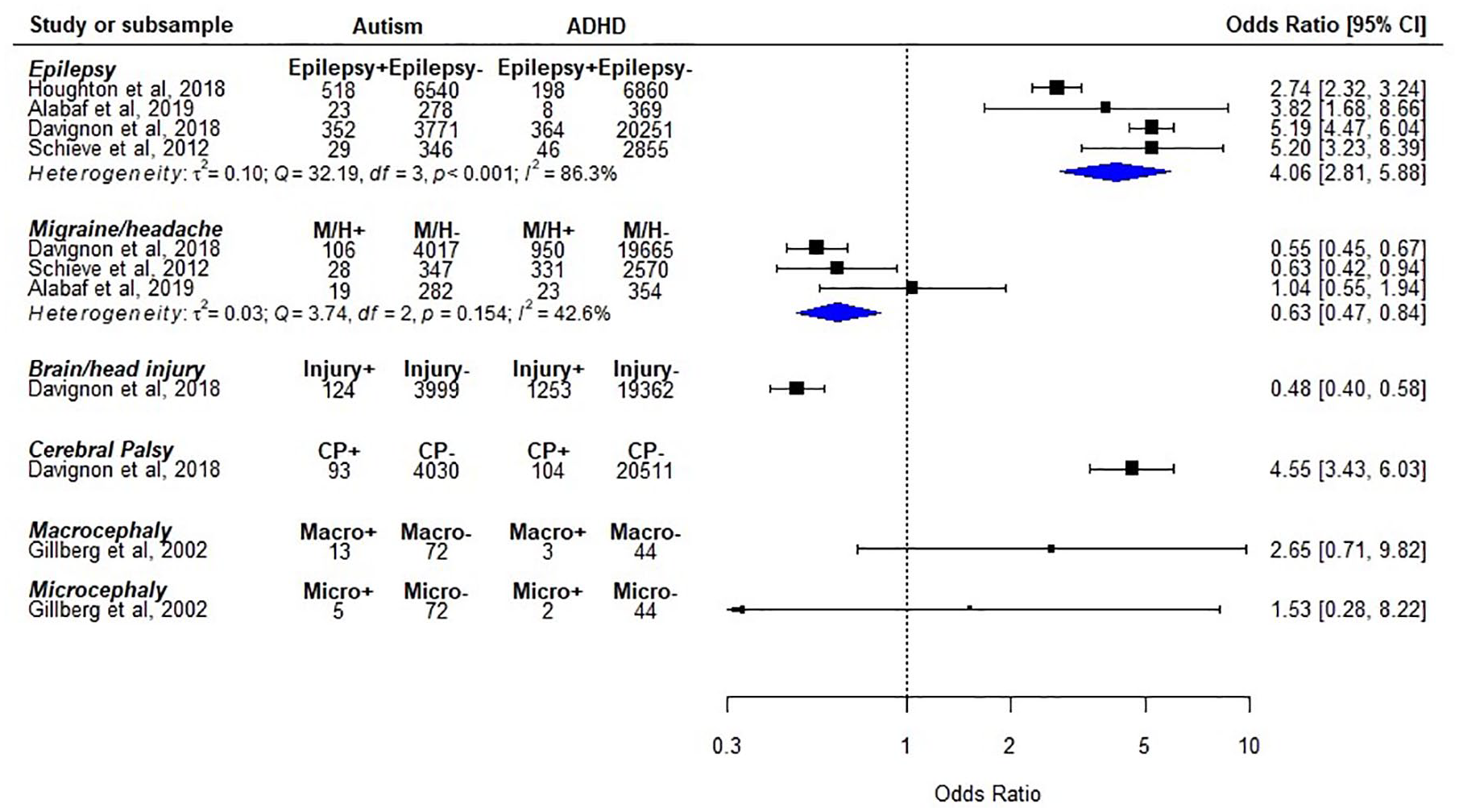
Neurological disorders in individuals with autism, as compared to those with attention-deficit/hyperactivity disorder.
Prevalence
The summary of study characteristics and estimated prevalence are presented in Table 2. Significant heterogeneity was detected in all the analyses of six neurological disorders. For epilepsy, a total of 31 studies reported the proportion of participants with ID in their samples and were included in the meta-regression analysis with ID as the explanatory variable. In the model with data transformation, the results of omnibus test suggested that ID significantly contributed to the heterogeneity across studies (R2 = 41.84%, QM (df = 1) = 19.43, p < 0.001). The regression coefficient showed that the rate of epilepsy increased 0.34% with 1% increase of participants with ID in the sample (data transformed). Meta-analytic scatterplot with regression line derived from back-transformed data are shown in Figure 8.
Summary of prevalence analyses.
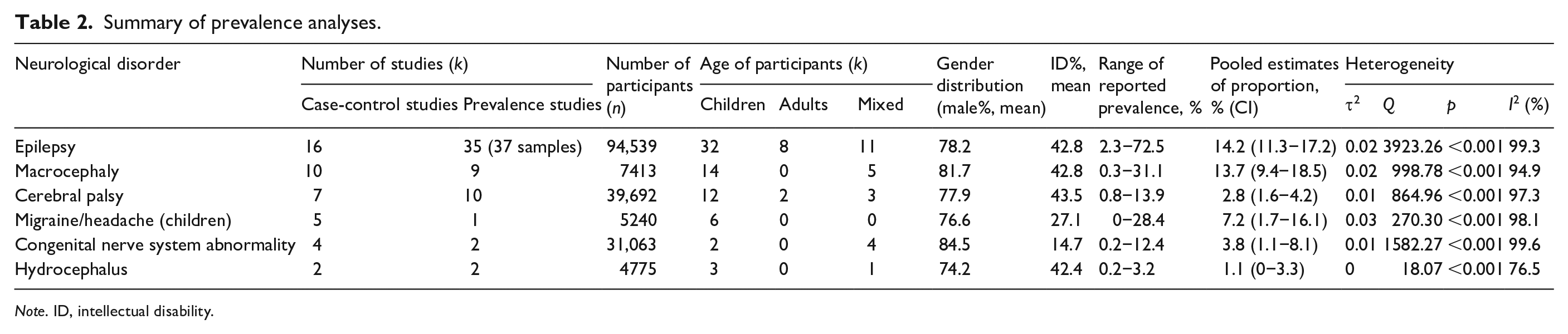
Note. ID, intellectual disability.
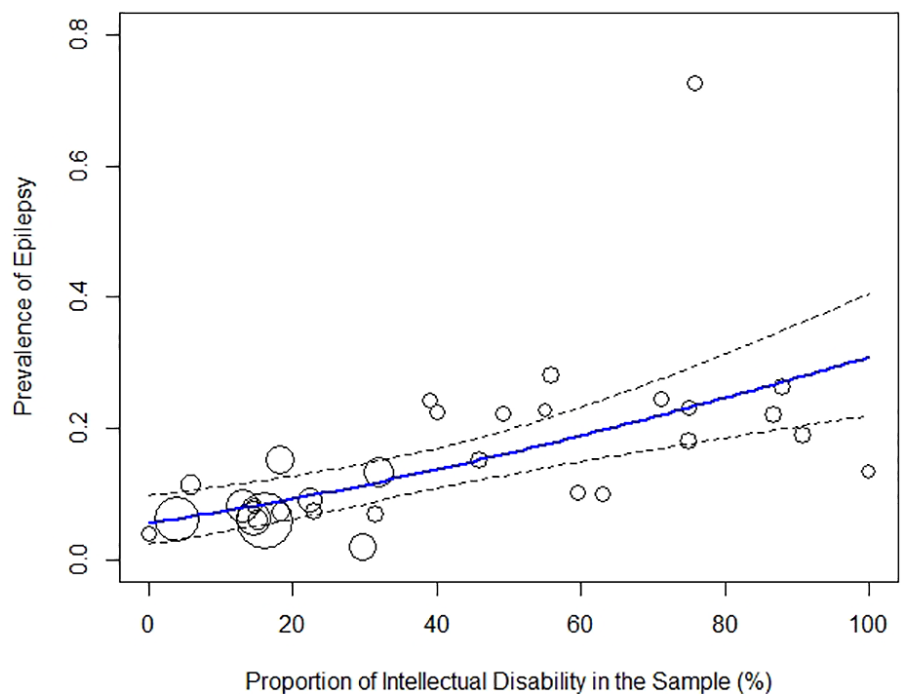
Meta-analytic scatterplot of prevalence studies of epilepsy in autism (k = 31).
Cohort studies
Figure 9 summarizes cohort studies investigating neurological disorders as a factor increasing likelihood of autism. Three studies focused on epilepsy as the exposure and all reported higher incidence of autism in the epilepsy group. However, heterogeneity across studies diminishes our confidence in the pooled estimate. Two studies focused on encephalopathy (periventricular leukomalacia, intraventricular hemorrhage, and newborn encephalopathy following fetal distress Badawi et al., 1998]) and suggested that encephalopathy could be a factor increasing the likelihood of subsequent autism. Nevertheless, studies evaluated macrocephaly as the exposure reported inconsistent results. One study (Chang et al., 2018) reported increased incidence of autism after traumatic brain injuries, while another study (Aagaard et al., 2018) did not find a significant association between microcephaly and autism.
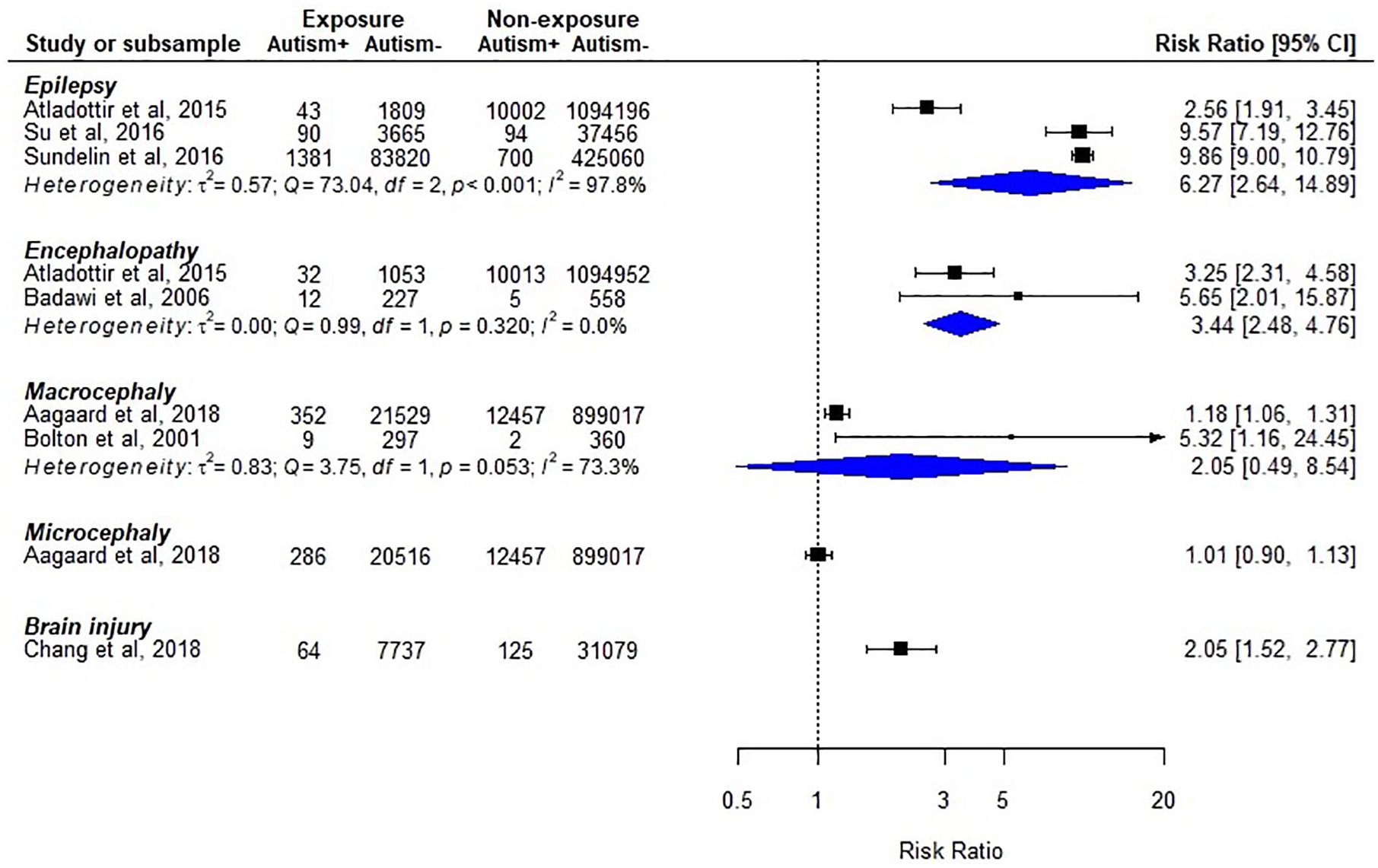
Cohort studies of neurological disorders as risk factors for autism.
Generalizability and risk of bias
Although the studies in this systematic review were distributed worldwide and covered diverse ethnicity and healthcare systems, some of the relatively rare diseases were studied in a limited number of regions. Regarding the validity of diagnoses, a majority of studies used medical records, clinical assessments, or validated instruments to identify individuals with autism and neurological disorders. While the characteristics of the included participants were usually reported, some studies did not provide demographics or level of intelligence of the samples. In addition, most studies did not include information about possible confounders, such as pre- and perinatal risk factors. Furthermore, some case-control studies did not perform individual matching, which might have introduced selection bias. The risk of bias of included studies are summarized in Figure 10.
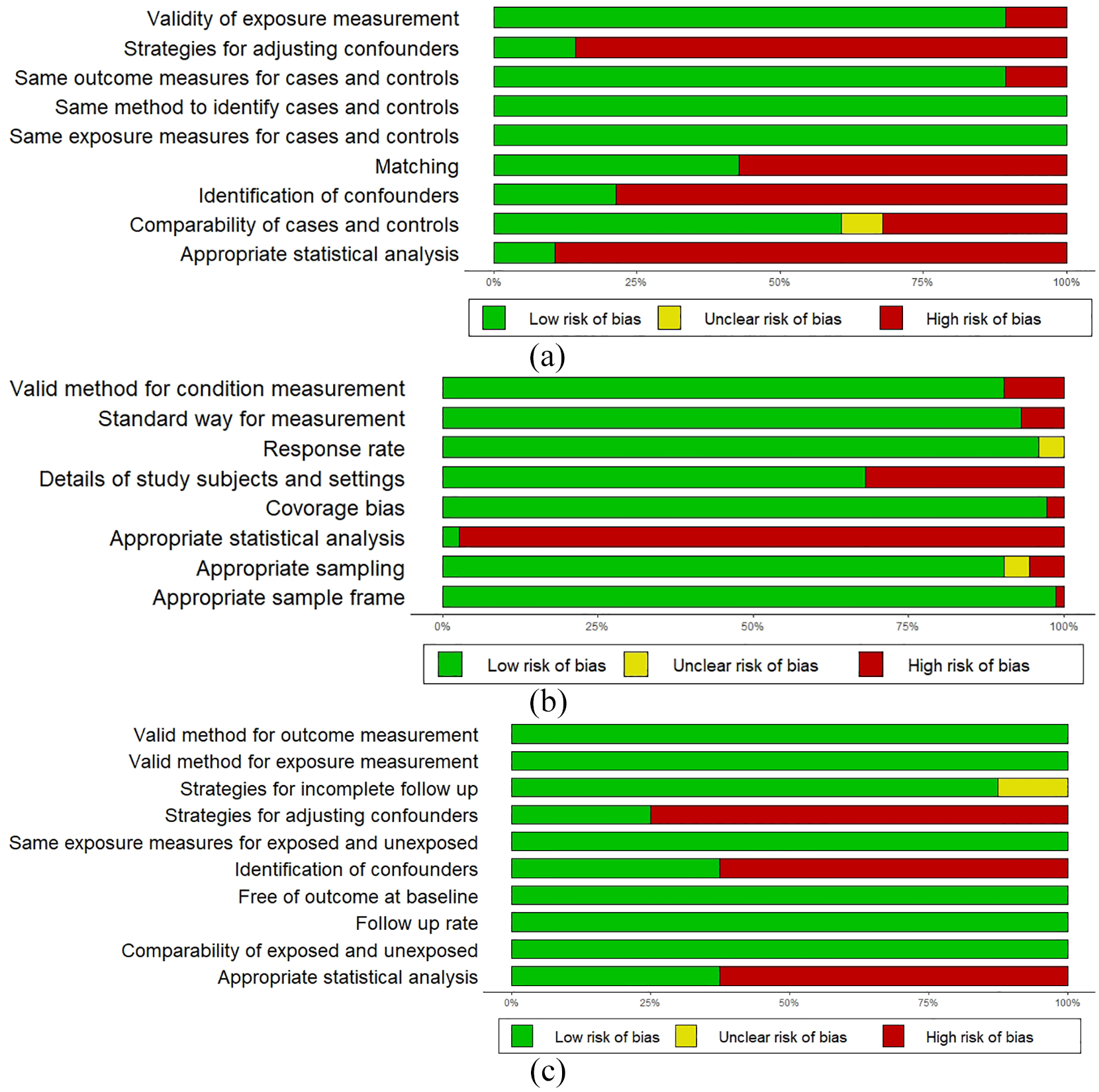
The risk of bias summary: (a) case-control studies (k = 28), (b) prevalence studies (k = 51), and (c) cohort studies (k = 8).
Discussion
This is the first systematic review and meta-analysis to present a comprehensive overview of the association of neurological disorders and autism across ages. The results indicate that autistic individuals are more likely than their counterparts to have a range of neurological complications, including epilepsy, macrocephaly, hydrocephalus, cerebral palsy, migraine/headache, and congenital abnormalities of the nervous system. Prevalence estimates ranged from 1.1% (0%–3.3%; hydrocephalus) to 14.2% (11.3%–17.2%; epilepsy). Compared to controls with ADHD, autistic individuals also had a higher probability of epilepsy. In addition, there was a temporal association of epilepsy and encephalopathy in early childhood with subsequent autism diagnosis. Our findings underscore that autism is a complex NDC, linked to increased susceptibility for neurological health issues. Hence, awareness of neurological complications in autism is imperative for a comprehensive individualized clinical management and tailored support provision.
While this review included studies covering most of the commonly reported neurological disorders in autism, several important gaps and inconsistencies in the literature were also identified. For example, with the exception of epilepsy and congenital abnormalities of nervous system, case-control studies yielded inconsistent results. Furthermore, the pooled prevalence estimates generally lacked precision. Importantly, no eligible studies were found for several large categories of neurological disorders, such as infectious diseases, inflammatory diseases, and the degenerative diseases of nervous system. Furthermore, studies focusing on children constituted more than half of the included articles, while only 10% focused explicitly on adults. In terms of the comparison between autism and other NDCs, only studies involving comparators with ADHD, LD, and ID, were identified, not motor or specific learning conditions. Finally, only a limited number of neurological disorders were investigated in the few identified cohort studies.
Substantial heterogeneity (variation in results across studies, indicating diminished confidence on the pooled estimates and a need for greater understanding of factors influencing the associations) was found across studies in most of the meta-analyses, which in part could be explained by the methodological diversity of the included studies. For example, the population characteristics varied due to different source populations, methods of sampling, and method used for case and exposure identification. In addition, some studies had small sample sizes which may have resulted in higher variability of the estimates. Furthermore, the wide variance in sample characteristics probably arises from the diversity of autism phenotypes, involving symptom presentation and severity, co-existing NDCs and psychiatric conditions, cognitive capacities, underlying etiologies, and resulting functional impairment (Bölte et al., 2019; Grove et al., 2015; Gyawali & Patra, 2019).
While there still is uncertainty regarding the underlying mechanism of the association between neurological disorders and autism, our results clearly show that autistic individuals are at increased risk for neurological complications. The immediate clinical and practical significance of this finding is further underscored by the literature showing that autistic individuals with certain neurological disorders exhibit a higher risk of social functioning challenges, poorer intervention outcomes, and increased mortality (Eriksson et al., 2013; Hirvikoski et al., 2016; Ko et al., 2016; Pickett et al., 2011). For several good reasons, clinicians should therefore be aware of common neurological complications in autism in their daily practice. First, clinical management might be complicated by neurological disorders. For instance, there is a risk for polypharmacy (such as the administration of both psychotropic medications and antiepileptic drugs) and the resulting increased risk of drug interactions and adverse drug reactions which may manifest as behavioral problems (Besag, 2018; Frye et al., 2011). Second, some neurological disorders might otherwise go undetected and untreated. For instance, it is presumably not widespread knowledge among clinicians that children with autism have an increased risk of migraine and headache. This finding might possibly reflect the shared serotonergic system alterations in autism and migraine (Casanova, 2008; Garbarino et al., 2019; Hamel & Currents, 2007). In addition, pediatric headache is associated with depression, anxiety, and other mental conditions, which frequently co-occur in autism (Jacobs et al., 2016). The identification of headache and correct differential diagnosis in autistic individuals can be difficult due to limited communication skills, particularly verbal abilities in young children and those with ID. Children who are unable to convey physical symptoms verbally may exhibit sleep disturbances or increased irritability (McGuire et al., 2016; Miller et al., 2003). Therefore, if such problem behaviors appear, they may be related to physical discomfort, and clinicians should consider migraine and headache as a possible underlying cause.
Another neurological disorder which could be easily missed in autism is absence epilepsy for its lack of overt motor signs and brief symptom duration (Matricardi et al., 2014). Moreover, the staring spells in absence epilepsy could also mimic some behavioral manifestation in autism and ADHD, and thus, this symptom is difficult to clarify among those with linguistic difficulties. Since evidence shows that childhood absence epilepsy could be associated with impaired executive functions, lower learning performance, and emotional problems (Caplan et al., 2008; Verrotti et al., 2015), clinicians should be alert to the need of early detection and management for this comorbidity in autism.
More explicit guidelines might help to advance the clinical management and support provision for autistic individuals with neurological disorders. It also seems reasonable to recommend neurological check-up conducted in autism-friendly and autism-experienced environments, followed by adequate treatment. In addition, it is important to note that neurological complications in autistic children may be cared for by pediatricians or neurologists, prior to a diagnosis of autism. For instance, our results revealed overlap between autism and early onset and congenital neurological disorders, namely macrocephaly, hydrocephalus, cerebral palsy, and inborn abnormalities of the nervous system. Since children with early neurological disorders are prone to social and cognitive challenges, neuropediatric clinical settings should be prepared to also apply a neurodevelopmental perspective on their clients (Tuchman, 2017). Evidence suggests that early intervention targeting social-cognition and early parent involvement can be effective in improving outcomes in infants and toddlers at increased likelihood of autism (Estes et al., 2014; Green et al., 2017). It might therefore be beneficial to monitor the social communication development of young children with certain neurological disorders, in order to initiate appropriate interventions.
Future directions
Based on this systematic review, we suggest some possible directions for future research to explore the interplay between neurological systems and autism behavioral phenotypes. First, longitudinal studies across the lifespan are needed to better understand the clinical profiles and trajectories of neurological disorders in autistic individuals. For example, it has been reported that the age distribution of epilepsy incidence in autism is bimodal, with peaks in early childhood and adolescence (Deykin & Macmahon, 1979; Volkmar & Nelson, 1990). However, most of the currently available studies are cross-sectional, and thus not informative to examine age effects on neurological complications in autism. Second, high-quality cohort studies with neurological disorders as exposure and autism as the outcome and the reverse design are also needed to clarify the direction of the relationship between autism and neurological disorders. Third, studies focusing on the impact of neurological disorders on autism presentation, functioning, and intervention outcomes would be helpful for clinicians to adapt intervention strategies for different subgroups. Fourth, the association between autism and neurological disorders may also indicate common etiological mechanisms. For example, our findings demonstrated a substantial degree of coexistence between autism and epilepsy, with ID moderating this association. It has been proposed that autism, ID, and epilepsy share mechanisms at genetic, molecular, and anatomical levels (Tuchman, 2017). More research is needed to disentangle the interweaving of potential pathways contributing to the overlap of autism and neurological disorders.
Limitations
The results of this systematic review should be interpreted with caution, mainly due to risk of bias and heterogeneity across published studies. The most common risk of bias identified in the included studies were missing information about potential confounders and inadequate control for factors, such as preterm birth and postnatal insults, which could increase risk for neurological disorders (Cowan, 2002; Li et al., 2019). Therefore, we are unable to assess in which direction and to what extent those factors would influence the relation between a specific neurological disorder and autism. Overall, the clinical usefulness of the estimates provided here is restricted by the heterogeneity of the population. This is especially true when a common condition shows moderation effect, such as the effect of ID on the relation between autism and epilepsy. In addition, inconsistent measurements of neurological disorders across studies, such as epilepsy and seizures, and also headache and migraine, may be a source of classification bias. It is important to note that, by definition, having a single seizure is not the same as having epilepsy. Similarly, symptoms of headache do not necessarily indicate a specific diagnosis of headache or migraine. However, our subgroup analyses did not suggest that these disparities had a major impact on the estimates. Furthermore, unrepresentative study populations may have affected the estimates. It should also be noted that several studies used sibling controls or a twin sample, which might have increased the heterogeneity across studies due to the differences between related and random comparators. Finally, the limited number of eligible studies in combination with incomplete reporting of sample characteristics precluded proper analyses of heterogeneity.
Conclusion
This systematic review shows that autistic individuals are more likely to experience a range of neurological disorders than controls from the general population. Clinicians and other professionals working with individuals on the autism spectrum need to be attentive to neurological complications, their adequate assessment, and treatment. Moreover, neurological disorders diagnosed in early childhood should be viewed from a neurodevelopmental perspective due to increased likelihood of subsequent autism. Future research should explore the association between neurological disorders and autism with longitudinal designs to uncover possible etiological mechanisms.
Supplemental Material
Appendix_Figures_(1) – Supplemental material for Neurological disorders in autism: A systematic review and meta-analysis
Supplemental material, Appendix_Figures_(1) for Neurological disorders in autism: A systematic review and meta-analysis by Pei-Yin Pan, Sven Bölte, Preet Kaur, Sadia Jamil and Ulf Jonsson in Autism
Supplemental Material
Appendix_S1_Documentation_of_literature_search – Supplemental material for Neurological disorders in autism: A systematic review and meta-analysis
Supplemental material, Appendix_S1_Documentation_of_literature_search for Neurological disorders in autism: A systematic review and meta-analysis by Pei-Yin Pan, Sven Bölte, Preet Kaur, Sadia Jamil and Ulf Jonsson in Autism
Supplemental Material
Appendix_S2_Reasons_for_exclusion – Supplemental material for Neurological disorders in autism: A systematic review and meta-analysis
Supplemental material, Appendix_S2_Reasons_for_exclusion for Neurological disorders in autism: A systematic review and meta-analysis by Pei-Yin Pan, Sven Bölte, Preet Kaur, Sadia Jamil and Ulf Jonsson in Autism
Supplemental Material
Appendix_Table_S1-S3 – Supplemental material for Neurological disorders in autism: A systematic review and meta-analysis
Supplemental material, Appendix_Table_S1-S3 for Neurological disorders in autism: A systematic review and meta-analysis by Pei-Yin Pan, Sven Bölte, Preet Kaur, Sadia Jamil and Ulf Jonsson in Autism
Footnotes
Acknowledgements
The authors thank Magdalena Svanberg and Sabina Gillsund for their assistance with the literature search and Nikolas Aho for data extraction.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sven Bölte discloses that he has in the last 5 years acted as an author, consultant, or lecturer for Shire, Medice, Roche, Eli Lilly, and Prima Psychiatry. He receives royalties for text books and diagnostic tools from Huber/Hogrefe, Kohlhammer, and UTB.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Pei-Yin Pan is supported by Tri-Service General Hospital, the Ministry of National Defense, Taiwan (R.O.C), and the Swedish Research Council.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
