Abstract
Proficient use of spatial terms such as under, to the left of or in front of is a central component of daily communication and is important in the development of language and spatial cognition. Here we examine spatial language abilities in intellectually high-functioning individuals with autism spectrum disorder, an area previously overlooked in autism research. Twenty-five high-functioning individuals with ASD and 25 typically developing controls, matched for chronological age and cognitive abilities, completed a novel battery tapping a broad range of spatial language abilities. We report selective difficulties in the production of spatial terms and spatial description recall in high-functioning ASD. Overall verbal abilities did not account for the observed group differences. Crucially, however, the intensity of autism spectrum traits predicted individual performance in both spatial language production and spatial description recall. We discuss the theoretical implications of these findings and explore their significance for both clinical practice and intervention.
Lay abstract
How we think and talk about space is an essential ability, necessary for understanding the world around us. We recruit spatial thinking every day when finding our way or using tools but also in more advanced tasks, such as reading complex graphs or maps. We do so also in daily communication when we use spatial language, terms such as under, over, to the left of or in front of, and when we give instructions. Spatial terms appear in children’s early vocabularies and continue to develop until late childhood or even early adolescence. Because spatial language develops over many years, some spatial terms are mastered very early, whereas others take longer to acquire. In the current set of studies, we tested how intellectually high-functioning children and adults on the autism spectrum use and understand these early- and late-acquired spatial terms in comparison to typically developing age-matched individuals. We found that children and adults on the autism spectrum experience difficulties with the use of some spatial terms (e.g. near and far or out of and down off) but not with others, which are acquired early (e.g. in and on or over and under). We also found that remembering spatial terms from short stories was more difficult for the individuals on the autism spectrum compared with typically developing individuals. These results reveal difficulties that can profoundly affect everyday communication of children and adults on the autism spectrum but also open new directions of research on language development in autism spectrum disorders.
Keywords
Introduction
Spatial skills are a core component of cognitive development and have a unique role in predicting later performance in a number of areas, including mathematics and science (Rimfeld et al., 2017; Verdine et al., 2017). Furthermore, spatial thinking is ubiquitous in everyday life and necessary for successful locomotion, wayfinding and tool use. Even though visuospatial abilities have been considered a strength in autism spectrum disorder (ASD; Mitchell & Ropar, 2004; Mottron et al., 2006; Stevenson & Gernsbacher, 2013), a growing number of studies point to difficulties in spatial tasks in ASD. Importantly, these difficulties were also observed in intellectually high-functioning individuals with ASD (here referred to as HFA), who score within normal ranges on standardized tests of cognitive and language abilities. There is evidence for selective impairments in HFA in spatial working memory (Lai et al., 2017; Wang et al., 2017), visual perspective taking (Pearson et al., 2013; Shield et al., 2016), binding objects to locations (Ring et al., 2015) and spatial navigation (Lind et al., 2013, 2014; Ring, Gaigg, de Condappa, et al., 2018; Smith, 2015). Crucially, acquisition of these skills goes hand in hand with linguistic development and involves the mastery of spatial language, that is, verbal descriptions of spatial relations such as under, to the left of, north or towards.
Spatial language is a unique domain of language, which is tightly yoked to non-verbal spatial abilities (Coventry & Garrod, 2004; Hayward & Tarr, 1995; Landau & Hoffman, 2005; Landau & Jackendoff, 1993) and builds on the pre-linguistic concepts already present from the first months of life (Casasola, 2018). The acquisition of spatial terms is strikingly consistent across languages, with in/on, up/down, here/there and over/under mastered first, followed by projective prepositions front/behind and right/left, which are not fully acquired before age 5 or 6 (Clark, 1973; Durkin, 1981; Harris, 1972; Johnston, 1988; Johnston & Slobin, 1979; Kuczaj & Maratsos, 1975; Landau & Hoffman, 2005). Mastery of geocentric terms (north/south) typically requires even more time (but see Shusterman & Li, 2016). Path terms for describing dynamic spatial relations (motion events) appear by age 2 (Brown, 1973; Choi & Bowerman, 1991), with goal paths (into, onto) encoded more often than source paths (out of, down off, see Lakusta & Landau, 2005, 2012; Papafragou, 2010). The well-organized and orderly development of spatial language allows for a clear distinction between the early- and late-emerging linguistic abilities. Importantly, even in the presence of a severe spatial impairment, as in Williams syndrome, the early-emerging spatial terms can be adequately mastered; however, late-emerging terms, such as projective prepositions, appear to be more vulnerable to spatial, or more generally, developmental deficits (Landau & Hoffman, 2005; Landau & Zukowski, 2003).
Surprisingly, spatial language abilities have never been thoroughly studied in ASD. Only a few studies, although with limited testing batteries and samples, reported evidence suggesting difficulties in spatial language in this population. In these studies, low-functioning children with ASD showed deficits in the comprehension and production of selected spatial and temporal prepositions (Churchill, 1972; Perkins et al., 2006; Ricks & Wing, 1975). Some preliminary observations have been also made about spatial language difficulties in individuals on the high end of the spectrum. In an early study by Ohta (1987), a subset of the sample constituted intellectually high-functioning children with ASD. In this study, one of the tasks tapped into the comprehension of spatial terms. Participants were verbally instructed to move certain items to new locations, for example, ‘put the button on the box’, or ‘put the button next to the box’. Results pointed to a significantly lower performance in this task in the ASD group compared with the control group, a difference that could not be solely explained by the general intelligence quotient (IQ). Observed errors included semantic violations of the prepositions, for example, putting the button into the box instead of on the box or on top of the box instead of next to the box. More recently, Vulchanova et al (2012, 2013) reported two case studies of individuals with HFA, who also performed surprisingly poorly on the spatial language task where they were asked to combine spatial prefixes (which in Slavic languages are semantically related to spatial prepositions) with the verbs. Still, many aspects of spatial language abilities have been largely ignored, for example, is spatial language affected uniformly in HFA or are difficulties limited to only certain types of spatial terms? Do these difficulties extend to comprehending and memorizing spatial descriptions, an ability essential when receiving verbal descriptions of locations or directions? Finally, are these difficulties related to changing perspectives in spatial language use, such as viewer-centred (e.g. ‘to my left’) versus environment-centred descriptions (e.g. ‘in front of the building’)?
Given that language deficits in the spatial domain can have profound consequences for education, as well as daily communication about objects’ locations, tool manipulation or navigation, the identification of such difficulties could lead to changes in the intervention targets in HFA. Moreover, the uneven cognitive and language profiles observed in ASD, with ‘peaks’ and ‘troughs’ within certain domains (Bernardino et al., 2012; Charman et al., 2011; Dawson et al., 2007; Lincoln et al., 1995; Mayes & Calhoun, 2003; Vulchanova et al., 2013), offer a means of addressing important theoretical questions. Specifically, can spatial language show an uneven breakdown, with some spatial language abilities being impaired while others are intact? Furthermore, are some components of spatial language more vulnerable in atypical linguistic or cognitive development? Finally, what type of developmental mechanism could account for this potential selective breakdown and could it explain the puzzle of uneven linguistic and cognitive development in ASD? Employing a novel spatial language battery, we present the first comprehensive test of spatial language abilities in HFA, attesting selective difficulties in the production of spatial terms and in the recall of spatial descriptions, consistent with an uneven cognitive profile.
Methods
Participants
Twenty-five intellectually high-functioning individuals with ASD (7 females, age range: 9–27, M = 17.9, standard deviation (SD) = 5.9) and 25 typically developing (TD) controls (11 females, age range: 9–27, M = 17.8, SD = 5.3), all native speakers of Norwegian, participated in the study (see Appendices 1 and 2 for the complete information about individual participants’ age and gender). The participants were recruited through the national and local branches of the Autism Society in Norway and local schools. Only individuals who had received a formal diagnosis of ASD or Asperger syndrome from an authorized psychologist in Norway (according to the DSM-4 criteria, and as such, automatically qualified for the diagnosis of Autism Spectrum Disorder under the 5th edition of the Diagnostic and Statistical Manual of Mental Disorders, DSM-5; American Psychiatric Association, 2013) were included in the HFA group. We included only individuals without intellectual disability (Full Scale IQ scores higher than 70, according to the DSM-5 cut-off point for intellectual disability; see Appendix 1 for the complete list of the individual Full Scale IQ scores in the HFA group).
Groups were matched on chronological age and Perceptual Reasoning (Wechsler IQ Scales; Wechsler, 2003, 2008; Norwegian standardization editions: Wechsler, 2009, 2011, respectively; see Table 1 and Figure 1). To compare the language abilities of the participants, we employed the overall Verbal Comprehension subscale (Wechsler IQ Scales). In addition, we assessed participants’ expressive language using the Speaking subscale (Sentence Combining and Multiple Meanings subtests) from the Test of Language Development–Intermediate: 4th Edition (TOLD-I:4; Hamill & Newcomer, 2008). We also obtained additional information about the ASD symptomatology in the HFA group and possible autism spectrum traits in the TD group using the autism-spectrum quotient (AQ; Baron-Cohen et al., 2001; Norwegian translation) and the Childhood Autism Spectrum Test (CAST; Scott et al., 2002; Norwegian translation) questionnaires (see Appendices 1 and 2 for the complete list of the individual AQ and CAST scores in the HFA and TD groups). The AQ and CAST questionnaires are brief instruments that allow measurement as to where any given individual lies on the continuum of autism spectrum traits (Baron-Cohen et al., 2001). In the present sample, the average proportions of the questionnaire scores differed significantly between the groups, with the HFA participants scoring reliably higher than controls (see Table 1).
Descriptive characteristics of the HFA and TD groups.
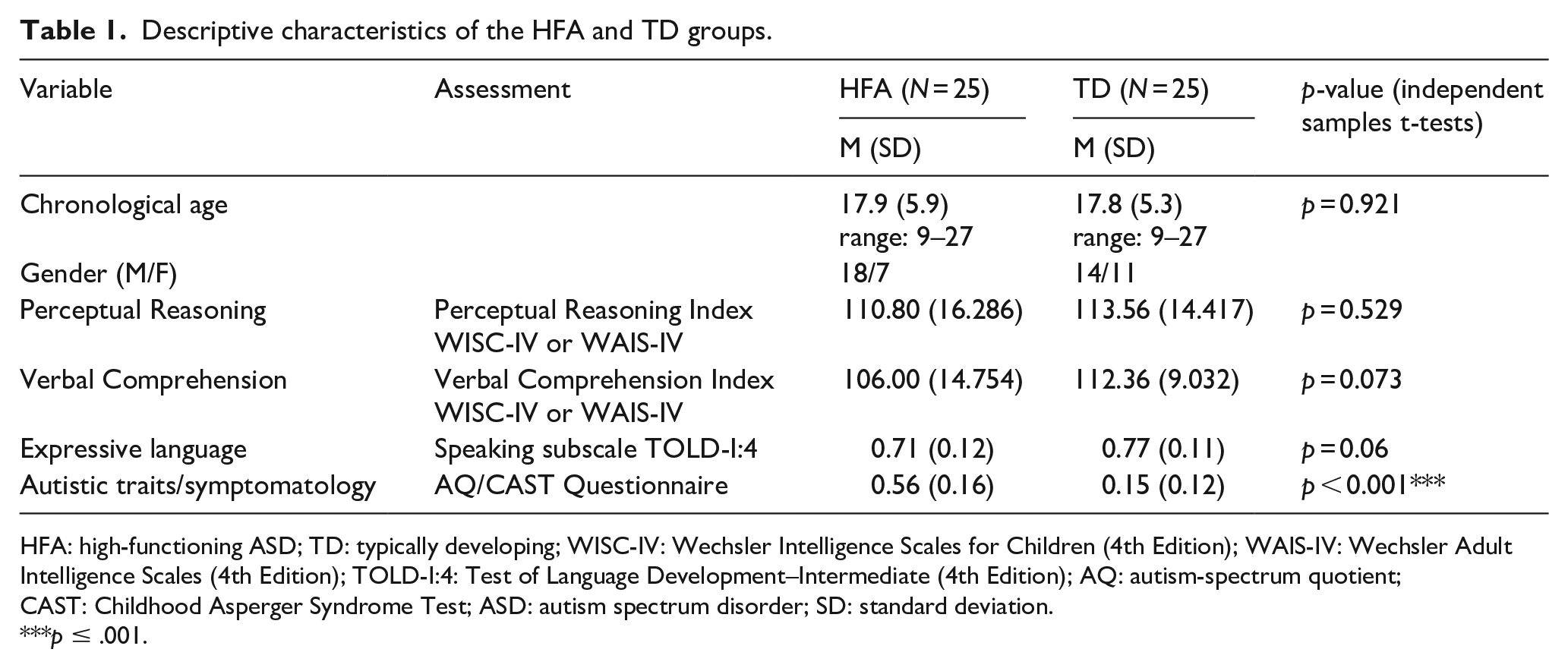
HFA: high-functioning ASD; TD: typically developing; WISC-IV: Wechsler Intelligence Scales for Children (4th Edition); WAIS-IV: Wechsler Adult Intelligence Scales (4th Edition); TOLD-I:4: Test of Language Development–Intermediate (4th Edition); AQ: autism-spectrum quotient; CAST: Childhood Asperger Syndrome Test; ASD: autism spectrum disorder; SD: standard deviation.
p ≤ .001.
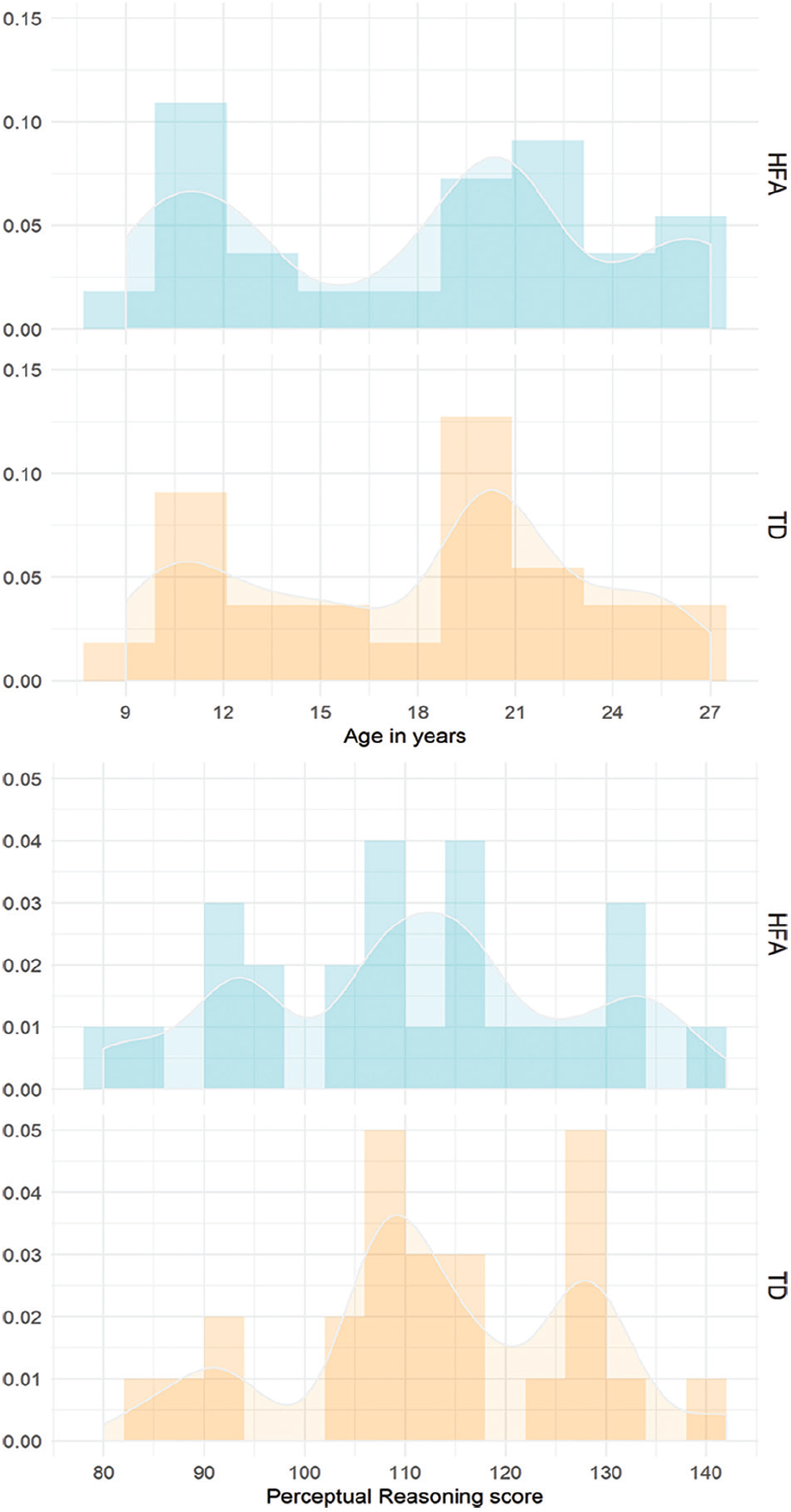
Distribution of age (upper panel) and Perceptual Reasoning scores (lower panel) in the HFA and TD groups. The y-axis represents density.
Materials
We adapted a battery of spatial language tests developed at the University of East Anglia (Markostamou & Coventry, 2020a, 2020b; Markostamou et al., 2015). The battery includes the Spatial Naming Test (SNT), the Spatial Verbal Memory (SVM) task and the Rotating Board Spatial Referencing (RBSR) task (the order corresponds to the order of administration).
The SNT is an analogue to the Boston Naming Test (Kaplan et al., 2001) and tests production of locative and directional/path prepositions. It consists of 30 pictures with simple geometrical shapes that represent different types of spatial relations (see Figure 2). The participant’s task was to name as accurately as possible the red ball’s position or its direction of movement in relation to the black cube, as distinguishable from the black balls’ locations. Part A (15 items) included relations denoted by locative prepositions: in, on, to the right of, on the left of, beside, above, under, below, behind, in front of, far to the left of, near to the left of, between, among and in the middle. Part B (15 items) included motion events, which target directional and path prepositions: downwards, upwards, to the right, across, into, onto, towards the side, out of, away from, down of, around, over, under, through, and along.
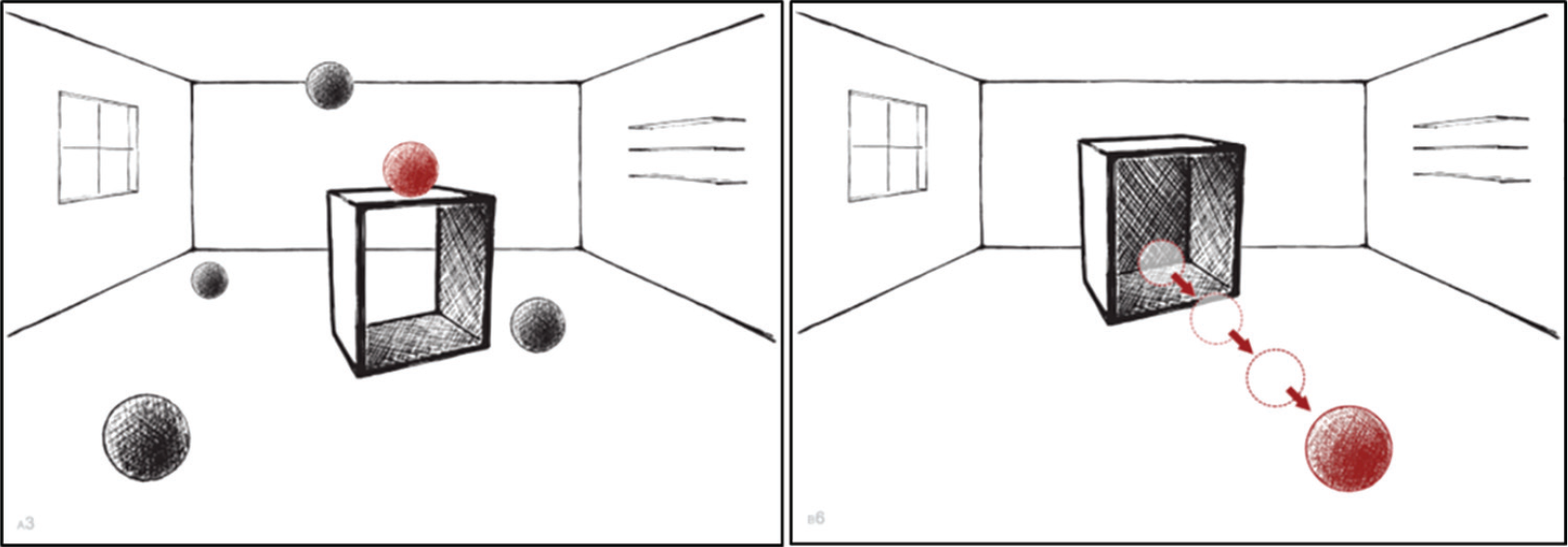
Example stimuli from Spatial Naming Test: part A (locative prepositions; left panel) and part B (directional/path prepositions; right panel).
The SVM task consists of two short stories told from egocentric (route description) and allocentric (survey description) perspectives matched for number of words and propositions. The egocentric (route description) stories described the spatial locations of the landmarks from the perspective of an agent, for example, ‘When he saw the blue lake in front of him, he turned left’, whereas the allocentric (survey description) stories described the spatial locations of the landmarks from the overhead, bird’s-eye view, for example, ‘The City Hall is in the centre of the town’. The stories were translated into Norwegian and pre-recorded by a native Norwegian speaker (see Appendix 3 for the original and translated versions of the stories). The participant’s task was to listen to the story and subsequently verbally recall everything they remembered from the story (as close as possible to how it was told). Around 25 minutes after immediate recall, participants were asked to retell the stories one more time (delayed recall).
The Rotating Board Spatial Referencing (RBSR) task tested comprehension of spatial terms from different spatial perspectives: intrinsic (object centred), absolute (geocentric, environment centred) and relative (egocentric: viewer centred and other person centred). Materials consisted of a rotating board with a red ball mounted on the outer rotating ring with the inner space in the middle reserved for the reference object (a cup or a toy car, depending on the condition; see Figure 3). The participant’s task was to judge the statements about the ball’s position as ‘true’ or ‘false’. More specifically, the experimenter on each trial (16 trials per condition) moved the ball to one of its pre-defined positions (see the locations numbered 1–8 in Figure 3) and read out loud a statement about the ball’s position, for example, ‘The ball is behind and to the right of the cup’. The participant responded ‘true’ or ‘false’ to every statement. In the relative condition, participants were judging whether the statements were true or false from their perspective (viewer centred) or from the perspective of the mini-figure on the other side of the board (other person centred). In the intrinsic condition, the participants were judging whether the statements were true or false from the perspective of an object with a natural front and back (a toy car). In the absolute condition, participants were judging as true or false statements such as ‘The ball is southeast of the cup’ with an arrow pointing to the north as the reference (see Figure 3).
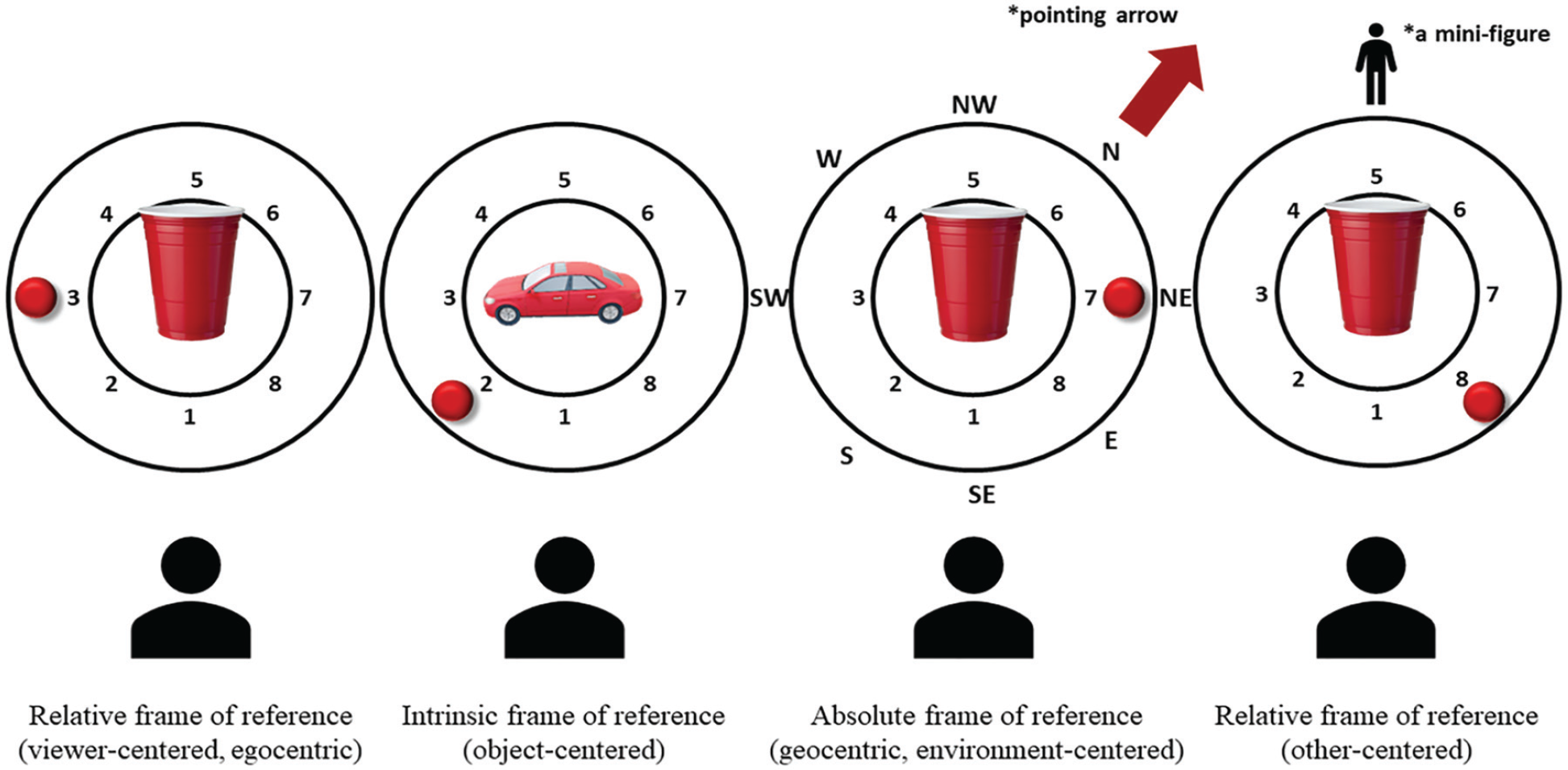
Rotating Board Spatial Referencing. Four task conditions, relative: viewer centred, intrinsic: object centred, absolute: environment centred and relative: other centred. Note that the boards, shown in plan view here, were presented horizontally (flat) in front of the participant. Numbers and letters that mark the locations in the picture were not visible to the participants.
Procedure
The study was conducted in compliance with the Regional Committees for Medical and Health Research Ethics (REK) in Norway (reference number: 2015/1642; project title: ‘Spatial language and spatial cognition in Autism Spectrum Disorder’). Participants (older than 18 years) or their parents (for participants younger than 18 years) filled out and signed the consent form for voluntary participation in the study. Participant assent was also obtained for children under the age of 18. All participants and the parents were informed about participation requirements and study procedures, and detailed instructions were given before each task. The total amount of time required to complete the spatial language battery was about 30–40 minutes. Background measures were collected independently in a separate testing session (total time about 2.5 hours) and included the Wechsler Intelligence Scale (WISC-IV for participants under 16 years old and WAIS-IV for participants over 16 years old), Test of Language Development (TOLD-I:4) AQ (for participants over 16 years) and the CAST (the parents of participants younger than 16 years) questionnaires. Testing took place at The Norwegian University of Science and Technology and at the University of Oslo. At the end of the testing procedure, all participants were invited to choose a gift (from among board games, puzzles, bags, cinema or water park tickets) in compensation for their participation in the study.
Results
Spatial Naming Test
All collected answers from the SNT were rated by two independent raters (a professional linguist and a linguist in training; both native speakers of Norwegian) on a 5-rank scale with respect to the semantic content of the response, that is, how accurately it corresponded to the content of the picture (very accurate, accurate, acceptable, barely acceptable or not acceptable). To ensure non-biased rating, the answers were arranged in alphabetical order under every item and not assigned to any subject codes, so that the raters were blind to the age, gender or diagnostic classification of the participants. Rated answers were subsequently scored by the experimenter (full score = 1, if the answer was rated as very accurate or accurate, half score = 0.5, if the answer was rated as acceptable, and 0 score, if the answer was rated as not acceptable).
The raters showed full agreement in 58% of the rated items and disagreement on 4% of the rated items. For the rest of the items (38%), the raters were in partial agreement, that is, one rater indicated an item as accurate and the other one as acceptable. In the cases of partial agreement or disagreement, the highest rating decided about the score for the particular answer (i.e. if at least one rater indicated that the answer was accurate, it received a score of 1; similarly if at least one rater indicated that the answer was acceptable, it received a score of 0.5).
We first looked at the overall performance between the groups in the task. We ran a 2 × 2 analysis of variance (ANOVA) on the average test scores with Preposition Type (locative, directional/path) as a within-subject factor and Group (HFA and TD) as between-subject factor. We added Verbal Comprehension and Expressive language scores as a covariate in the analysis to control for possible group effects caused by differences in verbal abilities (see relatively low p-value in Verbal Comprehension and Expressive language comparisons, Table 1). The analysis revealed a significant main effect of Group, F(1,49) = 19.643, p < 0.001,
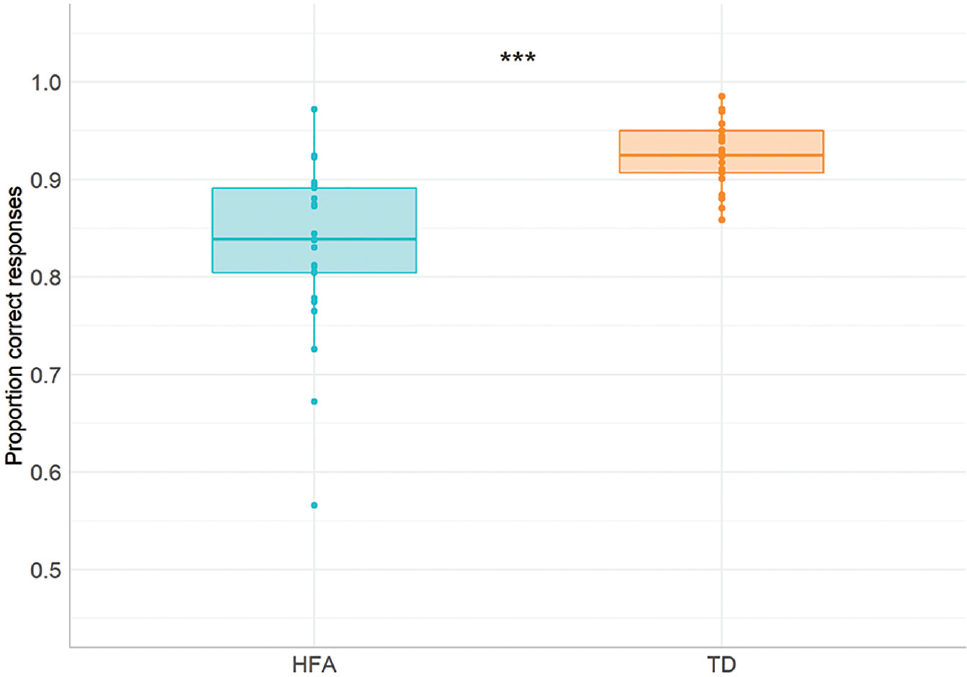
Proportion correctly named items in the Spatial Naming Test in the HFA and TD groups.
Next, we looked at the accuracy scores on single-test items to investigate whether spatial language production was affected uniformly or selectively (see Appendix 4 for the complete summary of the comparisons). While on some items, both groups were at ceiling (e.g. in, under, upwards), the highest discrepancies between the groups’ scores were in proximal (near/far) and source path terms (out of/down off/away from). After collapsing these item scores into two categories, Mann–Whitney U tests confirmed that in comparison to controls, the HFA group made significantly more errors both in the Proximity, U = 113, p < 0.001, and Source Path category, U = 166, p = 0.003 (see Appendices 5 and 6 for a qualitative description of error types).
Spatial Verbal Memory Task
Participants’ answers from both stories in the SVM task were recorded and later transcribed. Each story was divided into 25 items: 10 spatial items (e.g. ‘towards’, ‘in front of him’, ‘on his right’) and 15 non-spatial items (e.g. ‘started walking’, ‘the City Hall’, ‘the Museum’). Every correctly recalled item received 1 point. We calculated the proportions of correctly recalled items separately for spatial and non-spatial content of the stories and conducted full-factorial analyses on the proportions.
We ran a 2 × 2 ×2 × 2 ANOVA, with Reference Frame (egocentric and allocentric), Recollection Time (immediate and delayed) and Item Type (spatial and non-spatial) as within-subject factors and Group (HFA and TD) as a between-subject factor on the proportions of correctly recalled items, with Verbal Comprehension and Expressive language as covariates. We observed a significant main effect of Group, F(1,49) = 4.193, p = 0.047,
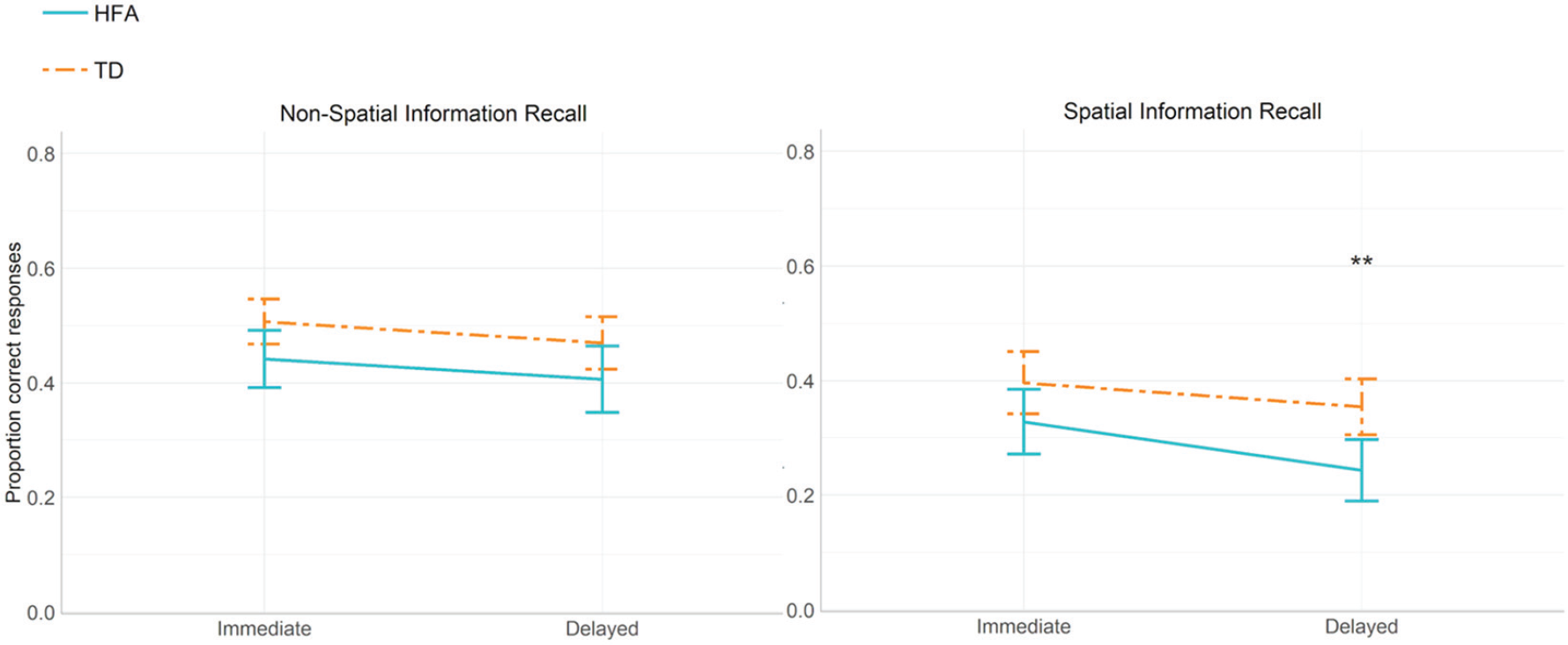
Proportion correctly recalled non-spatial and spatial items in the Spatial Verbal Memory task (immediate and delayed recall); error bars represent ±2 SEM.
Finally, in order to control for possible differences in working memory abilities between the TD and HFA groups, we compared the Working Memory subscale scores (Wechsler IQ test) between the groups and used the working memory scores as a predictor for performance of participants with HFA in the delayed recall of the SVM task. The analyses showed no significant differences in the working memory scores between the groups, t = –1.351, p = 0.183 and no significant effect of working memory scores on the delayed recall in the HFA group, R2 = 0.0259, F(1,24) = 1.2, p = 0.28.
Rotating Board Spatial Referencing Task
Four participants (two HFA and two TD) withdrew before the completion of the last task, and therefore, the group statistics are different for the RBSR task (see Table 2).
Descriptive characteristics of the HFA and TD groups: Rotating Board Spatial Referencing Task.
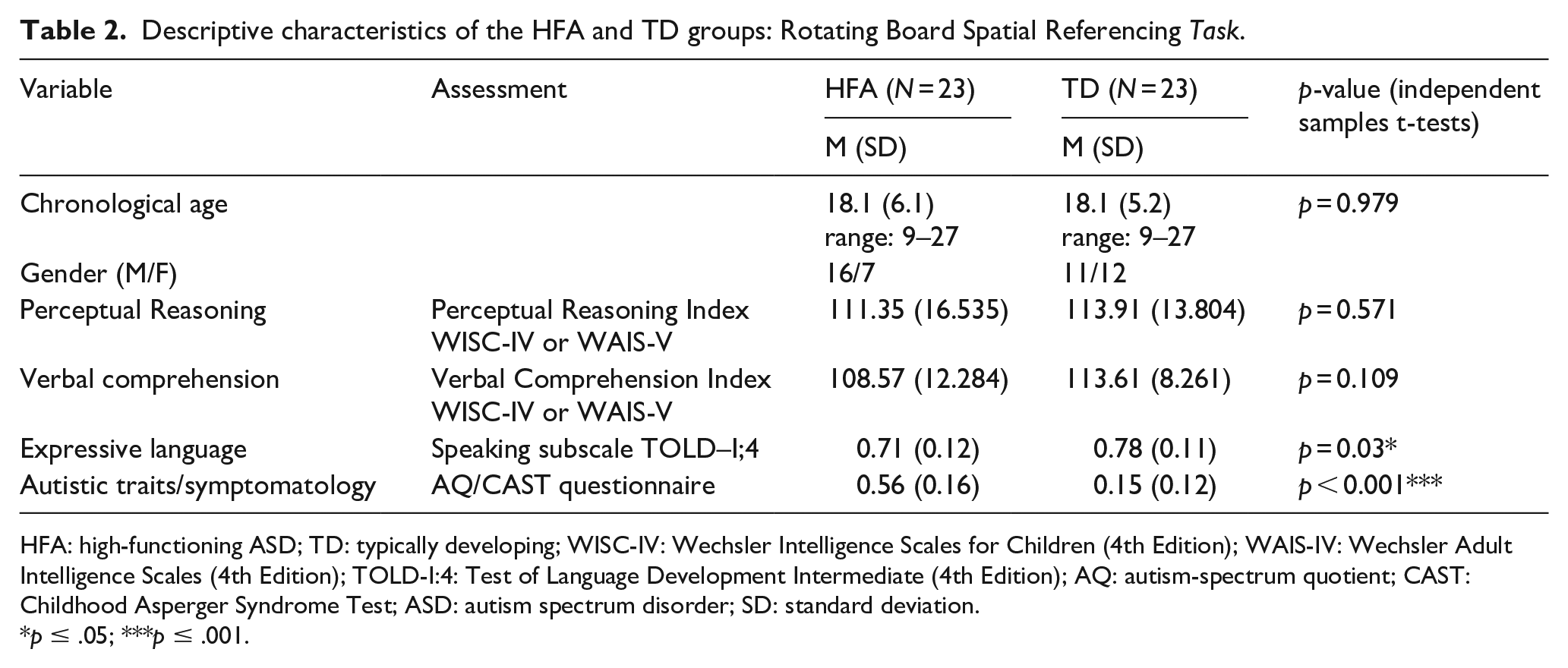
HFA: high-functioning ASD; TD: typically developing; WISC-IV: Wechsler Intelligence Scales for Children (4th Edition); WAIS-IV: Wechsler Adult Intelligence Scales (4th Edition); TOLD-I:4: Test of Language Development Intermediate (4th Edition); AQ: autism-spectrum quotient; CAST: Childhood Asperger Syndrome Test; ASD: autism spectrum disorder; SD: standard deviation.
p ≤ .05; ***p ≤ .001.
We first compared the overall accuracy on the task and ran non-parametric tests for between subject comparisons, because of strongly skewed distributions of the scores (with skewness of −1.82, SE = 0.35, and kurtosis of 3.37, SE = 0.69). Mann–Whitney U test on the scores showed no reliable differences overall between the groups on the task (U = 197, p = 0.137; see Figure 6). In order to investigate whether individuals with HFA showed selective difficulties with only one and not all of the task conditions, we calculated the proportions of correct responses for each condition. Mann–Whitney U tests revealed a significant difference between the groups in the egocentric (viewer centred) condition (U = 357, p = 0.008), with the HFA group scoring significantly lower compared with the TD group (note, however, that HFA participants scored still around 90% in that condition, which indicates an overall successful performance in that condition despite observed group difference). None of the other comparisons reached significance. 1
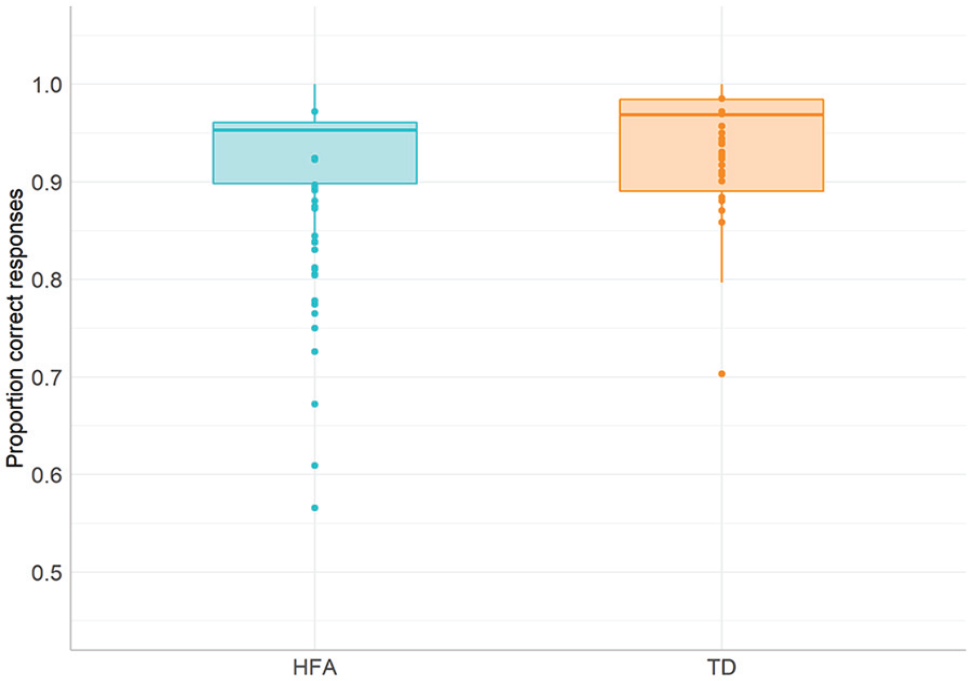
Average accuracy scores in HFA and TD groups in the Rotating Board Spatial Referencing task.
In order to identify what types of errors contributed to the group differences in the egocentric condition, we identified the items with the highest discrepancy in the number of errors between the groups. Items ‘in front of’, ‘in front of and to the left of’ and ‘behind’ showed the highest percentage of errors in the HFA group (13%, 13% and 9%, respectively), while TD participants’ scores were at ceiling. Qualitative analysis of the incorrect answers revealed that participants with HFA interpreted the direction within front/back axis differently compared with the TD group. Instead of using the mirror reflection of the axis, where Front is on the same side of the reference object as the viewer, a translation strategy was used, where Front is placed on the opposite side of the reference object. 2 However, this strategy was not applied systematically throughout this condition.
Performance predictors
Finally, we investigated whether the level of autism spectrum traits, as measured by the scores obtained on the AQ/CAST questionnaires, or participants’ age could account for the differences in task performance (see Figure 7). We ran multiple regression analyses with the average proportion of AQ/CAST scores and age as predictors, separately for the scores in the SNT, SVM and RBSR tasks. The results of the regression analyses indicated that the two predictors explained 40% of the variance in the SNT task (R2 = 0.396, F(2,46) = 15.1, p < 0.001), with both AQ/CAST score (β = –0.19, t = –4.96, p < 0.001) and age (β = 0.006, t = 3.22, p = 0.002) significantly predicting task performance.
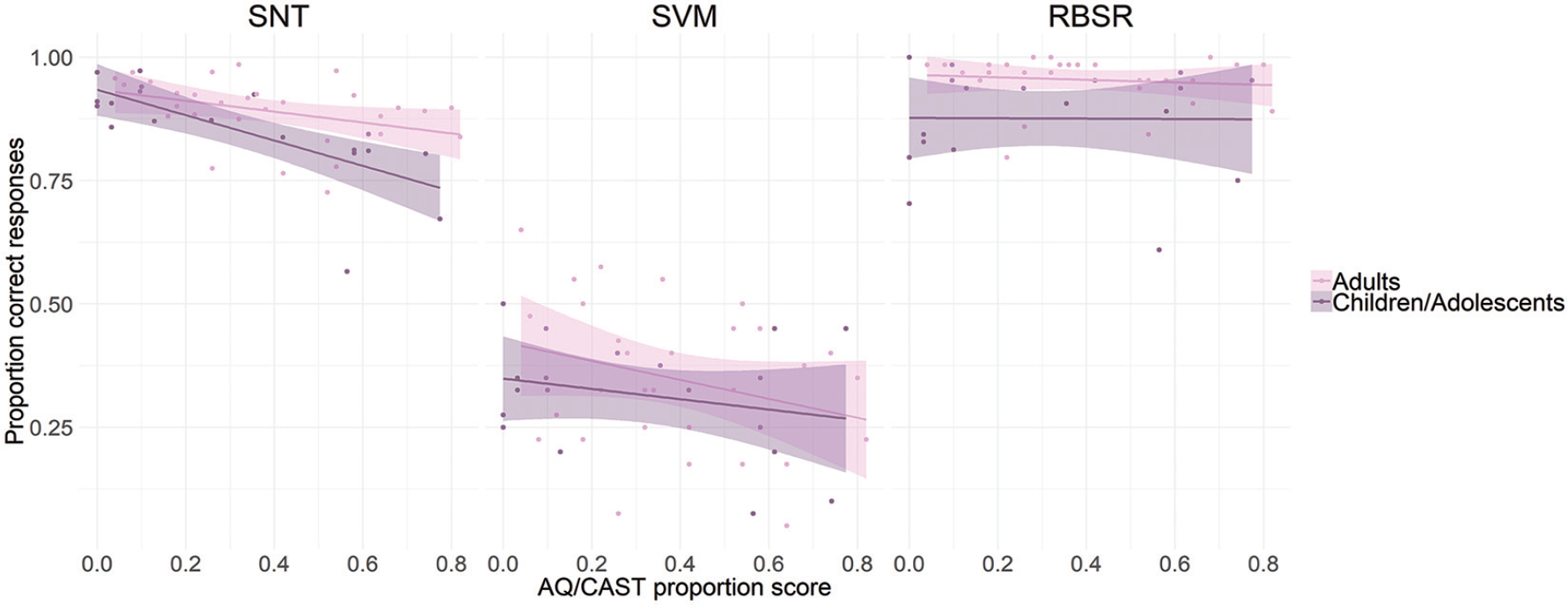
The relationship between task accuracy and proportion obtained score in the AQ/CAST questionnaire. AQ/CAST scores significantly predicted performance in the SNT and SVM task (left and middle panel) but not in the RBSR task (right panel). The relationship has been plotted separately for two age groups (for visualization purposes only): children/adolescents (9–17 years) and adults (18–27 years); shaded area represents ±2 SEM.
In the SVM task (Spatial Delayed condition), the model accounted for 14% of the variance (R2 = 0.142, F(2,46) = 3.55, p = 0.037), where only AQ/CAST score (β = –0.20, t = –2.58, p = 0.013), but not age (β = 0.003, t = 1.10, p = 0.278), significantly predicted task performance.
In the RBSR task, the model explained 11% of the variance (R2 = 0.111, F(2,43) = 2.7, p < 0.079), where only age (β = 0.005, t = 2.3, p = 0.026), and not AQ/CAST score (β = –0.009, t = –0.173, p = 0.864), accounted for task performance. In sum, the level of autism spectrum traits predicted performance in the SNT and SVM task and age-predicted participants’ performance in the SNT and RBSR task.
Discussion
The current study revealed selective difficulties in HFA in the spatial language domain. Specifically, individuals with HFA scored lower than controls on spatial language production and spatial description recall; however, these difficulties were not distributed uniformly but rather clustered in the areas of projective prepositions (left/right, front/back), source path terms (out of/down off), proximal terms (near/far) and delayed recall of spatial content. These findings provide the first evidence for selective deficits in a broad range of spatial language abilities in ASD that are also positively associated with autism spectrum traits. Finally, contrary to previous evidence suggesting difficulties with perspective taking in ASD (Pearson et al., 2013; Shield et al., 2016), the HFA participants in the current study showed overall similar performance to controls on the RBSR task.
Our results provide new, clinically relevant insights into the characteristics of the linguistic profiles in HFA. The findings indicate selective deficits in the spatial language domain, which lead to an uneven profile that resembles the uneven profiles reported previously for other aspects of cognition and language in HFA. For example, despite their relatively large vocabularies, individuals with HFA have been shown to display selective deficits in lexical processing (e.g. the use of idiosyncratic meanings and the absence of a shape bias in word learning, see Tek et al., 2008; Tek & Naigles, 2017; Volden & Lord, 1991). Similarly, individuals with HFA were shown to master most aspects of grammar but nevertheless display subtle difficulties with some syntactic elements, such as double complement constructions or personal and reflexive pronouns (Eigsti et al., 2007; Janke & Perovic, 2015, 2017; Kjelgaard & Tager-Flusberg, 2001; Perovic et al., 2013a, 2013b). Such selective and nuanced deficits in language are often disregarded in clinical work and absent from intervention targets, as they seemingly do not have a significant effect on the everyday functioning of individuals with HFA. Even though these difficulties often do not pass the impairment threshold in standardized tests, they should be nevertheless addressed in both clinical work and basic research, as they can have a profound impact on the overall functioning of individuals with HFA (Eigsti & Schuh, 2017), and they are often one of the strongest predictors of outcomes even on the high end of the spectrum (Szatmari et al., 2003).
Beyond their obvious clinical significance, the current results also carry significant theoretical implications. First, they provide new evidence for a dissociative nature of spatial language, that is, a possibility for impairment in some subsystems (e.g. production of projective, proximal and source path terms), while other subsystems remain intact. Second, they can give new insights into possible mechanisms behind this selective breakdown. Here, a critical question is whether one developmental mechanism, such as a developmental delay (i.e. a later acquisition of certain spatial language abilities compared with the timing in typical development) or arrest (i.e. a failure to acquire certain spatial language abilities at any point in development) could account for all selective deficits in spatial language in HFA.
Many of the deficits we observed in our HFA population corresponded to late-emerging abilities in spatial language and show parallels with other developmental disorders, for example, problems with projective prepositions and source paths have been previously observed in both younger children and individuals with Williams syndrome (Durkin, 1981; Harris, 1972; Johnston, 1984; Landau & Hoffman, 2005; Landau & Lakusta, 2006; Landau & Zukowski, 2003). This can indicate a delay in the mastery of terms, which are acquired later in development and which appear more challenging in the type of mapping between the language and the visuospatial representations. In further support of the delay hypothesis, the present study showed that both AQ/CAST scores and age predicted participants’ production of spatial terms. This suggests that irrespective of the intensity of autism spectrum traits within our population, performance could still improve with age. Accordingly, as relational memory (e.g. binding objects and locations in memory) shows a protracted development but single-item memory develops early (Ngo et al., 2017), the difficulties we observed in the recall of spatial descriptions could also result from a developmental delay or arrest. Interestingly, we found significant group differences only in the delayed, and not immediate, recall of spatial content. This difference was not accounted for by general working memory abilities in the HFA group. One possible explanation for this finding is that as verbal and visuospatial components are involved in memorizing spatial descriptions, but to different degrees (Brunyé & Taylor, 2008; De Beni et al., 2005), the participants with HFA might have engaged verbal working memory in the immediate recall (see Williams et al., 2005), while only delayed recall relied on relational memory. As a result, only delayed recall of spatial content posed difficulties in the HFA group as it relied on a cognitive ability that is acquired later in development. This would indicate that, even though individuals with HFA might not differ in their overall working memory abilities from the TD individuals, they might show difficulties in binding separate items in memory from language, an ability that is foundational to encoding and retrieving relational information from spatial language.
However, some of the observed deficits, such as the omissions of proximal terms and the lack of group differences in the RBSR, are difficult to reconcile with the developmental delay hypothesis. Proximal terms (near/far) are acquired relatively early in development and often even spontaneously chosen over projective prepositions by Williams syndrome individuals or typical children (Landau & Hoffman, 2005). Thus, observed omissions of proximal terms in the HFA group might result from a different mechanism, for example, a failure to attend to the distance in the spatial configuration between the located and the reference object (or interpreting it as less salient). Alternatively, the omissions might also point to a more specific problem with proximity in ASD (cf. the use of here and this for distal locations in ASD; Hobson et al., 2010). Given that proximal terms are imprecise (not defining the exact distance from the reference object), highly context-dependent and subjectively evaluated (Durkin, 1981), they might pose a particular challenge in ASD (Jolliffe & Baron-Cohen, 1999; Lopez & Leekam, 2003; Vermeulen, 2015). Another possibility is that, in the current study, the proximity items required more advanced use of proximal terms. Indeed, it has been previously shown that the use of near and far changes over time and reaches adult-like level only later in development, showing a surprisingly protracted learning trajectory (Durkin, 1981).
Different mechanisms could also account for the lack of group differences in the RBSR task in the present study. One possibility is that the HFA group compensated by applying an inversion strategy (i.e. inverting the left-right and front-back axis), instead of rotating their mental position – a type of strategy observed in children in mental rotation tasks, which facilitates performance (see Vander Heyden et al., 2017). In this way, participants could arrive at the normalized response by applying alternative strategies, a mechanism previously observed in HFA with ‘optimal outcomes’ (e.g. Eigsti et al., 2016). Another possibility is that, as language codes space in a coarse manner as opposed to detailed perceptual coding (see Jackendoff, 1983; Landau & Jackendoff, 1993), the comprehension of spatial terms can, in fact, be easier than fine-grained visual comparisons in different spatial perspectives. This interpretation could explain the differences between the current study, which tested linguistic performance in a perspective taking task, and other studies that tested non-linguistic perspective taking in HFA (Pearson et al., 2013; Ring, Gaigg, Altgassen, et al., 2018; Shield et al., 2016). Since the current results do not provide sufficient evidence to resolve between these possibilities, future research should use more fine-grained measures, which tap into the online processes involved in the task.
In sum, the present findings not only provide novel evidence for spatial language difficulties in individuals on the autism spectrum but also indicate a dissociative rather than uniform nature of these difficulties and point to several possible (but not mutually exclusive) mechanisms underlying this selective breakdown. That is, some of the observed deficits could be accounted for by a developmental delay hypothesis, while others most probably result from different mechanisms, such as impairments in integrating linguistic and non-linguistic information or deficits in spatial cognition.
These findings suggest intriguing questions for further research. Specifically, do some of the observed deficits in spatial language in HFA reflect an underlying deficit in spatial cognition or arise on the intersection of language and cognition? Furthermore, what regulates the integration of linguistic and non-linguistic spatial information and which aspects of this process (if any) are affected in HFA? Finally, to what extent are some of observed deficits, such as difficulties with proximity terms, specific to ASD? Although further research is necessary, the current study provides the first evidence for a range of selective difficulties in spatial language in HFA, revealing the dissociative nature of the spatial language domain as well as yielding important insights for future clinical work.
Footnotes
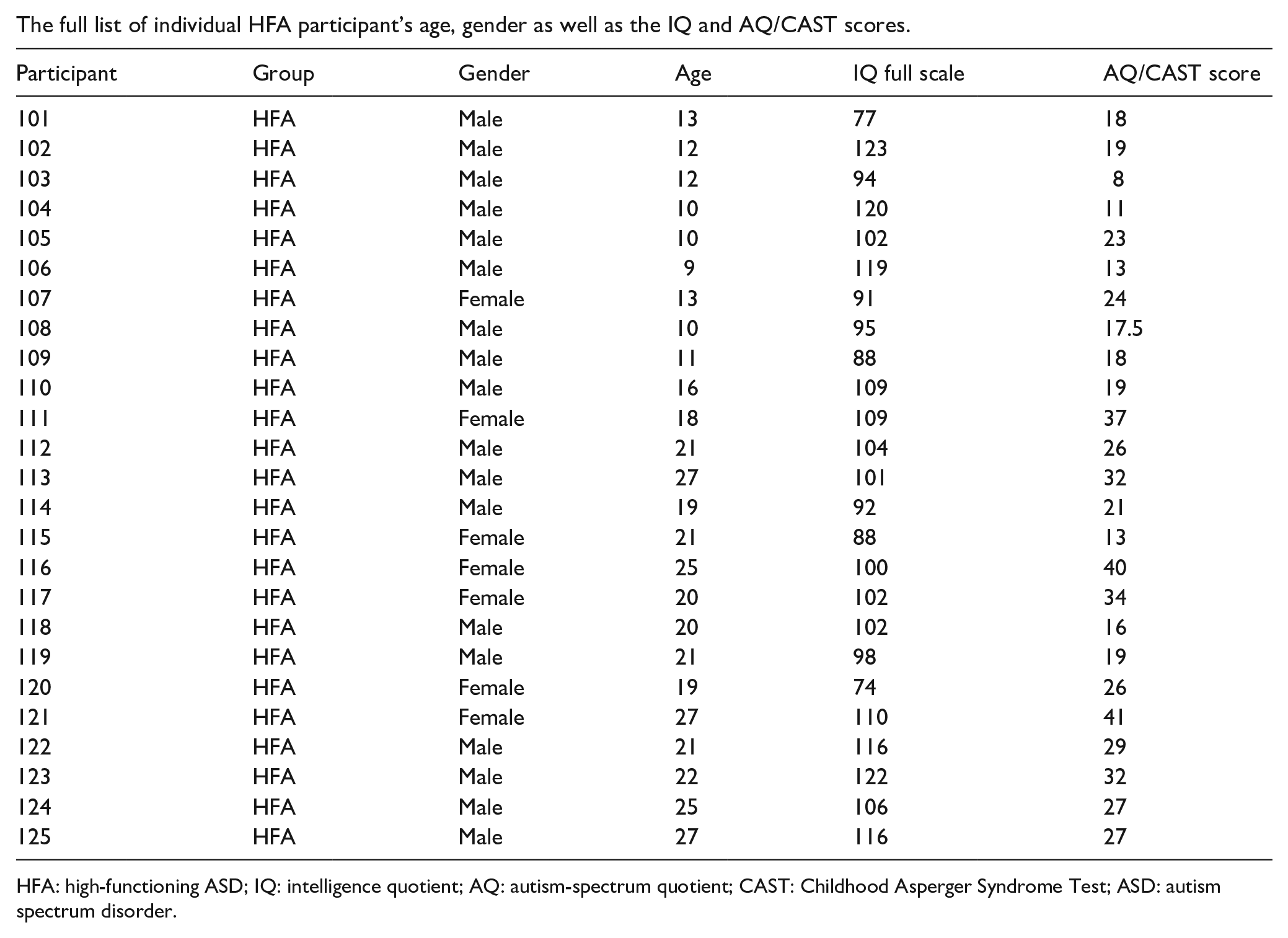
Appendix 1
The full list of individual HFA participant’s age, gender as well as the IQ and AQ/CAST scores.
| Participant | Group | Gender | Age | IQ full scale | AQ/CAST score |
|---|---|---|---|---|---|
| 101 | HFA | Male | 13 | 77 | 18 |
| 102 | HFA | Male | 12 | 123 | 19 |
| 103 | HFA | Male | 12 | 94 | 8 |
| 104 | HFA | Male | 10 | 120 | 11 |
| 105 | HFA | Male | 10 | 102 | 23 |
| 106 | HFA | Male | 9 | 119 | 13 |
| 107 | HFA | Female | 13 | 91 | 24 |
| 108 | HFA | Male | 10 | 95 | 17.5 |
| 109 | HFA | Male | 11 | 88 | 18 |
| 110 | HFA | Male | 16 | 109 | 19 |
| 111 | HFA | Female | 18 | 109 | 37 |
| 112 | HFA | Male | 21 | 104 | 26 |
| 113 | HFA | Male | 27 | 101 | 32 |
| 114 | HFA | Male | 19 | 92 | 21 |
| 115 | HFA | Female | 21 | 88 | 13 |
| 116 | HFA | Female | 25 | 100 | 40 |
| 117 | HFA | Female | 20 | 102 | 34 |
| 118 | HFA | Male | 20 | 102 | 16 |
| 119 | HFA | Male | 21 | 98 | 19 |
| 120 | HFA | Female | 19 | 74 | 26 |
| 121 | HFA | Female | 27 | 110 | 41 |
| 122 | HFA | Male | 21 | 116 | 29 |
| 123 | HFA | Male | 22 | 122 | 32 |
| 124 | HFA | Male | 25 | 106 | 27 |
| 125 | HFA | Male | 27 | 116 | 27 |
HFA: high-functioning ASD; IQ: intelligence quotient; AQ: autism-spectrum quotient; CAST: Childhood Asperger Syndrome Test; ASD: autism spectrum disorder.
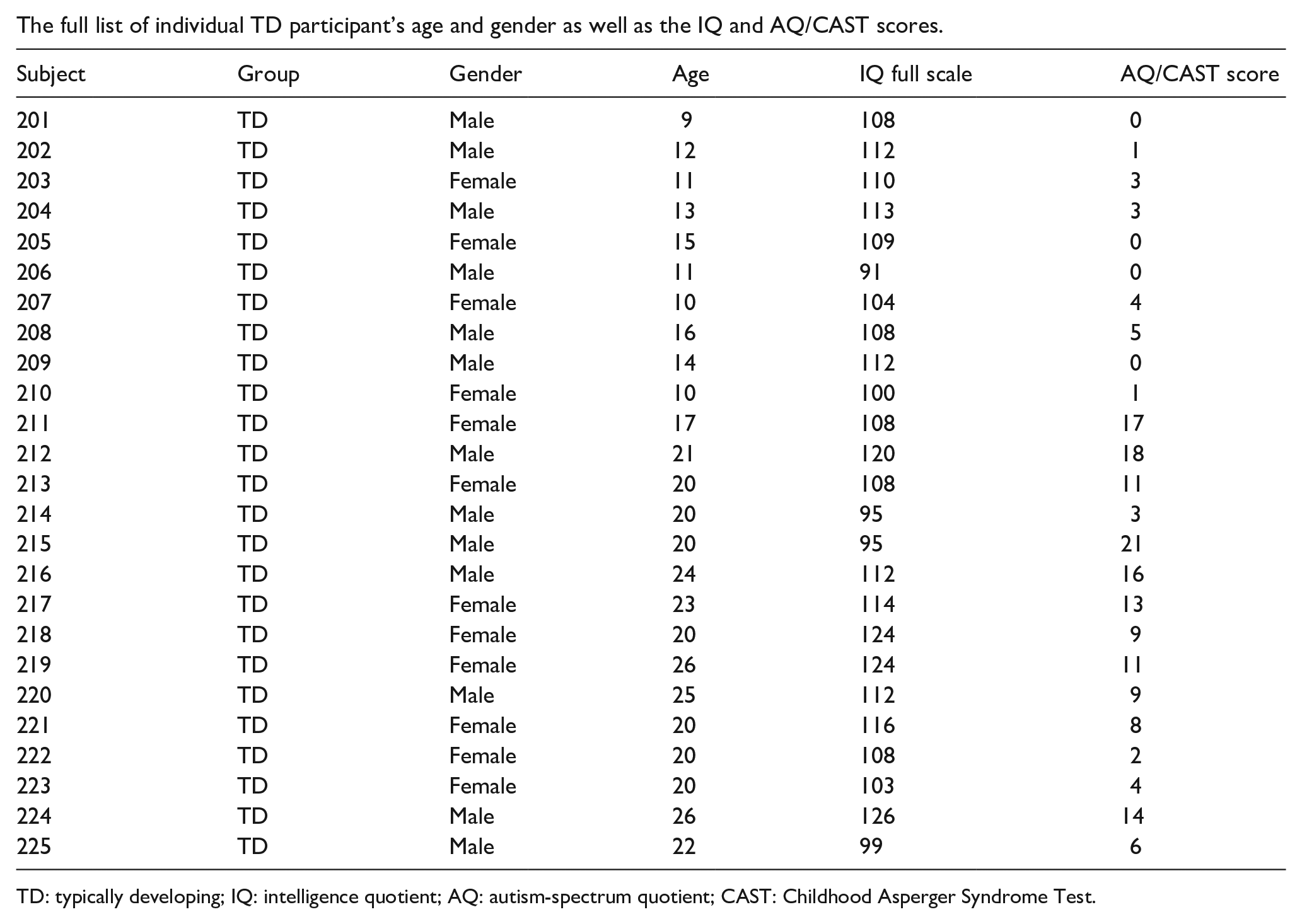
Appendix 2
The full list of individual TD participant’s age and gender as well as the IQ and AQ/CAST scores.
| Subject | Group | Gender | Age | IQ full scale | AQ/CAST score |
|---|---|---|---|---|---|
| 201 | TD | Male | 9 | 108 | 0 |
| 202 | TD | Male | 12 | 112 | 1 |
| 203 | TD | Female | 11 | 110 | 3 |
| 204 | TD | Male | 13 | 113 | 3 |
| 205 | TD | Female | 15 | 109 | 0 |
| 206 | TD | Male | 11 | 91 | 0 |
| 207 | TD | Female | 10 | 104 | 4 |
| 208 | TD | Male | 16 | 108 | 5 |
| 209 | TD | Male | 14 | 112 | 0 |
| 210 | TD | Female | 10 | 100 | 1 |
| 211 | TD | Female | 17 | 108 | 17 |
| 212 | TD | Male | 21 | 120 | 18 |
| 213 | TD | Female | 20 | 108 | 11 |
| 214 | TD | Male | 20 | 95 | 3 |
| 215 | TD | Male | 20 | 95 | 21 |
| 216 | TD | Male | 24 | 112 | 16 |
| 217 | TD | Female | 23 | 114 | 13 |
| 218 | TD | Female | 20 | 124 | 9 |
| 219 | TD | Female | 26 | 124 | 11 |
| 220 | TD | Male | 25 | 112 | 9 |
| 221 | TD | Female | 20 | 116 | 8 |
| 222 | TD | Female | 20 | 108 | 2 |
| 223 | TD | Female | 20 | 103 | 4 |
| 224 | TD | Male | 26 | 126 | 14 |
| 225 | TD | Male | 22 | 99 | 6 |
TD: typically developing; IQ: intelligence quotient; AQ: autism-spectrum quotient; CAST: Childhood Asperger Syndrome Test.
Appendix 3
Appendix 4
Proportions correct responses in the Spatial Naming Task per single item.

| Item | Target preposition | Group | N | Mean | SD | SE |
|---|---|---|---|---|---|---|
| A1 | i; inni [in] | HFA | 25 | 0.960 | 0.138 | 0.0277 |
| TD | 25 | 1.000 | 0.000 | 0.0000 | ||
| A2 | til høyre for [to the right of] | HFA | 25 | 1.000 | 0.000 | 0.0000 |
| TD | 25 | 1.000 | 0.000 | 0.0000 | ||
| A3 | på; oppå [on] | HFA | 25 | 1.000 | 0.000 | 0.0000 |
| TD | 25 | 1.000 | 0.000 | 0.0000 | ||
| A4 | over [above; over] | HFA | 25 | 0.840 | 0.374 | 0.0748 |
| TD | 25 | 0.980 | 0.100 | 0.0200 | ||
| A5 | bak [behind] | HFA | 25 | 0.880 | 0.332 | 0.0663 |
| TD | 25 | 0.960 | 0.200 | 0.0400 | ||
| A6 | under [under] | HFA | 25 | 1.000 | 0.000 | 0.0000 |
| TD | 25 | 1.000 | 0.000 | 0.0000 | ||
| A7 | under; langt under [below] | HFA | 25 | 0.920 | 0.277 | 0.0554 |
| TD | 25 | 0.980 | 0.100 | 0.0200 | ||
| A8 | foran [in front of] | HFA | 25 | 0.940 | 0.220 | 0.0440 |
| TD | 25 | 1.000 | 0.000 | 0.0000 | ||
| A9 | langt |
HFA | 20 | 0.900 | 0.308 | 0.0688 |
| TD | 25 | 0.880 | 0.332 | 0.0663 | ||
| A9 | HFA | 23 | 0.804 | 0.292 | 0.0608 | |
| TD | 24 | 0.979 | 0.102 | 0.0208 | ||
| A10 | nær |
HFA | 22 | 0.841 | 0.358 | 0.0764 |
| TD | 23 | 1.000 | 0.000 | 0.0000 | ||
| A10 | HFA | 21 | 0.810 | 0.370 | 0.0807 | |
| TD | 22 | 1.000 | 0.000 | 0.0000 | ||
| A11 | inntil |
HFA | 19 | 0.842 | 0.375 | 0.0859 |
| TD | 20 | 1.000 | 0.000 | 0.0000 | ||
| A11 | HFA | 23 | 0.848 | 0.279 | 0.0583 | |
| TD | 25 | 0.920 | 0.187 | 0.0374 | ||
| A12 | mellom [between] | HFA | 25 | 0.960 | 0.200 | 0.0400 |
| TD | 25 | 1.000 | 0.000 | 0.0000 | ||
| A13 | blant [among] | HFA | 25 | 0.300 | 0.323 | 0.0645 |
| TD | 25 | 0.400 | 0.382 | 0.0764 | ||
| A14 | i midten av [in the middle of] | HFA | 25 | 0.820 | 0.284 | 0.0569 |
| TD | 25 | 0.920 | 0.187 | 0.0374 | ||
| A15 | HFA | 23 | 0.609 | 0.476 | 0.0992 | |
| [ |
TD | 22 | 0.795 | 0.367 | 0.0783 | |
| foran; |
HFA | 17 | 0.529 | 0.329 | 0.0799 | |
| [in front of; |
TD | 23 | 0.826 | 0.324 | 0.0675 | |
| B1 | nedover [downwards] | HFA | 25 | 0.960 | 0.200 | 0.0400 |
| TD | 25 | 1.000 | 0.000 | 0.0000 | ||
| B2 | oppover [upwards] | HFA | 25 | 1.000 | 0.000 | 0.0000 |
| TD | 25 | 1.000 | 0.000 | 0.0000 | ||
| B3 | mot høyre [to the right of] | HFA | 25 | 0.960 | 0.138 | 0.0277 |
| TD | 25 | 1.000 | 0.000 | 0.0000 | ||
| B4 | tvers over (fra venstre mot høyre) | HFA | 25 | 0.880 | 0.299 | 0.0597 |
| (across (from the left to the right of)) | TD | 25 | 0.980 | 0.100 | 0.0200 | |
| B5 | inn i [into] | HFA | 25 | 0.920 | 0.277 | 0.0554 |
| TD | 25 | 0.980 | 0.100 | 0.0200 | ||
| B6 | ut av; ut fra [out of] | HFA | 25 | 0.800 | 0.382 | 0.0764 |
| TD | 25 | 0.960 | 0.200 | 0.0400 | ||
| B7 | bort fra; vekk fra [away from] | HFA | 25 | 0.460 | 0.455 | 0.0909 |
| TD | 25 | 0.740 | 0.357 | 0.0714 | ||
| B8 | rundt [around] | HFA | 25 | 1.000 | 0.000 | 0.0000 |
| TD | 25 | 0.960 | 0.200 | 0.0400 | ||
| B9 | over [over] | HFA | 25 | 1.000 | 0.000 | 0.0000 |
| TD | 25 | 1.000 | 0.000 | 0.0000 | ||
| B10 | under [under] | HFA | 25 | 0.920 | 0.277 | 0.0554 |
| TD | 25 | 0.960 | 0.200 | 0.0400 | ||
| B11 | gjennom [through] | HFA | 25 | 0.960 | 0.200 | 0.0400 |
| TD | 25 | 0.960 | 0.200 | 0.0400 | ||
| B12 | opp på [onto] | HFA | 25 | 0.760 | 0.411 | 0.0823 |
| TD | 25 | 0.920 | 0.236 | 0.0473 | ||
| B13 | ned av; ned fra [down off] | HFA | 25 | 0.760 | 0.293 | 0.0586 |
| TD | 25 | 0.920 | 0.187 | 0.0374 | ||
| B14 | langs (til venstre foran) (along (to the left in front of)) | HFA | 25 | 0.600 | 0.433 | 0.0866 |
| TD | 25 | 0.880 | 0.261 | 0.0523 | ||
| B15 | mot siden av; til sides for [towards the side of] | HFA | 25 | 0.440 | 0.441 | 0.0881 |
| TD | 25 | 0.660 | 0.426 | 0.0852 |
HFA: high-functioning ASD; TD: typically developing; ASD: autism spectrum disorder.
Note that some of the items were scored twice when it was possible to name two distinct components of a spatial relationship in the picture, for example, ‘far to the left side of’, with a proximity component ‘far’ and axial horizontal component ‘left side of’ (see e.g. item A9). In such cases, participants would get 1 point for correctly naming the proximity and 1 point for correctly identifying direction within horizontal axis. We observed highest differences in the scores in proximity components in items A9 and A10 (‘
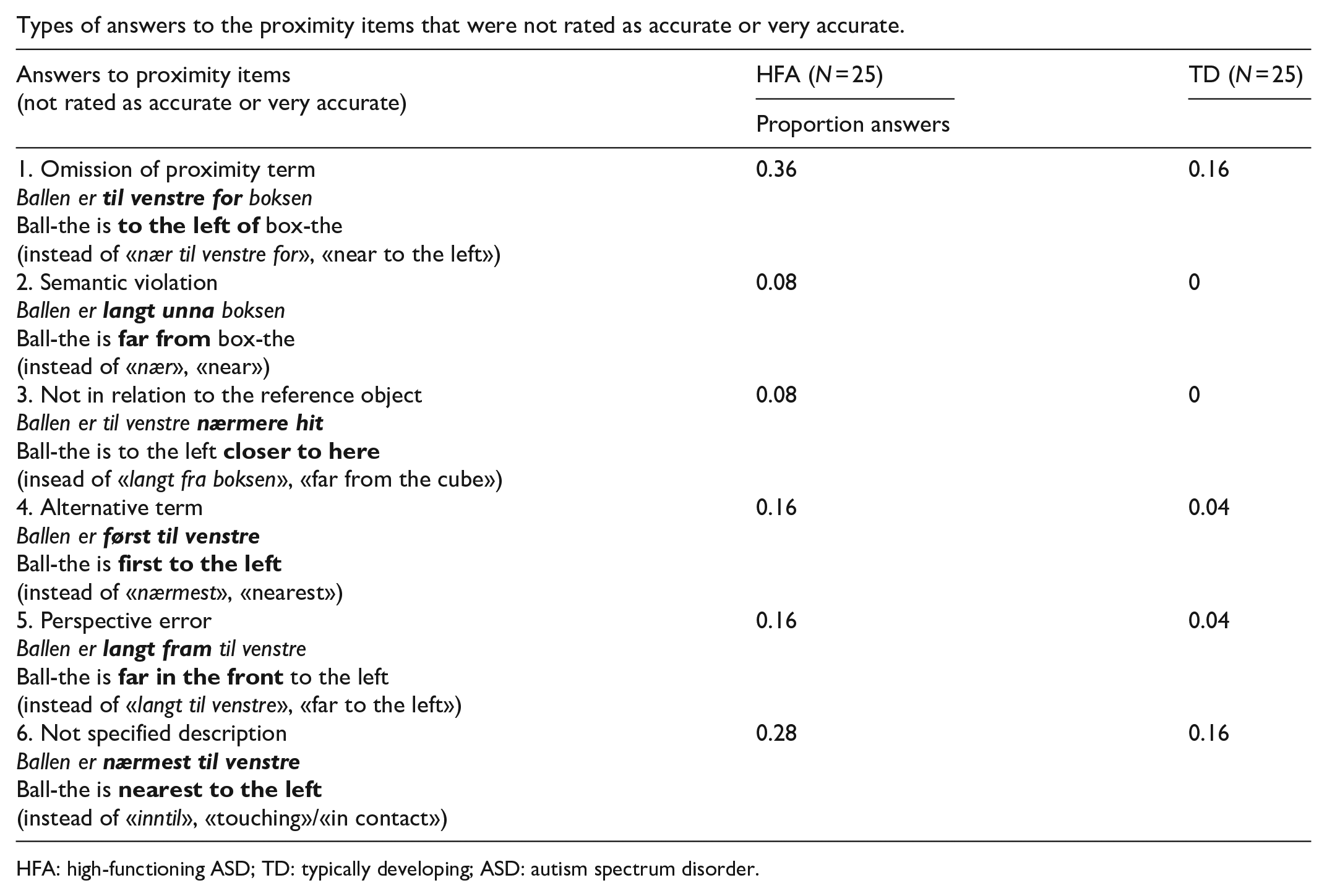
Appendix 5
Types of answers to the proximity items that were not rated as accurate or very accurate.
| Answers to proximity items |
HFA (N = 25) |
TD (N = 25) |
|---|---|---|
| Proportion answers | ||
| 1. Omission of proximity term Ballen er Ball-the is (instead of «nær til venstre for», «near to the left») |
0.36 | 0.16 |
| 2. Semantic violation Ballen er Ball-the is (instead of «nær», «near») |
0.08 | 0 |
| 3. Not in relation to the reference object Ballen er til venstre Ball-the is to the left (insead of «langt fra boksen», «far from the cube») |
0.08 | 0 |
| 4. Alternative term Ballen er Ball-the is (instead of «nærmest», «nearest») |
0.16 | 0.04 |
| 5. Perspective error Ballen er Ball-the is (instead of «langt til venstre», «far to the left») |
0.16 | 0.04 |
| 6. Not specified description Ballen er Ball-the is (instead of «inntil», «touching»/«in contact») |
0.28 | 0.16 |
HFA: high-functioning ASD; TD: typically developing; ASD: autism spectrum disorder.
Appendix 6
Types of answers to the source path items that were not rated as accurate or very accurate.
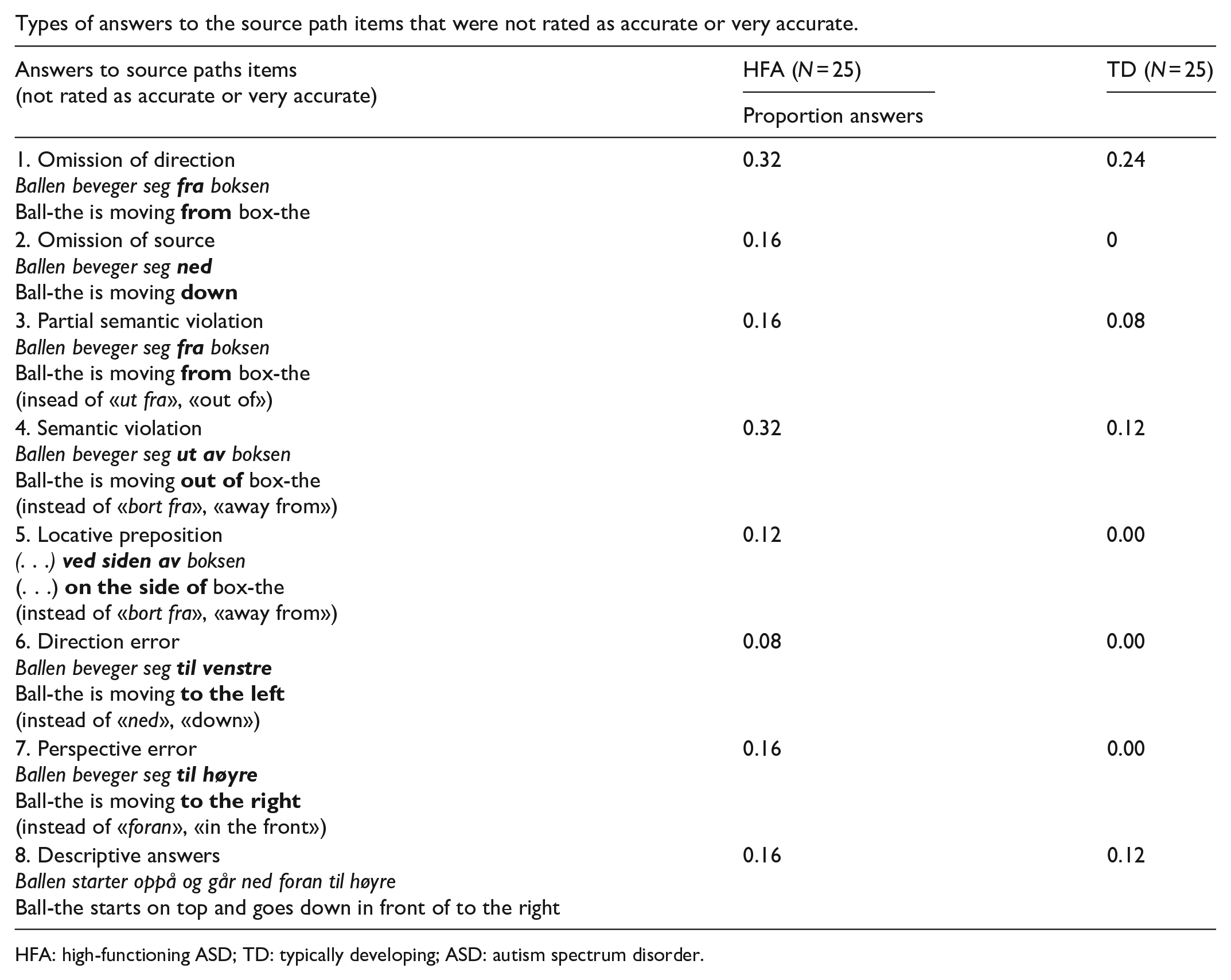
| Answers to source paths items |
HFA (N = 25) |
TD (N = 25) |
|---|---|---|
| Proportion answers | ||
| 1. Omission of direction Ballen beveger seg Ball-the is moving |
0.32 | 0.24 |
| 2. Omission of source Ballen beveger seg Ball-the is moving |
0.16 | 0 |
| 3. Partial semantic violation Ballen beveger seg Ball-the is moving (insead of «ut fra», «out of») |
0.16 | 0.08 |
| 4. Semantic violation Ballen beveger seg Ball-the is moving (instead of «bort fra», «away from») |
0.32 | 0.12 |
| 5. Locative preposition (. . .) (. . .) (instead of «bort fra», «away from») |
0.12 | 0.00 |
| 6. Direction error Ballen beveger seg Ball-the is moving (instead of «ned», «down») |
0.08 | 0.00 |
| 7. Perspective error Ballen beveger seg Ball-the is moving (instead of «foran», «in the front») |
0.16 | 0.00 |
| 8. Descriptive answers Ballen starter oppå og går ned foran til høyre Ball-the starts on top and goes down in front of to the right |
0.16 | 0.12 |
HFA: high-functioning ASD; TD: typically developing; ASD: autism spectrum disorder.
Acknowledgements
We are very grateful to all participants and their families for participating in the study as well as Autism Society in Norway for their assistance in the recruitment process. We would like to cordially thank Bruno Laeng for his invaluable help and support in data collection at the University of Oslo. We also thank Ioanna Markostamou for her helpful comments on the administration guidelines for the test battery. We would also like to acknowledge Randi Alice Nilsen, Lene Nysæter and Maria Boer Johannessen for their help with the ratings and transcriptions of the participants’ responses. Finally, we would like to thank Barbara Landau for her advice on the categorization of the items in the production task.
Author contributions
A.B., V.V. and M.V. adapted the experimental tasks with the help of K.R.C; A.B. carried out the experiment; A.B. and K.R.C. conducted the statistical analyses; A.B. wrote the manuscript with support from M.V., V.V. and K.R.C. All authors reviewed the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study was conducted in compliance with the Regional Committees for Medical and Health Research Ethics (REK) in Norway (reference number: 2015/1642; project title: “Spatial language and spatial cognition in Autism Spectrum Disorder”).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the research has been funded by the Norwegian University of Science and Technology (NTNU Trondheim, Norway). The authors have not received any other financial support for the research, authorship, and/or publication of this article.
