Abstract
This study investigated whether reduced visual attention to an observed action might account for altered imitation in autistic adults. A total of 22 autistic and 22 non-autistic adults observed and then imitated videos of a hand producing sequences of movements that differed in vertical elevation while their hand and eye movements were recorded. Participants first performed a block of imitation trials with general instructions to imitate the action. They then performed a second block with explicit instructions to attend closely to the characteristics of the movement. Imitation was quantified according to how much participants modulated their movement between the different heights of the observed movements. In the general instruction condition, the autistic group modulated their movements significantly less compared to the non-autistic group. However, following instructions to attend to the movement, the autistic group showed equivalent imitation modulation to the non-autistic group. Eye movement recording showed that the autistic group spent significantly less time looking at the hand movement for both instruction conditions. These findings show that visual attention contributes to altered voluntary imitation in autistic individuals and have implications for therapies involving imitation as well as for autistic people’s ability to understand the actions of others.
Autism spectrum condition (ASC), referred to herein as autism is a neurodevelopmental condition characterised by deficits in social communication and social interaction as well as repetitive and restricted behaviour (American Psychiatric Association, 2013). Voluntary imitation, the ability to purposefully copy another person’s actions is also altered, with consistent evidence of both reduced propensity to imitate and reduced accuracy of imitation in autistic children and adults (Edwards, 2014; Vivanti & Hamilton, 2014). Voluntary imitation is essential for learning new actions and behaviours from others, transferring cultural knowledge and strengthening affiliative bonds (Farmer, Ciaunica, & Hamilton, 2018; Nadel, 2014). Due to this link between imitation and social behaviours, it has been proposed that altered imitation in autistic individuals could contribute to some of their social deficits (Rogers & Pennington, 1991). Indeed, some emerging therapies use imitation training to improve broader social behaviours such as reciprocity, joint attention and using emotions in an interactive manner (Ingersoll, 2012; Ingersoll, Berger, Carlsen, & Hamlin, 2017). However, at present, the cause of altered imitation in autism remains unknown, limiting our understanding of why imitation training might be beneficial and how it can be refined or personalised. This study investigated whether reduced attention to the observed action might account for altered imitation in autistic adults.
A number of studies show that both autistic children and adults display a specific pattern of imitation: although they are more able to imitate the end goal of an action, they are less proficient at imitating the style or kinematics (e.g. amplitude and speed) of the action, particularly when a visual goal is absent or the action lacks meaning (Carmo, Rumiati, Siugzdaite, & Brambilla, 2013; Cossu et al., 2012; Hamilton, Brindley, & Frith, 2007; Hayes, Andrew, Elliott, Gowen, & Bennett, 2016; Hobson & Hobson, 2008; Hobson & Lee, 1999; Rogers, Bennetto, McEvoy, & Pennington, 1996; Rogers, Young, Cook, Giolzetti, & Ozonoff, 2010; Stone, Ousley, & Littleford, 1997; Vanvuchelen, Roeyers, & De Weerdt, 2007; Vivanti, Nadig, Ozonoff, & Rogers, 2008; Vivanti, Trembath, & Dissanayake, 2014; Wild, Poliakoff, Jerrison, & Gowen, 2012; Williams, Whiten, & Singh, 2004). This pattern was quantitatively demonstrated in our earlier work, where autistic and non-autistic participants observed and then imitated a video of a hand producing sequences of goal-directed (simple visual targets) and goal-less pointing movements that differed in speed, amplitude or elevation (Wild et al., 2012). Imitation was measured using motion tracking and quantified according to how much participants modulated their movement (e.g. the degree to which they changed the amplitude of their movement when observing elevated compared to direct hand movements). Compared to non-autistic participants, autistic individuals modulated their movements less, particularly for the goal-less condition. This pattern of results can be understood in the context of goal-directed theories of imitation. It has been suggested that there are two routes for imitation; a semantic route for meaningful, or goal-directed actions (processed by occipital-temporal areas, hippocampus and inferior parietal), and a direct route, for actions which are novel, or do not have a goal (processed by occipital-temporal areas and inferior frontal gyrus) (Hamilton, 2008; Rumiati & Tessari, 2002; Tessari, Canessa, Ukmar, & Rumiati, 2007). The goal-directed theory of imitation (GOADI; Bekkering, Wohlschlager, & Gattis, 2000; Wohlschlager, Gattis, & Bekkering, 2003) suggests that in order to imitate, the observer creates a cognitive hierarchy of goals for the action during observation and then performs an imitative action based on those goals. This implies that the end-point of an action would be preferentially imitated using the semantic route (i.e. the final goal of the action), whereas the way it is reached may not be imitated as well (i.e. the means or style of the action). In goal-less imitation, the movement style would move up the hierarchy to become a goal, leading to better imitation of movement kinematics as a result of visuomotor mapping of the action in the direct route (Wild, Poliakoff, Jerrison, & Gowen, 2010). Based on the dual route model, the pattern of imitation in autistic individuals showing relatively intact goal-directed imitation but reduced imitation of style suggests that the semantic route for imitation is functioning, but that it is the direct visuomotor route which is impaired as detailed in a model proposed by Hamilton (2008).
These findings raise the possibility that imitation deficits could be due to altered processes involved in visuomotor integration (VMI) 1 , consistent with the common occurrence of motor difficulties in autistic individuals (Gowen & Hamilton, 2013). In such a case, one might expect motor and imitation ability to be correlated and for motor ability to explain much of the imitation deficit. Although correlations between imitation and various measures of motor ability have been reported in autistic participants (Biscaldi et al., 2015; Dadgar et al., 2017; Smith & Bryson, 1998; Stieglitz Ham, Corley, Rajendran, Carletta, & Swanson, 2008; Vanvuchelen et al., 2007) when motor difficulties are controlled for or entered as a variable in a regression model, they do not fully account for imitation impairments (Dewey, Cantell, & Crawford, 2007; Dowell, Mahone, & Mostofsky, 2009; Dziuk et al., 2007; Macneil & Mostofsky, 2012; Rogers et al., 1996; Rogers, Hepburn, Stackhouse, & Wehner, 2003; Rogers et al., 2010; Stieglitz Ham et al., 2011; Vivanti et al., 2008; Wild et al., 2012). While a direct test of the role of VMI in imitation deficits remains to be conducted, it seems likely that other factors could also influence imitation accuracy in autistic individuals.
One factor that has been shown to influence imitation in non-autistic individuals is visual attention towards the action with studies showing that directing participant attention to the observed movement improves accuracy of imitation (Hayes, Roberts, Elliott, & Bennett, 2014; Janelle, Champenoy, Coombes, & Mousseau, 2003). Using a similar imitation paradigm to our earlier work (Wild et al., 2012), we investigated the effect of instructing non-autistic participants to attend to the observed hand movement (Bek, Poliakoff, Marshall, Trueman, & Gowen, 2016). Participants first performed a block of imitation trials where general instructions to imitate the action were given. They then performed a second block where they were explicitly instructed to attend closely to the characteristics of the movement (attention group) or received no further instructions (control group). Imitated movements in the attention group were closer in duration, peak velocity and amplitude to the observed model compared with the control group.
Eye tracking findings from our previous work (Wild et al., 2012) suggest that reduced imitation of movement kinematics could result from decreased visual attention to, and tracking of, the movement to be imitated. Compared to the non-autistic group, the autistic group spent relatively more time gazing at goals then the area between the goals where the hand movement occurred, together with less pursuit of the hand movement compared to the non-autistic group, although this was for a small sample only (
The aim of this work was to examine the role of visual attention on the degree of voluntary imitation in autistic adults by (1) comparing imitation performance between two instruction conditions (general vs attention) and (2) recording where participants looked during observation of the action to be imitated. We used a similar paradigm to in our previous work (Wild et al., 2012) where participants observed, then imitated sequences of hand movements that varied in speed or vertical amplitude during a general and explicit attention instruction block. Imitation was measured by the degree to which participants modulated their movements between the different heights and speeds of the observed movements. If altered visual attention causes reduced imitation in autistic individuals, we would expect reduced modulation in the general instructions condition compared to the non-autistic group but similar levels of modulation across both groups in the attention condition. In regards to eye tracking, we would expect autistic participants to show less time fixating on the hand than the non-autistic group in the general instruction condition but to become more similar to the comparison group in the attention instruction. However, if altered VMI causes reduced imitation, one would expect that modulation should remain reduced for the autistic group in the attention condition, regardless of whether there is increased time looking at the hand in the attention condition.
Materials and methods
Stimuli and procedures were based on previous studies (Bek et al., 2016; Wild et al., 2012).
Participants
A total of 22 autistic and 22 typical participants matched on age, sex, handedness and full-scale intelligence quotient (IQ) were recruited through the laboratory database,
Participant demographics.
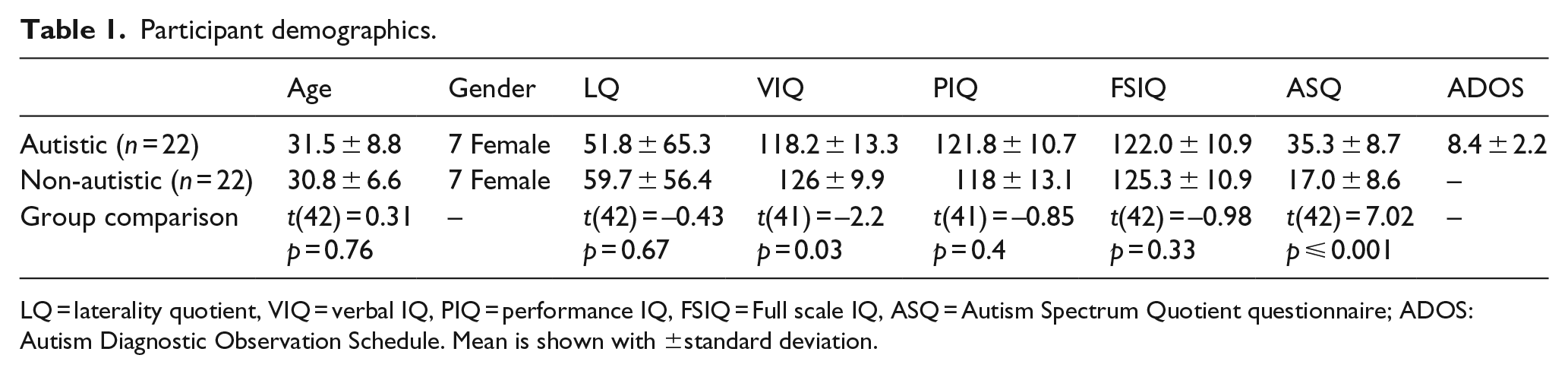
LQ = laterality quotient, VIQ = verbal IQ, PIQ = performance IQ, FSIQ = Full scale IQ, ASQ = Autism Spectrum Quotient questionnaire; ADOS: Autism Diagnostic Observation Schedule. Mean is shown with ±standard deviation.
Apparatus
Participants were seated at a wooden table in a lab cubicle facing the display screen (1920 × 1080 px, active viewing area – 52.8 cm × 26.9 cm), which was positioned 82.1 cm (±4.4 cm) from the participant’s eyes. A Polhemus FASTRAK motion tracker was used for kinematic data collection. A single motion sensor was attached to the distal phalange of the index finger of the dominant hand. Movement was sampled at 120 Hz in X, Y, Z coordinates. Participants kept their non-dominant hand on their knee under the table. An EyeLink 1000 Plus eye-tracker (SR research) collected eye movement data, using the desktop mounted, remote mode setting (head free-to-move) with a sampling frequency of 500 Hz and typical accuracy of 0.25–0.50.
Stimuli
Visual stimuli were presented using Experiment Builder (SR Research). Stimuli were identical to that used in previous work (Wild et al., 2012) and consisted of short video clips showing a human hand, termed the ‘demonstrator hand’, visible just beyond the wrist performing short pointing movement sequences with an index finger on a black surface (Figure 1(a)). There were four pointing locations 150 mm apart in a horizontal line (Figure 1(b)). In each video clip, the index finger was positioned on one of the four locations, then moved to a second location and then to a third location (two movements). In order to guide the demonstrator during filming, small marks that were not visible on camera were drawn onto the table surface. A metronome was used during recording to pace the demonstrator’s hand, but all clips were played silently during the experiment.
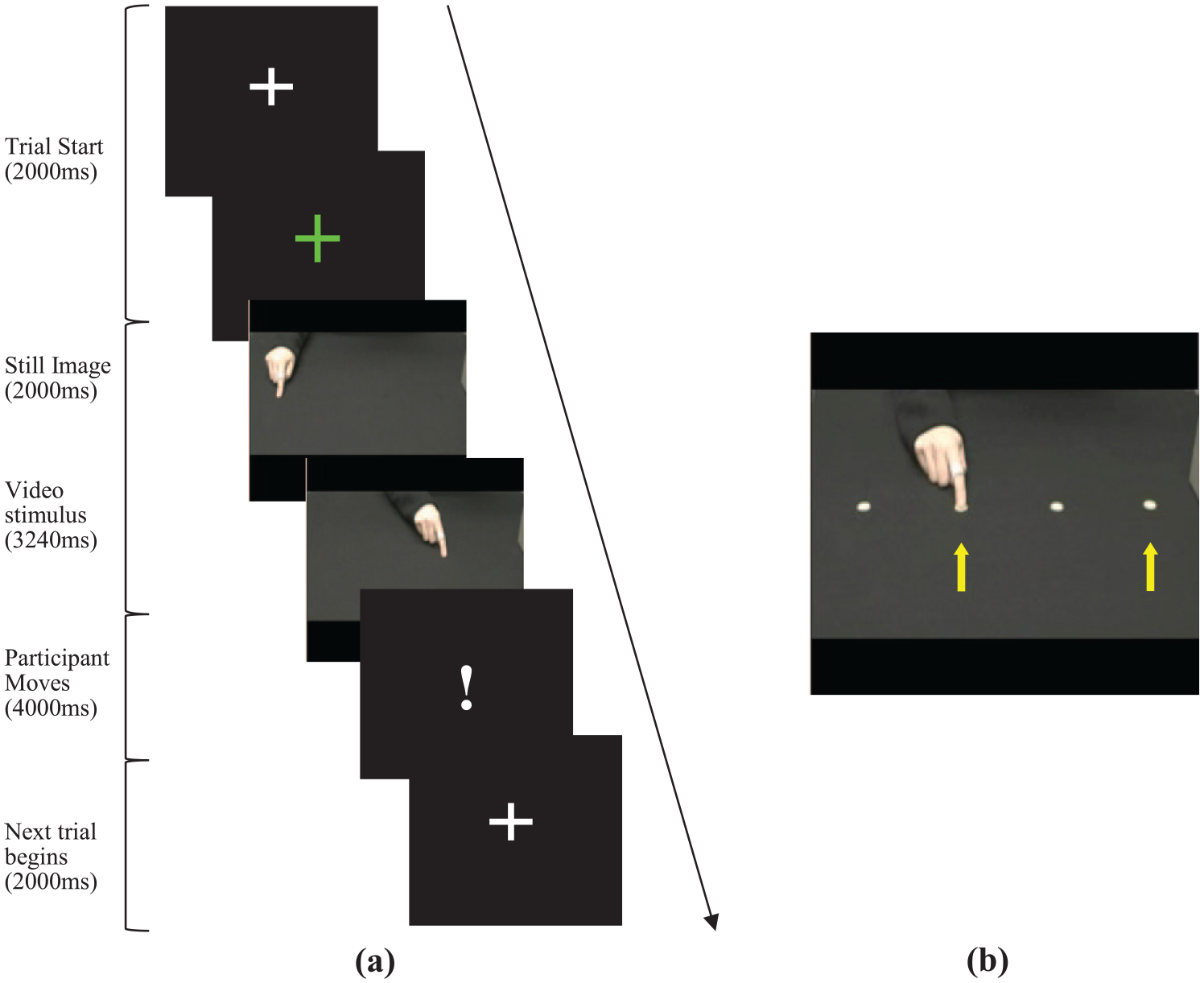
(a) Time-lapse diagram of experiment. Each trial began with a fixation cross, followed by a still image of a hand and a movie clip of a hand sequence. Participants imitated when the exclamation mark appeared. (b) Four pointing locations where movement sequences could begin and end. Hand and eye analysis was conducted for the movement between locations 2 and 4 only (yellow arrows). Note that visual targets are for description purposes only and were not displayed during the video clips.
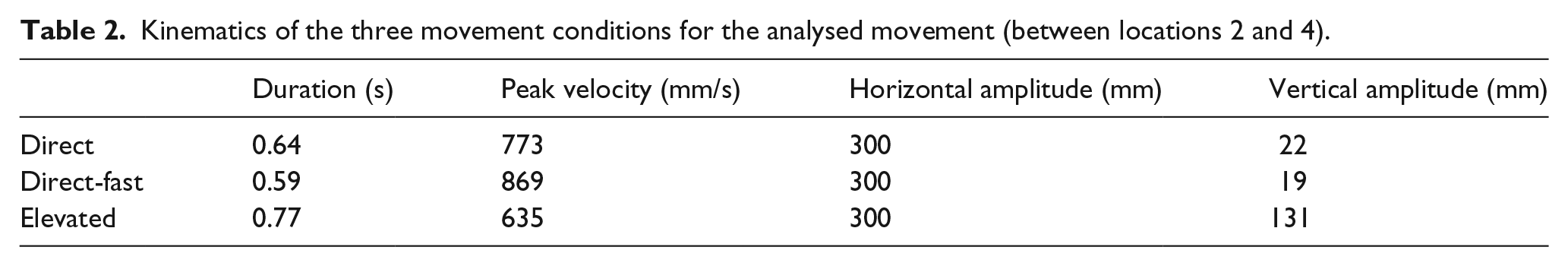
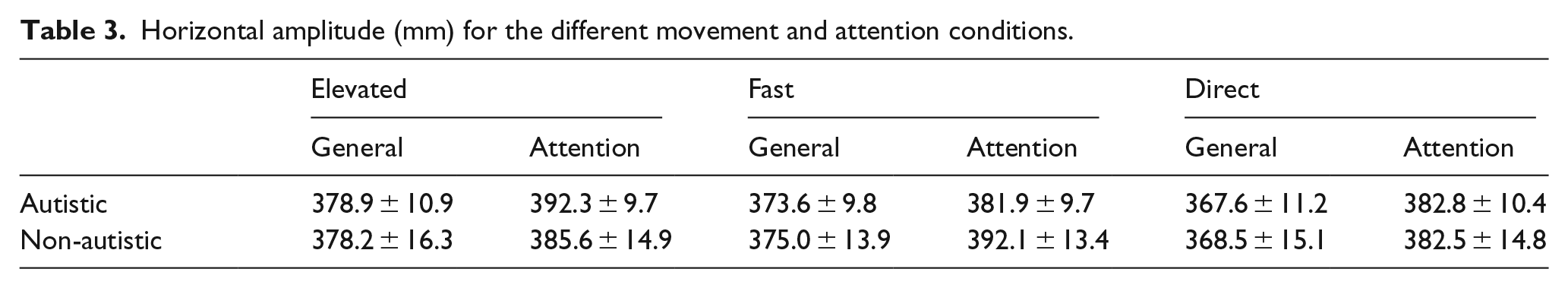
The kinematics of the demonstrator’s hand movements were also measured using Polhemus LIBERTY motion tracker during recording of the video clips, to allow a direct comparison between the demonstrator and participant imitation modulation. A single movement, between locations 2 and 4 (Figure 1(b)) was selected for kinematic and eye movement analyses. This movement was selected because this was the largest movement in the sequences, so produced more data to analyse. The hand in the video clips was recorded in three different conditions (elevated, direct and direct-fast) that differed according to the height and speed (Table 2).
Kinematics of the three movement conditions for the analysed movement (between locations 2 and 4).
Procedure
Each trial (Figure 1(a)) started with a white fixation cross that flashed green (2000 ms), followed by a still image of the hand in the start position of the movement sequence that was displayed for 2000 ms, allowing the participant to move their hand to the equivalent start location on the table. A video clip of the movement sequence was then played (3240 ms), followed by an exclamation mark coupled with a sound signal (beep, 1000 ms) signalling to the participants that they should imitate the movement.
The experiment was split into two blocks. Participants first completed a ‘general instruction’ block with the following instruction presented on the screen and read to them: ‘Watch the video clip carefully and then copy what you saw as best you can’. After that they completed an ‘attention instruction’ block, with the instruction to: Pay close attention to the specific movement made. For example, look at how fast the action is carried out, and the size of the movement, so how high the hand is lifted, and exactly where the movement occurs from and to. (Bek et al., 2016)
Each block consisted of 24 trials containing the 2–4 sequence and 12 oddball trials containing different movement sequences, to reduce predictability. The 24 2–4 trials were split into eight elevated, eight direct and eight direct-fast. The trials in each block were presented in a randomised order. During the experiment, eye movements of a single eye were tracked, and a 9-point calibration procedure was performed at the beginning and in the middle of each block.
Analysis
Kinematic data
Trials containing the 2–4 movement were analysed using MATLAB R2017a (Mathworks Inc). Movement data were filtered with a 120 Hz Butterworth filter and movement onset and offset times were determined when movement velocity rose above or fell below 10% of the peak velocity for six consecutive samples (48 ms). Movement amplitude, duration and peak velocity were calculated for each test trial. The level of imitation was measured as modulation, the degree to which participants adapted their movement elevation and speed to changes in elevation and speed in demonstrated videos. For example, to measure the amount of vertical amplitude modulation, the difference was calculated between direct and elevated trials. Similarly, for duration and velocity modulation, the difference between direct and direct-fast trials was calculated. This was calculated so that positive values resulted when vertical amplitude was greater in elevated than direct trials and when duration/velocity were, respectively, greater/lower in direct compared to direct-fast trials. This method was chosen as opposed to direct comparison with the features of the observed movement to reduce factors related to video presentation such as decreased visual information and between participant inconsistencies in interpreting imitation from video stimuli (e.g. some participants may match the spatial position of the hand on the screen, whereas others might copy the actual size of the movement regardless of spatial position on the screen).
Outlier removal was performed at the individual and group level and based on the non-recursive procedure recommended by Van Selst and Jolicoeur (1994). Almost 5% of trials (53 autistic, 54 non-autistic) were removed at the individual level and four participants removed at the group level (one autistic and one non-autistic for vertical modulation, one non-autistic for peak velocity modulation and one autistic participant for duration modulation). The two groups remained matched on age, sex, handedness and full scale IQ. Modulation was compared between participant groups and instruction block using a mixed ANOVA (group × instruction). The mean modulation for each group was also compared to the demonstrator’s modulation using one sample
Eye data
Eye data were analysed using Data Viewer (SR Research) and MATLAB R2015a (Mathworks Inc). Trials in which missing data (blinks and pupil/corneal reflection loss) were more than 1/3 of the total trial duration were removed from analyses. In addition, participants whose data contained more than 1/3 of removed trials were also removed from further analysis. This resulted in the removal of 5.8% of trials (97 in autistic and 26 in non-autistic participants) and the removal of five autistic participants. Reasons for missing data were reflection from glasses and substantial head movement. All eye data analysis was carried out collapsed across the different speed and elevation conditions and for the 2–4 movement only. For each of the below analyses, outlier removal was performed at the individual and group level (Van Selst & Jolicoeur, 1994). For percentage of time spent on the dynamic interest area (IA), this resulted in the removal of 23 (2.18%) autistic and 31 (2.94%) non-autistic trials and 2 non-autistic participants. For fixation distance from the IA, this resulted in the removal of 33 (3.13%) autistic and 35 (3.31%) non-autistic trials and 2 non-autistic participants. The two groups remained matched on age, sex, handedness and full-scale IQ after removal of outlier participants.
Percentage of time spent on dynamic IA
Visual attention to the moving hand was investigated by defining a dynamic circular IA that was constantly centred on the fingertip in the video throughout the movement (Figure 2). This enabled us to assess the location of participant’s eyes relative to the moving hand. The position and size of the IA was experimentally derived from the proportion of fixation duration to the finger and hand across participants [see Supplemental material, Appendix A for details of method]. It remained the same size for all participants and for all time points throughout the videos. The mean percentage time of eye gaze spent in this IA was calculated for each individual across trials and analysed using a mixed ANOVA (group × condition).
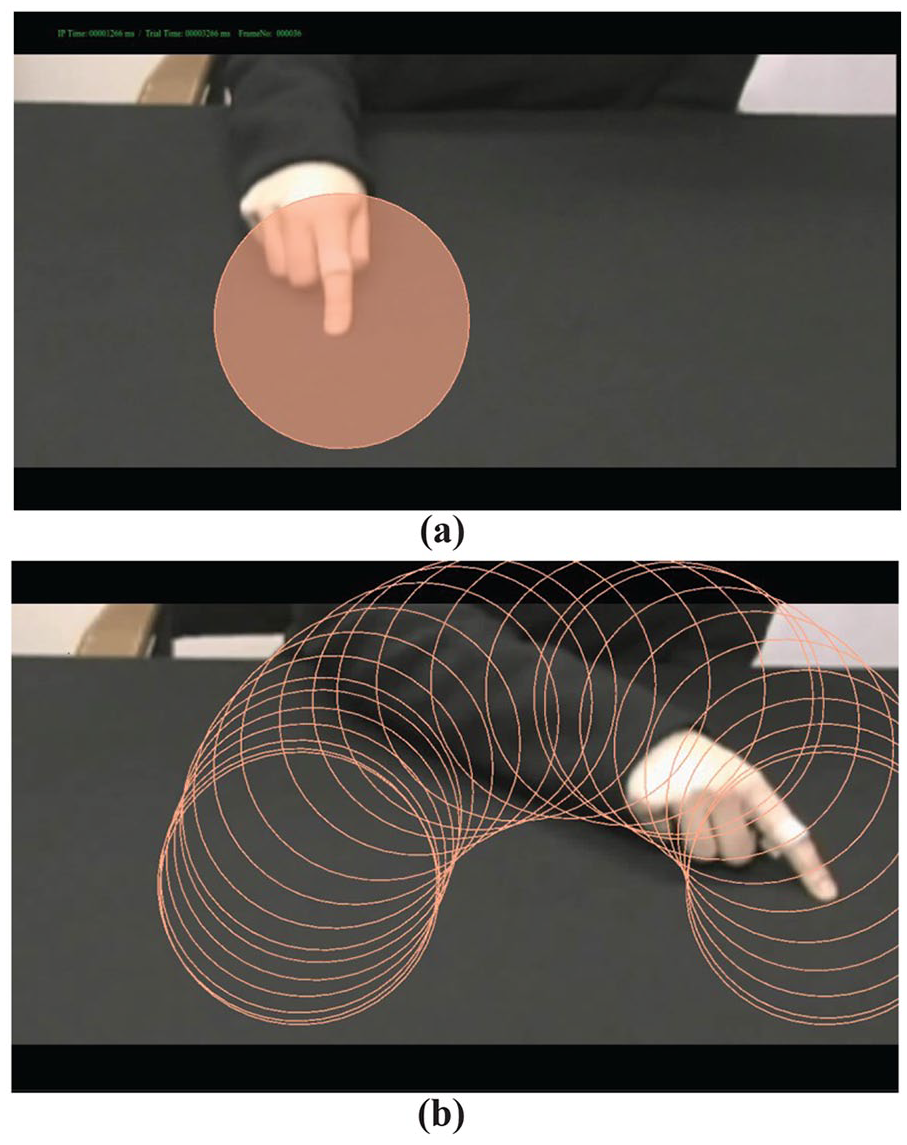
(a) Dynamic interest area (IA). It covered 14% of total screen area and had a mean radius of 5.2° of visual angle. (b) Path of dynamic IA during the 2–4 movement.
Fixation distance from centre of IA
When performing an IA analysis, some fixations fall near to the centre of the IA and some fall near the outside margin, and they are different in respect to the participant’s attention. Similarly, fixations outside the IA might be just next to it or further away. Therefore, an additional measure of fixation distance from the fingertip (centre of IA) was calculated in degrees and analysed using a mixed ANOVA (group × condition) to provide a fuller picture of the attention to the fingertip.
Comparison between kinematic and eye data
In order to assess whether imitation ability was related to visual attention, the difference between general and attention instructions was calculated for vertical modulation, the percentage of time spent on the dynamic interest area and the fixation distance from the centre of the IA. Correlations were performed between the vertical modulation change and the two eye movement measures. If the change in imitation ability is related to a change in visual attention to the hand, we would predict that more time spent on the dynamic interest area and gazing closer to the centre of the IA should positively correlate with larger vertical modulation. Due to the earlier outlier removal, this analysis was conducted on 16 autistic and 20 non-autistic participants. Correlations were performed across the two groups, as power was too low to perform separate group analysis.
Bayes factor analysis
We additionally calculated the Bayes factor for kinematic and eye data using JASP (JASP Team, 2018) with the default prior setting. The Bayes factor (BF10) expresses the relative evidence in support of the alternative hypothesis compared to the null hypothesis, given the observed data. BF10 ⩾ 3 suggests increasingly strong evidence in favour of the alternative hypothesis, while BF10 ⩽ 0.33 suggests increasingly strong evidence in favour of the null. When BF10 = 1, the data do not support either hypothesis (Wagenmakers et al., 2018). For analyses with >2 repeated factors, the BF inclusion factor is reported for interactions and can be interpreted similarly to BF10 (Wagenmakers et al., 2018). Bayesian statistics are particularly informative in exploring the extent to which data support the null hypothesis, which is not possible using frequentist statistics.
Community involvement statement
Autistic people and family members were involved in the design, interpretation and dissemination of the study, through the
Results
Kinematic data
Imitation modulation of duration and peak velocity was absent in both the non-autistic and autistic participants. Consequently these data, together with discussion around reasons for lack of modulation is included in Supplemental material, Appendix B only.
Vertical amplitude modulation (direct versus elevated)
Figure 3 shows how vertical height was modulated for the two instruction conditions and groups. It can be seen that the non-autistic group has greater vertical modulation than the non-autistic group, particularly for the general instruction condition. Levene’s test for equality of variances was not significant for either block (
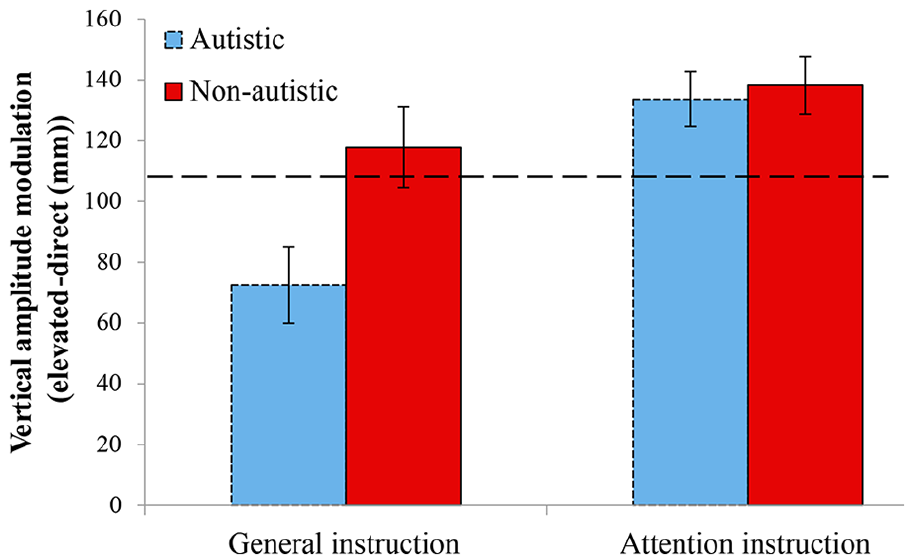
Imitation modulation for autistic (blue) and non-autistic (red) groups during the general and attention instruction conditions for vertical modulation. Error bars = standard error.
A mixed ANOVA (group × instruction) showed that the group effect was not significant (
Comparison to the demonstrator (110 mm) using one sample
Horizontal amplitude
Levene’s test for equality of variances was not significant for either block (
Horizontal amplitude (mm) for the different movement and attention conditions.
Eye data
Percentage of time on dynamic IA
Figure 4(a) shows that the autistic group spent less time dwelling on the finger IA compared to the non-autistic group. Levene’s test for equality of variances was significant for the general instruction block (
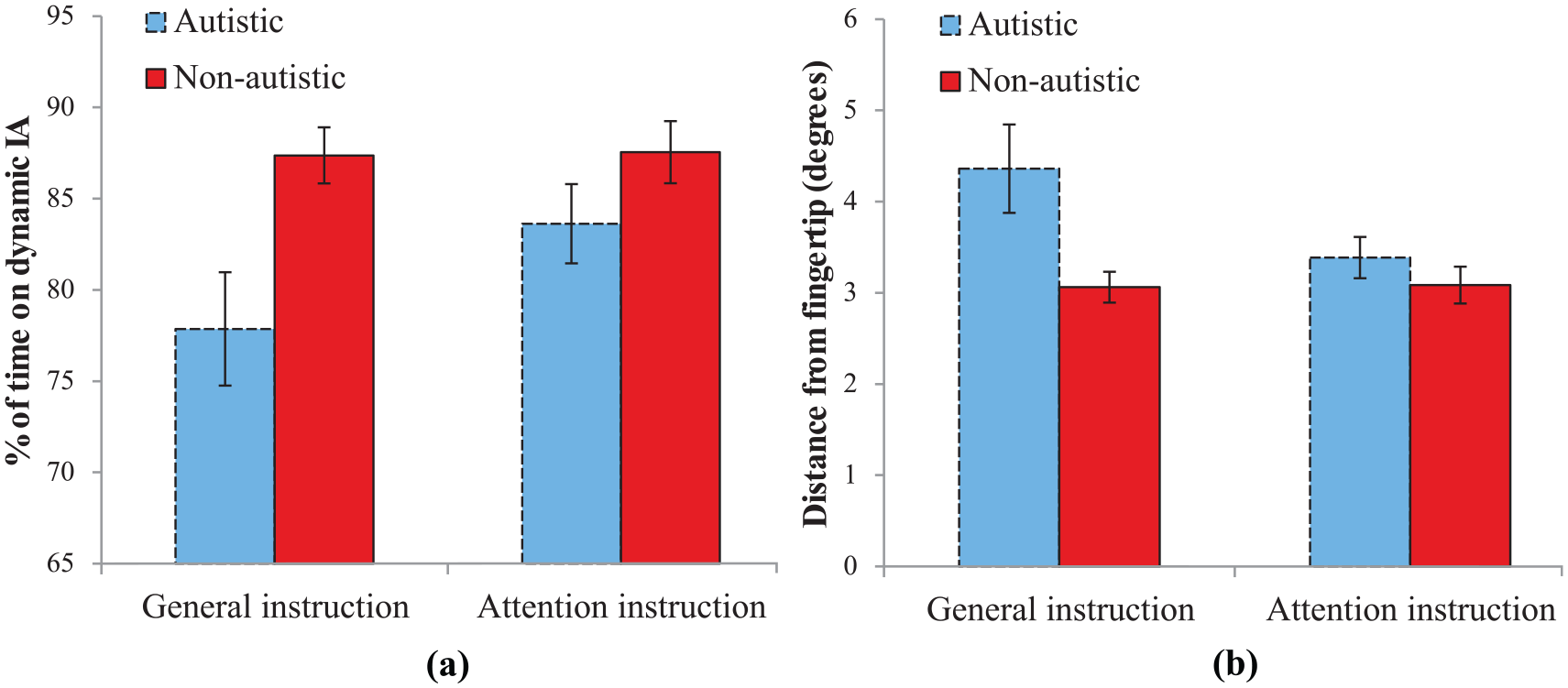
Results of eye analysis for autistic (blue) and non-autistic (red) groups during general and attention instructions: (a) percentage of time on dynamic IA and (b) distance from fingertip. Error bars = standard error.
A mixed ANOVA (group × instruction) showed a main effect of group (
Fixation distance from the centre of the IA (fingertip)
Fixation distance from the fingertip showed that the autistic group fixated further away than the non-autistic group and also decreased the distance to the fingertip between general and attention instructions condition (Figure 4(b)). Levene’s test for equality of variances was significant for the general instruction block (
The mixed ANOVA (group × instruction) confirmed that autistic participants fixated significantly further away from the fingertip than non-autistic participants (
Comparison between kinematic and eye data
A positive correlation was found across participants between the change in vertical modulation between general and attention instructions and the change in time spent on IA (Spearman’s Rho = 0.49,
Discussion
In this study, we examined how instructions to attend to an observed movement influenced imitation ability and eye gaze in autistic compared to non-autistic adults. Findings revealed that attention instructions increased the degree to which the autistic group modulated the amplitude of their own movement to levels that were similar to the non-autistic group. This is the first study to show that explicit instructions to attend to movement features of an observed action increased the level of imitation in autistic adults in a task where participants had already been asked to imitate the action. These findings have implications for understanding why imitation is affected in autistic individuals as well as for their ability to learn from and understand the actions of others.
In the general instruction condition, the autistic group modulated their movements significantly less between the observed direct and elevated movement compared to the non-autistic group (and the demonstrator movement), replicating previous findings demonstrating reduced imitation of action kinematics in autistic individuals. In contrast, the non-autistic group showed equivalent imitation modulation to the demonstrator, consistent with previous work showing high fidelity of imitation even for unnecessary or irrational actions such as our elevated movement (Forbes & Hamilton, 2017; McGuigan, Makinson, & Whiten, 2011; Wild et al., 2012). However, following instructions to attend to the movement, the autistic group showed equivalent imitation modulation to the non-autistic group. Moreover, both groups produced greater imitation modulation than the demonstrator in the attention condition, that is, they ‘overimitated’ the difference between the direct and vertical movement. These findings concur with studies demonstrating that the performance of autistic individuals across a range of tasks including false belief and automatic imitation tasks becomes similar to non-autistic individuals when accompanied by explicit instructions of what to do or where to look (Callenmark, Kjellin, Ronnqvist, & Bolte, 2014; McIntosh et al., 2006; Senju, 2013). Importantly, our results have identified that explicit instruction to attend to the kinematics of an observed movement increases imitation in autistic adults in a voluntary imitation task, where they have already been instructed to imitate as accurately as they can. This highlights that task instructions need to be highly specific for autistic individuals in order for them to perform in a similar way to non-autistic participants.
The eye movement findings partly support the hypothesis that imitation ability in autistic individuals is related to visual attention to the observed movements. Consistent with our previous smaller study (Wild et al., 2012), the autistic group spent significantly less time looking at the hand movement, and their gaze was significantly further away from the fingertip. Although non-significant, there was a clear pattern for the autistic group to increase the time spent looking at the hand and decrease the distance to the fingertip in the attention condition. The positive correlation between the change in vertical amplitude modulation and the change in percentage of time spent tracking the hand between the two instruction conditions further supports the idea that visual attention contributes towards imitation performance. It is possible that attention instructions alerted autistic participants to the relevant features, but they used a different strategy compared to the non-autistic group, where they tracked the movement less but still obtained enough visual information to increase imitation modulation. This may have been through the use of peripheral attention or the use of directed saccades to the relevant movement features (e.g. top of vertical movement). This possibility is supported by a meta-analysis identifying reduced smooth pursuit gain (ability to match eye velocity with target velocity) in autistic individuals (Johnson, Lum, Rinehart, & Fielding, 2016) together with our previous findings of less pursuit of the hand movement (Wild et al., 2012). Given our ambiguous findings, future work should investigate eye movement strategies further by examining smooth pursuit, saccades and fixations.
The results of this study suggest that for our imitation task, attention rather than motor coordination is the key determinant of imitation level. This interpretation is supported by the observation that although the autistic group had poorer coordination than the non-autistic group measured using the Developmental Coordination Disorder (DCD)/Dyspraxia Checklist (Kirby, Edwards, Sugden, & Rosenblum, 2010) and a pointing task (Supplemental material, Appendix C), they still showed similar levels of imitation modulation compared to the non-autistic group. Furthermore, there were no significant correlations between these motor tasks and imitation modulation (Supplemental material, Appendix C). However, it is important to highlight that the contribution of visuomotor processes should not be ruled out, as a number of studies have observed correlations between imitation and motor performance (Dadgar et al., 2017; Smith & Bryson, 1998; Stieglitz Ham et al., 2008; Vanvuchelen et al., 2007), and our imitation task was relatively simple and repetitive, so unlikely to fully load VMI. Indeed, greater differences in imitation between autistic and non-autistic groups have been found for actions that are motorically more complex (Chetcuti, Hudry, Grant, & Vivanti, 2017; Rogers et al., 1996).
Our findings suggesting that failure to imitate the style of a movement is due to reduced attention towards that movement fit with theories proposing that autism can be seen in terms of diminished social motivation, including reduced visual orienting to human stimuli because they do not experience social reward (Chevallier, Kohls, Troiani, Brodkin, & Schultz, 2012; Dawson et al., 2004). This is supported by two recent meta-analyses of eye-tracking studies identifying that autistic individuals have overall reduced visual attention to social stimuli and increased attention to non-social stimuli, particularly if there are multiple people or interactions (Chita-Tegmark, 2016; Frazier et al., 2017). This might suggest that the reduced imitation modulation and visual attention to the action which we observed in the presence of a single hand and no face would be magnified in more natural settings. Other theories, such as the social top–down response modulation (STORM) model (Wang & Hamilton, 2012), propose that imitation deficits in autism occur due to atypical top–down modulation of imitation in response to social cues such as facial expression and eye gaze. Our results advance the model by demonstrating that imitation can be modulated by explicit instructions to social cues. It should be noted that Vivanti et al. (2014) draw the important distinction between propensity (frequency) to imitate and accuracy of imitation. In line with these authors, we suggest that there is an initial social drive to engage with another person that relates to propensity to imitate and social measures such as joint attention as found by Vivanti et al. Our task bypasses this initial social drive, as participants are instructed to imitate. Once an individual has chosen to engage with another person, imitation accuracy is dependent on attention to the kinematic cues of the action. Without explicit instructions to attend to the movement kinematics, autistic individuals attend less to the movement kinematics leading to reduced accuracy of imitation compared to non-autistic people.
The findings of this study have implications for how autistic people learn and obtain socially relevant information from the observation of other peoples’ actions (Gowen, 2012). There is an emerging body of work highlighting the importance of observing action kinematics for the ability to judge vitality form (e.g. how a movement is performed – whether it is rough or gentle) (Di Cesare, Dio, Marchi, & Rizzolatti, 2015) and predict the actions and intentions of others (Abernethy & Zawi, 2007; Ansuini, Cavallo, Bertone, & Becchio, 2015; Ansuini et al., 2016; Becchio, Koul, Ansuini, Bertone, & Cavallo, 2018; Cavallo, Koul, Ansuini, Capozzi, & Becchio, 2016; Diersch, Cross, Stadler, Schütz-Bosbach, & Rieger, 2012). Observing kinematics is also important when learning new actions as instructors often enhance or exaggerate features in a movement sequence in order to highlight the important elements to imitate (Brand, Baldwin, & Ashburn, 2002; McEllin, Knoblich, & Sebanz, 2018). Exaggerated movements may also be important for communicating intentions during joint actions, in order to optimise the success of the action (Pezzulo et al., 2019; Vesper, Schmitz, Safra, Sebanz, & Knoblich, 2016) or to provide a social signal when used by the imitator (Krishnan-Barman & Hamilton, 2019). Analysis of action kinematics is particularly important when there are fewer contextual cues (e.g. objects), the goal is unclear or participants have little experience with the observed actions (Nicholson, Roser, & Bach, 2017). Our results, together with previous findings that autistic individuals imitate in a goal-directed way (i.e. emulate actions), suggest that they do not pay attention to body kinematics and consequently will miss out on these social and teaching-related kinematic cues. Indeed, there is evidence that autistic individuals are less able to use kinematic features, as they are impaired at recognising vitality form (Di Cesare, 2017; Rochat et al., 2013) and are less able to predict the goal of an action from grasp kinematics (Turi, Muratori, Tinelli, Morrone, & Burr, 2017) compared to non-autistic groups. Moreover, since kinematic profiles are key to the ‘readability’ of actions by others (Koul, Cavallo, Ansuini, & Becchio, 2016), our results also suggest the possibility that autistic individuals may be less inclined to show communicative kinematics in their actions leading to difficulties performing joint actions and teaching others. Importantly, the current findings highlight that instructions to attend to relevant features of a movement may be useful when developing therapies that help autistic people to interpret actions or use or directly teach imitation (Ingersoll, 2012; MacDonald & Ahearn, 2015; Plavnick & Hume, 2014).
When interpreting our results, there are some limitations to consider. It was necessary to always place the attention instruction after the general instruction, as it would be difficult to prevent participants using this strategy when given the general instruction condition. Although our results did show a learning effect (greater imitation modulation over time), there are several findings that argue against learning effects being the sole explanation for increased imitation modulation in the attention instruction condition (see Supplemental material, Appendix D). First, the increase in imitation modulation between the general and attention instruction condition was more than twice the size as that between the first and the last four trials in the general block and produced a much larger Bayes likelihood of being different (32 times vs 3.2 times more likely). Second, the difference in imitation modulation between the two instruction conditions was more than three times higher in the study participants compared to two new autistic participants who performed the general attention condition twice (Supplemental material, Appendix D). Third, the findings from these two additional participants are in line with our previous results showing no order effects for non-autistic participants (
Although full-scale and performance IQ were matched between autistic and non-autistic participants, the verbal IQ was lower in the autistic group. However, it is unlikely that this caused the difference in imitation modulation between the groups, as verbal IQ and imitation for the general instruction were not correlated (
Conclusion
Instructions to attend to the kinematic features of an observed action increased imitation of movement amplitude in autistic adults to similar levels as non-autistic adults. Eye movement analysis revealed that attention instructions are likely to cue attention to the relevant kinematic features, but that the time spent tracking the observed action remains lower than that of non-autistic individuals. For our simple imitation task, attention rather than motor skills were a significant contributing factor to lower levels of imitation in the autistic group.
Supplemental Material
AUT882810_Supplemental_material_Appendix_A – Supplemental material for Instructions to attend to an observed action increase imitation in autistic adults
Supplemental material, AUT882810_Supplemental_material_Appendix_A for Instructions to attend to an observed action increase imitation in autistic adults by Emma Gowen, Andrius Vabalas, Alexander J Casson and Ellen Poliakoff in Autism
Supplemental Material
AUT882810_Supplemental_material_Appendix_B – Supplemental material for Instructions to attend to an observed action increase imitation in autistic adults
Supplemental material, AUT882810_Supplemental_material_Appendix_B for Instructions to attend to an observed action increase imitation in autistic adults by Emma Gowen, Andrius Vabalas, Alexander J Casson and Ellen Poliakoff in Autism
Supplemental Material
AUT882810_Supplemental_material_Appendix_C – Supplemental material for Instructions to attend to an observed action increase imitation in autistic adults
Supplemental material, AUT882810_Supplemental_material_Appendix_C for Instructions to attend to an observed action increase imitation in autistic adults by Emma Gowen, Andrius Vabalas, Alexander J Casson and Ellen Poliakoff in Autism
Supplemental Material
AUT882810_Supplemental_material_Appendix_D – Supplemental material for Instructions to attend to an observed action increase imitation in autistic adults
Supplemental material, AUT882810_Supplemental_material_Appendix_D for Instructions to attend to an observed action increase imitation in autistic adults by Emma Gowen, Andrius Vabalas, Alexander J Casson and Ellen Poliakoff in Autism
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: A.V. was funded by the UK Engineering and Physical Sciences Research Council under their Doctoral Training Partnership with the University of Manchester.
Ethics approval and consent to participate
Participants gave written informed consent, and the study was approved by the University of Manchester Research Ethics Committee (Ref: 2017-2541-4204).
Supplemental material
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
