Abstract
Understanding the factor structure of autistic symptomatology is critical to the discovery and interpretation of causal mechanisms in autism spectrum disorder. We applied confirmatory factor analysis and assessment of measurement invariance to a large (
Introduction
A key constraint in efforts to elucidate the biology of autistic syndromes involves an (as yet) imperfect understanding of how and why the observed clinical features of the autistic syndrome—traditionally including deficits in reciprocal social behavior, communicative impairment, and stereotypic behavior/restricted interests—co-vary (i.e. “travel” together) in nature. Although some studies in the general population have suggested that inherited influences on the three classic domains of autism spectrum disorder (ASD) symptoms are substantially (Happe and Ronald, 2008) or partially independent of one another (Ronald et al., 2006a, 2006b, 2008), important caveats to such analyses are that they can be confounded by measurement methods that do not ascertain autistic symptoms and traits with adequate specificity (e.g. capturing social variation related to antisocial behavior or communicative variation related to Specific Language Impairment (SLI)), or across the full range in which autistic traits and symptoms manifest themselves in nature.
Furthermore, heterogeneity in the mechanisms that confer susceptibility to autistic symptomatology can give rise to disparate estimations of both heritability and symptom structure, depending on the characteristics by which a given sample is ascertained. Although it is now estimated that a substantial proportion of cases of autistic syndromes are familial (Constantino et al., 2013; Klei et al., 2012)—and that familial autistic syndromes share causal influences with milder (subclinical) manifestations of autistic symptomatology (Constantino et al., 2006, 2010; Piven et al., 1997; Robinson et al., 2011)—sporadic autistic syndromes influenced by rare, highly penetrant, de novo mutations account for at least 10%–15% of cases, many of which reflect symptom patterns that are uniquely influenced by their respective (and disparate) genetic origins. Thus, samples preferentially incorporating sporadic (simplex) autism versus familial (multiplex) autism or general population subjects might understandably manifest some elements of contrasting factor structure (Virkud et al., 2009).
The Social Responsiveness Scale (SRS) is a quantitative measure of traits and symptoms that, in aggregate, distinguish autistic syndromes from other variations in human behavior, and that are highly heritable. SRS measurements of the traits that have historically characterized the “autism triad” have exhibited—consistently and across cultures (Bolte et al., 2008; Constantino et al., 2004; Kamio et al., 2012)—very strong intercorrelations. These and other studies, using other measures, have supported the collapse of separate social and communication criterion domains featured in
Given the possibility that, because of the diversity of causal pathways, factoral separation or parsimony in autism may only be relative (rather than absolute), or may be distinct for subpopulations of subjects (e.g. representing simplex versus multiplex autism as discussed above), it is important to continue to examine data from large populations encompassing the full diversity of manifestations of this axis of human social variation. The availability of quantitative indices of such variation furthermore makes it possible to move beyond the question of “how many factors are there” to consider the
Several recent studies have helped motivate our analytic approach. Using quantitative data from the Interactive Autism Network (IAN) National Volunteer Register, Frazier et al. (2012) demonstrated the ability of two empirically separable
The present study
The primary aim of the present study was to conduct confirmatory factor analysis of the SRS–Second Edition (SRS-2) and test for measurement invariance of its structure across age (age < 10 years vs age ≥ 10 years, child vs adult), sex, ASD diagnosis within autism-affected families (ASD vs non-ASD siblings), and across methods of adult report (self-report vs other report). A second exploratory aim was to examine sex differences and age-related changes in the factors derived from the SRS-2.
The present study employed exploratory and confirmatory factor methods to evaluate single-factor, two-factor, and multifactor models in an attempt to elucidate the broad (single- vs two-factor solution) and specific (possible multifactor solution) constructs measured by the SRS-2. The broad structure of the SRS-2 addresses whether variation in autism symptom ratings can be parsimoniously described using a single factor or whether two factors corresponding to proposed
Stability of measurement is a precondition to evaluating the effects of demographic factors, such as age and sex, on autism symptom factors. Previous studies, including the original standardization studies of the SRS, have suggested significant influences of sex (females have lower scores) but minimal influence of age on SRS scores. More recent studies have suggested significant sex and age effects modified by diagnostic status (ASD vs non-ASD). The present study attempts to advance this literature by evaluating the effects of age, sex, and ASD diagnosis on empirically identified broad and specific SRS-2 factors.
Methods
Participants
SRS-2 data were obtained from three distinct samples—the IAN (
The Washington University Human Research Protection Office reviewed the protocol (HRPO number: 201209133) and deemed it appropriate and not subject to Institutional Review Board (IRB) oversight because it involved exclusively anonymized data.
Symptom measurement
The SRS-2 is a 65-item, ordinally scaled (1 = “not true” to 4 = “almost always true”) quantitative assessment of the severity of autism traits. Its predecessor, the SRS, has been one of the most frequently used quantitative measures of autism symptoms, with very strong measurement properties in healthy and autism-affected samples (Constantino and Gruber, 2005). For 4- to 18-year-olds, the SRS-2 represents exactly the same item set as the SRS. The present study capitalizes, in part, on the availability of data derived from the self-report and other-report versions of the
Statistical analyses
Packet development
It is challenging to identify a parsimonious factor solution using 65 ordinally scaled items because many small item clusters (overlaps in content between only 2 and 3 items) will contribute minor common variance, resulting in high-dimensional solutions with little theoretical or clinical value. For this reason, we developed an a priori strategy for developing item packets, which reflect collections of items that share similar content and can be submitted as indicators to subsequent factor analyses. Packets are advantageous for evaluating the broad structure of autism symptoms by reducing the probability of identifying factors that are difficult to replicate. Using packets as indicators in factor analyses also increases the likelihood that any fine-grained factors or sub-factors generalize beyond specific item content or wording. Empirical studies have supported the use of item packets in similar contexts (Little et al., 2002; Nasser and Takahashi, 2003; Nasser and Wisenbaker, 2003). To develop item packets, we conducted an exploratory factor analysis using all 65 SRS-2 items. The analysis used principal axis factoring with promax rotation in SPSS version 20 (IBM Corporation, 2011). It should be noted that ordinally scaled items typically require a weighted least squares estimation with missing value (WLSMV) estimator to appropriately correct for bias in the use of items with ordinal distributions. However, the WLSMV estimator is computationally intensive, particularly when higher factor number solutions (>3 factors) are applied to a large number of items (65). Because the focus of this procedural step was not on identification of autism symptom structure but rather to reduce the item set into conceptually similar packets, a principal axis factoring approach was implemented.
Packet development was conducted in a combined child clinical and child population sample (combined child sample). This combined sample was used because this afforded the largest sample size and greatest variance in SRS-2 scores. Having sufficient score variance is crucial to identifying the number and specificity of factors (Floyd and Widaman, 1995). To develop packets, 10 factors were extracted and rotated. The 10 factors were chosen based on initial inspection of eigenvalues suggesting that little common variance remained after extracting 8–10 factors. The number of factors extracted was then progressively decreased until the final factor solution included factors with at least 3 items with primary loadings (>.30). This process resulted in an eight-factor solution with every SRS-2 item showing at least one primary loading. The eight factors had at least 3 distinct item loadings that were easily interpreted (factor 1: emotion recognition (ER), factor 2: odd behavior/perseverative thinking, factor 3: social avoidance (SA), factor 4: repetitive mannerisms (RM), factor 5: separation anxiety, factor 6: interpersonal relatedness (IR) items, factor 7: insistence on sameness (IS), factor 8: conversational impairment). SRS-2 items were then assigned to one factor based on their highest loading. Using this procedure, each factor consisted of 4–13 items, all with salient loadings. Next, within each factor, items were randomly selected for the first packet until the packet consisted of at least 3 total items (the number of items per packet was dependent on the total number of items available for each factor). After the first packet had at least 3 items, subsequent packets were created by randomly selecting any remaining items until the second packet had at least 3 items. The procedure continued until all of the items assigned to that factor were placed into packets. Across the eight factors, 19 item packets were created with 3–5 items per packet. Internal consistency of item packets was good to excellent (α = .72–.93), particularly given the small number of items per scale.
Exploratory and confirmatory factor analyses
The 19 item packets were submitted to exploratory and confirmatory factor analyses using maximum likelihood estimation in Mplus version 5.2 (Muthén and Muthén, 2007). Exploratory factor analyses were conducted to examine the relative improvement in fit with additional factors extracted. Exploratory factor analyses tend to provide a more conservative estimate of whether additional factors provide sufficient reliable variance to yield useful clinical and research information (Fabrigar et al., 1999; Frazier and Youngtrom, 2007). After evaluating exploratory solutions to determine the relative increment in variance explained across additional factors, a set of confirmatory factor models was estimated. These a priori models included a single-factor model, a two-factor model dividing all of the social communication/interaction (SCI) packets and the restricted/repetitive behavior (RRB) packets (
Measurement invariance
To examine measurement equivalence across ages (age < 10 years vs age ≥ 10 roughly contrasting prepubertal children from those representing preadolescence and higher, and caregiver-report of children vs adult other-report), sexes, ASD diagnosis within families affected by ASD (ASD-affected siblings vs unaffected siblings of ASD-affected children) and reporters (adult self-report vs adult other-report), a series of multigroup confirmatory factor analyses were computed. To elaborate, multigroup confirmatory factor analyses permit simultaneously estimating factor structure in two prespecified groups (e.g. males and females). In the simplest model (often called the baseline or configural model), separate estimates of factor loadings, intercepts (corresponding to indicator/packet means), and residual variances (corresponding to measurement error and specific variance attributed to each indicator/packet) are generated for each group. This model assumes that the factor structure is not equivalent across groups. After this model is estimated, a series of increasingly more restrictive models representing gradually more equivalent structures between groups are estimated in a stepwise fashion. If at any step a substantial decrease in model fit is observed, then the groups are determined to be nonequivalent on the parameters fixed at that step. For example, if in the second step (weak invariance), model fit decreases after fixing factor loadings across groups, then the groups do not have equivalent relationships between indicators and factors.
As stated above, the baseline model for these analyses estimated separate factor loadings, indicator (packet) intercepts, and residual variances. This model evaluated
Model fit
Confirmatory factor models were evaluated using six indices: chi-square, the comparative fit index (CFI), the Tucker–Lewis Index (TLI), the root-mean-square error of approximation (RMSEA), the Akaike Information Criterion (AIC) (Akaike, 1987), and the Bayesian Information Criterion (BIC) (Schwarz, 1978). CFI and TLI values from .90 to .92 were considered adequate fit, .92 to .95 good fit, and >.95 excellent fit (Marsh et al., 2004). RMSEA values <.10 were considered adequate fit and <.08 were judged as good fit (Kline, 1998). AIC and BIC are useful for comparing the relative fit of non-nested models, with lower values indicating better fit. For model comparison, differences between models were considered significant if the difference in BIC was >5 (Kass and Raftery, 1995).
Model comparisons for measurement invariance analyses were based on empirical work indicating that a drop in CFI or TLI >.01 or an increase in RMSEA >.01 implies measurement nonequivalence (Chen, 2007; Cheung and Rensvold, 2002; Vandenberg and Lance, 2000). For the present study, if any of these indices fell beyond this cutoff, the more restrictive model was considered to have reduced fit. Chi-square differences tend to be overly sensitive to group differences resulting in false rejections of measurement invariance (Cheung and Rensvold, 2002; Little, 1997). For this reason, chi-square difference was not evaluated.
Age and sex effects on SRS-2 factors
To examine age and sex effects, confirmatory factor models were reestimated with factors regressed on age and sex in a combined child and adult population sample. Additionally, in the child clinical sample, factors were regressed on age, sex, ASD diagnosis, and the interactions of age and sex with ASD diagnosis. The latter terms evaluate whether age and sex effects on symptom levels differ across non-ASD and ASD-affected siblings. These analyses were conducted using both the two-factor model based on
Results
Exploratory factor analysis
Exploratory factor analysis of the 19 item packets in the combined child clinical and population sample indicated that the largest improvement in fit occurred from 1 to 2 factors (Δχ2(18) = 9362.21,
Confirmatory factor analysis
Table 1 presents results of confirmatory factor analyses across selected models in the combined child sample (clinical plus population), adult other-report, and adult self-report samples. Results of the combined child sample indicated that the two-factor
SRS-2 confirmatory factor analytic model results in children, adults other-report, and adults self-report.
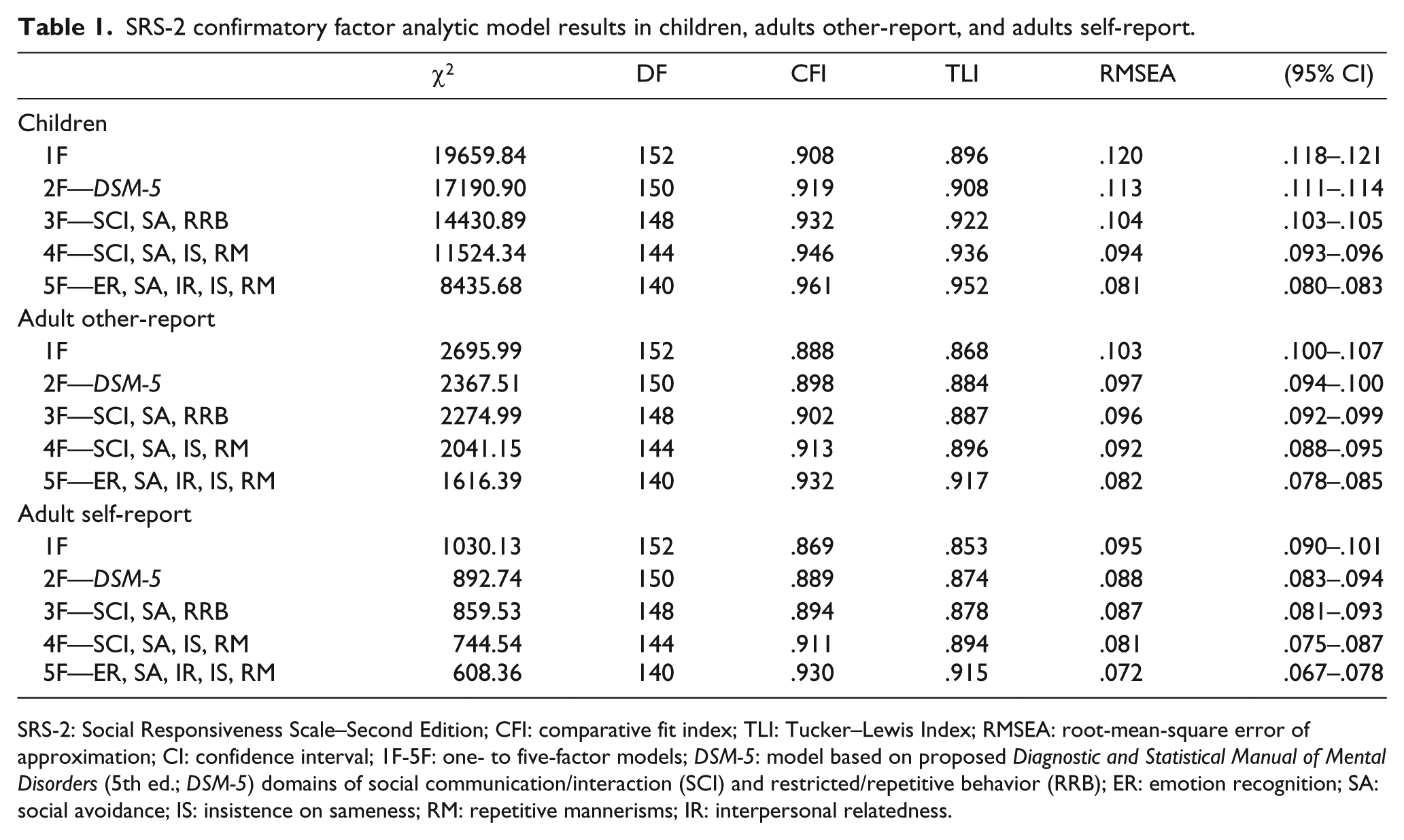
SRS-2: Social Responsiveness Scale–Second Edition; CFI: comparative fit index; TLI: Tucker–Lewis Index; RMSEA: root-mean-square error of approximation; CI: confidence interval; 1F-5F: one- to five-factor models;
Two additional hierarchical post hoc models were estimated. The first model (Hierarchical—1F) specified a single hierarchical factor measured by the five specific factors. The second model (Hierarchical—2F) specified two hierarchical factors, corresponding to
Inter-trait correlations
Very high correlations were observed between SCI and RRB domains in the two-factor
Measurement invariance
Because of the utility of the two-factor model in measuring
Measurement invariance of the two-factor
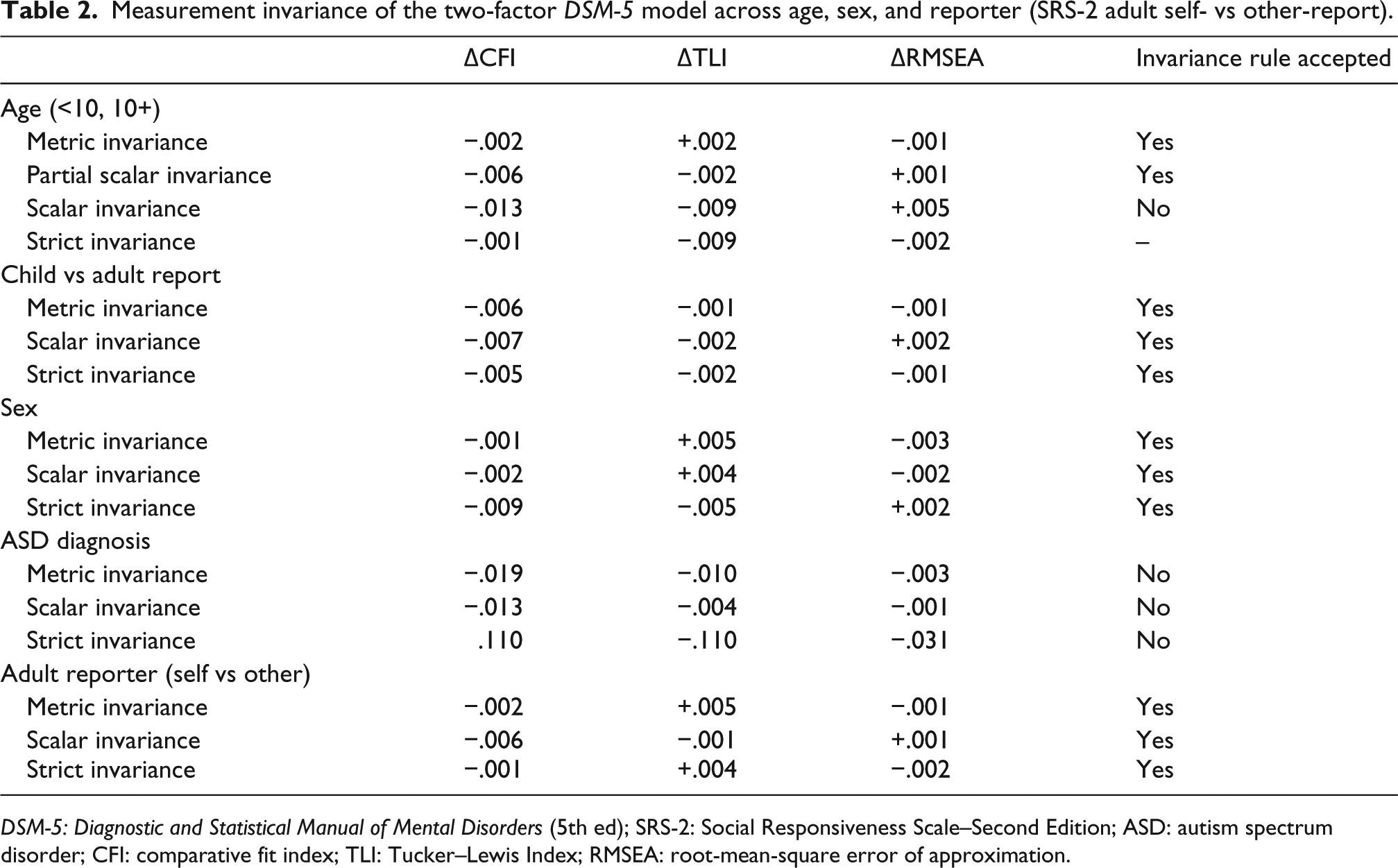
Age effects
Table 3 presents the effects of age, sex, and their interaction with ASD diagnosis on
Relationship between age, sex, ASD diagnosis, and their interaction with
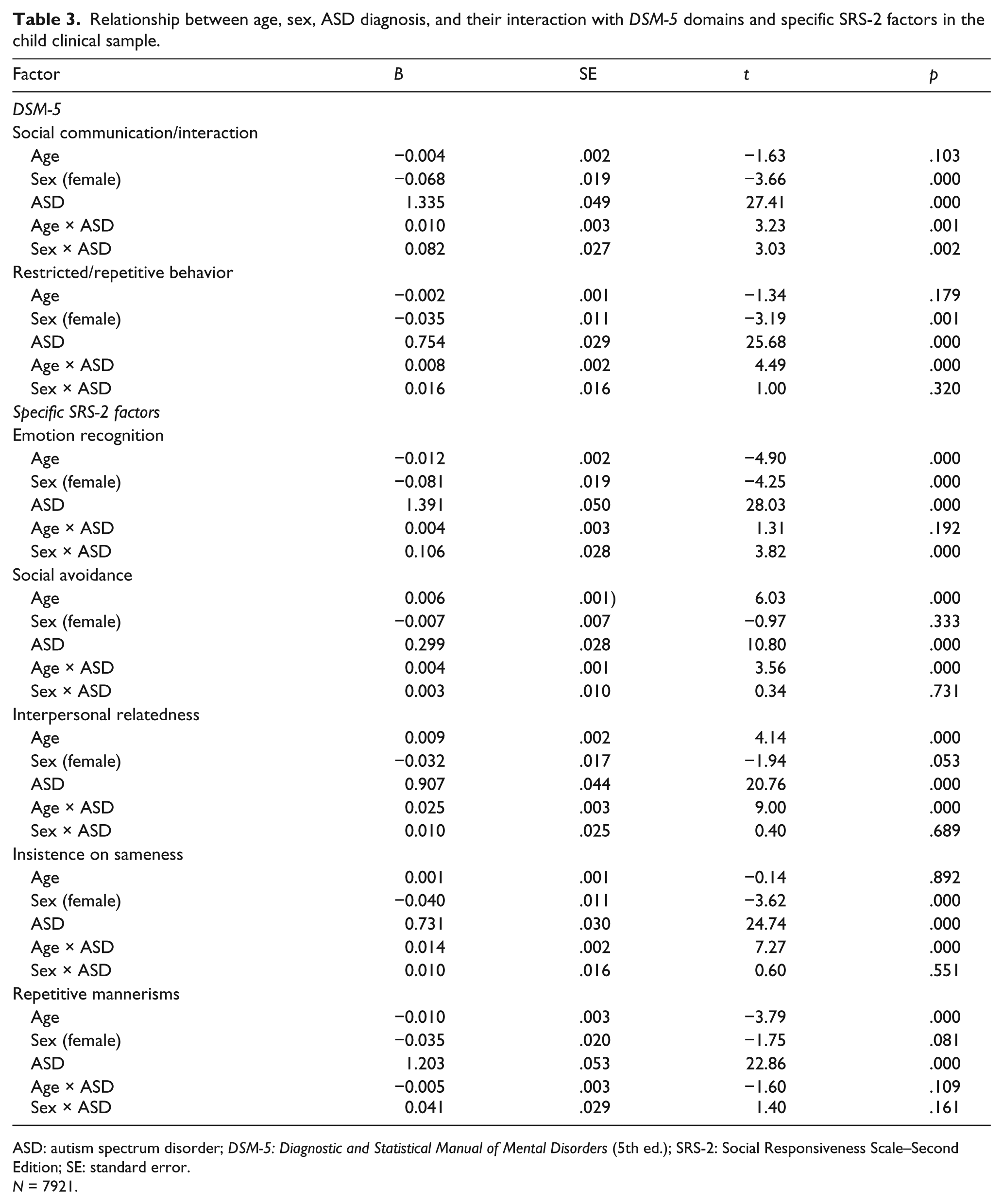
ASD: autism spectrum disorder;
Figures 1 and 2 present age and sex effects on
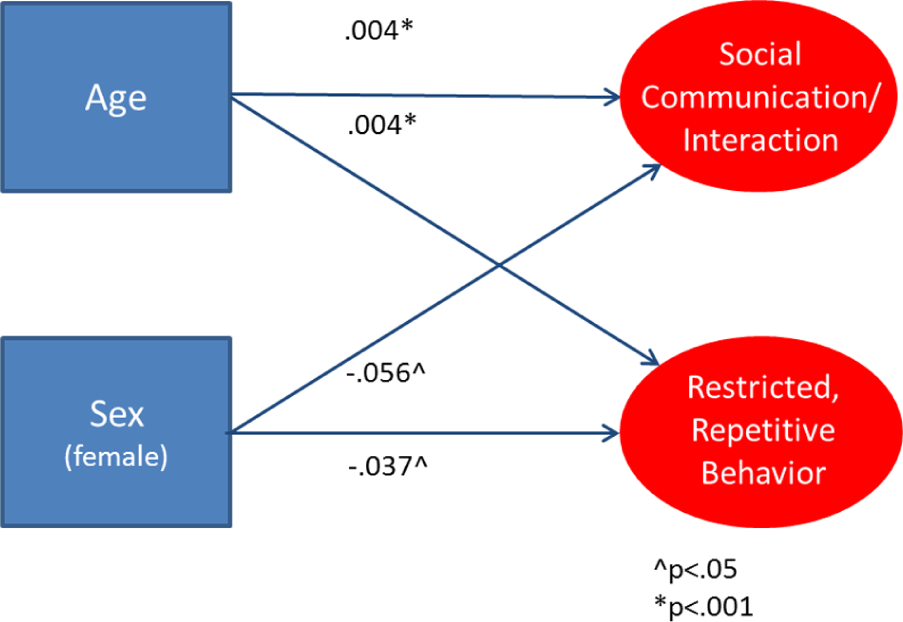
Influence of age and sex on
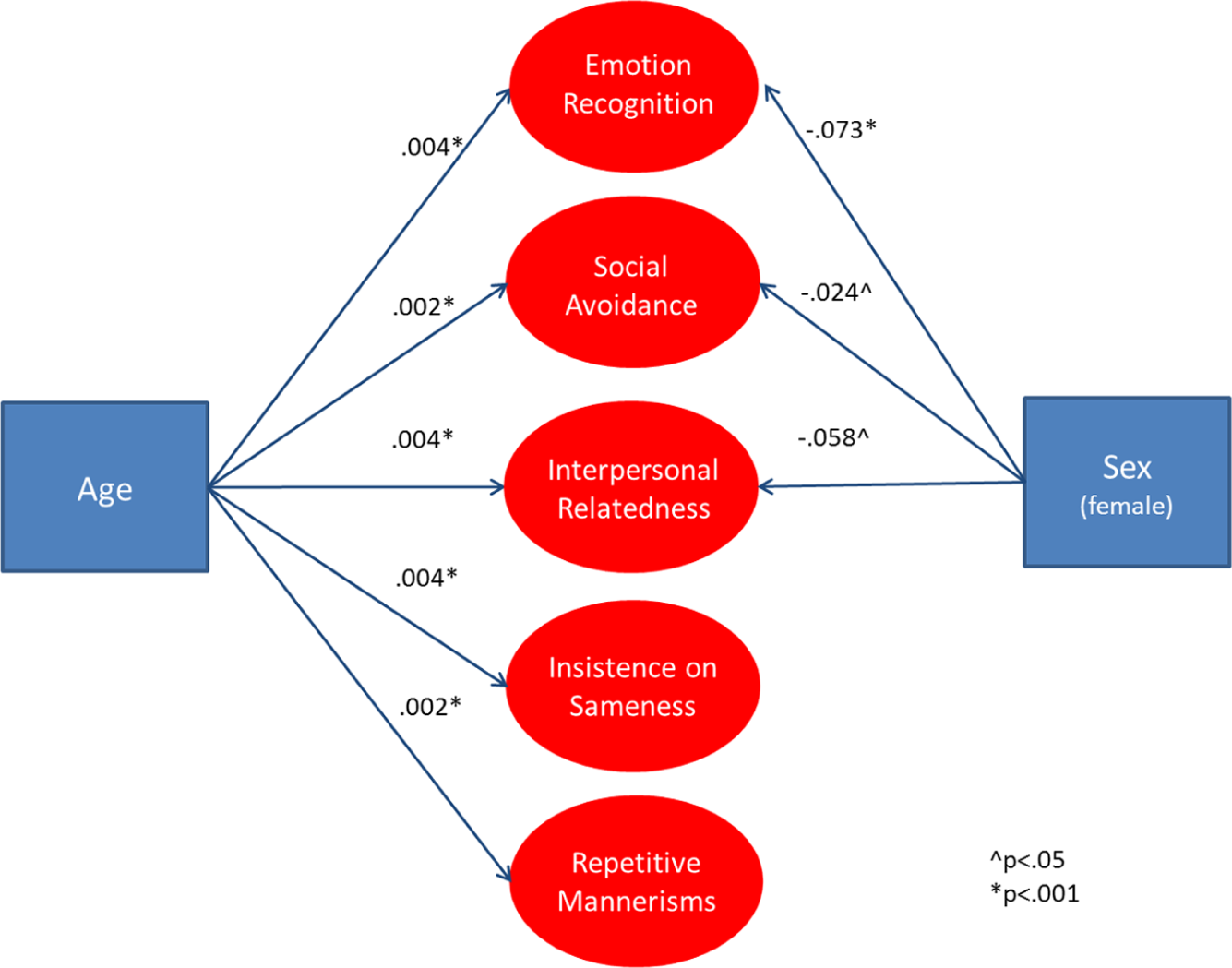
Influence of age and sex on specific SRS-2 factors in the population.
Sex effects
Overall, females had lower SCI symptom levels than males. However, ASD-affected females show higher SCI symptom levels than ASD-affected males, whereas non-ASD siblings show the opposite pattern. Higher SCI symptom level in ASD-affected females was driven by substantially worse ER. No significant differences in SA or IR difficulties were observed. Overall, females had lower RRB symptom levels than males, and this was largely attributable to significantly lower levels of IS. No sex differences were observed for RM and there were no interactions with ASD diagnosis for either IS or RM.
In the combined child and adult population sample, females had lower overall SCI (
Group heritability and sibling scores
In order to place these results in the context of previously published data that involved use of the total SRS score, we reanalyzed SRS data originally published with respect to total SRS score in Constantino et al. (2007) and in Constantino and Todd (2000), for neither of which there was any overlap with the primary data analyzed in this report. Table 4 presents mean item scores for affected versus unaffected children in ASD-affected families, including total SRS scores (as previously published for those data sets in Constantino et al., 2007) and scores for each of the
Average item scores for DSM-5 domains and specific SRS-2 factors, separately for ASD and non-ASD siblings from Constantino et. al. (2007).
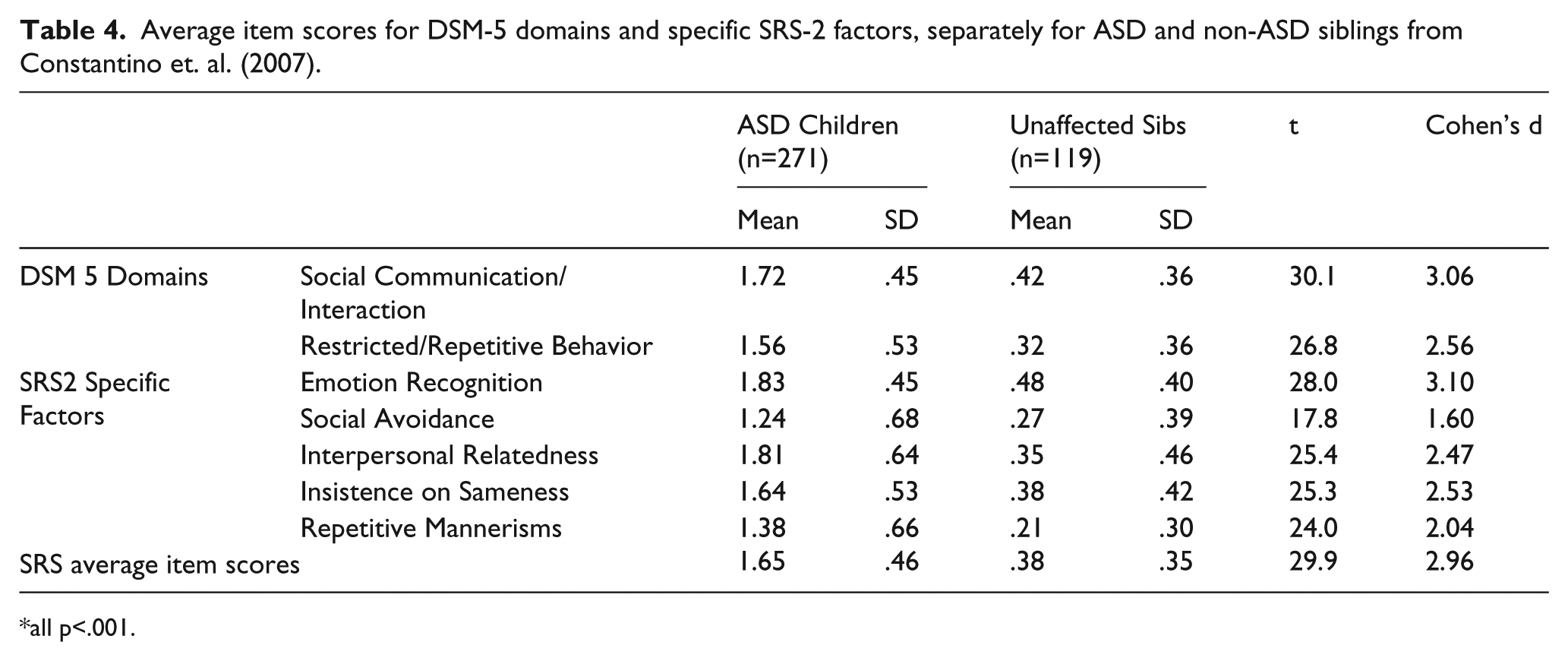
all p<.001.
Discussion
The present study extends previous findings on the factor structure of autism, using some of the largest samples harnessed to date for examination of quantitative autistic traits. The results provide strong evidence of separable, but highly correlated, autistic traits corresponding to the
Regardless of the most appropriate interpretation, observations of measurement nonequivalence and categorical distinctions between ASD and non-ASD siblings will require confirmation in additional samples. These findings may indirectly reflect the role of diverse, individually rare, de novo mutations suspected of strongly influencing the development of a significant minority of clinical autistic syndromes (10%–15%) (see Sanders et al., 2012), but having less of a role in quantitative trait variation in unaffected individuals and in familial forms of ASD. Although the genetic causes of subclinical autistic traits profoundly overlap with those of familial autistic syndromes (Constantino et al., 2010; Klei et al., 2012; Robinson et al., 2011), they do not necessarily overlap with those of sporadic forms of autism (Virkud et al., 2009). As our understanding of the causes of autism evolves, it is likely time to move from positing categorical or dimensional models to conceptualizing the autism spectrum as a hybrid influence both by categorical and dimensional components.
The statistical power afforded by the present study permitted identification of specific SRS-2 factors which are respectively encompassed by the broader SCI and RRB domains—including what we have termed in this report as ER, SA, IR, IS, and RM. Interestingly, these facets did not have straightforward relationships with one another, indicating that a simple hierarchical structure with specific factors measuring broad SCI and RRB domains may not be tenable. Additional research with multiple instruments may be needed to further understand the complexity of these relationships. Even with this important caveat, empirically derived factors may complement use of the SRS total score and the original theoretically derived SRS treatment scales. For example, additional facets of autism that exhibit subtle factoral separation may be helpful in etiologic and neurobiologic research for identifying specific risk or protective factors or neural circuit abnormalities. Empirically derived factors may be particularly useful in treatment settings where treatment effects may differentially influence one aspect of SCI or RRB but not all, or show specific temporal patterns. For example, social skills programs for high-functioning ASD may improve ER and decrease SA more rapidly, but IR difficulties may only subside slowly after critical improvements in the other domains have occurred. Social skills interventions may yield less improvement to IS or RM unless these are also specifically targeted.
Sex and ASD diagnosis moderated symptom levels across specific SRS-2 factors, highlighting the complexity of autism trait emergence and reaffirming literature identifying sex differences in the manifestation of autistic symptoms in diagnosed populations (Carter et al., 2007; Hartley and Sikora, 2009; Lord et al., 1982). For example, poorer ER in ASD-affected females may prove to be a critical underlying factor in the observation that girls who garner an ASD diagnosis tend to exhibit more severe phenotypes than their male counterparts (Carter et al., 2007; Dworzynski et al., 2012; Lord et al., 1982), and the observation of increasingly disparate sex ratios at higher cognitive levels in ASD (Lord and Schopler, 1985). Similarly, lower RRB levels in females are congruent with recent empirical work using both interview-based and observational methods (Mandy et al., 2012). Lower RRB levels in females are not likely accounted for by under-identification, particularly given that proportionally more females are identified at lower cognitive levels. Investigations that carefully sample males and females with ASD across the range of cognitive severity are needed to determine whether sex differences in symptom levels are independent of ascertainment and diagnostic biases, and may provide important clues to specific biological pathways through which many (but not all) females are spared full phenotypic expression of genetic liability to many forms of familial autism (Constantino and Charman, 2012).
Age also had a complex relationship with SRS-2 factors. Most prominently, SA and IR difficulties appear to be more prevalent in older youth, but these effects were much more pronounced in ASD-affected youth than in their unaffected siblings. The pattern of findings suggests that the accumulation of negative social experiences as a result of impaired ER and perspective taking may lead to greater SA and IR difficulties with age. However, it is important to keep in mind that developmental inferences are tenuous with cross-sectional data and these effects may simply represent sampling differences across age. RRB constructs also showed a complex relationship with age. Levels of IS were higher in older youth with ASD, but levels of RM were lower with age in ASD-affected youth.
Examination of these phenomena in prospective longitudinal data sets involving children with autism and their unaffected siblings is under way, and can provide further clarification of these findings. Specifically, the
Resolution of empirically derived SRS-2 factors presents an opportunity to provide more nuanced behavioral phenotypes with which to associate genetic and/or neurobiologic variation, and to derive refined estimates of heritability. Twin correlations in
Age and sex effects reinforce the development and application of demographically corrected normative scores, which have proven critical in measuring the familial aggregation of quantitative autistic traits (Constantino et al., 2010), and which contextualize the derivation of diagnostic boundaries across sex, age, and culture (see Kamio et al., 2013). The parsimonious nature and adequate fit of the two-factor model implies that scoring these broad autism traits in the SRS-2 may be helpful in diagnostic contexts where separate measurements of
Limitations and future directions
As with all factor analytic studies, results were limited by the available indicators submitted for analysis. Fortunately, the SRS-2 contains a diverse array of autism-associated traits that are widely sampled in a unimodal distribution in the general population; thus, the likelihood that primary constructs were missed or under-identified is relatively low. For example, use of SRS-2 item packets identified many of the SCI and RRB constructs that have been identified in previous literature. Use of item packets may also lead to loss of minor factors. However, the large size and diverse nature of the item pool, as well as careful attention to higher dimensional factor solutions, mitigates this risk.
While inter-trait correlations were quite high, even for specific SRS-2 constructs obtained from the five-factor solution, it is always important to consider whether these relationships are free of rater bias. This was directly tested for total SRS scores and found to be absent in the general population twin sample reported by Constantino and Todd (2003). Furthermore, in SRS research in clinical populations, subtle rater
Given the very large sample sizes examined (total
Footnotes
Acknowledgements
This work was also supported by grants HD 068479 and HD 042541 to Dr. Constantino, as well as grant 8370 from Autism Speaks. IAN, the Interactive Autism Network is a web project of Kennedy Krieger Institute, sponsored by the Autism Speaks Foundation, Inc.
Funding
This work was made possible in part by the Case Western Reserve University/Cleveland Clinic CTSA Grant Number UL1 RR024989 provided by the National Center for Research Resources and the National Center for Advancing Translational Sciences, National Institutes of Health. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of NIH.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
