Abstract
Background
Previously, we have demonstrated that Apolipoprotein A-I (ApoA-I) could inhibit the secretion of Hepatitis B virus (HBV), suggesting that stimulation of ApoA-I may block particle production. In the present study, we evaluated the anti-HBV effect of RVX-208, a small-molecule stimulator of ApoA-I gene expression.
Methods
RVX-208 was used to treat HepG2.2.15 cell, a HepG2 derived cell line stably producing HBV virus. Real-time PCR was performed to examine the HBV DNA levels. Magnetic particles, which were coated with anti-HBS or anti-HBE antibody, were used to examine the HBsAg and HBeAg levels in the supernatant of cultured HepG2.2.15 cells in combination with the enzyme conjugates that were prepared with horseradish peroxidase labelled anti-HBS or anti-HBE antibody in a double antibody sandwich manner. RNA-seq, immunoblots and real-time PCR were used to analyze the functional mechanism of RVX-208.
Results
RVX-208 could elevate the ApoA-I protein levels in HepG2.2.15 cells. In the meantime, RVX-208 significantly repressed HBV DNA, HBsAg and HBeAg levels in the supernatants of HepG2.2.15 cells. RNA-seq data revealed that RVX-208 treatment not only affected the cholesterol metabolism, which is closely related to ApoA-I, but also regulated signalling pathways that are associated with antiviral immune response. Moreover, mechanistic studies demonstrated that RVX-208 could activate cGAS-STING pathway and upregulate the transcription of a series of interferons, pro-inflammatory cytokines and chemokines with antiviral potential that are at the downstream of cGAS-STING pathway.
Conclusion
Our study demonstrated that RVX-208, an inducer of ApoA-I, could suppress HBV particle production through activation of cGAS-STING pathway.
Introduction
Hepatitis B is one of the most common liver diseases globally. 1 Hepatitis B virus (HBV) causes liver damage mainly through chronic infection, and the chronic HBV infection would eventually lead to cirrhosis and hepatocellular carcinoma. With the introduction of hepatitis B vaccination, prevalence of chronic HBV infection has declined dramatically, with a reduction of 31.3% in all-age prevalence and 76.8% in children younger than 5 years between 1990 and 2019. However, until 2019, the estimated global, all-age prevalence of chronic HBV infection was still 4.1%, which means about 316 million people were infected with HBV. 2 Antiviral drugs are needed for a large portion of chronic hepatitis B patients with active HBV replication. However, current drugs against HBV, which mainly include pegylated interferon α (Peg-IFN-α) nucleos(t)ide analogues (e.g. tenofovir and entecavir), are limited. 3 Therefore, it is of great importance to develop novel drugs to suppress HBV infection.
Apolipoprotein A-I (ApoA-I), the major protein component of high density lipoprotein (HDL), has been proved to play vital roles in regulation of cholesterol trafficking, inflammation and immune response. 4 In a previous study, it was shown that ApoA-I could interact with HBx protein, a multifunctional viral regulator of the HBV gene products, and the interaction between HBx and ApoA-I impaired the lipid-binding ability and aggregation properties of ApoA-1, leading to an accumulation of cellular cholesterol, which finally promoted HBV budding process. Interestingly, this study also showed that overexpression of ApoA-I could significantly inhibit HBV secretion, suggesting a possibility to suppress HBV infection through stimulation of ApoA-I. 5
RVX-208, a novel, orally active Bromo and Extra Terminal (BET) protein inhibitor, has been demonstrated to be an inducer of ApoA-I. 6 Based on the previous study, 5 we hypothesize that RVX-208, a drug developed to treat atherosclerosis, may also hold the potential to suppress HBV particle production through upregulation of ApoA-I. In this study, we showed that RVX-208 could indeed repress HBV DNA copies, HBsAg levels and HBeAg levels in cell culture supernatants of HepG2.2.15 cells, without impairing the cell viability of HepG2.2.15 cells. Moreover, we found that RVX-208 could activate a potent antiviral signalling pathway, cGAS-STING pathway, which then elevate the expressions of a series of antiviral interferons, cytokines and chemokines. Taken together, our study demonstrated that RVX-208 could suppress HBV particle production through activation of cGAS-STING pathway.
Materials and methods
Cell culture
HepG2.2.15, which is derived from HepG2 cell line and can stably produce HBV virus, 7 was cultured with Dulbecco’s modified Eagle’s medium (DMEM) (HyClone, Thermo Scientific) supplemented with 10% fetal bovine serum (FBS) (HyClone), 200 μg/mL G418 (Gibco, Thermo Scientific) at 37°C and 5% CO2.
MTT assay
HepG2.2.15 cells were seeded in a 96-well plate (5 × 103 cells/well). After RVX-208 treatment for indicated times, 20 μL of MTT solution (Invitrogen, Thermo Scientific) was added to the cells, followed by incubation at 37°C for 4 h. The supernatant was discarded before 100 μL of dimethyl sulfoxide (DMSO) solution (Invitrogen, Thermo Scientific) was added. The optical density in each well was measured at a wavelength of 490 nm with a microplate reader, and cell viability rates were calculated.
CCK-8 assay
HepG2.2.15 cells were seeded in a 96-well plate (2 × 103 cells/well). After RVX-208 treatment for indicated times, 10 μL of CCK-8 solution (Beyotime Biotechnology, Shanghai, China) was added to the cells, followed by incubation at 37°C for 2 h. The optical density in each well was measured at a wavelength of 450 nm with a microplate reader, and cell viability rates were calculated.
Enzyme-linked immunosorbent assay (ELISA)
Apolipoprotein A-I levels in the supernatant of cultured HepG2.2.15 cells were assessed with the sandwich ELISA Kit (Cusabio Biotech, Wuhan, China) according to the protocol. The optical density (OD) value was measured at the wavelength of 450 nm using Multiskan FC photometer (Thermo Scientific).
Real-time PCR
Hepatitis B virus DNA was extracted from the supernatant of cultured HepG2.2.15 cells by DNA sample release reagent (Sansure Biotech, Changsha, China) according to the instructions of the manufacturer. Extracted HBV DNA was then quantified by real-time qPCR following the instructions of the manufacturer (Liferiver Biotech, Shanghai, China). Total DNA of HepG2.2.15 cells were extracted using FastPure Blood/Cell/Tissue/Bacteria DNA Isolation Mini Kit (Vazyme, Nanjing, China). Total RNA of HepG2.2.15 cells were extracted using Trizol (Invitrogen, Thermo Scientific). cDNA was prepared using HiScript III All-in-one RT SuperMix Perfect for qPCR (Vazyme, Nanjing, China). Real-time PCR was performed using ChamQ SYBR Color qPCR Master Mix (Vazyme, Nanjing, China) following the manufacturer’s instructions. The primer sequences are listed in Table S1.
Specific small interfering RNA and transfection
Specific small interfering RNAs (siRNAs) targeting ApoA-I and STING were purchased from GenePharma (Shanghai, China). The sequences were as follows: siApoA-I: 5’-CCAACAGCUGAACCUGAAU-3’; siSTING: 5’-GCCUCAUUGCCUACCAGGATT-3’. The negative control siRNA was provided by GenePharma (Shanghai, China). The transient transfection of siRNA was performed using GenmuteTM Reagent (SignaGen Laboratories, Maryland, USA) according to the manufacturer’s instructions.
Magnetic particle chemiluminescence
The levels of HBV surface antigen (HBsAg) and HBV e antigen (HBeAg) in the supernatant of cultured HepG2.2.15 cells were detected by magnetic particle chemiluminescence kit (Autobio, Zhengzhou, China). The double antibody sandwich method was used for detection. Magnetic particles were coated with anti-HBS or anti-HBE antibody, and the enzyme conjugate was prepared with horseradish peroxidase labelled anti-HBS or anti-HBE antibody. The antibody-antigen-antibody-enzyme complex is formed through immune reaction, which catalyzes the luminescence substrate to emit photons, and the luminescence intensity is proportional to the HBsAg and HBeAg content.
Immunoblot analysis
Primary antibodies were from Cell Signaling Technology (phospho-TBK1 Ser172, 1:1000, rabbit monoclonal, #5483; phospho-IRF3 Ser396, 1:1000, rabbit monoclonal, #29047) and Zen Bioscience (STING, 1:1000, rabbit monoclonal, #R25931; GAPDH, 1:1000, mouse monoclonal, #390035), and Secondary antibodies were from Zen Bioscience (Goat anti-mouse IgG-HRP, 1:5000, #511103; Goat anti-rabbit IgG-HRP, 1:5000, #511203).
RNA sequencing
The detailed procedures were described previously. 8 Briefly, total RNA extracts of RVX-208 treated and control HepG2.2.15 cells were used for RNA-seq to identify the differentially expressed mRNAs. Differential expression analysis between two groups was performed with DESeq R package. To control the False Discovery Rate (FDR), the p values were adjusted with the Benjamini-Hochberg method. The cutoff value of differentially expressed mRNAs was set as |log2[fold change (FC)]|>1 and FDR <0.05.
Statistical analysis
Statistical analyses were conducted and graphics were generated using Prism version 9 (GraphPad Software). All statistical tests were two-sided, and p < .05 was considered statistically significant. All quantitative data were recorded as mean ± standard error of measurement (SEM).
Results
RVX-208 decreased HBV particle production of HepG2.2.15 cells
To determine whether RVX-208 could suppress HBV particle production, we first examined the effect of RVX-208 on cell viability of HepG2.2.15 cells to find a proper antiviral concentration of RVX-208. We first performed MTT assay and found that RVX-208 did not impair the cell viability of HepG2.2.15 cells after treatment of 24 h until the concentration was raised to 120 μM (Figure 1(a)). Because each viability test is based on a specific biochemical read-out, which may be affected by the drug of interest. In order to minimize false negative outputs, we also performed CCK-8 assay to examine the cell viability of HepG2.2.15 cells after RVX-208 treatment. Likewise, the results also showed that RVX-208 did not significantly affect the cell viability of HepG2.2.15 cells at the concentration of 100 μM (Figure 1(b)). Therefore, we chose the concentration of 100 μM, which was the highest concentration of RVX-208 that did not show significant inhibitory effect on cell viability of HepG2.2.15 cells, to perform subsequent experiments. The effect of RVX-208 on cell viability of HepG2.2.15 cells. A-B, MTT assay (a) and CCK-8 assay (b) were performed to examine the cell viability of HepG2.2.15 cells after RVX-208 treatment of different concentrations for 24 h. Each experiment was repeated for three times. **p < .01, ***p < .001. ns, not significant.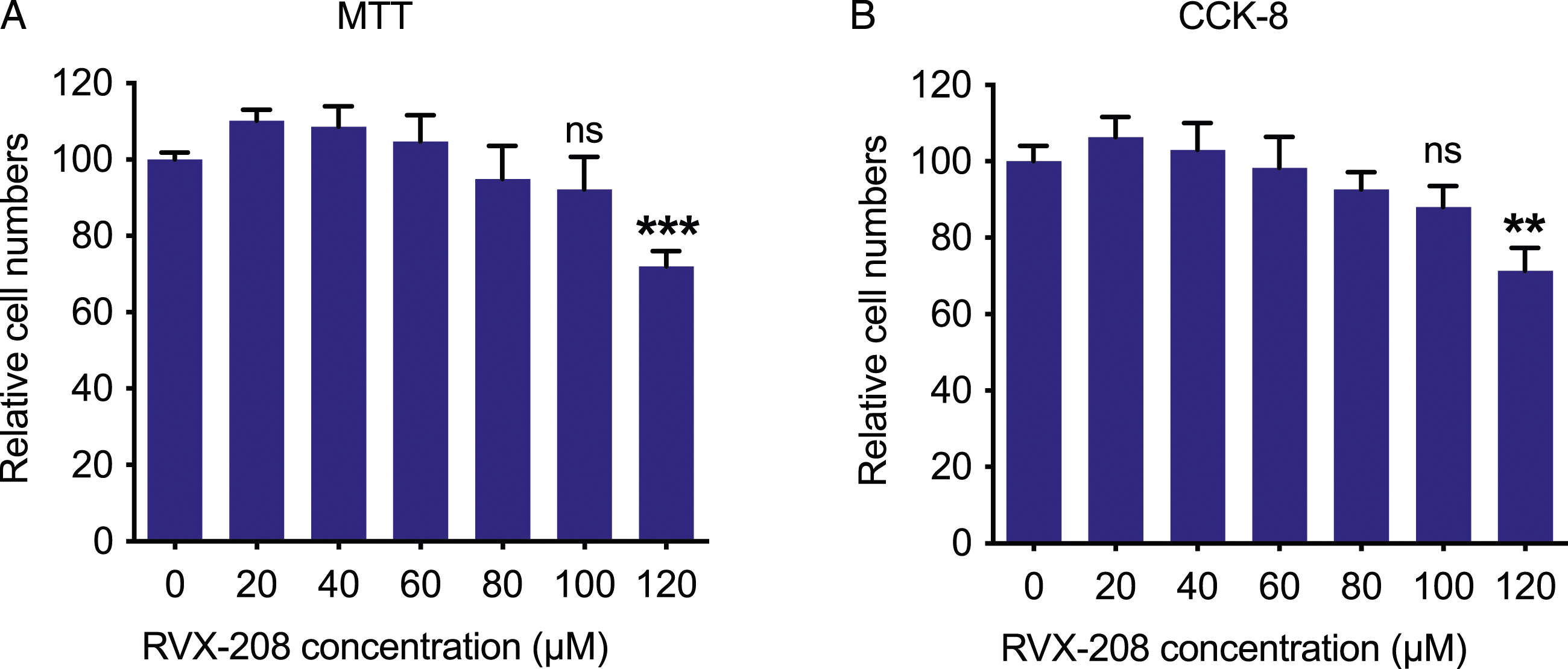
Next, to validate the efficacy of RVX-208, we also examined ApoA-I levels in the supernatant of cultured HepG2.2.15 cells. As shown in Figure 2, RVX-208 elevated ApoA-I levels in a dose-dependent manner (fold change = 1.92 at 50 μM, fold change = 3.20 at 100 μM), and the effect of RVX-208 becomes evident from 4 h (fold change = 1.97) and lasted until 24 h (fold change = 3.14). These results indicated that RVX-208 functioned as an inducer of ApoA-I in HepG2.2.15 cells. The effect of RVX-208 on ApoA-I protein levels in the supernatant of cultured HepG2.2.15 cells. A-B, ELISA was performed to examine ApoA-I levels in the supernatant of cultured HepG2.2.15 cells after RVX-208 treatment of different concentrations (treatment time 24 h) (a) or different times (concentration = 100 μM) (b). Each experiment was repeated for three times. ***p < .001.
After that, we studied the effect of RVX-208 on HBV particle production of HepG2.2.15 cells. We found that RVX-208 significantly repressed the copy numbers of HBV DNA at a concentration of 100 μM (Figure 3(a)). In addition, the suppressive effect of RVX-208 on HBV DNA copies could not be observed until the treatment time was extended to 24 h (Figure 3(b)). To determine whether ApoA-I is essential for the anti-HBV effect of RVX-208, we knocked down the expression of ApoA-I in HepG2.2.15 cells treated with RVX-208 and found that the suppressive effect of RVX-208 on HBV DNA copies was abolished (Figure 3(c) and (d)). To verify that RVX-208 could indeed decrease HBV particle production, we then examined the levels of HBsAg and HBeAg in the supernatant of cultured HepG2.2.15 cells. Consistently, RVX-208 also evidently repressed the levels of HBsAg and HBeAg at a concentration of 100 μM and a treatment time of 24 h (Figure 4(a)–(d)). Taken together, these results showed that RVX-208 treatment significantly suppressed HBV particle production of HepG2.2.15 cells through upregulation of ApoA-I expression. The effect of RVX-208 on HBV DNA copies in the supernatant of cultured HepG2.2.15 cells. A-B, Real-time PCR was performed to examine HBV DNA levels in the supernatant of cultured HepG2.2.15 cells after RVX-208 treatment of different concentrations (a) or different times (b). (c) ELISA was performed to examine ApoA-I levels in the supernatant of cultured HepG2.2.15 cells after RVX-208 and siApoA-I or negative control siRNA (siNC) treatment. (d) Real-time PCR was performed to examine HBV DNA levels in the supernatant of cultured HepG2.2.15 cells after RVX-208 and siApoA-I or negative control siRNA (siNC) treatment. Each experiment was repeated three times. *p < .05, **p < .01, ***p < .001. ns, not significant. The effect of RVX-208 on HBsAg and HBeAg levels in the supernatant of cultured HepG2.2.15 cells. A-B, Magnetic particle chemiluminescence was performed to examine HBsAg levels in the supernatant of cultured HepG2.2.15 cells after RVX-208 treatment of different concentrations (a) or different times (b). C-D, Magnetic particle chemiluminescence was performed to examine HBeAg levels in the supernatant of cultured HepG2.2.15 cells after RVX-208 treatment of different concentrations (c) or different times (d). Each experiment was repeated three times. *p < .05, **p < .01, ***p < .001. ns, not significant.

RVX-208 suppressed HBV particle production through activation of cGAS-STING pathway
Previously, it has been shown that ApoA-I could inhibit HBV secretion.
7
As an inducer of ApoA-I, it is quite possible that RVX-208 decreased HBV particle production of HepG2.2.15 cells via inhibition of HBV secretion. To identify whether RVX-208 suppresses HBV levels in other mechanisms, we performed RNA-sequencing with RVX-208 treated and control HepG2.2.15 cells to investigate alterations of gene expressions upon RVX-208 treatment. We identified 1945 upregulated genes and 1743 downregulated genes (Figure 5(a)). Gene Ontology (GO) analysis indicated that RVX-208 could regulate a series of biological processes that are associated with cholesterol transport and metabolism (Figure 5(b)), which was quite consistent with the fact that RVX-208 is an inducer of ApoA-I and ApoA-I is a regulator of cholesterol trafficking and metabolism. Interestingly, GO analysis also revealed that RVX-208 regulated genes were associated with immune system process, which could be linked with the antiviral effect of RVX-208. To identify possible downstream signalling pathways associated with immune response, we also performed Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis with RNA-seq data. As indicated by KEGG analysis, that JAK-STAT pathway and TNF pathway were among the most remarkable pathways enriched (Figure 5(b)). Through bioinformatic analysis, we found that both IL-6, the initiator of JAK-STAT pathway,
9
and TNF-α, the initiator of TNF pathway,
10
were the target genes of NF-κB
11
; therefore, we hypothesized that the antiviral effect of RVX-208 might be related to activation of NF-κB. Alteration of gene expressions in HepG2.2.15 cells after RVX-208 treatment. (a) Volcano plot of differential mRNA profiles between RVX-208 treated and control HepG2.2.15 cells. (b) Functional annotation by GO and KEGG analysis.
Recently, a series of publications have demonstrated that NF-κB can be activated by cGAS-STING pathway, a key mediator of inflammation in the setting of infection with potent antiviral effect.12–14 To determine whether RVX-208 could exert its antiviral effect through activation of cGAS-STING pathway, we first performed immunoblot analysis to examine the activation status of two key factors of cGAS-STING pathway, TBK1 and IRF3. As shown in Figure 6(a), RVX-208 induced phosphorylation of both TBK1 and IRF3 in a dose-dependent manner. Phosphorylation of TBK1 and IRF3 is a hallmark of activation of cGAS-STING pathway, which will stimulate the expression levels of various interferons, pro-inflammatory cytokines and chemokines with antiviral potential.
12
To verify the activation of cGAS-STING pathway by RVX-208, we examined several downstream genes of cGAS-STING pathway with antiviral potential. It was shown that RVX-208 treatment significantly increased the mRNA levels of IFN-β, IFN-γ, IL-6, IL-1β, TNF-α, MCP1, MX-1 and ISG-15 (Figure 6(b)), indicating that RVX-208 activated the cGAS-STING pathway. To determine whether the cGAS-STING pathway mediated the anti-HBV effect of RVX-208, we knocked down the expression of STING in HepG2.2.15 cells treated with RVX-208 and found that the suppressive effect of RVX-208 on HBV DNA copies was eliminated (Figure 6(c) and (d)), suggesting that RVX-208 indeed suppressed HBV particle production through activation of cGAS-STING pathway. RVX-208 repressed HBV particle production by activation of cGAS-STING pathway. (a) Phosphorylation levels of TBK1 and IRF3 in HepG2.2.15 cells after RVX-208 treatment were examined by immunoblots. (b) mRNA levels of mRNA levels of IFN-β, IFN-γ, IL-6, IL-1β, TNF-α, MCP1, MX-1 and ISG-15 in HepG2.2.15 cells after RVX-208 treatment were examined by real-time PCR. (c) Protein expression level of STING in HepG2.2.15 cells after RVX-208 and siSTING or negative control siRNA (siNC) treatment was examined by immunoblots. (d) Real-time PCR was performed to examine HBV DNA levels in the supernatant of cultured HepG2.2.15 cells after RVX-208 and siSTING or negative control siRNA (siNC) treatment. Each experiment was repeated for three times. *p < .05, **p < .01, ***p < .001.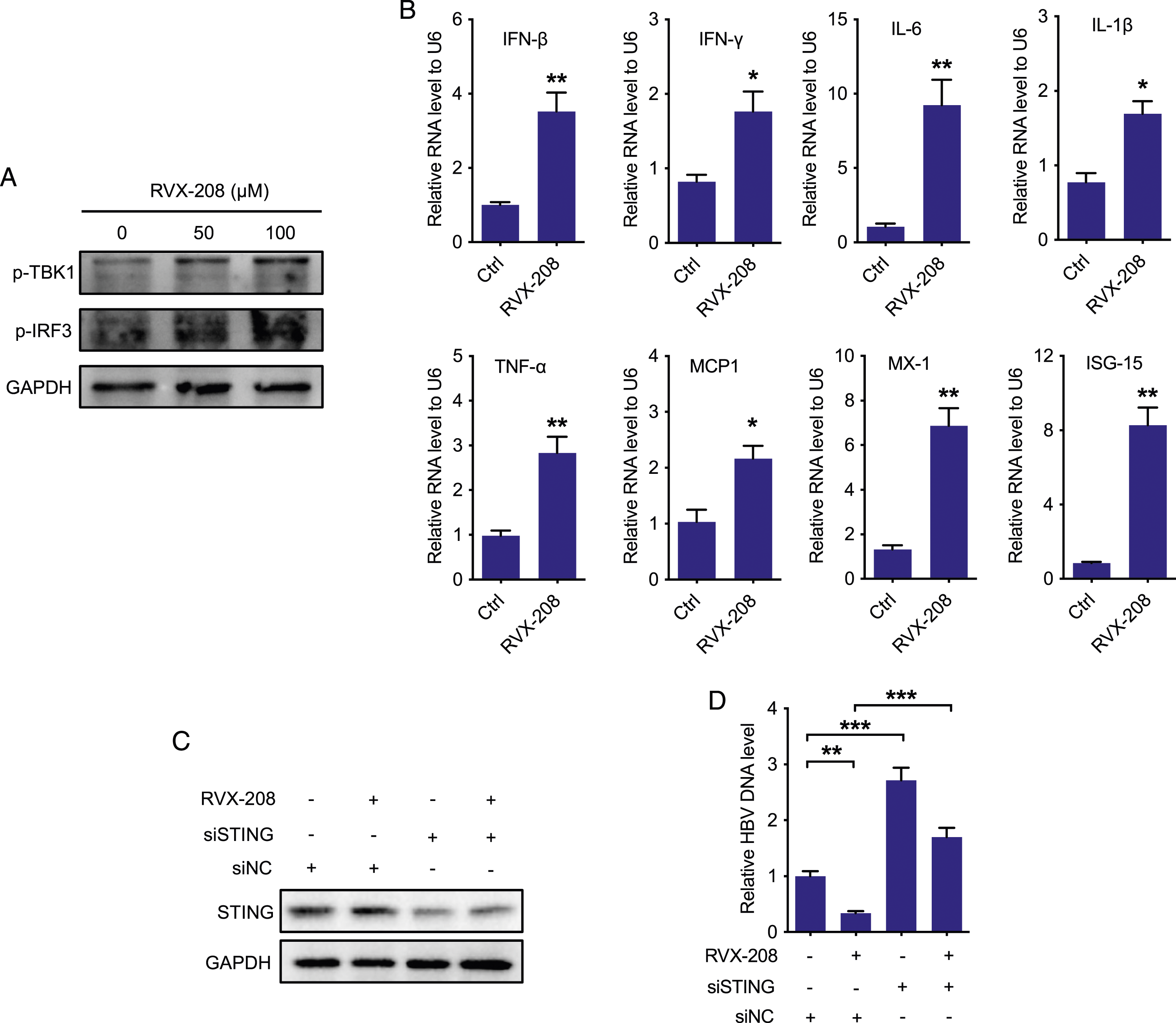
Discussion
Hepatitis B virus infection remains a heavy burden for public health. Most HBV-infected patients develop chronic hepatitis B because current available anti-HBV drugs cannot eradicate the virus. Meanwhile, current drugs against HBV are limited. Therefore, development of novel anti-HBV drugs is in urgent need. In this study, we proved that RVX-208, an inducer of ApoA-I, held the potential to serve as an anti-HBV drug. RVX-208 (also known as apabetalone) is an orally available small molecule, which is now under development for the treatment of various diseases including coronary artery disease, rheumatoid arthritis, and chronic kidney disease.15–17 Some of the studies with RVX-208 have reached clinical trials, such as a phase III clinical trial called ‘BETonMACE’, which uses RVX-208 to treat high-risk patients with coronary artery disease (CAD) and type 2 diabetes mellitus (DM), 18 suggesting that RVX-208 is a safe compound with great clinical potential. Interestingly, a recent study revealed that RVX-208 could attenuate SARS-CoV-2 infection through reducing ACE2 expression, 19 and this study has led to a clinical trial already (NCT04894266), which supported our finding that RVX-208 is of anti-viral potential. However, until now, there is no report about the anti-HBV effect of RVX-208, indicating the novelty of our study.
In a previous study, it was shown that overexpression of ApoA-I could inhibit HBV secretion. 7 In this study, we also observed that RVX-208 treatment significantly decreased HBV DNA copies, HBsAg levels and HBeAg levels in the supernatant of cultured HepG2.2.15 cells, indicating that RVX-208 repressed HBV particle production through stimulating the expression levels of ApoA-I. More importantly, we demonstrated that the antiviral effect of RVX-208 could also be mediated by cGAS-STING pathway. We showed that RVX-208 induced activation of cGAS-STING pathway, which resulted in upregulation of a series of antiviral interferons, cytokines and chemokines. This finding indicated that RVX-208 had double antiviral effects: on the one hand, it blocked HBV secretion by elevating ApoA-I levels to regulate cholesterol metabolism; on the other hand, it also suppressed HBV particle production by activation of cGAS-STING pathway.
cGAS-STING pathway has emerged as a crucial mediator of antiviral effect of immunity. Upon virus infection, cGAS-STING pathway detected the foreign DNA and stimulated the transcription of antiviral type I interferons. 13 cGAS-STING pathway has been reported to mediate immune defense against various viruses (both DNA and RNA viruses), such as herpes simplex virus 1 (HSV1), 20 vesicular stomatitis virus (VSV) 21 and SARS-CoV-2. 22 As a small DNA virus, replication of HBV has also been demonstrated to be significantly repressed by activation of cGAS-STING pathway. 23 In this study, our data indicated that RVX-208 could inhibit HBV particle production by activation of cGAS-STING pathway, which further supported the notion that cGAS-STING pathway played a crucial role in immune defense of HBV. However, our data did not confirm that cGAS-STING pathway played a vital role in RVX-208-mediated inhibition of HBV particle production. We will address this question in our future study and elucidate the molecular mechanism underlying RVX-208-mediated activation of cGAS-STING pathway in the meantime.
Although our study provided novel findings that may inspire anti-HBV drug development, and identified possible molecular mechanisms for the anti-HBV effect of RVX-208, several questions need to be answered in the future. We only showed that RVX-208 could activate cGAS-STING pathway, however, what was the direct target of RVX-208 that played crucial roles in stimulating cGAS-STING signalling remained unknown. The factor that bridged RVX-208 and cGAS-STING pathway might be ApoA-I, or some other downstream target genes of BET transcriptional regulators. Also, the factor might be a novel target of RVX-208 that had nothing to do with BET family. We plan to answer this question in our future study, possibly through chemical proteomic approaches. 24
In conclusion, our study demonstrated that RVX-208, an inducer of ApoA-I, held the potential to suppress HBV particle production. Moreover, we showed that RVX-208 exerted its anti-HBV effect not only via ApoA-I mediated regulation of cholesterol metabolism, but also through activation of cGAS-STING pathway.
Supplemental Material
Supplemental Material - RVX-208, an inducer of Apolipoprotein A-I, inhibits the particle production of hepatitis B virus through activation of cGAS-STING pathway
Supplemental Material for RVX-208, an inducer of Apolipoprotein A-I, inhibits the particle production of hepatitis B virus through activation of cGAS-STING pathway by Dan Shu, Lin Cheng, Kefei Yuan, Dan Liu and He Wei in Antiviral Therapy
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the 2021 annual foundation of Chengdu Medical College (CYZYB21-11), the Science and Technology Major Program of Sichuan Province (2022ZDZX0019), and 1.3.5 project for disciplines of excellence, West China Hospital, Sichuan University (ZYGD22006).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
