Abstract
Although favipiravir is a promising drug for coronavirus disease 2019, some adverse effects, including skin lesions, have been reported. A 56-year-old female who was prescribed favipiravir by a filiation team following a positive severe acute respiratory syndrome coronavirus 2 polymerase chain reaction test presented to our hospital. After examination, favipiravir and paracetamol were prescribed. She represented to the hospital with facial swelling and itchy rashes on her forearm. Angioedema and urticaria were diagnosed. Favipiravir was discontinued. Steroid and antihistaminic therapy were administered for angioedema. To our knowledge, this is the first reported case of favipiravir-induced angioedema and urticaria in Turkey.
Keywords
Introduction
Coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome CoV 2 (SARS-CoV-2), was first reported in Wuhan, China, and has rapidly spread worldwide. As a result, many studies have been conducted to determine an effective treatment for the disease; however, no drugs have been approved by the Food and Drug Administration. After high clinical and radiological improvement rates and faster viral clearances were achieved with favipiravir in China, it was approved for COVID-19 therapy, with or without hydroxychloroquine, based on clinician decision and disease severity [1,2]. However, data on the adverse effects of favipiravir are limited.
Herein, we report a case of favipiravir-induced angioedema and urticaria in
Case Report
A 56-year-old female was prescribed favipiravir (day 1, 1,600 mg twice a day; day 2–5, 600 mg twice a day) after she was tested positive for COVID-19 by the filiation team who visited her home. Because she had a 10-year history of hypertension and had taken amlodipine, she visited our hospital to confirm whether favipiravir was necessary. The patient had no known history of drug allergy or any other allergy. Sub-pleural, localised ground-glass opacities were observed on thorax tomography (Figure 1). Favipiravir and paracetamol were then prescribed. Multiple, bilateral sub-pleural ground-glass opacities.
The patient, however, was readmitted on day 3 of favipiravir use with facial swelling, especially on the lips and around the eyes, and rashes on her forearm (Figure 2). An allergist diagnosed the patient with favipiravir-induced angioedema and urticaria. Favipiravir was discontinued, and methylprednisolone and antihistaminic treatment were administered. Clinical signs regressed in a few hours and completely disappeared within the following 2 days. She did not receive alternative therapy and was followed up with supportive treatment throughout the isolation period. Adverse effects of favipiravir use reported on day 3: Facial swelling, especially on the lips and around the eyes, and rashes on the forearm.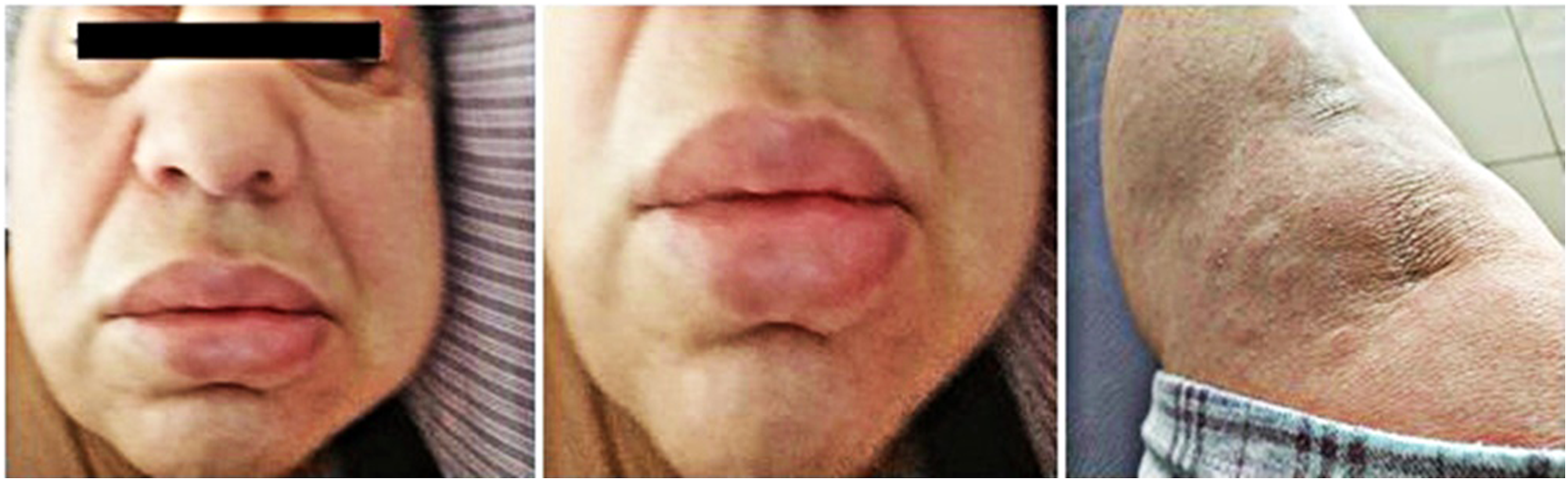
Discussion
Urticaria and angioedema are some of the most common dermatologic emergencies. Urticaria is a vascular reaction induced by various immunological or non-immunological stimuli. Drugs are the most common cause of urticaria [3]. To the best of our knowledge, this is the first case of favipiravir-induced angioedema and urticaria reported in the literature.
Favipiravir, a nucleotide analogue, halts viral replication by selectively and potently inhibiting the RNA-dependent RNA polymerase. It has proven effective against a broad range of influenza viruses and many other RNA viruses [4,5]. Because the discovery of a new and specific antiviral agent against SARS-CoV-2 would take a significant amount of time, favipiravir, which has been proven to be effective against RNA viruses, was first used in Wuhan, China, the epicentre of the pandemic. After a few months of favipiravir use, high clinical and radiological improvement rates and faster viral clearances were achieved in the early clinical studies in China. It is currently being used in Japan, Russia, Ukraine and India [6]. Likewise, favipiravir is approved for COVID-19 therapy in Turkey [2]. Although its adverse effects are generally well-tolerated, elevated transaminases, diarrhoea, nausea hyperuricaemia and neutropenia may be occasionally observed [4,5]. Skin lesions (urticarial lesion, livedo or necrosis, maculopapular eruption, vesicular rash and pseudo-chilblain) in patients with COVID-19 have been reported in the literature with a rate of 2%–20% [7]. According to a recently published review, favipiravir-induced skin lesions are rare [6], but angioedema and urticaria have not been declared yet.
COVID-19 continues to rapidly spread worldwide, suggesting the inefficacy of the current antiviral drugs available. Although favipiravir is currently used to treat COVID-19 in many countries and is generally well-tolerated, adverse effects, including severe hypersensitivity reactions, should be considered, and patients should be informed.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
