Abstract
We report a case of an infant with HIV receiving raltegravir granules for oral suspension and rifampicin-based TB prophylaxis. Raltegravir trough levels remained subtherapeutic and viral load increased during concurrent rifampicin therapy despite using double-dosed raltegravir. Even after rifampicin therapy, a higher dose was needed. This highlights the importance of therapeutic drug monitoring and dose adjustments of raltegravir in infants with rifampicin as comedication.
Introduction
Tuberculosis (TB) is the leading cause of death among children living with human immunodeficiency virus (HIV) worldwide [1]. Treatment optimization of both HIV and TB is challenging because of drug–drug interactions (DDI) and dose recommendations for managing these DDI in paediatric populations are often based on limited clinical data [2].
Rifampicin (RIF) is the cornerstone in the treatment of TB but notorious for causing DDIs as it is a potent inducer of drug metabolizing enzymes, such as uridine 5′-diphosho (UDP)-glucuronosyltransferase 1A1 (UGT1A1) [3]. Raltegravir (RAL), an integrase strand-transfer inhibitor, is registered for treatment of HIV in children from birth [4]. RAL is predominantly metabolized by UGT1A1, resulting in a DDI with RIF.
Limited pharmacokinetic (PK) data in infants and children suggest that twice the usual pediatric dose of the chewable RAL formulation is needed to achieve adequate trough levels whilst on concurrent RIF treatment [5]. Krogstad et al. included 13 infants with HIV aged 4 weeks to <2 years, receiving RAL and RIF simultaneously for pulmonary TB [5]. Doubling the dose of RAL to 24 mg/kg/day b.i.d. in the presence of RIF was safe and resulted in all infants achieving therapeutic plasma concentrations (RAL trough level >0.033 mg/L). To our knowledge, no data is currently available on the use of RAL granules for oral suspension (instead of chewable tablets) during concurrent RIF use for primary TB prophylaxis. Here, we report the individual PK data of RAL before, during and after RIF therapy for primary TB prophylaxis in an infant with HIV using RAL granules for oral suspension.
Case description
A woman (originally from Brazil, aged 29) with HIV was presented at the Amsterdam University Medical Center, Amsterdam, The Netherlands, at 36 weeks of gestation with an HIV viral load of 30,314 copies/mL. At diagnosis, an intensive combination antiretroviral therapy (cART) regimen was started. The viral load dropped substantially to <40 copies/mL 1 day before labour at 40 weeks of gestation. The neonate (birth weight: 3015 g) was healthy and not breastfed. The initial postnatal HIV prophylaxis (PNP) regimen included AZT (8 mg/kg/day b.i.d.), lamivudine (3TC) (4 mg/kg/day b.i.d.) and lopinavir/ritonavir (LPV/r) (32/8 mg/kg/day b.i.d.). At 6 days of life, LPV/r was switched to RAL granules for oral suspension (1.5 mg/kg/day q.d. for 2 days followed by 6 mg/kg/day b.i.d.) due to gastrointestinal adverse effects. PNP was discontinued after 4 weeks according to the local guidelines. At 2 months of age, HIV RNA PCR was positive (viral load: 1.0*107 copies/mL) and the cART (AZT, 3TC, RAL) regimen was restarted after which the viral load dropped to undetectable levels within 5 weeks. The infant’s mother was diagnosed with active TB at age five and a half months. Hence, RIF (10 mg/kg/day q.d.) and isoniazid (10 mg/kg/day q.d.) for primary TB prophylaxis was started and continued for 2 and 3 months, respectively. Cultures for TB and interferon-γ release assay were negative. Dosing of RAL prior, during and after RIF therapy was guided by therapeutic drug monitoring (TDM). Trough levels >0.022 mg/L were considered to be therapeutic. This PK target is derived from multiple studies in which a correlation was shown between RAL trough levels below 0.022 mg/L and viral failure in adults living with HIV using once daily dosing of RAL [6,7]. Even though this target has not been confirmed in subjects using twice daily dosing of RAL, it is a widely accepted reference [8,9]. This target level is used in clinical practice in the Netherlands. RAL trough levels dropped to undetectable concentrations within 2 weeks after RIF initiation and remained subtherapeutic (trough level (C12): 0.017 mg/L) after increasing the RAL dose from 11 mg/kg/day b.i.d. to 25 mg/kg/day b.i.d. (twice the usual dose) (Figure 1). Subsequently, a detectable viral load was measured and RAL dose was increased to 50 mg/kg/day b.i.d.. Two weeks after RIF discontinuation, RAL dose was lowered to 17 mg/kg/day b.i.d, as the enzyme induction by RIF was suspected to be negligible at this stage. However, RAL trough levels remained subtherapeutic (Figure 1) (C12: 0.012 mg/L and C12: 0.017 mg/L). Again, RAL dose was increased to 50 mg/kg/day b.i.d., which is four times the registered dose and therapeutic levels of RAL were achieved (C12: 0.04 mg/L). This dose was continued; no subtherapeutic RAL levels or detectable viral loads were measured thereafter. The UGT1A1 genotype was wild-type (UGT1A1*1/*1; normal enzymatic activity). No other drugs or supplements known to potentially interfere with RAL PK were used. Treatment adherence and administration of RAL oral suspension was believed to be good. A training was provided at the start of RAL therapy to make sure preparation and administration of RAL oral suspension went according to the instructions. No adverse events of RAL were reported.
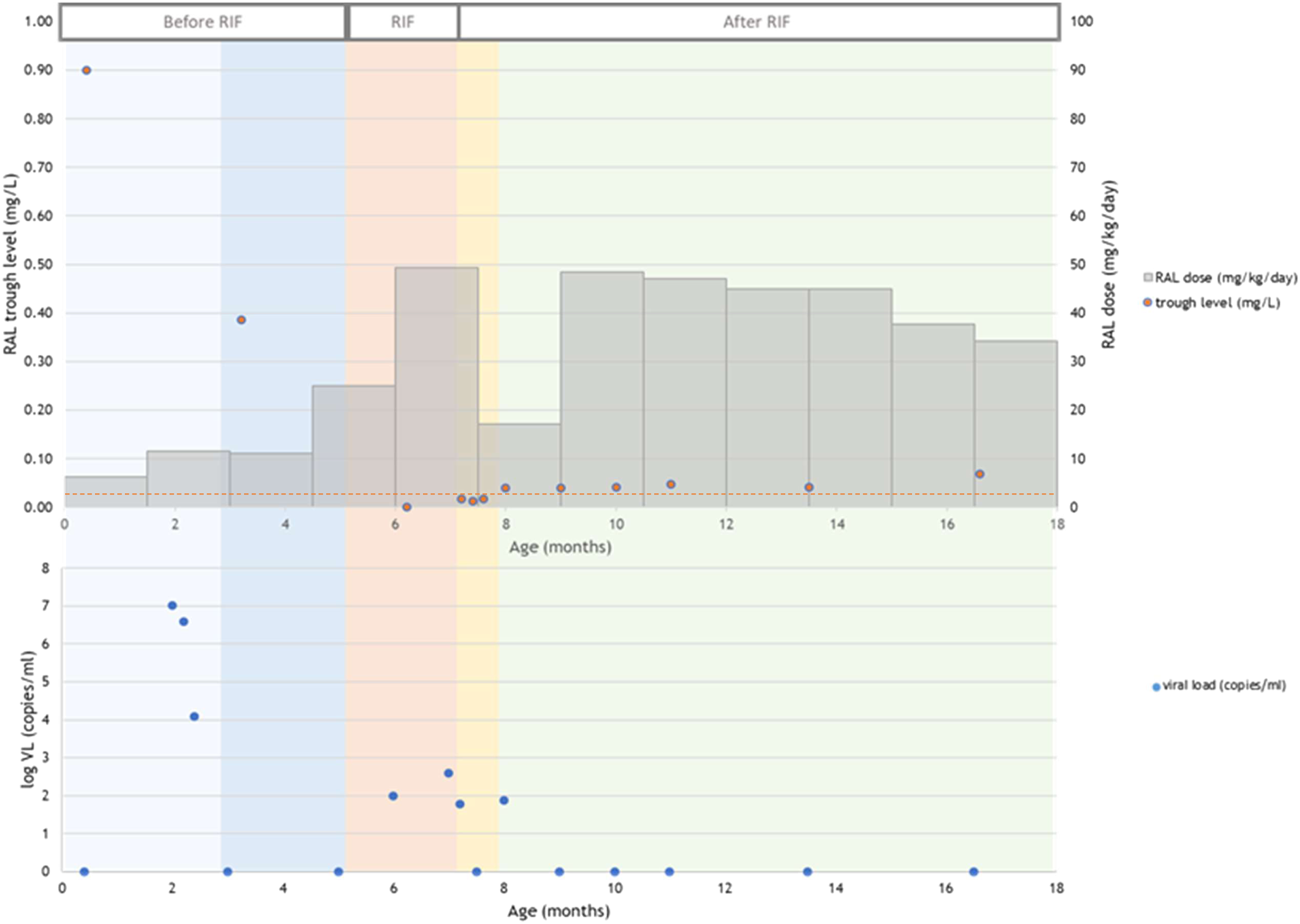
Pharmacokinetic profile of RAL in a patient before, during and after RIF. The light blue field represents the first 2 months of age, the dark blue field represents the period before RIF was started, the orange fields represents the period during RIF and the yellow field represents 1 month after RIF was discontinued. The green box represents the period >1 month after RIF was discontinued. The grey boxes represent the daily RAL dose at each time point; the brown dots represent the RAL trough concentrations and the blue dots display the viral load (logarithmic scale); the brown dotted line represents the RAL target trough level of 0.022 mg/L. Abbreviations: RAL, raltegravir; RIF, rifampicin.
PK modelling
RAL trough plasma concentrations were measured repeatedly (Figure 1). Extensive RAL sampling was performed 6 months after RIF discontinuation at 0, 2 and 6 h after the morning drug dose. These samples were used to determine the individual PK parameters by Bayesian estimation using a previously published 2-compartment model with first-order absorption for RAL in children [8]. To enable changes in the individual PK parameters over time caused by for instance maturation of drug metabolizing enzymes or DDIs, several occasions were specified. Occasions were defined as first and second month after birth, third to fifth month after birth, during RIF treatment, 1 month after RIF discontinuation and longer (up to 10 months) after RIF treatment had ended. As inter-occasion variability (IOV) was not included in the published model, an IOV of 30% was assumed to allow for changes in individual PK parameters over time. Based on the estimated individual PK parameters and target trough level, the optimal RAL dosage per occasion was estimated. Analyses were performed with NONMEM (version 7.4.2, ICON Development Solution, Gaithersburg, MD, USA).
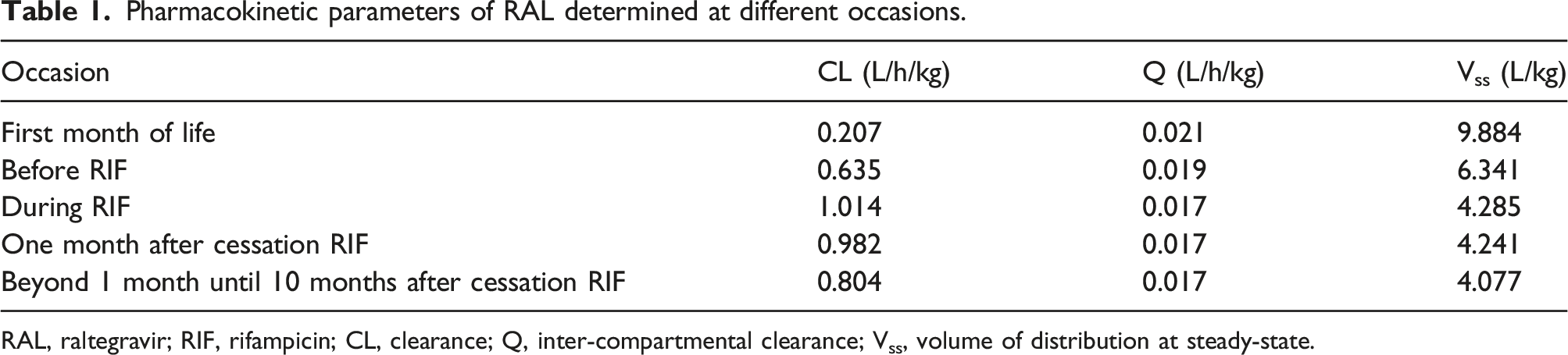
A notable increase in RAL clearance was observed during RIF use (Figure 2, Table 1). Clearance remained higher after RIF discontinuation as compared to prior to RIF use, presumably due to enzyme maturation. Overall, individual clearance of RAL was 157% higher as compared to the mean clearance for a patient of this bodyweight as predicted by the model. Based on the simulations a RAL dose of 34 mg/kg/day b.i.d. (three times the usual dose) would have been needed during co-administration of RIF to maintain adequate trough levels. From 1 month after RIF discontinuation, a RAL dose of 19 mg/kg/day b.i.d. (1.6 times the usual dose) would have been needed. Clearance per kg bodyweight of RAL in a patient before, during and after RIF. The blue field represents the period before RIF was started, the orange field represents the period during RIF and the yellow field represents 1 month after RIF was discontinued. The green box represents the period >1 month after RIF was discontinued. The black dots represent RAL clearance estimated by Bayesian estimation. Pharmacokinetic parameters of RAL determined at different occasions. RAL, raltegravir; RIF, rifampicin; CL, clearance; Q, inter-compartmental clearance; Vss, volume of distribution at steady-state.
Discussion
This case-report is, to our best knowledge, the first to describe the DDI between RAL granules for oral suspension and RIF for TB prophylaxis in an infant living with HIV. Based on PK stimulations, three times the daily registered RAL dose was needed to overcome the DDI with RIF to maintain therapeutic trough levels. Where available, TDM-guided dosing of RAL during concurrent RIF use and up to at least 4 weeks after RIF treatment is discontinued is needed to prevent potential treatment failure in infants with HIV and TB.
Krogstad et al. showed that doubling the dose of RAL chewable tablets to 24 mg/kg/day b.i.d. while concomitantly receiving RIF resulted in adequate PK levels in infants [5]. The difference in RAL dose adjustments between their results and ours (a three-fold increased dose (34 mg/kg/day b.i.d.) of RAL granules for oral suspension) may be explained by RAL’s high intra- and interpatient PK variability, the latter being up to 164% for RAL trough levels in adults [10]. This may be partially explained by differences in UGT1A1 genotype as RAL trough levels were found to be modestly higher in UGT1A1*28/*28 carriers and slightly lower for UGT1A1*36 carriers, as compared to wild-type homozygous carriers [11]. A second explanation is the concomitant effect of UGT1A1 enzyme induction by RIF and UGT1A1 enzyme maturation on clearance, making it difficult to assess the effect of RIF on RAL PK in our patient. On average, UGT1A1 enzyme maturation reaches its full potential at 4 months of age, but may take longer [12].
The impact of RIF on RAL PK is higher in healthy adults as compared to patients with a HIV/TB coinfection [13,14]. Since our patient had no active TB and therefore a lower burden of disease, this may partially explain the higher RAL dose needed in our patient, as compared to the patients with a HIV/TB coinfection of Krogstad et al [5].
A limitation of our study is that the high intra- and interpatient variability complicates characterization of the PK of an individual patient. Although the applied population PK model estimated the observed levels well, PK parameters might differ in time. Also, no RIF levels were determined during RAL therapy, so we were not able to rule out very high RIF levels in this infant potentially causing stronger induction.
During RIF therapy, four times (50 mg/kg/day b.i.d.) the registered RAL dose was safely administered. However, based on model simulations a three-fold increase (34 mg/kg/day b.i.d.) of the registered dose would have been adequate. Two times the registered dose, as previously suggested, resulted in subtherapeutic trough levels and a detectable viral load in our patient. Our patient exhibited a higher RAL clearance without RIF coadministration as compared to the mean population clearance described by the PK model, but still fits within the high interpatient variability seen in RAL PK parameters [8].
Conclusion
We presented a case of a 6-month old infant receiving RIF-based TB prophylaxis who required a three-fold higher RAL dose as compared to the registered dose when using concurrent RIF and up to at least 4 weeks after RIF discontinuation to safely achieve adequate RAL trough concentrations and HIV viral load suppression. This clinically relevant DDI was manageable by applying TDM-guided dosing based on RAL trough concentrations, improving HIV treatment outcome. Additional pharmacokinetic studies are needed to rule out differences in DDI magnitude between RAL chewable tablets and RAL oral granules for suspension with RIF.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
