Abstract
Background
Relationships between distinct antiretroviral therapy (ART) adherence patterns and risk of drug resistance are not well understood.
Methods
We conducted a nested case–control analysis within a longitudinal cohort study of individuals initiating efavirenz-based ART. Primary outcomes of interest, measured at 6 and 12 months after treatment initiation, were: 1) virologic suppression, 2) virologic failure with resistance, and 3) virologic failure without resistance. Our primary exposure of interest was ART adherence, measured over the 6 months before each visit with electronic pill monitors, and categorized in three ways: 1) 6 months average adherence; 2) running adherence, defined as the proportion of days with average adherence over
Results
We analyzed data from 166 individuals (107 had virologic failure during observation and 59 had virologic suppression at 6 and 12 months). Average adherence was higher among those with virologic suppression (median 83%, IQR 58–96%) versus those with virologic failure with resistance (median 35%, IQR 20–77%, pairwise P < 0.01) and those with virologic failure without resistance (median 21%, IQR 2–54%, pairwise P < 0.01). Although treatment gaps generally predicted virologic failure (P < 0.01), they did not differentiate failure with and without drug resistance (P > 0.6).
Conclusions
Average adherence patterns, but not the assessed frequency of treatment gaps, differentiated failure with versus without drug resistance among individuals initiating efavirenz-based ART. Future work should explore adherence-resistance relationships for integrase inhibitor-based regimens.
Introduction
Incomplete antiretroviral therapy (ART) adherence causes virologic failure and drug resistance in individuals living with HIV [1,2]. However, the relationship between adherence patterns and resistance is poorly understood, with existing literature limited to modeling or clinical studies of early ART regimens [3,4]. There is also a lack of data demonstrating how adherence differentially influences virologic failure with and without resistance. Such data is particularly important in sub-Saharan Africa, where pharmacogenetic studies show unique drug metabolism patterns for non-nucleoside reverse transcriptase inhibitors (NNRTIs), which remain in use in many countries [5].
We sought to respond to this literature gap by analyzing adherence data collected among individuals initiating efavirenz-based ART [6]. We characterized adherence patterns using pill monitors among participants categorized into three outcomes: 1) achieving virological suppression, 2) experiencing failure with drug resistance, and 3) experiencing failure without drug resistance. We hypothesized that intermediate adherence would differentiate failure with resistance from virologic suppression and virologic failure without resistance.
Methods
Participants and procedures
Data were derived from the Monitoring Early Treatment Adherence (META) study, an observational cohort of ART-naïve adults initiating once-daily efavirenz/tenofovir/emtricitabine and followed for 12 months in South Africa and Uganda [6]. ART adherence was dispensed according to local guidelines and measured electronically using a real-time monitor (Wisepill Technologies, South Africa). Blood was collected at baseline, 6 months, and 12 months for viral load monitoring.
Sub-study design
We conducted a nested case–control analysis within the META study. Cases were participants who experienced a viral load >1,000 copies/mL at either 6 or 12 months. Controls, matched by country and CD4 stage (>350 versus <200 cells/μL), were sampled randomly from participants who achieved virologic suppression (<40 copies/mL) at both 6 and 12 months. Participants who met case criteria at both 6 and 12 months were included in analyses at both time points. A total of 166 participants were selected for sequencing, with an approximately 2:1 case: control ratio (107:59). Sequencing was performed for all participants at the baseline visit and at all time points when the viral load was >1,000 copies/mL.
Statistical analyses
Our primary clinical outcomes of interest were categorized at the 6- and 12-month visits as: 1) virologic suppression, 2) failure with resistance, or 3) failure without resistance. We excluded individuals with drug resistance at enrollment.
Our primary exposure of interest was ART adherence over the 6 months before each visit, and measured as: average adherence, defined as number of pill monitor opening events observed divided by the number of opening events expected (capped at 100% per day); running adherence, defined as the proportion of days with average adherence over 9 days of less than or equal to 10%, 20%, and 30% (Supplementary Index); [7] and number of 3-, 7-, and 28-day treatment gaps. Statistical methods are detailed in the Supplemental Index.
Ethical statement
This study was approved by institutional review boards at Mass General Brigham, Mbarara University of Science and Technology, the Uganda National Council for Science and Technology, the University of Cape Town, and the Western Cape Provincial Department of Health.
Results
A flow diagram demonstrating selection of participants is displayed in Supplementary Figure 1. Age, sex, CD4 count, and pregnancy status for the participants selected for 6- and 12-month analyses were comparable between cases and controls (Supplemental Table 1). Drug resistance mutations for those failing therapy are displayed in Supplemental Figure 2.
Descriptive statistics and group comparisons for different adherence measures by study visit

Three groups include virologic suppression (1. VS), virologic failure with resistance (2. VF with resistance), and virologic failure without resistance (3. VF without resistance). 6- and 12-month P-values were based on Kruskal–Wallis test with DSCF pairwise adjustment. Combined mean estimates, 95% confidence intervals (CIs), and P-values were based on small-sample adjusted GEEs. IQR: interquartile range.
aIf an overall test P-value was not <0.05, we did not proceed with pairwise comparisons adjusted for multiplicity.
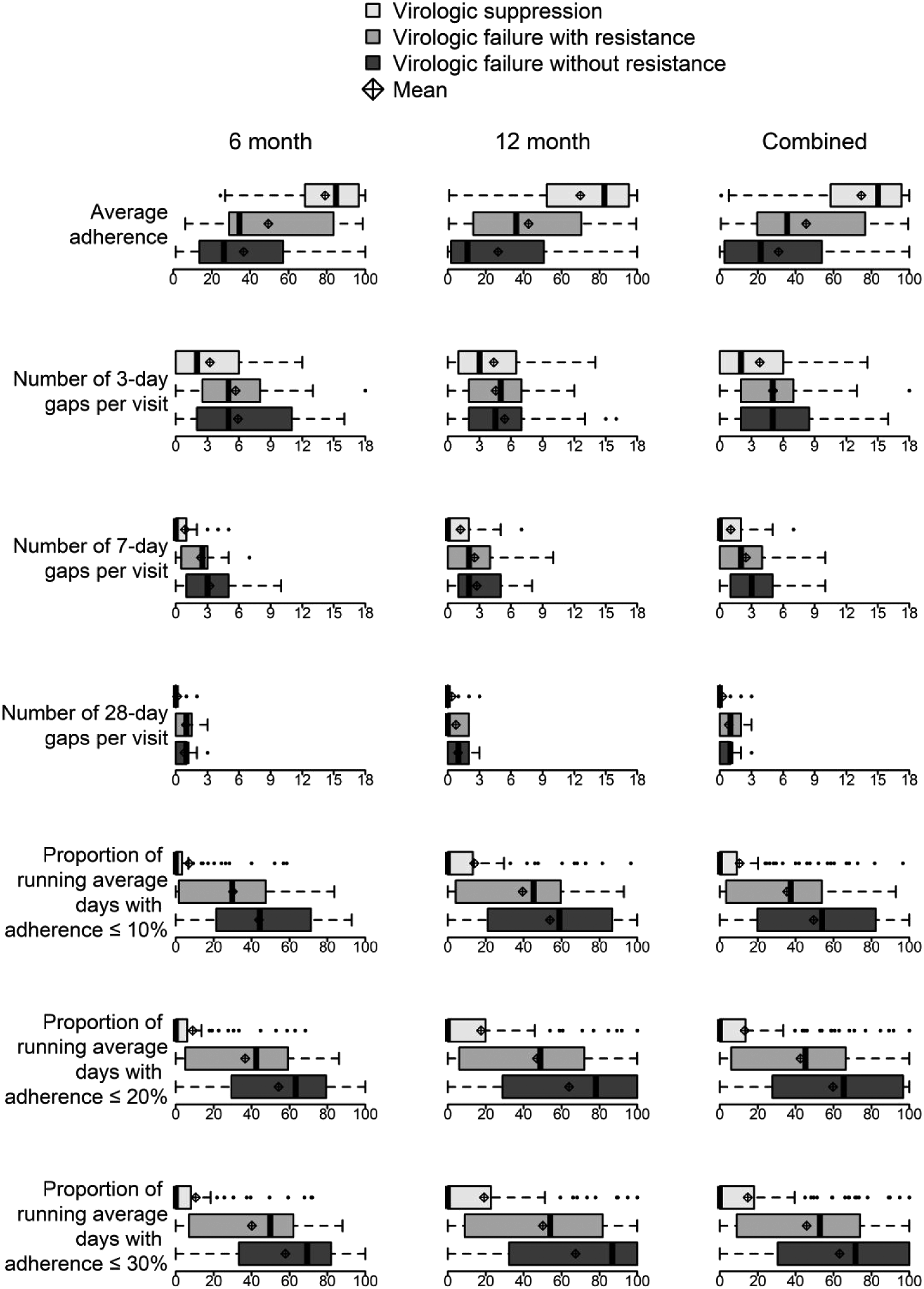
Boxplots of adherence measures by study visit. The boxplot compares virologic outcomes (boxes) by adherence measures (vertical axis) for the 6 months, 12 months, and combined analyses in percentage (horizontal axis). Bold vertical lines denote medians, boxes denote interquartile ranges (IQRs), dashed whiskers extend to 1.5 times IQR or bounds of adherence measure, small dots denote observations beyond whiskers, and diamond-plus symbols denote means. Boxes are colored by group: virologic suppression (light gray), virologic failure with resistance (gray), and virologic failure without resistance (dark gray)
Discussion
Among participants in a prospective observational cohort initiating efavirenz-based ART, average and running adherence differed among the three clinical outcomes of virologic status and drug resistance. Although longer treatment gaps were associated with a higher likelihood of virologic suppression, there were no differences in treatment gaps between those failing with versus without drug resistance. Our results suggest that average adherence and running adherence may differentiate whether people taking efavirenz-based ART fail with or without drug resistance.
Our data related to average and running adherence is consistent with existing literature. Without treatment pressure, mutations attenuate viral fitness compared with wild-type virus [8] Viral dynamic models using drugs with half-lives of 4–12h suggest that a mutation-selection window of drug concentration exists between low and high levels of adherence, during which resistant strains of virus are favored [4,8,9]. Although there is overlap in adherence ranges among each of the clinical outcomes, our data suggest that an intermediate average adherence value (i.e., 50–75%) most favors drug resistance for efavirenz (40–55h half-life). Adherence rates below 50% or above 75% are common in those who experience failure without resistance or are suppressed, respectively. Analogous to average adherence, an intermediate value for running adherence, was associated with failure with resistance.
Our results did not demonstrate a relationship between treatment gaps and the risk of failure with and without resistance. By contrast, in a modeling study, treatment gaps of longer than 4 days for a theoretical drug (12h half-life) had low risk of causing resistance because intermediate drug concentrations were not sustained during these long gaps [4]. When doses were missed consecutively, drug concentrations were at an intermediate level for less time, and thus the virus could not mutate to outcompete wild-type virus. Nonetheless, we found that increasing treatment gaps increased the risk of failing with drug resistance (compared to achieving suppression). Others have also shown that individuals with drug resistance are more likely to have treatment gaps compared to suppressed individuals [10–12]. For example, Oyugi et al. and Meresse et al. found significant relationships between treatment interruptions and drug resistance in patients taking NNRTI-based ART [11,12]. However, our study is the first to our knowledge to compare treatment gap patterns between those with virologic failure with and without resistance. Additional work is needed to understand how adherence patterns uniquely impact resistance for drugs with longer half-lives like efavirenz.
Our results should be interpreted in light of limitations. Although this analysis is among the largest studies to include objective adherence data and resistance testing, our dataset was limited to approximately 100 observations each at 6 months and 12 months. We used real-time electronic adherence monitoring, which depends on participant usage of pill bottles and can under- or overestimate true adherence [13,14]. Future studies should measure plasma drug concentrations to understand their relationship with clinical outcomes. Furthermore, our analysis can only be generalized to treatment-naïve populations achieving suppression rather than treatment-experienced populations maintaining suppression. For example, we have shown much lower rates of average adherence are needed to maintain suppression [15], and a recent clinical trial demonstrates that suppressed individuals can generally maintain suppression with lower average adherence and 3-day treatment gaps [16]. Finally, interrogating a single ART regimen limits the generalizability of our findings. Each regimen is differentially impacted by adherence patterns due to variable pharmacodynamics [10,17]. For example, dolutegravir, an integrase strand transfer inhibitor, was found to be less susceptible to virologic failure even in the context of suboptimal average adherence [18,19]. A study by Parienti et al. found no statistically significant association between treatment interruption and virologic failure for boosted protease inhibitor-based regimens [20]. This contrasted with a study on nevirapine, a NNRTI with a shorter half-life than efavirenz, for which treatment gaps were a risk factor for virologic failure [10]
In summary, our data suggest that average and running adherence patterns may differentiate failure with versus without drug resistance among individuals initiating ART. Although our study is among the first to compare treatment gap patterns between those failing with and without resistance, we did not identify treatment gaps that differentiated failure with and without drug resistance for those on an efavirenz-based regimen. Further studies assessing how combined objective adherence measures can be used to improve clinical outcomes will be valuable to corroborate these findings, particularly for dolutegravir-based regimens now in widespread use in sub-Saharan Africa.
Supplemental Material
Supplemental Material - Antiretroviral therapy adherence patterns, virological suppression, and emergence of drug resistance: A nested case–control study from Uganda and South Africa
Supplemental Material for Antiretroviral therapy adherence patterns, virological suppression, and emergence of drug resistance: A nested case–control study from Uganda and South Africa by Anisha Tyagi, Yao Tong, Dustin J Rabideau, Zahra Reynolds, Tulio De Oliveira, Richard Lessells, Gideon Amanyire, Catherine Orrell, Stephen Asiimwe, Benjamin Chimukangara, Jennifer Giandhari, Sureshnee Pillay, Jessica E Haberer, and Mark J Siedner on behalf of the META Study Investigators in Antiviral Therapy
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JEH has received consultation fees from Merck. All other authors have no conflicts of interest or financial disclosures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Bill and Melinda Gates Foundation (OPP1056051).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
