Abstract
Background
Etravirine (ETR) is approved as a component of second or third-line antiretroviral treatment (ART) for children living with HIV. We assessed the outcomes of ETR-based ART in children in routine care in Europe and Thailand.
Methods
Data on children aged <18 years at ETR start were pooled from 17 observational cohorts. Characteristics at ETR start, immunological and virological outcomes at 12 months, discontinuations, adverse events (AEs) and serious adverse events (SAEs) were described. Follow-up was censored at ETR discontinuation, death or last visit.
Results
177 children ever received ETR. At ETR start, median [IQR] age was 15 [12,16] years, CD4 count 480 [287, 713] cells/mm3, 70% had exposure to ≥3 ART classes and 20% had viral load (VL) <50 copies/mL. 95% received ETR in combination with ≥1 potent drug class, mostly protease inhibitor-based regimens. Median time on ETR was 24 [7, 48] months. Amongst those on ETR at 12 months (n=141), 69% had VL<50 copies/mL. Median CD4 increase since ETR start (n=83) was 147 [16, 267] cells/mm3. Overall, 81 (46%) discontinued ETR by last follow-up. Median time to discontinuation was 23 [8, 47] months. Common reasons for discontinuation were treatment simplification (19%), treatment failure (16%) and toxicity (12%). Eight children (5%) had AEs causally associated with ETR, all dermatological/hypersensitivity reactions. Two were SAEs, both Stevens–Johnson Syndrome in children on regimens containing ETR and darunavir and were causally related to either drugs; both resolved following ART discontinuation.
Conclusion
Children receiving ETR were predominantly highly treatment-experienced, over two-thirds were virally suppressed at 12 months.
Introduction
Second- and third-line antiretroviral treatments (ART) are needed for children and adolescents living with HIV who experience treatment failure and have limited ART options, due to multidrug resistance or drug intolerance [1–3]. Etravirine is a second-generation non-nucleoside reverse-transcriptase inhibitor (NNRTI) approved for use as part of second or third-line ART in children and adolescents aged ≥6 years [4]. Approval was extended to children ≥2 years weighing ≥10 kg in the USA in 2018 [5] and in Europe in 2020 [4]. ETR is included as a potential component of second and subsequent line regimens in previous and current international paediatric HIV treatment guidelines [6–8].
Clinical outcome data for ETR in treatment-experienced children and adolescents are limited to single-arm trials and observational studies with <50 patients. These reported 33%–78% viral suppression <50 copies/mL at 48 weeks [9–14]. Common adverse reactions included gastrointestinal disorders and rashes. This study pooled data from the European Pregnancy and Paediatric Infections Cohort Collaboration (EPPICC) to assess the characteristics, effectiveness and safety of ETR in routine care across Europe and Thailand.
Methods
Individual patient data were pooled from 17 cohorts from 11 European countries and Thailand, for children/adolescents starting ETR aged <18 years, with followed-up data until 31/01/2018. Individual cohorts gained local ethics approval. Data were collected using the HIV Cohort Data Exchange Protocol (www.hicdep.org) and included demographics, clinical and ART history, CD4, viral load (VL), biochemistry results and adverse events (AEs).
Children were categorised into five mutually exclusive groups based on age, treatment history and initial ETR dose for weight: (1) licenced; (2) unlicensed; (3) off-label, ART inexperienced (no prior protease inhibitor (PI) or NNRTI exposure); (4) off-label, age < 6 years; and (5) missing weight/dose. Treatment-experienced children ≥6 years, receiving an ETR dose within ±20% of European dose recommendations for weight at ETR start (twice daily ETR dosing: 100 mg (≥16 to <20 kg), 125 mg (≥20 to <25 kg), 150 mg (≥25 to <30 kg) and 200 mg (≥30 kg)) were assigned to the ‘licenced’ group [4].
Follow-up time in ETR-related clinical trials was excluded. Characteristics at ETR start and frequency and reasons for ETR discontinuation were described. Amongst those on ETR at 12 months, immunological response (change in CD4 cell count) and virological suppression (VL<50 & <400 copies/mL) were summarised. Rates of Division of AIDS (DAIDS) [15] grade ≥3 laboratory AEs were estimated for the licenced dose group (all other groups had sample sizes <20), for absolute neutrophil count (ANC), total cholesterol, triglycerides, alanine transaminase (ALT), total bilirubin, fasting and non-fasting plasma glucose, pancreatic amylase, pancreatic lipase, aspartate aminotransferase (AST), alkaline phosphatase (ALP) and low-density lipoprotein cholesterol (LDL). Rates were reported overall and at <12, 12–24 and >24 months after ETR start. Follow-up time for laboratory AEs was censored at first AE within each time-period or 30 days after ETR discontinuation. Clinical AEs and serious AEs (SAEs) were described and rates of specific events reported. Follow-up time was censored at the earliest of: discontinuation of ETR, death or last visit. STATA version 16 (College Station, TX: StataCorp LLC) was used for all analyses.
Results
Characteristics at etravirine (ETR) start and at 12 months.
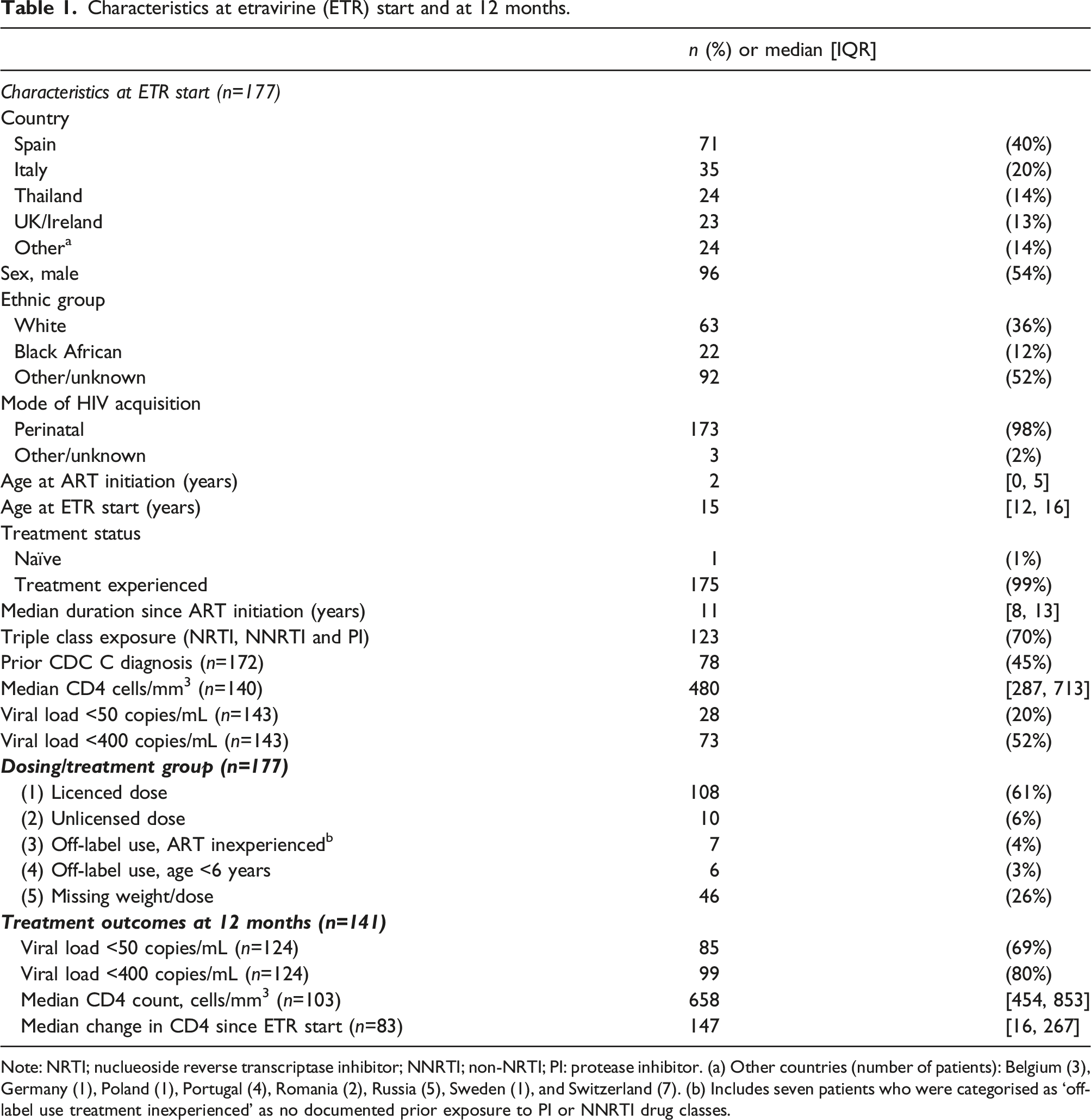
Note: NRTI; nuclueoside reverse transcriptase inhibitor; NNRTI; non-NRTI; PI: protease inhibitor. (a) Other countries (number of patients): Belgium (3), Germany (1), Poland (1), Portugal (4), Romania (2), Russia (5), Sweden (1), and Switzerland (7). (b) Includes seven patients who were categorised as ‘off-label use treatment inexperienced’ as no documented prior exposure to PI or NNRTI drug classes.
Median follow-up after ETR initiation was 24 [7, 48] months. Amongst patients on ETR at 12 months (n=141), 69% (85/124 with data) had VL<50 copies/mL and 80% (99/124) <400 copies/mL, median CD4 (n=103) was 658 [454, 853] cells/mm3 and median CD4 change from ETR start (n=83) was 147 [16, 267] cells/mm3 (p<0.001).
DAIDS grade ≥3 rates of laboratory AEs for the licenced dose group are shown in Figure 1. Rates were highest in the first 12 months on ETR (range, 0.5–4.2 per 100 person-years) and declined thereafter for raised ALT, ALP, bilirubin, total cholesterol, LDL and reduced absolute neutrophil count. For raised pancreatic amylase, rates were highest at 12–24 months and declined thereafter. For raised triglycerides, rates remained relatively constant. No grade ≥3 AEs were reported for raised AST, fasting and non-fasting plasma glucose or pancreatic lipase across all groups (data not shown). Incidence rates (95%CIs) per 100 person-years of follow-up for DAIDS grade ≥3 laboratory adverse events after ETR start, both overall and by time-period in children 6-<18 years taking a licensed dose (N=96 with lab data).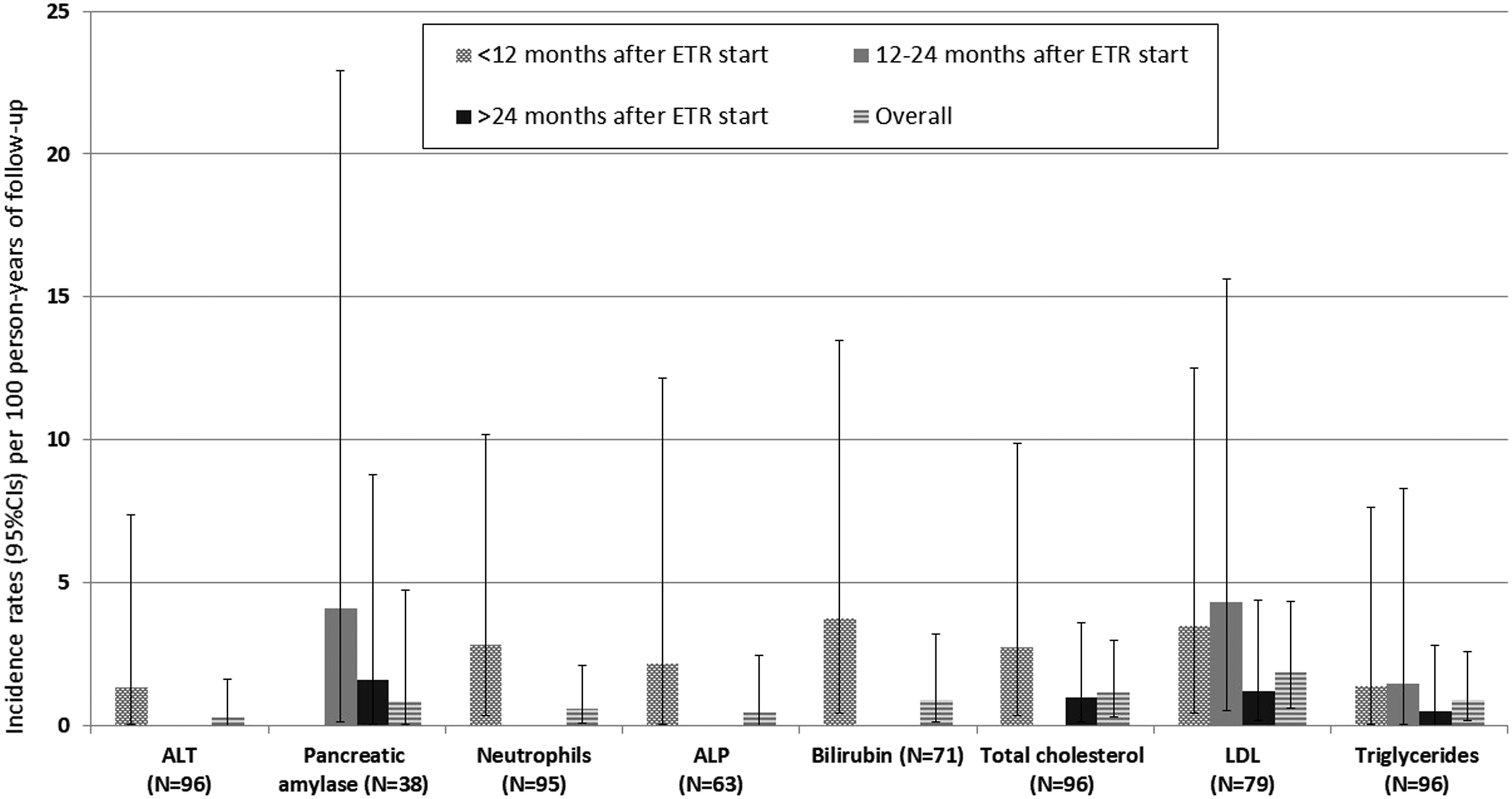
Amongst 138 (78%) patients with clinical data, eight clinical AEs (in eight patients), were reported as possibly, probably or definitely causally related to ETR. All were cutaneous/hypersensitivity reactions occurring within 1 month of ETR start in the licenced group. Two were serious, both Stevens–Johnson Syndrome (SJS), of which one was considered life-threatening. Both patients were on regimens containing ETR and darunavir and were reported as causally related to either drugs. Both events resolved after temporary discontinuation of all ART. The remaining AEs were five rash/erythema and one generalised hypersensitivity/urticaria reaction. Of these, five events resolved (two patients stopped ETR due to the event) and one had unknown resolution status. Amongst patients with clinical data, 5.8% [95%CI 2.7, 10.7] reported skin/hypersensitivity reactions whilst on ETR, of which two (1.4% [95%CI 0.2, 4.7]) were SJS, occurring at a rate of 0.5/100 person-years [95% CI 0.1, 2.1].
Three patients died, two whilst on ETR and one within 3 weeks of stopping. Two were AIDS-associated events and one was an HIV-related metastatic adenocarcinoma. None were considered ETR-related.
Timing of and reasons for ETR discontinuation.

aOf those stopping for toxicity, six were due to hypersensitivity reactions, three due to gastrointestinal toxicity and one due to lipodystrophy.
bOf those stopping in the ‘other’ category, reasons comprised: one due to availability of a more effective treatment, one due to a change in treatment not due to side effects/poor adherence/contra-indication, one due to ETR becoming unavailable and one as study treatment completed.
Discussion
The World Health Organization has highlighted the need for more data on outcomes on paediatric second and third-line ART [16]. This cohort study is one of the largest paediatric studies of ETR in a ‘real world setting’ to date and predominately includes highly treatment-experienced adolescents. The majority received ETR combined with PI-based optimised background regimens. Amongst those on ETR at 12 months, nearly 70% were virally suppressed<50 copies/mL and there was a substantial increase in the median CD4 cell count. These findings are comparable to the PIANO trial, an open-label single-arm ETR trial with optimised background regimen in treatment-experienced children and adolescents aged 6–<18 years (n=101) [9]. At 48 weeks, 56% of children and 68% of adolescents had VL<50 copies/mL and median change in CD4 cell count from baseline was 156 [141, 178] cells/mm3. A Spanish observational cohort (N=23) of treatment-experienced children/adolescents reported similar findings; 78% had VL<50 copies/mL at a median of 48 weeks [10]. The recent P1090 trial on ETR in treatment-experienced children aged 1–6 years reported that among children on the final dose, 75% of those aged 2–6 years and 33.3% of those aged 1–2 years had VL<400 copies or ≥2 log reduction from baseline at week 48 [14].
Approximately half of the participants in our study discontinued ETR by end of follow-up. The most common reason was treatment simplification reflecting availability of new drugs, including integrase inhibitors [17].
DAIDS grade ≥3 laboratory events occurred infrequently, and clinical AEs were uncommon. Two (1.4%) patients experienced Stevens–Johnson Syndrome, a severe and potentially life-threating hypersensitive reaction. Both were taking darunavir concomitantly and the events were reported as causally related to either/both drugs. Both events resolved after temporary discontinuation of ART. Previous studies have reported associations between both ETR and darunavir and SJS/other cutaneous hypersensitivity reactions [18,19]. The prevalence of SJS in our cohort was higher than that previously reported in adults on ETR (<0.1%) [4].
This study has some limitations. The median duration of follow-up on ETR was relatively short at ∼2 years and we did not have data on HIV drug resistance to assess how this may affect treatment response. However, our study includes data from cohorts across Europe and Thailand and contributes important information on outcomes in children/adolescents on advanced therapy. ETR is now approved for children aged ≥2 years, although there remains scarce data in this younger age group and further monitoring is needed. Paediatric ETR use has some disadvantages including twice-daily dosing, lack of a fixed-dose combination formulation and multiple interactions with other antiretrovirals [4,8]. Nonetheless, ETR remains an option to combine with optimised background regimens for treatment-experienced children and adolescents.
Footnotes
Acknowledgements
We thank all the children and adolescents for their participation in these cohorts, and the staff members who cared for them. We thank all the cohort data managers for providing their data and the EPPICC/Penta coordination team: Charlotte Duff, Anna Tostevin (University College London, London, UK), Daniel Gomezpena, Laura Mangiarini and Alessandra Nardone (Penta Foundation, Padova, Italy). We also thank the etravirine teams at Janssen-Cilag International NV for their input to study reports and review of the manuscript, in particular Magda Opsomer, Annemie Van Bael (Janssen-Cilag International NV, Belgium).
Author Contributions
E Chappell, IJ Collins, A Judd, A Lyons and C Giaquinto were responsible for the study concept and design. L Thompson undertook statistical analyses. A Lyons wrote the first draft. All other co-authors provided data for the study. All co-authors participated in discussions about the design of the study. They also critically reviewed the article and approved the final version submitted.
Group Authorship
The European Pregnancy and Paediatric Infections Cohort Collaboration (EPPICC) Study Group
Writing Group
Alex LYONS (MRC Clinical Trials Unit at UCL, University College London, London, UK), Lindsay THOMPSON (MRC Clinical Trials Unit at UCL, University College London, London, UK ), Elizabeth CHAPPELL (MRC Clinical Trials Unit at UCL, University College London, London, UK), Luminita ENE (Clinical Department of Infectious Diseases (HIV Department), Dr. Victor Babeş Hospital for Infectious and Tropical Diseases, Bucharest, Romania), Luisa GALLI (Infectious Disease Unit, Meyer Children's Hospital, Department of Health Sciences, University of Florence, Florence, Italy) Tessa GOETGHEBUER (Hopital St Pierre, Brussels, Belgium), Gonzague JOURDAIN (Institut de recherche pour le développement (IRD)-PHPT, Marseille, France; Faculty of Associated Medical Sciences, Chiang Mai University, Chiang Mai, Thailand), Antoni NOGUERA-JULIAN (Infectious Diseases and Systemic Inflammatory Response in Pediatrics, Infectious Diseases Unit, Department of Pediatrics, Sant Joan de Déu Hospital Research Foundation, Barcelona, Spain), Christian R. KAHLERT (Children’s Hospital of Eastern Switzerland and Cantonal Hospital, Infectious Diseases and Hospital Epidemiology, St Gallen, Switzerland), Christoph KöNIGS (Department for Children and Adolescents, Division for Stem Cell Transplantation and Immunology, University Hospital Frankfurt/Main, Germany), Pope KOSALARAKSA (Department of Pediatrics, Khon Kaen University, Khon Kaen, Thailand), Pagakrong LUMBIGANON (Department of Pediatrics, Khon Kaen University, Khon Kaen, Thailand), Magdalena MARCZYNSKA (Medical University of Warsaw, Hospital of Infectious Diseases, Warsaw, Poland), Laura MARQUES (Centro Hospitalar e Universitário do Porto, Porto, Portugal), Marissa NAVARRO (Hospital General Universitario “Gregorio Marañón”, Madrid, Spain; Universidad Complutense, Madrid, Spain; Instituto de Investigación Sanitaria Gregorio Marañón (IISGM), Spain; Red de Investigación Translacional en Infectología Pediátrica, RITIP, Madrid, Spain), Lars NAVER (Karolinska University Hospital and Karolinska Institutet, Stockholm, Sweden), Liubov OKHONSKAIA (Federal Budgetary Institution “Republican Clinical Infectious Hospital” of the Ministry of Health of the Russian Federation, Saint-Petersburg, Russian Federation), Filipa PRATA (Hospital de Santa Maria/CHULN, Lisbon, Portugal), Thanyawee PUTHANAKIT (Department of Pediatrics, Faculty of Medicine, Chulalongkorn University and HIVNAT, Thai Red Cross AIDS Research Center, Bangkok, Thailand ), Jose TOMAS RAMOS (Departamento de Salud Pública y Materno-infantil. Universidad Complutense. Hospital Clínico San Carlos, Instituto de Investigación Sanitaria del Hospital Clínico San Carlos (IdISSC), Madrid. Spain), Anna SAMARINA (The City HIV centre, St. Petersburg City AIDS Center, St Petersburg, Russian Federation), Claire THORNE (University College London Great Ormond Street Institute of Child Health, London, United Kingdom), Evgeny VORONIN (Federal Budgetary Institution “Republican Clinical Infectious Hospital” of the Ministry of Health of the Russian Federation, Saint-Petersburg, Russian Federation), Anna Turkova ((MRC Clinical Trials Unit at UCL, University College London, London, UK; Great Ormond Street Hospital, London, UK), Carlo GIAQUINTO (Paediatric European Network for the Treatment of AIDS (Penta), Padova, Italy), Ali JUDD (MRC Clinical Trials Unit at UCL, University College London, London, UK), Intira J COLLINS (MRC Clinical Trials Unit at UCL, University College London, London, UK).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AL report grant funding via Penta Foundation from Janssen-Cilag. IJC and AJ report grant funding via Penta Foundation from ViiV Healthcare, AbbVie, Janssen-Cilag and Gilead. EC reports grant funding via Penta Foundation from ViiV Healthcare. CT reports grant funding via Penta Foundation from ViiV Healthcare and Merck.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Janssen-Cilag International, and also received support from the European Union’s Horizon 2020 research and innovation programme under Grant Agreement No 825579 for the REACH study. The MRC Clinical Trials Unit at UCL is supported by the Medical Research Council (programme number MC_UU_12023/26).
Participating cohorts
Hospital St Pierre Cohort, Brussels, Belgium; German Paediatric and Adolescent HIV cohort, Germany; Italian Register for HIV infection in children, Italy; Paediatric Cohort, Poland; Centro Hospitalar do Porto, Portugal; Hospital de Santa Maria/CHLN, Portugal: “Victor Babes” Hospital Cohort, Romania; Federal State-owned Institution “Republican Clinical Infectious Diseases Hospital” of the Ministry of Health of the Russian Federation, St Petersburg, Russia; The City HIV Centre, St Petersburg, Russia; CoRISpe-cat, Catalonia, Spain; CoRISpeS-Madrid cohort, Spain; Karolinska University Hospital, Stockholm, Sweden; Swiss HIV Cohort Study (SHCS) and the Swiss Mother and Child HIV Cohort Study, Switzerland; HIVNAT, Bangkok, Thailand; Kohn Kaen University, Thailand; Programme for HIV Prevention and Treatment (PHPT) Study Group, Thailand; National Study of HIV in Pregnancy and Childhood, UK and Ireland; Collaborative HIV Paediatric Study, UK and Ireland.
