Abstract
Background
This study compared the effectiveness and durability of DTG/RPV with commonly prescribed 3-drug regimens (3-DR) in people living with HIV (PLWH) in a real-world setting.
Methods
Antiretroviral therapy (ART)-experienced, virologically suppressed PLWH who initiated DTG/RPV or a 3-DR in 2018 were identified in the OPERA® database and followed through 6/30/2019. Virologic failure (two consecutive viral loads (VL) ≥ 200 copies/mL or single VL ≥ 200 copies/mL with regimen modification/discontinuation) and maintained virologic suppression (last VL test < 50 or < 200 copies/mL) were described. Kaplan–Meier methods were used to estimate time to virologic failure and treatment discontinuation. Risk of virologic failure was adjusted for age, sex, race/ethnicity, risk of infection, region, baseline CD4 cell count, history of substance abuse or syphilis, and mortality risk score at baseline in a Cox model.
Results
PLWH initiating DTG/RPV were older and more likely to be Hispanic or have comorbidities than 3-DR initiators. DTG/RPV users experienced fewer discontinuations (15%) and were more likely to be suppressed at study end (98%) than 3-DR users (28% and 96%, respectively). Virologic failure was uncommon; rates per 100 person-years did not differ between the DTG/RPV (1.45, 95% CI: 0.69, 3.03) and 3-DR (2.63, 95% CI: 2.21, 3.14) groups. The risk of virologic failure did not differ significantly between the groups in adjusted Cox models (adjusted hazard ratio 1.32, 95% CI: 0.61, 2.89).
Conclusions
The findings of this real-world OPERA® study suggest that DTG/RPV can be a viable alternative to standard 3-DRs for ART-experienced, virologically suppressed PLWH.
Introduction
Triple antiretroviral (ARV) therapy (ART) (two nucleoside reverse transcriptase inhibitors [NRTIs] plus a third agent from either the non-nucleoside reverse transcriptase inhibitor [NNRTI], protease inhibitor [PI], or integrase strand transfer inhibitor [INSTI] classes) has long been the standard of care for people living with HIV (PLWH) [1]. Effective treatments prolong life expectancy, but usually with increasing comorbidities related to age and ART [2], resulting in higher poly-pharmacy and patient adherence concerns [3,4]. Additionally, there are toxicity concerns, especially as they relate to NRTIs and PIs [5–9]. As such, strategies are needed to reduce drug exposure and ART-related toxicities and comorbidities. Switching from an effective regimen to an alternative one might be due to adverse events, drug–drug or drug–food interactions, pill burden, cost, or the desire to simplify a regimen. Newer, more powerful ARVs have introduced the potential for effective therapy while using fewer drugs [10,11].
Dolutegravir/rilpivirine (DTG/RPV) was the first single tablet (50 mg/25 mg), once-daily regimen containing only 2 ARVs to be approved (late 2017) for the treatment of HIV in adults who are virologically suppressed. Current guidelines recommend DTG/RPV as a 2-drug regimen (2-DR) switch strategy for virologically suppressed PLWH without prior virologic failure or drug resistance [1,12,13]. Trials have established the long-term efficacy, safety, and tolerability of DTG/RPV. The SWORD-1 and SWORD-2 open-label, phase 3, non-inferiority studies enrolled virologically suppressed PLWH who were on their first or second ART regimen. At 48 weeks, 95% of PLWH in both the DTG/RPV and current ART regimen groups were virologically suppressed; the single tablet DTG/RPV was non-inferior to current ART and had a safety profile that was expected based on the individual components DTG and RPV [14]. The week 48 results were reproduced in patients who switched to DTG/RPV after week 52 (i.e., the late switch group). At week 100, 89% of the early switch group (95% confidence interval [CI]: 86, 92) and 93% (95% CI: 91, 95) of the late switch group were virologically suppressed; the safety profile continued to be favorable in both groups [15]. PLWH in both the early and late switch groups maintained virologic suppression at 148 weeks (84%) and 96 weeks exposure (90%), respectively, and continued to tolerate DTG/RPV in follow-up analyses. Only 6% of the early switch group and 3% of the late switch group experienced grade 2–4 adverse events (AEs) and only 4 (<1%) serious AEs were drug related. No integrase resistance was identified among 11 PLWH on DTG/RPV who met confirmed virologic withdrawal criteria (viral load ≥ 50 copies/mL followed by viral load ≥ 200 copies/mL) through week 148; NNRTI resistance-associated mutations were identified in only 6 PLWH (<1%) [16].
Clinical trials have established the efficacy, safety, and tolerability of DTG/RPV. The objective of this study was to evaluate the effectiveness (i.e., virologic suppression or failure) and durability (i.e., treatment discontinuation) of a 2-DR of DTG/RPV compared to guideline-recommended 3-drug regimens (3-DR) in ART-experienced, virologically suppressed PLWH in a real-world setting.
Methods
Study design and population
Prospectively captured clinical data from the electronic health records (EHR) of 105,643 PLWH in the Observational Pharmaco-Epidemiology Research and Analysis (OPERA®) cohort were evaluated for inclusion in the study; PLWH were residents of 148 cities in the United States (US) and Puerto Rico. The OPERA® cohort database obtains annual institutional review board (IRB) approval from Advarra IRB, including a waiver of informed consent and authorization for the use of protected health information. The study population included people with HIV-1, but not HIV-2, infection who were aged 13 years or older, ART-experienced, and virologically suppressed to < 50 copies/mL on or prior to initiation of the regimen of interest. The regimens of interest included a 2-DR of DTG/RPV or a commonly prescribed 3-DR containing DTG, elvitegravir [EVG], raltegravir [RAL], darunavir [DRV], RPV, or atazanavir [ATV] plus two NRTIs, boosted or unboosted, initiated between 1 January 2018 and 31 December 2018; included PLWH with no exposure to the regimens of interest prior to initiation. Each person contributed person-time from initiation of the 2-DR or 3-DR until occurrence of an outcome of interest or one of the following censoring events: (a) regimen discontinuation, (b) death, or (c) study end (30 June 2019).
Study outcomes
Effectiveness outcomes of virologic failure and suppression were evaluated, as well as durability of the ART regimen. Virologic failure was defined as either two consecutive HIV RNA viral loads ≥ 200 copies/mL or a single viral load ≥ 200 copies/mL and regimen modification or discontinuation. The time to virologic failure was calculated as the difference between the date of the second component of virologic failure and date of ART initiation. Maintenance of virologic suppression was calculated among PLWH with ≥ 1 viral load test result during follow-up and was defined as having the last viral load test < 50 copies/mL or < 200 copies/mL. With respect to regimen durability, treatment discontinuation was defined as the stop date recorded for any component of the regimen (i.e., modification or discontinuation). The time to treatment discontinuation was calculated as the difference between the first stop date for a regimen component and date of ART initiation.
Statistical analysis
Descriptive statistics for baseline demographic and clinical characteristics, as well as virologic and discontinuation outcomes, were calculated; medians with interquartile ranges (IQR) were used for continuous variables and proportions were used for categorical variables. Statistical comparisons of patient characteristics by regimen groups (i.e., 2-DR vs. 3-DR) were made using Pearson’s chi-square for categorical variables and Wilcoxon rank-sum test for continuous variables. Kaplan Meier methods, including log-rank tests, were used to estimate time to virologic failure and time to treatment discontinuation. Multivariable Cox Proportional Hazards models were used to model time to virologic failure; covariates included in the adjusted model, identified a priori based on the literature, were age, sex, race, ethnicity, risk of infection, region, baseline CD4 cell count, history of substance abuse, history of syphilis, and Veterans Aging Cohort Study (VACS) Mortality Index Score at baseline. CD4 cell count and the VACS index were modeled continuously, with quadratic terms to introduce flexibility. All other variables were modeled categorically. A sensitivity analysis was conducted to assess the impact of adherence associated with multiple tablet regimens in the 3-DR group in which analyses were repeated in a population restricted to individuals on a single tablet regimen.
Results
Study population
A total of 6069 ART-experienced, virologically suppressed PLWH initiated the 2-DR of DTG/RPV (n = 545) or a guideline-recommended 3-DR (n = 5524) between 1 January 2018 and 31 December 2018. More than half of PLWH in the 3-DR group initiated regimens containing EVG (34%) or DTG (31%) (Figure 1). The most frequent 3-DRs, which represented over 80% of all 3-DRs, were EVG/c/FTC/TAF (32%), DTG/ABC/3TC (17%), RPV/FTC/TAF (16%), DTG/FTC/TAF (12%) and DRV/c/FTC/TAF (6%). The most common ART regimen prior to the regimens of interest was EVG/c/FTC/TAF in both groups (2-DR: 23%; 3-DR: 10%). Distribution of core agents in the 3-drug regimens.
Baseline demographic and clinical characteristics of ART-experienced people living with HIV initiating a 2-drug or 3-drug regimen.

3-DR, 3-drug regimen; AIDS, acquired immunodeficiency syndrome; ART, antiretroviral therapy; DTG, dolutegravir; IQR, interquartile range; IVDU, intravenous drug use; MSM, men who have sex with men; RPV, rilpivirine; µL, microliter; VACS, Veterans Aging Cohort Risk Index.
aMissing: n = 3.
bMissing: n = 53.
cDTG/RPV initiators were more likely to have a baseline history of cardiovascular disease, cancer, endocrine disorders, renal disease, and hypertension than 3-DR initiators, who were more likely to have a history of substance abuse.
dVACS Mortality Index: Scored by summing pre-assigned points for age, CD4 count, HIV-1 RNA, hemoglobin, platelets, aspartate and alanine transaminase, creatinine, and viral hepatitis C infection. A higher score is associated with a higher risk of 5-year all-cause mortality [22]. Missing: n = 791.
Effectiveness: Virologic outcomes
Virologic failure among ART-experienced people living with HIV initiating a 2-drug or 3-drug regimen.

CI, confidence interval; DTG, dolutegravir; n, number; RPV, rilpivirine.
aAdjusted for age (13–25, 26–49, 50+), male sex, African American race, Hispanic ethnicity, risk of infection reported as men who have sex with men, region (South, Midwest, Northeast, West), baseline CD4 cell count (continuous, quadratic), history of substance abuse, history of syphilis, and VACS score at baseline (continuous, quadratic).
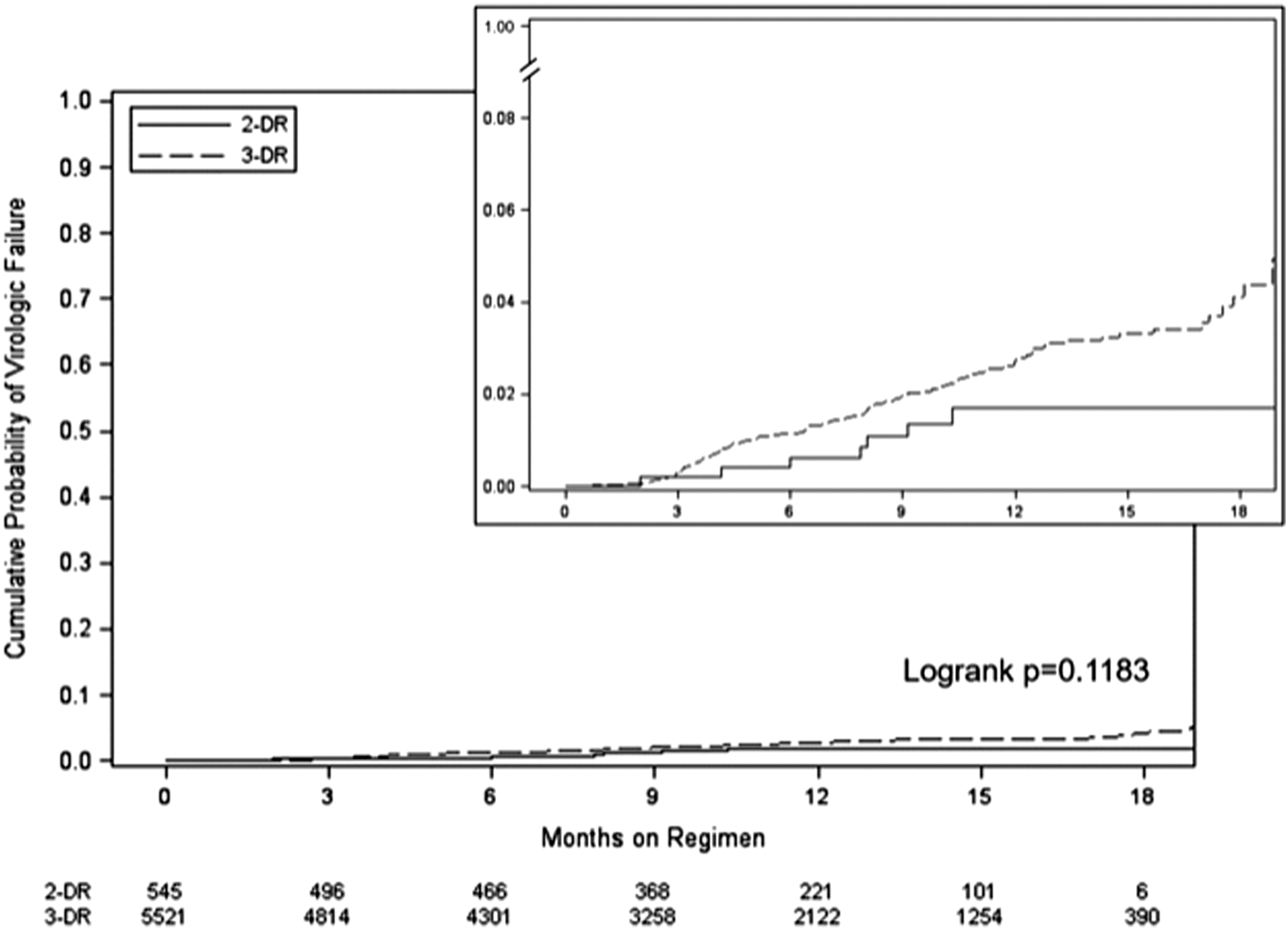
Unadjusted cumulative probability of virologic failure with a 2-drug regimen of dolutegravir and rilpivirine versus a 3-drug regimen.
Durability: Treatment discontinuation
The median follow-up (i.e., months on a study-eligible regimen) was similar between the groups (11 months, IQR: 7, 15). Seventy-three percent of PLWH were still on their ART regimen at study end, though PLWH initiating DTG/RPV experienced significantly fewer discontinuations (n = 82, 15%) than PLWH initiating a 3-DR (n = 1,544, 28%) (Figure 3). The median time to discontinuation was also similar between the 2-DR (4 months, IQR: 2, 7) and 3-DR (5 months, IQR: 2, 9) groups. Among the 1626 PLWH who discontinued their ART regimen, almost half (47%) were virologically stable at the time of discontinuation. Deaths were rare and did not differ between groups, with 5 deaths in the 2-DR group experienced (0.9%) and 28 deaths in the 3-DR group experienced (0.5%; p = 0.2141). Unadjusted cumulative probability of discontinuation with a 2-drug regimen of dolutegravir and rilpivirine versus a 3-drug regimen.
Sensitivity analysis: Excluding multiple tablet regimens
In a sensitivity analysis excluding multiple tablet regimens, 71% of the 3-DR group (n = 3942) was compared to the 2-DR group (n = 545). As in the main analysis, no difference in the risk of virologic failure was observed (aHR: 1.09; 95% CI: 0.49–2.46) (Supplemental Material).
Discussion
In this real-world analysis, PLWH initiating DTG/RPV were older and more likely to be Hispanic, receive care in the southern US, or have comorbidities than PLWH initiating a 3-DR. DTG/RPV users experienced fewer discontinuations (15% vs. 28%) and were more likely to be suppressed at study end than 3-DR users, regardless of the viral load threshold considered (< 50 or < 200 copies/mL). Virologic failure was uncommon and rates per 100 person-years did not differ between the DTG/RPV 2-DR (1.45, 95% CI: 0.69, 3.03) and 3-DR (2.63, 95% CI: 2.21, 3.14) groups. Differences in the risk of virologic failure between the groups in adjusted Cox models were not significant (aHR 1.32, 95% CI: 0.61, 2.89).
Findings from this study in OPERA® are consistent with other evaluations of DTG/RPV in real-world settings. In the Dat’AIDS Cohort Study in France, 152 ART-experienced (41% INSTI-naïve), virologically suppressed (for a median 10 yrs) PLWH were switched to DTG/RPV and followed for a median 36 weeks. Virological failure occurred in only 3 (2%) individuals and only 19 (13%) individuals discontinued the 2-DR [17].Of the 104 ART-experienced PLWH in the DORIVIR Spanish Cohort who switched to DTG/RPV primarily due to toxicity (42%), convenience (28%), or drug interactions (17%), 79% were virologically suppressed at baseline. At 24 weeks, 95 (91%) remained on DTG/RPV and 92 (89%) were virologically suppressed to a viral load < 50 copies/mL (efficacy: 88% (intention to treat) and 97% (per protocol)). Discontinuations were not attributed to adverse events [18]. The TivEdO cohort included 145 PLWH who had switched to DTG/RPV; 73% were virologically suppressed to < 50 copies/mL at baseline and all were followed for a median 101 weeks. Half of the PLWH had previously failed at least one ART regimen and a vast majority switched to DTG/RPV for simplification (52%) or toxicity (37%) reasons. At week 96, 138 (95%) of PLWH were virologically suppressed and 123 (85%) had undetectable viral loads [19,20].
The 105,643 PLWH in the OPERA® cohort database at the time of this study represent approximately 8% of PLWH in the US [21] and included people from clinics of varying size, specialty (i.e., primary care or infectious disease), and geographic setting (e.g., urban and rural). EHR data from this large and diverse population of PLWH in care provided accurate and detailed information on DTG/RPV prescriptions in a real-world setting, virologic outcomes, and treatment discontinuation. The large OPERA® cohort provided a unique opportunity to evaluate over 500 PLWH who were prescribed a 2-DR of DTG/RPV during the study period and compare outcomes with over 5500 PLWH who were prescribed a variety of 3-drug regimens.
This study is not without limitations. Though the outcome of virologic failure was rare (2%), there was no resistance data available for the PLWH who experienced the outcome. The low rate of virologic failure, specifically among PLWH initiating DTG/RPV, increases the likelihood of false negative results (i.e., type II statistical error). Despite this low event rate limitation, this OPERA® cohort included a substantially larger group of PLWH initiating DTG/RPV (n = 545) than other observational studies (n = 104 to 152 in cohorts described above). Reasons for discontinuation of an ART regimen, which occurred among approximately a quarter of the included PLWH and was significantly more common in the 3-DR group, were not available. There were differences between groups at baseline, including a higher prevalence of substance abuse in the 3-DR group. These differences may represent a channeling bias where providers prescribed 3-DR to PLWH perceived as more likely to be non-adherent. However, this may be balanced by the higher prevalence of comorbidities and higher VACS scores in the 2-DR group. The Cox proportional hazards model evaluating virologic failure between the 2-DR and 3-DR groups was adjusted for important confounders, but a substantial number of PLWH did not have a VACS Index score available (n = 791). In a sensitivity analysis in which the VACS Index was dropped from the modeling, virtually identical results were observed (aHR: 1.33, 95% CI: 0.61, 2.91). However, unmeasured and residual confounding potentially remain. Finally, much has been written on the benefits of single tablet regimens for adherence. In this study, only 71% of the 3-DR consisted of a single tablet, in contrast to 100% of the 2-DRs. All participants were virologically suppressed at index, suggesting adequate levels of adherence in both groups. Nevertheless, we undertook a sensitivity analysis excluding multiple tablet regimens to control for potential differences in adherence. No difference in risk of virologic failure was observed.
Trial data and real-world evidence have suggested that DTG/RPV is non-inferior to standard 3-DRs and is well-tolerated. Among ART-experienced, virologically suppressed PLWH initiating DTG/RPV as a 2-DR or a standard 3-DR, there was no observed difference in the risk of virologic failure, which was uncommon, in a real-world setting. The findings of this OPERA® study suggest that a 2-DR of DTG/RPV can be a viable alternative to standard 3-DRs for ART-experienced, virologically suppressed PLWH who have comorbidities. Continued evaluation of DTG/RPV and other 2-DRs in observational settings is needed to evaluate longer-term effectiveness and durability.
Supplemental Material
sj-pdf-1-avt-10.1177_13596535211073235 – Supplemental Material for Dolutegravir/rilpivirine 2-drug regimen comparable to commonly prescribed 3-drug regimens up to 18-months in a real-world setting
Supplemental Material, sj-pdf-1-avt-10.1177_13596535211073235 for Dolutegravir/rilpivirine 2-drug regimen comparable to commonly prescribed 3-drug regimens up to 18-months in a real-world setting by Gerald Pierone Jr, Jennifer S Fusco, Vani Vannappagari, Laurence Brunet, Rachel P Weber, Michael Aboud, Jean van Wyk, Leigh Ragone, and Gregory P Fusco in Antiviral Therapy
Footnotes
Acknowledgments
This work was funded by ViiV Healthcare. This research would not be possible without the generosity of people living with HIV and their OPERA® caregivers. Additionally, we are grateful for the following individuals who are (or were at the time of the study) employed by Epividian, Inc. (Durham, NC, USA): Kathy Schulman (Analyst), Lito Torres (SAS programming), Jeff Briney (QA), Bernie Stooks (Database Architecture & Management), Judy Johnson (Medical Terminology Classification), and Rodney Mood (Site Support).
Author contributions
GP and JSF share the responsibility for the design of this study and protocol development. GP, JSF, and GPF contributed to the acquisition of data. JSF and LB are responsible for all the analyses, conducted according to the protocol. GP, JSF, LB, RPW, and GPF contributed to the interpretation of results. JSF, LB and RPW drafted the manuscript. All authors have critically reviewed and approved the manuscript and have participated sufficiently in the work to take public responsibility for its content.
Declaration of conflicting interests
The author(s) declared the following potential conflicts: GP is a member of the Epidemiology and Clinical Advisory Board for Epividian. JSF, LB, RPW, and GPF are employed by Epividian, Inc.; Epividian has had research funded by ViiV Healthcare, Merck & Co., Janssen Pharmaceutica, Gilead Sciences, and AIDS Healthcare Foundation. VV, MA, JvW, and LR are employed by ViiV Healthcare and hold stocks and shares in GSK as part of their employment. It is Epividian's policy to publish the results of all studies, regardless of the results, for full transparency; the sponsor of this study did not direct the study design, analysis, or submission of this manuscript for publication.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by ViiV Healthcare.
This research was presented in part at the Conference on Retroviruses and Opportunistic Infections (CROI 2020), Boston, USA, March 8-11, 2020.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
