Abstract
Background
To evaluate the impact of preemptive acyclovir treatment on herpes simplex virus (HSV) bronchopneumonitis in mechanically ventilated patients with HSV oropharyngeal reactivation.
Methods
Ancillary study of the Preemptive Treatment for Herpesviridae (PTH) clinical trial. Patients included in that trial from one centre (Pitié-Salpêtrière Hospital) and in whom at least one bronchoalveolar lavage (BAL) was performed for ventilator-associated pneumonia suspicion were included in the present study. Rate of HSV bronchopneumonitis, defined as clinical symptoms suggesting of pneumonia and presence of HSV in BAL fluid ≥105 copies of HSV/106 cells, were compared in patients who received either acyclovir or placebo.
Results
Eighty-three patients were included; 40 having received preemptive acyclovir and 43 having received a placebo, without differences between groups at admission or at randomization. The number of patients who developed HSV bronchopneumonitis was lower among acyclovir-treated patients than among placebo-treated patients (40% vs. 72%, respectively, p = .003). Results were similar when restricted to patients without HSV detected in the lower respiratory tract at randomization (31% vs. 61%, respectively, p = .03).
Conclusions
Preemptive acyclovir treatment in mechanically ventilated patients with HSV oropharyngeal reactivation reduces HSV bronchopneumonitis rate.
Introduction
In the recently published Preemptive Treatment for Herpesviridae (PTH) trial, preemptive use of acyclovir for mechanically ventilated (MV) patients with oropharyngeal herpes simplex virus (HSV) reactivation failed to demonstrate a positive impact on day-60 ventilator-free days (VFDs), although a trend towards lower day-60 mortality rate was found in patients treated with acyclovir as compared to those having received placebo. 1 Those results could be explained by the lack of acyclovir effectiveness, lack of HSV pathogenicity in MV patients, wrong outcome choice (day-60 mortality rate being more accurate than day-60 VFDs) 2 or inability of acyclovir to prevent HSV bronchopneumonitis. Indeed, the rate of HSV bronchopneumonitis was similar in the two groups. 1 Although the morbidity of HSV reactivation in the lower respiratory tract (and therefore its treatment) may be disputable, 3 HSV bronchopneumonitis, that is, lung parenchyma involvement, is associated with potentially increased morbidity and/or mortality.4,5 HSV bronchopneumonitis is defined by clinical signs suggesting of pneumonia, presence of HSV in the lower respiratory tract and histological criteria, that is, HSV-specific nuclear inclusions in cells collected during bronchoalveolar lavage (BAL). 4 However, the detection of those inclusions are subjected to intra- and inter-observer variability, may be difficult to search and frequently missed. 6 The use of virus load in BAL fluid has been described as a surrogate of histology: a cut-off of 105 copies of HSV/106 cells had a good accuracy to predict HSV bronchopneumonitis 4 and was associated with prognosis. 5 In the recent PTH trial, the authors only used the histological definition for HSV bronchopneumonitis diagnosis, 1 and therefore may have underdiagnosed some episodes.
We therefore performed this retrospective study to evaluate, within the PTH trial, the impact of preemptive acyclovir treatment on HSV bronchopneumonitis in patients with HSV oropharyngeal reactivation, HSV bronchopneumonitis being defined by clinical symptoms suggesting of pneumonia and the presence of HSV in BAL fluid ≥105 copies of HSV/106 cells.
Methods
All patients included in the PTH trial1,7 in one participating centre (Pitié-Salpêtrière hospital) were retrospectively reviewed and included. The PTH trial, a double-blind, placebo-controlled, randomised study was designed to evaluate the impact of preemptive acyclovir, given intravenously at a dose of 15 mg/kg/day during 14 days, on MV duration, assessed by the number of days alive and free from MV at day 60 (MV-free days). Results of this study have been published elsewhere. 1 Patients randomized in the study and suspected of having developed ventilator-associated pneumonia (VAP) underwent fiberoptic bronchoscopy and BAL. 8 BAL fluid was send to the bacteriological lab for bacterial culture and susceptibility testing, and to the virology lab for virus testing. In this, latter were tested for HSV genome and albumin gene were quantified using in-house real-time polymerase chain reactions (PCRs), as previously described, and HSV load was calculated and expressed in copies of HSV/106 cells collected by BAL.9,10 In patients with multiple episodes of VAP suspicion, BAL was repeated as frequently as needed.
We retrospectively defined HSV bronchopneumonitis as VAP suspicion plus HSV load in BAL ≥105 copies of HSV/106 cells. 4 Rate of HSV bronchopneumonitis were compared in both groups (acyclovir-treated and placebo-treated).
Ethics
The PTH trial was approved by an independent ethics committee (Comité de Protection des Personnes Sud Mediterranee 5). 1
Results
Characteristics and outcome of patients.
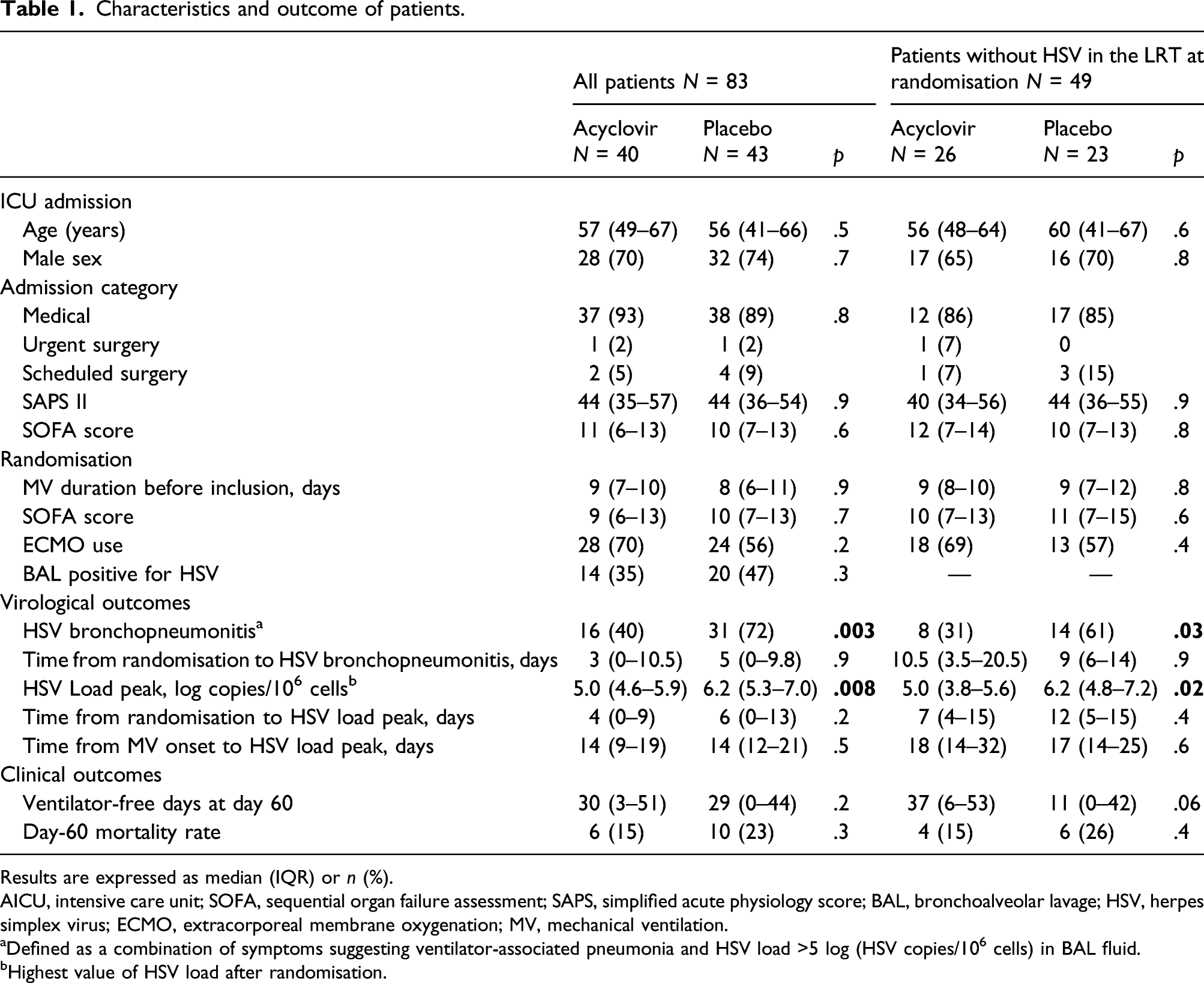
Results are expressed as median (IQR) or n (%).
AICU, intensive care unit; SOFA, sequential organ failure assessment; SAPS, simplified acute physiology score; BAL, bronchoalveolar lavage; HSV, herpes simplex virus; ECMO, extracorporeal membrane oxygenation; MV, mechanical ventilation.
aDefined as a combination of symptoms suggesting ventilator-associated pneumonia and HSV load >5 log (HSV copies/106 cells) in BAL fluid.
bHighest value of HSV load after randomisation.
Outcomes
Virological outcomes are displayed in Table 1. Patients having received preemptive acyclovir developed less frequently HSV bronchopneumonitis than those having received a placebo, and their peak of HSV load in the BAL fluid was lower than the peak for patients having received a placebo. Ventilator-free days at day 60 and day-60 mortality rates were similar in both groups.
Since having HSV reactivation in the respiratory tract at randomisation may have an impact on HSV bronchopneumonitis rate, we separately analysed the 49 patients without HSV in the lower respiratory tract at randomization (26 in the acyclovir arm and 23 in the placebo arm). Their characteristics at ICU admission and randomisation were similar (Table 1). The rate of HSV bronchopneumonitis was lower in patients having received acyclovir, as compared to placebo, as well as their highest HSV load in BAL fluid (Table 1). Mortality rate and day-60 ventilator-free days were not different, although trend towards higher ventilator-free days was observed in the acyclovir-treated patients.
Interestingly, 40% of patients having received acyclovir (31% of those without HSV in the lower respiratory tract at randomisation) developed HSV bronchopneumonitis despite this treatment (Table 1).
Discussion
In this ancillary study of the PTH trial, we found that acyclovir, given as preemptive treatment in patients with HSV oropharyngeal reactivation, allowed to reduce the incidence of HSV bronchopneumonitis. Moreover, although we were unable to show any significant effect on outcome, there was a trend toward higher number of MV-free days in the subgroup of patients without HSV reactivation in the lower respiratory tract at randomisation and treated with acyclovir. Since HSV bronchopneumonitis may be deleterious, 4 this decrease in its incidence might explain, at least in part, the non-significant difference in mortality rates observed in the PTH trial. 1 However, the use of acyclovir at a dosing of 15 mg/kg/day during 14 days did not suppress the risk of developing HSV bronchopneumonitis. Whether higher dose and/or prolonged duration of treatment may be more efficient remains to be determined.
Limitations of our work include the limited number of patients and the inclusion of only patients from one centre. Moreover, 16 patients included in the PTH study had no BAL and were therefore not included in the present ancillary study. However, it is unlikely that those patients developed HSV bronchopneumonitis; they were not sampled because they had no signs suggestive of pneumonia, which defines HSV bronchopneumonitis. 4
Conclusion
In conclusion, preemptive use of intravenous acyclovir, at a dosing of 15 mg/kg/day during 14 days, in mechanically ventilated patients with HSV oropharyngeal reactivation, may reduce the frequency of HSV bronchopneumonitis. Whether this decrease has an impact on outcome remains to be determined.
Footnotes
Authors contribution
AT and CEL conceived and designed the study, performed the analysis and wrote the first draft of the manuscript. SB, OB and DB performed the virological analysis. AT, MPdC, MS, NB, GH, AC and CEL included the patients. All authors made significant contribution, read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. None for the present study. The PTH trial was funded by the Direction de la Recherche Clinique et du Développement and the French Ministry of Health; Programme Hospitalier de Recherche Clinique 2011
Disclosure
DB reports having receiving fees from Biomérieux, Qiagen, Roche Diagnostics outside the submitted work. Other authors have no conflicts of interest to disclose. CEL reports having receiving fees from Bayer Healthcare, Thermo Fisher Brahms, Biomérieux, Faron, Carmat, Aerogen, Merck Sharp and Dohme, outside the submitted work.
