Abstract
The development of direct-acting antivirals (DAA) has revolutionized the treatment of chronic hepatitis C, enabling cure of hepatitis C virus (HCV) infection in more than 95% of cases. There are essentially no contraindications, so almost any patient can now be successfully treated. The result is the prevention or amelioration of cirrhosis, hepatocellular carcinoma (HCC), and extrahepatic manifestations. Consequently, the 2020 Nobel Prize in Medicine and Physiology was awarded for the discovery of HCV. Due to the high efficacy of therapy, even global HCV elimination is conceivable even without a vaccine. Here, we would like to venture a SWOT analysis of current HCV therapies aimed at HCV elimination.
Introduction and a brief historical review of the development of HCV therapy
In 1989, 31 years before the Nobel Prize was awarded, the identification of hepatitis C virus (HCV) as the causative agent of non-A and non-B hepatitis was published.
1
Even before that, interferon alfa (IFN) was used and led to normalization of transaminases and improvement of liver histology in some patients.
2
After the identification of HCV, it became possible to measure the success of therapy by the sustained elimination of HCV RNA from the serum, the sustained virological response (SVR). Since then, SVR rates have increased from 5 to 20% with IFN monotherapy and up to 40–50% with the combination of IFN + ribavirin (RBV) and finally to almost 100% today with direct-acting antiviral agents (DAA) (Figure 1). In the period between recombinant IFN monotherapy and DAA therapy, the development and approval of pegylated interferon alfa (PEG-IFN) improved the pharmacokinetics of IFN, allowing more convenient dosing intervals and higher SVR, especially in HCV genotype 1 (GT1) patients.3,4 In addition, the treatment duration was individualized based on viral kinetics and genotype.5,6 There was even a brief period where genetic testing for IL28b further refined therapy.7,8 Finally, a major breakthrough was achieved with the development of direct-acting antivirals (DAA) against HCV starting in 2003 (9). The main targets for DAA are the NS3/4A protease, the NS5B polymerase and the NS5A replication complex. Combinations of different DAA from these different classes allow for very effective treatments. The first DAA tested in humans was the protease inhibitor (PI) BILN-2061 (ciluprevir),9,10 but drug toxicity in animal models halted further development. Finally, in 2011, the first selective PI were approved for patients with HCV GT1. Boceprevir (BOC) and telaprevir (TLV) improved SVR rates up to 75% in naïve HCV GT1 patients and 29–88% in treatment-experienced HCV GT1 patients (reviewed in11,12). However, both PIs had to be combined with PEG-IFN + RBV, as monotherapy would lead to rapid development of drug resistance. Furthermore, these two PIs cannot be combined as they have the same target and cross-resistance occurs. Telaprevir had to be administered at least twice daily and BOC three times daily, and both PIs are associated with severe side effects, especially anemia and skin rash.13,14 In 2014, new DAAs were approved. Simeprevir (SMV) was the first once-daily PI. Sustained virological response rates for treatment-naïve GT1 patients increased to 80–81% with PEG-IFN + RBV plus SMV.15,16 Sofosbuvir (SOF) was then the first once-daily NS5B polymerase inhibitor available (approved 12/2013 by FDA and 1/2014 by EMA) and became the “final nail in the coffin for hepatitis C”.
17
The resistance barrier of SOF was much higher compared to the available PIs. Therefore, a combination of SOF and RBV was sufficient for some patients. Valid data were first published for genotypes 2 and 3 with SVR rates of 85–100% in treatment-naïve GT2/3 patients,
18
with the very first data coming from New Zealand.
19
SOF was then promptly combined with a PI or an NS5A inhibitor. Treatment with SOF + SMV resulted in an SVR of 92% in GT1,
20
which was later also confirmed in large real-world cohorts.
21
The combination of SOF with the NS5A inhibitor daclatasvir (DCV) or ledipasvir (LDV) were the first NS5A-based IFN-free combination therapies to also show an SVR of > 90%.22–25 Importantly, the combination of SOF + DCV (approved by the EMA 8/2014) and the fixed-dose combination of SOF/LDV as a single tablet (approved by the FDA 10/2014 and by the EMA 11/2014) showed an SVR of > 95% in GT1 patients with treatment failure on prior triple therapies PEG-IFN + RBV/PI.22,25 Sofosbuvir in combination with DCV also has activity against other genotypes, including GT3.
26
The so-called 3D regimen, ombitasvir (OBV), paritaprevir/r (PTV/r) and dasabuvir (DSV) (approved by the FDA in 12/2014 and by the EMA in 1/2015 for GT1 and GT4 patients), was the first combination to include DAAs against all three targets.27–30 In 2016, the fixed-dose combination elbasvir (EBR) plus grazoprevir (GZR) was approved and became the first regimen for patients with chronic kidney disease (CKD), a single-tablet regimen for GT1 and GT4.31,32 The first pangenotypic DAA combinations SOF plus velpatasvir (VEL)33,34 and glecaprevir (GLE) plus pibrentasvir (PIB)35,36 were approved in 2016 and 2017, respectively. Finally, the triple fixed-dose combination SOF/VEL plus the PI voxilaprevir (VOX) was approved in 8/2017, allowing retreatment of patients who have failed DAA therapy.
37
Development of chronic hepatitis C therapy. Sustained virological response (SVR) which is equivalent to cure has improved from around 5% with conventional recombinant interferon alfa monotherapy in the early 90s to > 95% today with DAA combinations (data for treatment-naïve GT1 patients). Indicated trials are not head-to-head and it is difficult to compare SVR between different studies because the populations had significant differences in genetic and socioeconomic backgrounds.
Having witnessed this great success, it is now time to conduct a SWOT analysis to be well prepared for the ultimate goal of HCV elimination (Figure 2). SWOT analysis of current DAA therapies aiming at HCV elimination.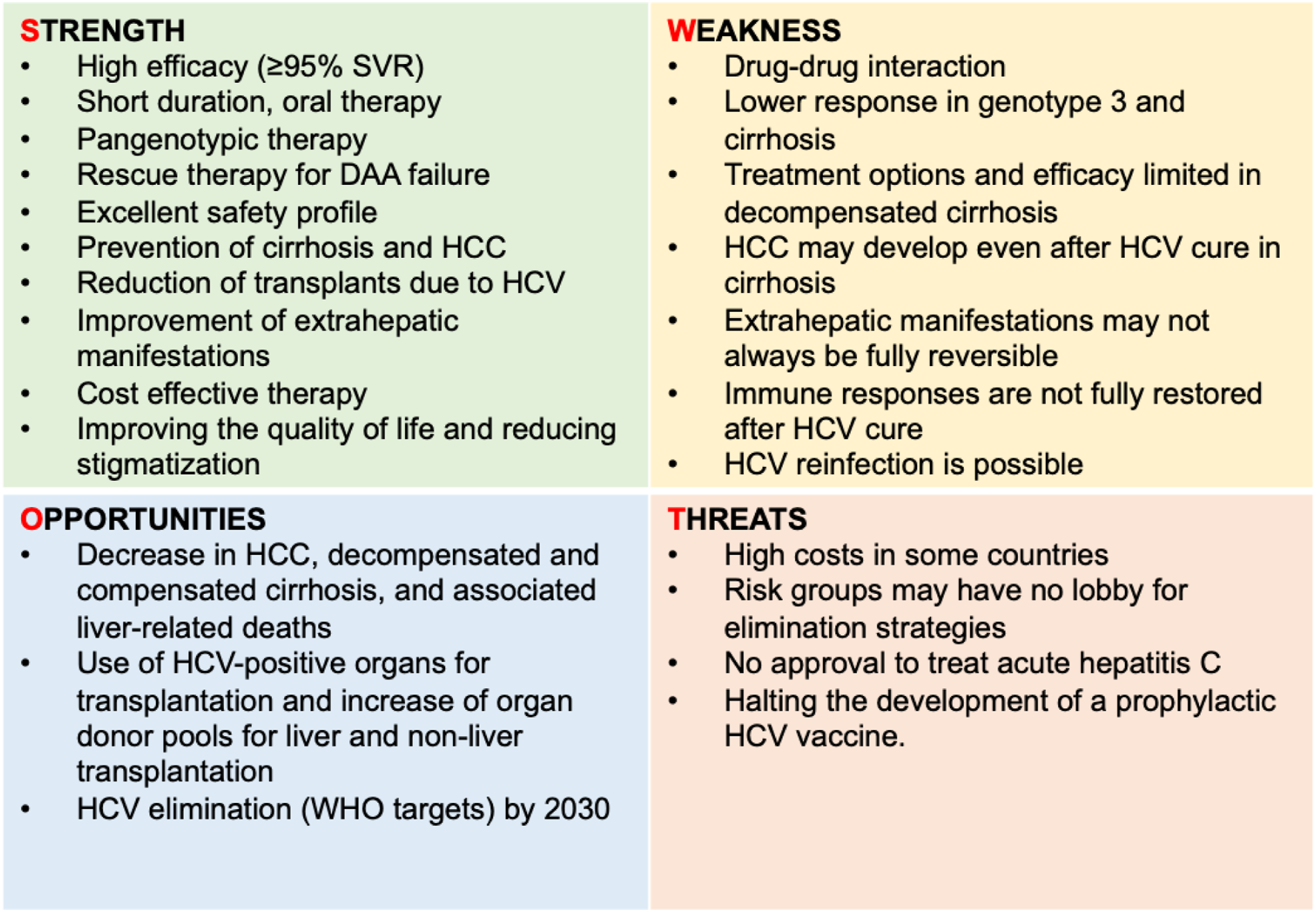
Strength of HCV therapy
All currently available and approved IFN-free DAA regimens are administered orally over a short period of time, usually 8–12 weeks, and have an excellent safety profile and similar efficacy of greater than 95%. 38 Real-Word Data have confirmed the high efficacy demonstrated in the phase III trials.39,40 Importantly, long-term benefits of SVR are the reduction of HCV-related hepatocellular carcinoma (HCC) and overall mortality. Eradication of HCV with DAA reduces the risk of HCC by more than 70%. 41 It has been shown that patients with SVR, even patients with compensated cirrhosis, have a similar life expectancy compared with the general population. 42 In patients with advanced and decompensated cirrhosis, SVR can lead to improvement of liver function tests,43,44 hepatic encephalopathy 45 and may reduce the need for liver transplantation.46,47 In addition, extrahepatic manifestations improve after successful HCV elimination (reviewed in48,49).
Preventing sequelae of chronic hepatitis C will also reduce or prevent long-term health care costs, such as expenses for treating the complications of cirrhosis including transplantation. However, the extent of cost-effectiveness certainly depends on the cost of DAA therapy, which can vary from country to country and can sometimes be very substantial, even in the four-digit range. Nevertheless, several studies from different areas of the world concluded that DAA therapy is highly cost-effective.50,50,51
In addition to preventing HCV-related clinical complications and economic reasons, eliminating HCV will reduce the stigma of infection among patients,52,53 and studies have shown that HCV cure is associated with improved quality of life (reviewed in54,55).
Currently, three pangenotypic DAA therapies are available as single-tablet regimens: SOF/VEL, GLE/PIB, and SOF/VEL/VOX.38,56 In countries where generic DAAs are available, the combination of SOF plus DCV could also be considered a pangenotypic regimen. 57 These pangenotypic combination therapies pave the way for elimination strategies by allowing simplified management, that is, without the need for genotyping. Both, latest EASL recommendations and AASLD guidelines promote a simplified strategy for naïve patients with chronic hepatitis C.38,56
Weakness of HCV therapy
Direct-acting antiviral agents can be inhibitors and substrates of P-glycoprotein (P-gp) and cytochrome P450 3A4, which are regularly involved in drug-drug interactions (DDI). 58 This was in particular relevant for first-generation PIs. 59 In addition, there are other mechanisms such as altered gastric pH that reduces the bioavailability of some DAAs.60,61 However, even current DAAs still exhibit some degree of DDI, and up to 40% of patients may be affected. The reduced DDI potential of modern DAA regimens compared with earlier DAAs is partially offset by changes in patient characteristics, particularly psychotropic drugs commonly taken by people who inject drugs (PWID). 62 Therefore, DDIs should not be underestimated. Careful evaluation of potential DDIs is essential to avoid adverse effects or unnecessary risk of treatment failure. In most cases, however, the drugs can be exchanged or even paused for the short term.
Another potential weakness of oral DAA therapy is reduced bioavailability due to reduced absorption in the presence of altered gastrointestinal pH, gastric emptying or intestinal transit time disorders and reduced absorption surface area. Thus, in patients who have undergone gastric surgery, the response to therapy may be impaired and thus the risk of DAA resistance may be increased. 63
Although pangenotypic therapies are available, patients with genotype 3 and cirrhosis have to be considered as a special challenge. Cirrhotic patients infected with genotype 3 who have NS5A-resistant associated substitution (RAS) Y93H achieve suboptimal SVR rates with SOF/VEL, and RBV is required to increase SVR to ≥ 95%. 64 Therefore, the latest EASL recommendations address this issue by recommending that patients infected with genotype 3 who have compensated cirrhosis should be treated with 12 weeks of SOF/VEL plus RBV if chosen. 38 Treatment recommendations for GLE/PIB are also more complex in genotype 3, as patients with treatment experience (prior PEG-IFN therapy) require 12 or, in the case of compensated cirrhosis, 16 weeks of therapy 38 although the data are limited in this situation.
There has been some concern for some time whether DAA therapy, especially SOF-based DAA therapy, can be used in patients with impaired renal function, as approximately 80% of SOF is excreted by the kidneys (after dephosphorylation to the nucleoside metabolite GS-331,007). Therefore, SOF has not been recommended for a long time in patients with a GFR < 30 mL/min/1.73 m2. However, there are now sufficient data to show that SOF-containing regimens are not only effective but also safe in patients with moderate to severe renal impairment, including patients with a GFR < 30 mL/min/1.73 m2 and patients with end-stage renal disease requiring hemodialysis,65–67 and thus treatment is not contraindicated in this setting. 38 Therefore, this weakness is now apparently resolved.68,69
In patients with decompensated (Child-Pugh B or C) or compensated (Child-Pugh A) cirrhosis with previous episodes of decompensation, PI-containing regimens are contraindicated 38 and SOF based treatments would ideally require additional RBV for an optimal response. 38 This could limit treatment options, especially for genotype 3 and patients after DAA failure as SOF/VEL/VOX contains a PI. As stated above, patients with decompensated cirrhosis may improve after HCV elimination43,44 but not all patients benefit, especially not those with higher MELD scores.70,71 Thus, patients with decompensated (Child-Pugh B or C) cirrhosis without HCC awaiting liver transplantation with a MELD score ≥ 18–20 should be transplanted first, without pretransplant antiviral treatment, and HCV infection should be treated and cured after liver transplantation. 38
As mentioned above as an important strength, DAA therapy reduces the risk of developing HCC and improves extrahepatic symptoms and quality of life. However, not all patients show the same benefit. Patients with advanced fibrosis or cirrhosis and certain risk factors (e.g., diabetes mellitus, older age, and low albumin) remain at risk for developing HCC even after HCV cure.72–75 Hepatitis C virus infection has been shown to induce epigenetic, signaling, and gene expression changes in the liver associated with altered innate liver immunity, which may persist after HCV cure and may explain persistent HCC risk in some patients. 76
In addition, quality of life does not improve in all patients, 77 suggesting that the long-term consequences of HCV may persist for at least some time, reminiscent of “Long COVID.” In this context, there are some research data suggesting that the altered immune response does not fully recover after HCV cure, for example, the interferon-associated cytokine milieu was not normalized, 78 which could be associated with fatigue or other extrahepatic manifestations. 79 Hepatitis C virus-specific cellular immune responses are also not fully recovered, leaving HCV a scar on the immune system,80–82 which may explain why patients can still be reinfected after HCV cure. So, in conclusion, there is a cure for the viral infection but not a complete biological cure in every case.
Opportunities of HCV therapy
In 2016, due to the great success of DAA therapies the World Health Assembly (WHA) unanimously adopted the resolution that viral hepatitis should be eliminated by 2030. Hepatitis C can be eliminated in principle as a public health threat (i.e., 90% reduction in new chronic infections, 65% reduction in mortality compared to a scenario in which interventions would continue at current levels). Mathematical modeling predicted that increasing SVR by DAAs to current levels of greater than 90% and increasing treatment uptake over the next 15 years will result in a 75% decrease in HCC, compensated and decompensated cirrhosis and liver-related mortality. 83
The ability to almost always cure HCV infection with DAA has led to consider transplantation of organs from HCV-positive donors, being not only anti-HCV but also HCV RNA positive. Several studies have shown excellent results in the setting of kidney, liver, heart, and lung transplantations (reviewed in 84 ). Pre-emptive treatment at the time of transplantation 85 or early treatment of acute HCV infection post-transplant 86 results in prevention of transplant hepatitis (reviewed in 84 ). Transplantation of HCV-positive organs offers the opportunity to increase the donor pool and thus transplantation rates, thereby reducing mortality on the waiting list. However, the intentional transmission of an infectious agent to recipients has significant ethical implications.
Threat to HCV therapy and HCV elimination efforts
The key to HCV elimination is to increase the uptake of treatment. Almost all patients can now be treated and cured with modern DAA therapies as described above, but often physicians are still reluctant to treat patients with active drug or excessive alcohol use, especially if the therapy is costly. These high-risk patients have no lobby in the health system, and networks to improve care for patients with addiction problems (drugs or alcohol), prison inmates or the homeless are insufficient but urgently needed if HCV elimination efforts are to succeed. In fact, 76% of high-income countries are not on track to meet WHO HCV elimination targets by 2030 and 60% are off track by at least 20 years. 87 However, especially for these risk groups, the therapy of chronic HCV infection represents a chance to eliminate one of the liver-toxic causes besides alcohol or drugs. Often, this can also be a reason to get the addiction problem under control. However, a prerequisite for starting therapy is good motivation of the patient and co-care by addiction experts, otherwise the treating physicians will experience negative examples and continue to hold back with screening and treatment in these risk groups. Lack of political will to establish the required infrastructure and networks taking care of those risk groups is a serious threat to the elimination strategy. In fact, political will was the highest predictor of when countries will achieve WHO elimination targets and was moved up in priority above all other factors. 88
If no screening is carried out in risk groups, there is a danger that patients will be diagnosed too late and become so-called late presenters. Hepatitis C virus infection should be diagnosed early, before HCV infection is diagnosed at later stages. Treatment for hepatitis C should ideally be initiated before cirrhosis develops.
Another threat in particular for high-risk groups is the fact that treatment is only approved for chronic but not acute hepatitis C. As of today, patients with acute hepatitis C or high-risk group patients usually must wait 6 months after initial diagnosis before treatment can be initiated. This carries the risk that patients will not return for therapy and thus are lost in the health care system. 89 These patients on the other hand may and will infect others with HCV. 90 However, DAA therapy is extremely effective and safe in this setting91–93 and an unrestricted DAA availability among HIV-positive men who have sex with men (MSM) has shown to reduce acute infections by 51%. 94 Thus, EASL recommends the treatment of acute hepatitis C, 38 but pharmaceutical companies are encouraged to take all measures to ensure that DAAs are officially approved for the treatment of acute hepatitis C as well.
Nevertheless, even if all the problems described above are resolved and the proposed measures are successful, doubts remain as to whether elimination is possible with DAA alone and without prophylactic vaccination.95,96 Since the success of DAA therapy has been so dramatic, most vaccination programs have not been pursued vigorously, in particular by big pharma. Yet there are still some consortia working on prophylactic vaccination, namely academic centers and innovative small biotech companies. Recently, a remarkable study was published evaluating a vector vaccine (recombinant chimpanzee adenovirus three vector vaccine followed by a recombinant modified vaccinia virus Ankara boost) encoding HCV nonstructural proteins in patients at risk for HCV infection due to recent injection drug use. Vaccination was unfortunately unsuccessful in preventing chronic HCV infection, but it demonstrated that HCV vaccination trials are feasible in principle and at least immune responses could be elicited and viral loads were lower in vaccinated individuals. 97 This should encourage consortia to continue working on vaccination. Promising preclinical data are certainly available. 96
Final conclusion
The history of hepatitis C is a prime example of successful basic, translational and clinical research that has become a masterpiece of modern translational research in medicine, from its initial definition as non-A, non-B hepatitis via first IFN based therapies towards the discovery of the virus and subsequent development of DAA therapies leading to ≥ 95% cure. This development may make it possible to eliminate an infectious disease for the first time without a prophylactic vaccine. To this end, we conducted a SWOT analysis, which shows that while this goal is possible, many challenges and threats remain to be tackled. The chapter of hepatitis C is not completely closed yet. Following viral discovery and development of highly efficacious well tolerated medical therapies HCV has now become a public health challenge.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MC received honoraraia from Abbvie, Falk, Gilead, GlaxoSmithKline, Jansen-Cilag, MSD Sharp & Dohme, Novartis, Roche, Spring Bank Pharmaceuticals, Swedish Orphan Biovitrum, outside the submitted work. MM received honoraria from Roche, BMS, Gilead, Novartis, Merck, Assembly Biosciences, Enyo Pharma, Curevac and Eiger outside this submitted work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
