Abstract
Background
Gilead Sciences, under Dr. John Martin’s leadership, created its Global Access Program to deliver high-quality, affordable medicines to treat and ultimately eliminate some of the world’s most challenging-to-treat, pervasive, life-limiting diseases not as philanthropy but based on a self-sustaining business model—a highly novel concept in the pharmaceutical realm. John was determined to bring together all key stakeholders from public health officials to doctors and patients around the globe to understand barriers and opportunities in HIV, viral hepatitis, and visceral leishmaniasis (VL) and in so doing, pushed the Gilead team to devise novel strategies to address healthcare disparities in resource-challenged geographies.
Purpose
This case study provides an overview of the evolution and impact of the Gilead Access to Medicines Program in providing treatments for HIV in low- and lower middle-income countries.
Introduction and Background
The Centers for Disease Control and Prevention (CDC) published its first scientific report describing the disease now known as AIDS in its Morbidity and Mortality Weekly Report on 5 June 1981 [1]. At that time and in the ensuing early years of the HIV/AIDS pandemic, an HIV diagnosis was widely considered to be a death sentence. This is in sharp contrast to today’s once-daily antiretroviral pill regimens which can make a virus both undetectable on tests and untransmittable to others. Though not curative, this intervention has meant that HIV patients are able to live fairly normal, long, healthy lives while experiencing low side effects. The pivotal technology that enabled this change was the evolution of highly active antiviral therapy or HAART [2]. The first antiretroviral (ARV) drug for HIV was introduced in 1987, and although this and subsequent drugs were shown to be effective, patient compliance was abysmal. The early drugs had to be taken multiple times per day and for extended periods of time with a range of negative side effects, leading to significant numbers of patients reducing or stopping treatment entirely.
In 2001, under the leadership of its late Chairman and CEO Dr. John Martin, Gilead Sciences’ Viread was approved by the FDA as an essential treatment option in the three-drug HAART regimen necessary to combat HIV/AIDS [3,4]. A mere 5 years later, Gilead transformed the treatment landscape with the FDA’s approval of the world’s first single-tablet regimen (STR), Atripla, which combined all three HAART HIV drugs into a single pill. Gilead continued to innovate in this space at a blistering pace, introducing a total of 6 HIV single-tablet regimens or STRs in the period of 2006 to 2018—Atripla, Complera, Stribild, Genvoya, Odefsey, and Biktarvy.
A range of treatment options meant that HIV could now be treated as a chronic illness, yet, despite being a global pandemic, the impacts were highly skewed. Estimates showed that in 2007, there were 33 million people living with HIV, 2.7 million new infections, and 2 million HIV-related deaths. Low- and lower middle-income countries were hit the hardest, accounting for more than 95% of new infections [5,6].
Gilead was acutely aware of this disparity and recognized that the development of innovative medicines for life-threatening diseases was only one aspect of improving public health. Under John Martin’s steadfast leadership, the Gilead Access Program invested in initiatives that promoted prevention, the strengthening of healthcare systems infrastructure, education, and financial support to the most vulnerable communities around the world (Figure 1). Gilead access to medicines scientific and social contract
Discussion
The Gilead Access Program has enabled access to essential and life-saving medicines for HIV, hepatitis B (HBV), hepatitis C (HCV) and visceral leishmaniasis for tens of millions of people in low- and middle-income countries. It is an innovative program and a prime example of John Martin taking his scientific training and experience and applying it to solving global health issues. The access program personifies John Martin’s passion, perseverance, his belief, and insistence that medical science should be directed to where need is greatest. John believed that it is not enough to discover a medicine and advance it in clinical trials; he believed it is equally important to ensure equitable access to all, including people living in resource-limited settings. That John’s pioneering vision for equitable access to Gilead’s medicines occurred prior to Gilead Sciences becoming profitable is a testament to his inherent goodwill and long-standing belief that life-saving treatments should be available to those in need, despite the prevailing notion that biopharma should focus on profits in core markets (Figure 2). Gilead 2003 access program announcement
It is hard to say when the planning for the Gilead Access Program started. As we have learned, John Martin would have an idea, and in a very calculated and methodical manner, he would begin to lay the groundwork for that idea. John probably started thinking about how to bring HIV therapy to resource-challenged settings in the mid 1990s as he saw the devastation the HIV/AIDS pandemic was causing, particularly on the continent of Africa. He understood how the unique science of nucleotide analogs could be part of a solution to end the suffering.
For us working closely with John, what felt like the beginning was a Friday afternoon in November of 2001, shortly after tenofovir disoproxil tumarate (TDF) was approved by the FDA in the United States with the brand name Viread [3,4]. John came to us informally and explained that Viread, with its once-daily dosing, high barrier to resistance and favorable safety and tolerability profile, was an ideal component of antiretroviral therapy (ART) for the developing world. John presented us with a straightforward and challenging goal: “figure out how to get it to as many patients as possible in developing countries as soon as possible.”
With this very firm directive, the team began to develop an Access to Medicines Program. We looked for successful examples among our competitors and quickly learned that none existed. Most of what we did see were public relations-based programs with little to no evidence that they delivered medicine. John was very clear that he was not interested in a corporate social responsibility (CSR) program or trying to generate positive PR—he wanted the access program to be real, to improve patients’ lives. John also did not want it to be philanthropy. He wanted a program that delivered high-quality, affordable medicines based on a self-sustaining business model (Figure 3). Barriers to access
The first versions of the access program from 2003 to 2006, while well intentioned, did not meet John’s directive. In 2006, only 30,000 people were receiving TDF-based regimens. The need was greater than 30 million. The reasons for this shortfall were numerous. The program provided TDF at no-profit prices, but that price was still far more than other generic HIV treatments such as D4T and AZT. At the time, Gilead supplied products on a named patient basis, but this proved to be very cumbersome and unscalable. Gilead also failed to recognize the need to obtain listing on the WHO essential medicines list, and local healthcare providers did not understand or have access to up-to-date medical information on TDF [5].
During this time, and always, we routinely traveled with John to all the major medical conferences, as well as trips to Africa, Asia, Latin America, and Eastern Europe. At these conferences and while traveling with John, we would meet with non-governmental organizations (NGOs) and patient advocates, visit healthcare providers, clinics, and ministries of health to understand access barriers. At night, John would convene us, often in the hotel lobby, and quiz us on what we had learned and steer us to solutions. It was through these experiences that we would learn the reasons for the failure of the first versions of the program.
Over time, John drove us to make the necessary changes to break down access barriers. He insisted that we establish an independent accountable team to lead the access program. John allocated regulatory resources to locally register Gilead products quickly and transparently and wherever needed, with the goal that low-income countries with the greatest need could obtain registration within 1 year of a new product being registered in the United States. Within the access team, John insisted on a separate medical team to communicate the data to WHO and local ministries of health. The access team developed a network of distribution partners around the world to assist in registrations, medical communications, pharmacovigilance/safety reporting, and procurement. Finally, John addressed the gap between Gilead’s no-profit pricing and the price of the generic alternatives (Figure 4). Gilead Brand and Generic Access to Medicines Model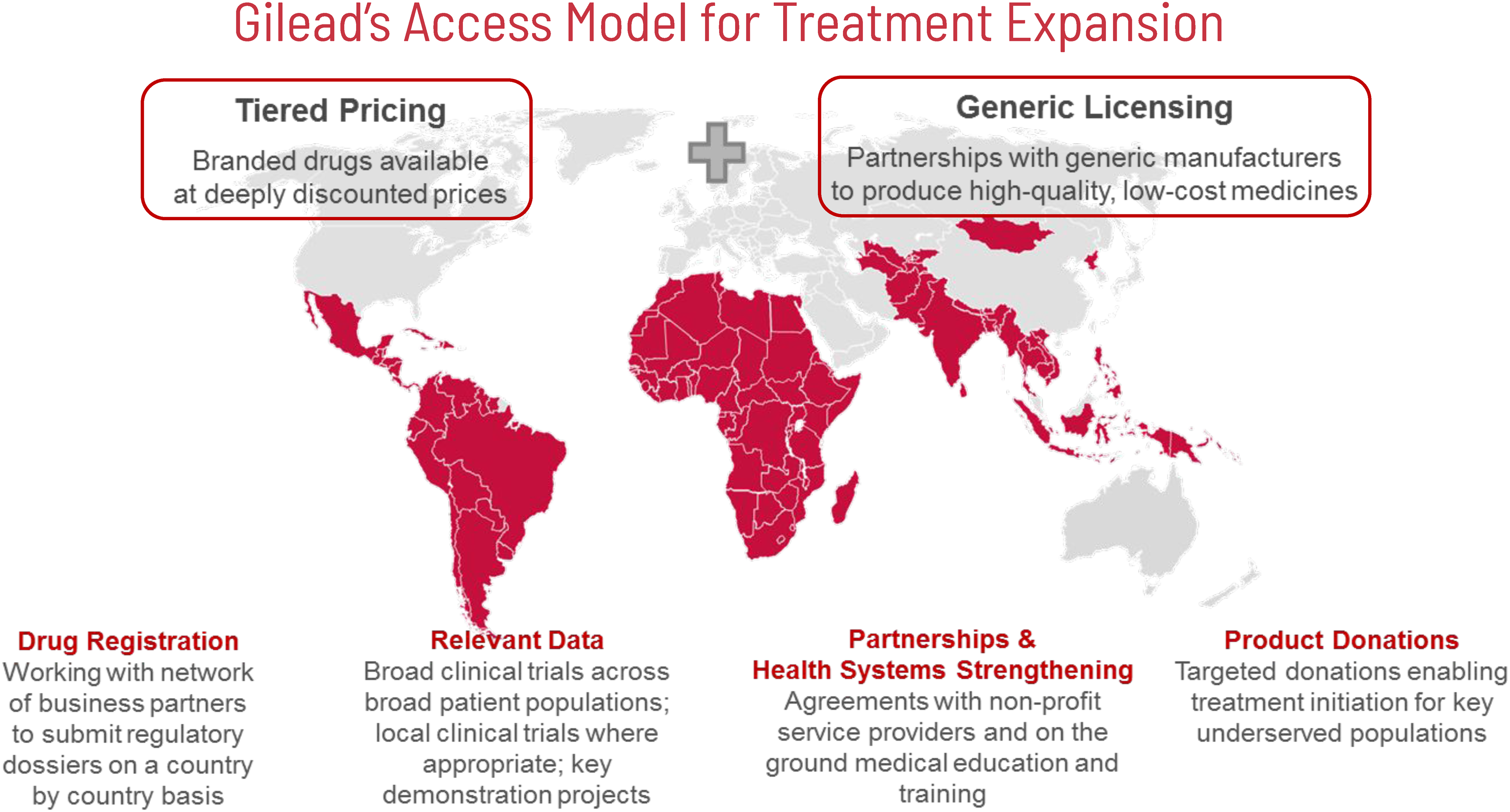
In 2006 and still today, most medicines for HIV and other diseases are supplied by Indian generic companies. Many NGOs and patient advocates encouraged John to allow Indian manufacturers to produce generic versions of Viread for distribution to countries hardest hit by the HIV/AIDS pandemic. John listened and challenged the teams to understand the opportunities and risks of having TDF licensed to and produced by manufacturers in India for the access program. John hypothesized that we could stimulate low-margin, high-volume, and high-quality production for distribution, while achieving the price reductions desired. He understood that we could vastly improve access if we structured the licenses to preserve the highly competitive generic marketplace (Figure 5). Voluntary licensing announcement, price reduction and patients on treatment 2007–2015
With John’s vision, the blueprint for the generic licensing model was drawn up. Gilead would license TDF to Indian generic companies; the license would include the right to manufacture API and finished products in India and distribute that product to over 116 countries. Gilead would provide a technology transfer and require WHO prequalification or FDA tentative approval of generic TDF. The licensees were free to develop their own proprietary competitive manufacturing processes and Gilead would remain hands-off and not audit or otherwise control their operations. The licensees were also free to set their own prices and pay Gilead a 5% royalty on finished product sales with the exception of pediatric formulations where we waived our royalties to accelerate the production of pediatric treatment options. Generic manufacturers could sell API to each other royalty-free, encouraging high-volume, low-cost API production among the most competitive producers. And perhaps most importantly, the license was available to any Indian company [7] (Figure 6). Voluntary licensing geographical scope, terms and conditions
The generic licensing combined with the internal operational model proved to be highly successful. From 2007 to 2018, the price of generic TDF per bottle went from $12.42 USD to $1.80 per month. TDF went from being registered in 15 low- and middle-income countries to more than 90 countries, and Gilead obtained listing on the Essential Medicines List and began comprehensive medical education programs in these markets. As a result, and due to the simultaneous increase in donor funding through PEPFAR [8] and The Global Fund [9], the number of patients on a TDF-containing regimen went from 30,000 to 12.5 million (Figure 7). Price reduction driven by generic entrants; number of patients on treatment 2006–2018
Conclusion
Since the original version of the Access Program, more indications such as HBV, pre-exposure prophylaxis (PrEP), and new products such as Gilead’s entire HIV, HBV, HCV portfolios and most recently, remdesivir were added. Under John’s direction, Gilead was the first company to enter the Medicines Patent Pool [10], with similar terms to its own licensing program. Gilead participated in and supported HCV elimination programs in Egypt, Georgia, Mongolia, and Brazil. The Access team supported the establishment of the West Bengal Indian Institute of Liver and Digestive Science (IILDS) and a reference lab in Senegal. With John, as he did with science, once we had solved one access challenge, he pushed us further (Figure 8). Health systems strengthening initiatives 2003–2018
John will be remembered for his genius and his foresight, and his incomparable vision for transforming science into solutions that changed people’s lives. Those of us who knew John well believe that one of John’s proudest achievements and motivations was the Access Program. Many people ask us how we were able to get support and approval for the Access Program as a for-profit company. To John, these issues—being a for-profit company and working to enable access to medicines around the world—were two sides of the same coin and in no way at odds with each other. John insisted on the Access Program and personally devoted his time to designing it and making it a success. It is a model that John wanted to endure and to evolve, to be applied not only to infectious disease but to noncommunicable diseases (NCDs) and other health innovations. For these reasons John Martin will be remembered for his seminal contributions in advancing global health and access to essential medicines.
Footnotes
Acknowledgments
We thank our former colleague Amy Flood for providing insight and expertise which greatly assisted with the writing of this article.
Author contributions
The authors collaborated on the writing of this case based on their decades-long oversight and leadership of the Gilead Access to Medicines Program.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Gregg Alton, Clifford Samuel, and Anand Reddi each worked at Gilead Sciences, Inc. Gregg as Chief Patient Officer, Clifford as Senior Vice President, Global Patient Solutions, Anand as Chief of Staff, International Operations and Corporate Affairs
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
