Abstract
Background
There is a paucity of data about the occurrence and risk factors of herpes simplex virus (HSV) reactivation among patients with severe COVID-19 presenting with acute respiratory distress syndrome (ARDS).
Methods
We performed a nested case-control study among a cohort of SARS-CoV-2 infected patients with ARDS. Between March and April 2020, all consecutive mechanically ventilated patients ≥18 years old with a positive PCR for SARS-CoV-2 on mucocutaneous samples were included in the study. We collected data on demographics, medical history, laboratory variables, administration of antivirals and other agents, respiratory and organ support procedures, microbiological results, and management of ARDS with prone positioning and the use of steroids. Univariate and multivariable Cox regression models were performed in order to identify predictors of HSV reactivation.
Results
Eighty-three patients with laboratory-confirmed SARS-CoV-2 infection were admitted to the ICU for mechanical ventilation. 18/83 (21.7%) patients developed mucocutaneous herpes simplex virus reactivation after a median of 17 days (IQR, 14–20). Prone positioning was the only independent risk factor for HSV reactivation (adj. hazard ratios, 1.60; 95% CI, 1.11–2.30; P = 0.009). All patients with mucocutaneous HSV reactivation were treated with antivirals. The outcome in terms of ventilator-associated pneumonia, catheter-related bloodstream infections, and in-hospital mortality was similar for patients with and without HSV reactivation.
Conclusions
HSV reactivation is frequent in COVID-19 patients with ARDS, especially if prolonged invasive mechanical ventilation with prone positioning is needed. Prompt testing for HSV and initiation of antiviral therapy should be performed in case of mucocutaneous lesions in this population.
Introduction
Herpes simplex virus (HSV) is a common virus that may affect critically ill patients undergoing mechanical ventilation [1,2]. Depending on age and socioeconomic status, 40–90% of the human population in different countries has antibodies to HSV [3]. Despite the presence of these antibodies, the virus reactivates following local stimuli (e.g. tissue lesion) or systemic stimuli (e.g. fever or impairment of the immune system). Reactivation of HSV has been associated with asymptomatic virus excretion in saliva, ulceration of the oral mucosa or herpes labialis, or more severe disease such as herpetic oesophagitis, tracheobronchitis or pneumonia in immunocompromised hosts [4,5]. Several studies have shown that HSV reactivation and active replication in the respiratory tract are common in mechanically ventilated critically ill patients even without underlying immunosuppression, with reported rates of 20 to 40% [1,2,5-7]. Whether HSV replication in the respiratory tract plays a significant role in the outcome of critically ill patients remains unclear [1,8]. It is also uncertain whether treatment with an antiviral agent is beneficial for these patients [9-11]. Little is known about HSV reactivation occurring in patients with COVID-19 and acute respiratory distress syndrome (ARDS).
The aim of this study was to investigate the occurrence and risk factors of HSV reactivation in a cohort of SARS-CoV-2 infected patients with ARDS receiving mechanical ventilation at the Regional Hospital of Locarno, Switzerland, during the first Swiss pandemic wave.
Methods
We performed a single-centre, nested case-control study to investigate the frequency and risk factors of HSV reactivation among patients with severe COVID-19 and ARDS who were admitted to the ICU at the Regional Hospital of Locarno, Switzerland, during the first wave of the Swiss COVID-19 pandemic between March and April 2020.
Local health and government officials in southern Switzerland responded to the COVID-19 pandemic by dedicating one public hospital (Regional Hospital Locarno, integrated in a multisite public hospital named Ente Ospedaliero Cantonale) exclusively to the care of patients with laboratory-confirmed SARS-CoV-2 infection. The pre-pandemic local ICU with eight beds was expanded to a unit with 45 beds in order to respond to the rapidly increasing number of COVID-19 patients requiring invasive respiratory support. We applied standard of care in accordance with the most recent guidelines to the entire cohort [12]. Specific treatments of COVID-19, including lopinavir/ritonavir, hydroxychloroquine, tocilizumab and remdesivir, were administered according to current national and international guidelines [13,14]. All consecutive mechanically ventilated patients ≥18 years old with a positive PCR for SARS-CoV-2 on mucocutaneous samples were included in this study. We collected data on demographics, medical history, laboratory variables, administration of antivirals and other agents, respiratory and organ support procedures, microbiological results, and management of ARDS with prone positioning and the use of steroids.
The primary endpoint was the frequency of mucocutaneous HSV reactivation after orotracheal intubation, defined as detection of HSV by qualitative specific PCR from a skin or mucosal lesion. We did not obtain HSV cultures. Secondary endpoints included the time to HSV reactivation from orotracheal intubation; risk factors of HSV reactivation according to demographic and clinical characteristics, disease severity (SOFA score at ICU admission), the use of steroids, prone positioning; and the impact of HSV reactivation on the incidence of ventilator-associated pneumonia (VAP), catheter-related bloodstream infections and in-hospital mortality.
Data were compared using the chi-square test or Fisher’s exact test for categorical variables, and the Mann-Whitney test for continuous variables. We used Kaplan-Meier curves to describe the cumulative incidence of HSV reactivation according to all variables collected, and the curves were compared using the log-rank test. Uni- and multivariable Cox regression analysis was used to investigate risk factors of HSV reactivation. All patients were censored at discharge from ICU if no HSV reactivation or death had occurred. All tests have been conducted two-sided and P-values <0.05 were considered statistically significant. All analyses were performed using commercially available software (STATA version 13.1 for Windows, StataCorp, College Station, TX, USA).
We followed the principles of the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for reporting our research [15]. The regional Ethics Committee of Canton Ticino, Bellinzona, Switzerland, approved the study protocol (Project ID: 2020-01429 CE 3665). Due to the observational nature of the study, the Ethics Committee waived the need for informed consent.
Results
General characteristics of the study population (n = 83) according to herpes simplex virus (HSV) reactivation.
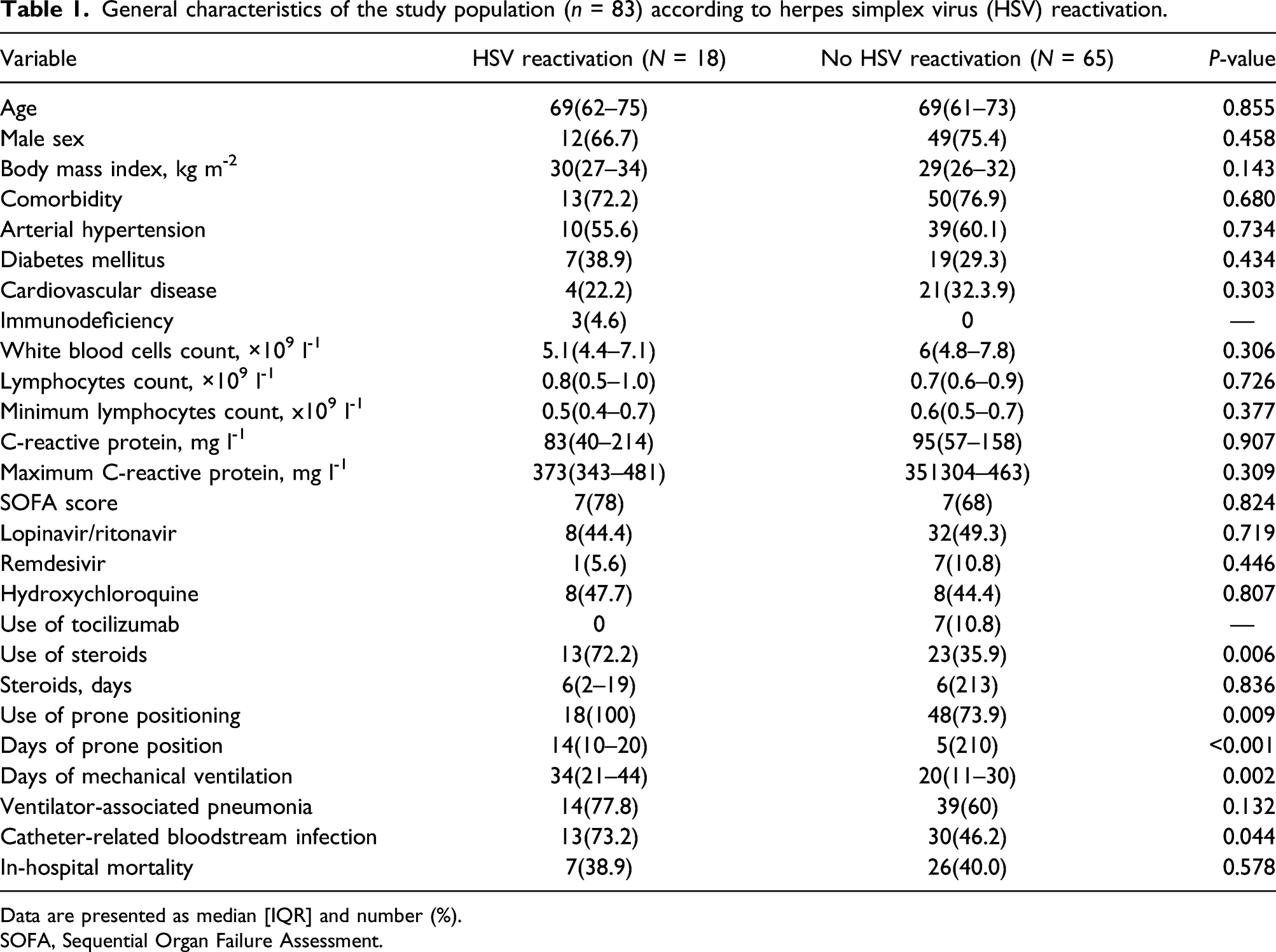
Data are presented as median [IQR] and number (%).
SOFA, Sequential Organ Failure Assessment.
Hazard ratios (HR) of herpes simplex virus reactivation, univariable and multivariable analysis.
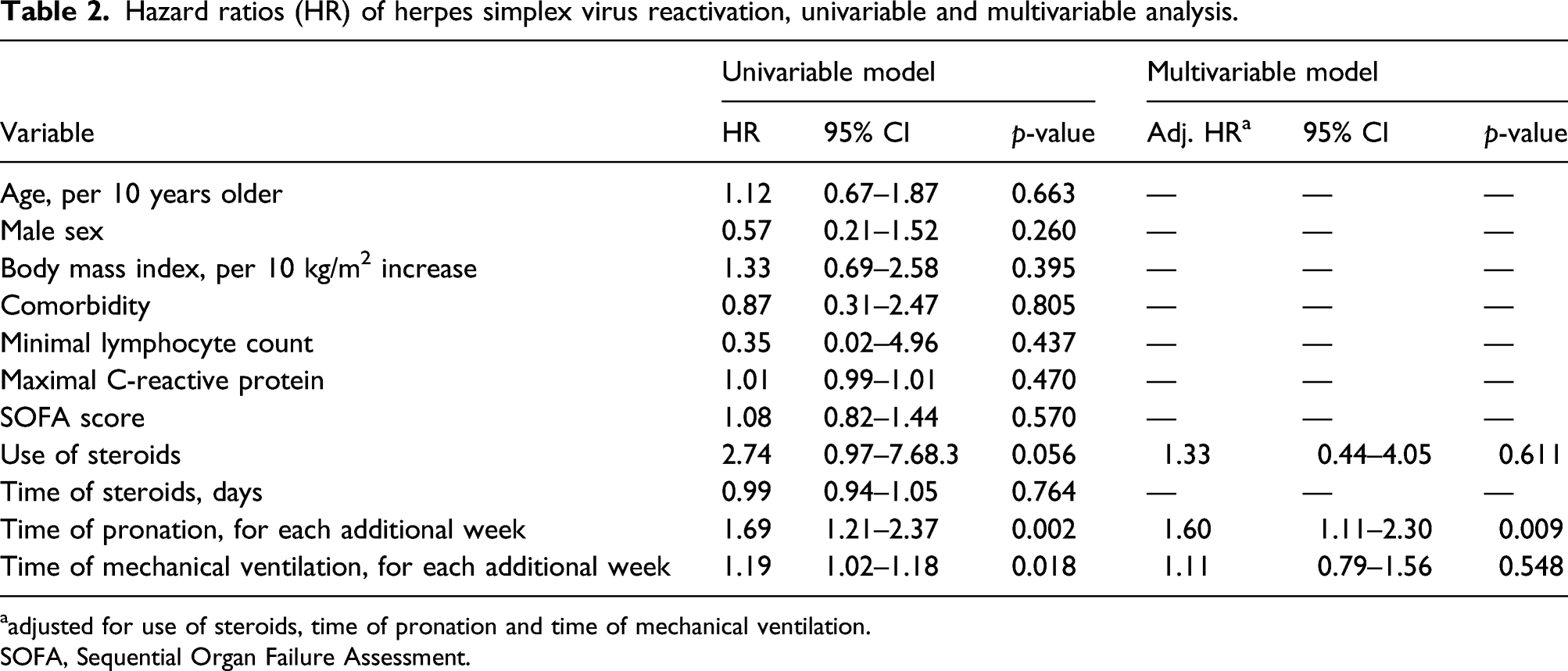
aadjusted for use of steroids, time of pronation and time of mechanical ventilation.
SOFA, Sequential Organ Failure Assessment.
Discussion
This prospective cohort study, involving 83 patients with severe COVID-19 and ARDS requiring mechanical ventilation during the first Swiss pandemic wave, illustrates an overall high rate of mucocutaneous HSV reactivation in critically ill COVID-19 patients. Moreover, we identified prone positioning, particularly the application of an elevated number of pronation cycles, as the only independent risk factor of HSV reactivation in our study population.
High prevalence of respiratory Herpesviridae reactivation has recently been described in 38 mechanically ventilated COVID-19 patients who were screened twice a week by specific PCR for HSV and CMV on tracheal aspirates without cytologic or histologic examination [16]. The clinical relevance of Herpesviridae detection in the lower respiratory tract remains controversial despite the association between antiviral treatment and lower ICU mortality has been described [1,2,4-6,8,9]. We focused on the occurrence of herpetic orofacial lesions that have been linked to a higher risk of bloodstream infections in the immunocompromised host. In our study, mucocutaneous HSV reactivation was more frequent in patients receiving steroids and in those with more prolonged mechanical ventilation. However, only prone positioning was an independent risk factor of HSV reactivation. We hypothesize that the repetitive trauma to the tissues occurring during prone position might have played a central role in the reactivation of the virus. In fact, the endotracheal tube may cause microtraumas to the orofacial region when a patient is turned in the bed and lies face down onto his anterior chest and abdomen.
Treatment with steroids and prolonged mechanical ventilation have been previously recognized as risk factors of HSV reactivation [1,4,6,7,9,10,16]. During the first Swiss COVID-19 pandemic wave national and international guidelines discouraged the administration of steroids, being an option at low dosage (i.e. hydrocortisone at a dose of 200 mg per day for 3–7 days) in case of septic shock not responsive to fluids and vasopressor therapy. This restrictive strategy might be the reason why steroids were not independently associated with HSV reactivation in our study.
Whether HSV reactivation in the setting of ARDS due to COVID-19 has an impact on the prognosis of these patients remains unclear and deserves to be further investigated. In contrast to previous studies [1,4,6-8,16], we did not observe any difference in the occurrence of ventilator-associated pneumonia, bloodstream infections or in-hospital death. This might be explained by the limited sample size of the study. On the other hand, we cannot exclude that antiherpetic treatment might have positively influenced the clinical course of the group with HSV reactivation in our population.
We acknowledge some limitations to our study. First, this is a single-centre study limiting the generalizability of our results. Second, we obtained an oropharyngeal swab or a swab from oral or skin lesions only in case of clinical suspicion for HSV reactivation, possibly underestimating its occurrence. Third, the study did not evaluate asymptomatic viral shedding, which could have influenced predictive factors. However, we concentrated on clinically relevant HSV reactivations rather than unspecific viral shedding of the lower respiratory tract. Fourth, due to the design of the study and the small sample size, we cannot draw any definitive conclusion about the utility of the antiherpetic treatment in case of prolonged prone positioning. The main strengths of this study are the prospective assessment of HSV reactivation and the choice of a clinically relevant endpoint. To our knowledge, this is the first study that investigates the problem of mucocutaneous HSV reactivation in patients with severe COVID-19.
In conclusion, mucocutaneous HSV reactivation is frequent in COVID-19 patients with ARDS, especially if prolonged invasive mechanical ventilation with prone positioning is needed. Our findings should be confirmed in a larger study.
Prompt testing for HSV and initiation of antiviral therapy should be performed at the occurrence of mucocutaneous lesions in this population.
Footnotes
Acknowledgements
We thank all the patients and the professionals working at our ICU for their collaboration during this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
