Abstract
Background
Hepatitis B virus (HBV) co-infection in human immunodeficiency virus (HIV)-positive individuals increases the risk of overall mortality, especially when HBV DNA levels are high. The role of CD4+ cell counts in this association is poorly defined. We aimed to determine whether HIV–HBV co-infection influences changes in CD4+ cell count before and during antiretroviral therapy and whether it affects mortality risk at levels of CD4+.
Methods
2052 HIV-positive participants from Côte d’Ivoire in a randomized-control trial assessing early or deferred ART were included. HBV-status was determined by hepatitis B surface antigen (HBsAg). Changes in CD4+ cell levels were estimated using a mixed-effect linear model. The incidence rates of all-cause mortality were estimated at CD4+ counts ≤350, 351–500, >500/mm3 and were compared between HBV-status groups as incidence rate ratios (IRR).
Results
At baseline, 190 (9%) were HBsAg-positive [135 (71%) with HBV DNA <2000 IU/mL, 55 (29%) ≥2000 IU/mL]. Follow-up was a median 58 months (IQR = 40–69). Between co-infection groups, there were no differences in CD4+ decline before ART initiation and no differences in CD4+ increase after ART initiation. After adjusting for sex, age, baseline HIV RNA level, and early/deferred ART arm, mortality rates were not significantly different between HBsAg-positive versus HBsAg-negative participants across strata of CD4+ levels. However, HBsAg-positive individuals with HBV-DNA ≥2000 IU/mL versus HBsAg-negative individuals had increased mortality rates at ≤350/mm3 (adjusted-IRR = 3.82, 95% CI = 1.11–9.70) and 351–500/mm3 (adjusted-IRR = 4.37, 95% CI = 0.98–13.02), but not >500/mm3 (adjusted-IRR = 1.07, 95% CI = 0.01–4.91).
Conclusion
Despite no effect of HBV-infection on CD4+ levels, HIV-HBV co-infected individuals with high HBV replication are at higher risk of mortality when CD4+ is <500/mm3.
Introduction
Co-infection with chronic hepatitis B virus (HBV) is found in approximately 8% of individuals living with human immunodeficiency virus (HIV) infection. 1 HIV–HBV co-infection is known to have broad consequences on the immunity of HIV-positive individuals. 2 HIV-positive individuals with both resolved and chronic HBV-infection have been shown to have an increased risk in progression to AIDS-defining illnesses in the absence of effective treatment. 3 With the initiation of potent antiretroviral therapy (ART), most studies have demonstrated that although HIV–HBV co-infection is no longer likely to affect progression to AIDS, 4 co-infection can be associated with slower immunoreconstitution, leading to lower CD4+ cell counts during long-term ART.5–7
Poor levels of immunity in HIV-positive individuals have severe clinical consequences. The relationship between lower CD4+ cell counts and overall death during the natural history of HIV infection has been clearly established. 8 During treatment with ART, faster increases in CD4+ cell counts immediately after ART initiation have been associated with long-term immunoreconstitution, 9 which lends to a lower risk of opportunistic infections and reduced short-term mortality.10–12 Given the association between HBV-infection and immunosuppression, HIV–HBV co-infection could increase the risk of overall mortality.
Indeed, several studies have shown an increased risk of overall mortality in individuals with HIV–HBV co-infection compared to HIV mono-infection despite treatment with effective anti-HBV and anti-HIV agents, although this association is inconsistent. 2 In a recent secondary analysis of a randomized clinical trial evaluating early and deferred ART in Côte d’Ivoire, HIV–HBV co-infected patients with high HBV DNA levels were at higher risk of death and lost to follow-up compared to HIV-positive individuals without HBV co-infection, regardless of treatment arm. 13 It was unclear, however, how immunosuppression was related to this finding. One hypothesis could be that HIV–HBV co-infected individuals with high HBV DNA levels may have had faster decreases in CD4+ cell count while not receiving ART and/or may have had slower increases in CD4+ cell count after ART initiation. Increased immunosuppression in these individuals could then have led to a higher risk in mortality. Another hypothesis could be that the association between HIV–HBV co-infection with high HBV replication and overall mortality may have been accentuated when CD4+ cell counts were lower.
We took advantage of prospectively collected data from the Temprano study 14 to determine whether there were any differences in CD4+ decline while not receiving ART and CD4+ immunoreconstitution after ART initiation between HIV–HBV co-infected and HIV mono-infected patients. We also aimed to evaluate the relative risk of mortality in HIV–HBV co-infected and HIV mono-infected patients according to particular levels of CD4+ cell count.
Methods
Study design and procedures
We included participants enrolled in the Temprano ANRS 12,136 trial. In brief, the Temprano trial was a randomized controlled, 2 × 2 factorial, superiority trial, conducted in nine clinics in Côte d’Ivoire. From March 2008 to July 2012, we included individuals based on the following criteria: HIV infection, aged ≥18 years, CD4+ count <800/mm3, absence of active TB or severe liver diseases, and no criteria for starting ART according to concurrent World Health Organization (WHO) guidelines. We excluded individuals based on the following criteria with respect to liver disease: plasma aspartate aminotransferase (AST), alanine aminotransferase (ALT) levels >4 times the upper limit of normal (ULN) or any other severe liver disease as assessed by the physician. Full description of the trial design is reported elsewhere. 14
We randomly assigned participants to one of the following arms: two “deferred ART” arms (arms 1 and 2), in which ART initiation was deferred until patients met criteria for initiation ART according to concurrent WHO guidelines; and two “immediate-ART” arms (arms 3 and 4), in which ART was immediately initiated at inclusion. Participants assigned to arms 2 and 4 additionally received 6-months isoniazid prophylaxis for tuberculosis. Participants assigned to all arms received ART containing tenofovir (TDF)/emtricitabine as the first-line regimen with the third agent being efavirenz, zidovudine, or lopinavir/ritonavir.
We followed participants for 30 months in the Temprano trial. We completed 30-month follow-up of the first Temprano study participant in September 2010. After this date, we asked all patients who reached the 30-month visit to continue follow-up in a post-trial phase (PTP). We continued this phase until the last patient completed their 30-month trial visit (closing date: 5 January 2015). Procedures of the Temprano study closely followed those of the PTP. Patients assigned to the deferred ART arms who had not started ART during the trial commenced ART during the PTP according to concurrent WHO criteria. The data analyzed in this study were collected between March 2008 and January 2015.
Included patients
For the present study, we considered all patients enrolled in the Temprano trial and PTP who had an available plasma hepatitis B surface antigen (HBsAg) and for those participants who were HBsAg-positive, an available plasma HBV DNA level.
The markers of HBV replication obtained in Temprano have been summarized elsewhere. 13 In brief, HBsAg serology was performed at baseline using an ELISA-based test (Monolisa® Bio-Rad, Marnes la Coquette, France). At inclusion, HBV DNA viral loads were quantified using an in-house polymerase chain reaction (PCR)-based assay (QuantiFast SYBR® Green PCR kit, Qiagen, Courtaboeuf, France; Light Cycler 480; Roche, Boulogne-Billancourt, France; detection limit: 12 copies/mL) 15 or a commercially available PCR assay (COBAS®Amplicor HBV Monitor, Roche Diagnostics, Meylan, France; detection limit = 60 IU/mL). Viral loads were reported in IU/mL in order to ensure comparability between assays [conversion factor: 1 IU/mL = 2.8 copies/mL]. 16
Statistical analysis
Patients were regrouped as HIV mono-infected (all HBsAg-negative participants) or HIV/HBV co-infected (all HBsAg-positive participants), the latter of which was further stratified on plasma HBV-DNA <2000 IU/mL and plasma HBV-DNA ≥2000 IU/mL at inclusion. The threshold of 2000 IU/mL was chosen based on levels that, along with ALT levels, differentiate chronic “infection” and chronic “hepatitis” among HBeAg-negative HBV mono-infected individuals, according to guidelines from the European Association for the Study of the Liver. 17
We first determined whether there were any differences between HBV-infection groups with respect to CD4+ cell count changes over time, that is, while not receiving ART (i.e., “off-ART”) and after ART initiation (i.e., “on-ART”). Using mixed-effect non-linear regression, we parameterized a model whereby the decrease in CD4+ cell count off-ART was a linear function over time and the increase in CD4+ cell count on-ART was a square-root function over time. Follow-up in this analysis ended at death, LTFU, or 5 January 2015, whichever occurred first. Estimates specific to each HBV-infection group were obtained from an interaction term between HBV-infection group and follow-up times for each period. A Wald χ2 test of this interaction term was used to determine whether the slopes of CD4+ cell counts were different between HBV-infection groups. A random-intercept and random-coefficient for time were used to account for variation between individuals with respect to CD4+ T cell count at the beginning of each time period and during follow-up, with an identity covariance structure between random-intercept and random-coefficient. Models were both unadjusted and adjusted a priori for CD4+ cell count at the beginning of each time period, HIV RNA level at inclusion (>5.0 versus ≤5.0 log10 copies/mL), age (>35 versus ≤35 years), and sex.
We then determined whether there were any differences between HBV-infection groups with respect to the incidence rate (IR) of all-cause mortality while at specific strata of CD4+ cell count. The time at risk in this analysis began at randomization and ended at death, LTFU, or 5 January 2015, whichever occurred first. We defined three levels of CD4+ cell count at ≤350/mm3, 351–500/mm3, and >500/mm3. We determined the time spent in a given CD4+ cell count stratum, assuming CD4+ to be constant between two measures of CD4+. We estimated the time-updated CD4+ cell count-specific IR of all-cause mortality per 100 person-years by dividing the number of events that occurred in each CD4 cell count stratum by the time spent in the corresponding stratum (for non-deceased patients) or the time between entry in the stratum and event (for deceased patients). 95% confidence intervals (CI) of IRs were calculated assuming a Poisson distribution if the number of events was lower than 50 and normal approximation otherwise. IR were compared between HBV-infection groups within CD4+ cell count strata by an Exact Poisson regression model using median unbiased estimates (with the mid-P-value rule) and log follow-up time as an offset. Incidence rate ratios (IRR) were estimated from this model and were given as both unadjusted and adjusted a priori for HIV RNA level at inclusion (>5.0 versus ≤5.0 log10 copies/mL), age (>35 versus ≤35 years), sex, and early versus deferred ART.
All reported p-values were two-sided and no adjustments for multiple comparisons were applied. Statistical analyses were performed using SAS (v9.4, Cary, North Carolina), and STATA (v15.0, College Station, Texas).
Funder, registration, and ethics
The Temprano trial protocol was approved by the Côte d'Ivoire National Ethics Committee for Health Research. It was registered at ClinicalTrials.gov (NCT00495651). Signed inform consent was provided prior to participating in the trial. The sponsor had no role in the conduct of the study and interpretation of the data.
Results
Description of the study population
Baseline and follow-up characteristics.

This included 516 patients randomized to immediate-ART and IPT, 514 to immediate-ART and no IPT, 511 to deferred ART, and IPT and 511 to deferred ART and no IPT. N: number in population; n: number in groups; IQR: interquartile range; BMI: body mass index; IPT: isoniazid preventive therapy; WHO: World Health Organization; ART: antiretroviral therapy; TDF: tenofovir; FTC: emtricitabine; ZDV: zidovudine; LPV/r: lopinavir/ritonavir; ALT: alanine transaminase; ULN: upper limit of normal. BMI: body mass index; Ag: antigen; Ab: antibodies. HBsAg: hepatitis B surface antigen.
ap-value for comparison between three groups (or two groups when first column is empty), using Pearson χ2 or Fisher Exact test for categorical variables and Kruskal–Wallis test for continuous variables.
bAmong 125 patients (missing data, n = 65).
c222 patients never started ART because their CD4 cell count remained >500/mm3 during the entire follow-up.
dMain ART regimen was TDF–FTC plus EFV. Among patients who started ART with TDF–FTC plus LPV/r, the reason for not receiving an EFV-based regimen was dual HIV-1/2 infection, previous nevirapine-use for preventing mother-to-child transmission, declining to use effective contraception, and other.
eOther regimens were ZDV–lamivudine (3 TC)–LPV/r (n = 29), ZDV–3TC–nevirapine (n = 15), ZDV–3TC–EFV (n = 8), 3TC–abacavir–LPV/r (n = 2), DDI 3 TC EFV (n = 2), stavudine (D4T)–3TC–EFV (n = 1), D4T–3TC–LPV/r (n = 1), TDF-FTC-3TC-D4T (n = 1), TDF-FTC-NVP (n = 1).
Patients were followed for a median 58 months (IQR = 40–69). Median follow-up was no different between HBsAg-negative (58 months, IQR = 41–69), and HBsAg-positive patients (60 months, IQR = 38–69) (p = 0.77), even with baseline HBV DNA viral load <2000 IU/mL (60 months, IQR = 39–69, p = 0.89) or ≥2000 IU/mL (60 months, IQR = 37–70, p = 0.87).
No difference in CD4+ cell count changes during follow-up between HIV–HBV co-infection and HIV mono-infection
Off-ART
Of the 1022 individuals randomized to receive deferred ART, 971 had at least two measures of CD4+ cell counts while not receiving ART. Off-ART median follow-up was 18 months (IQR = 11–31) in this group. In HBsAg-negative patients, mean CD4+ count was 477/mm3 (95% CI = 467–486) at baseline (n = 890), decreasing to 426/mm3 (95% CI = 414–437) after 12 months without ART (n = 729) and 372/mm3 (95% CI = 359–386) at the end of off-ART follow-up (n = 602). In HBsAg-positive patients, mean CD4+ count was 452/mm3 (95% CI = 421–482) at baseline (n = 81) and similarly decreased to 395/mm3 (95%CI = 364–425) after 12 months without ART (n = 65) and 358/mm3 (95% CI = 312–403) at the end of off-ART follow-up (n = 50). Based on the mixed-effect non-linear regression model, there were no significant differences in CD4+ cell decrease while not receiving ART between HBsAg-negative and HBsAg-positive patients, both unadjusted (p for interaction = 0.63) and adjusted for baseline CD4+ level, HIV RNA level, age, and sex (p for interaction = 0.73) (Figure 1(A)). Modeled CD4+ T cell count decrease while not receiving antiretroviral therapy (ART). Mean CD4+ T cell counts estimated as linear decrease off-ART over time (lines) along with their 95% confidence intervals (shaded) using a mixed-effect regression model. Estimates are adjusted for baseline CD4+ level, HIV RNA level, age, and sex. Curves are stratified by hepatitis B surface antigen (HBsAg)-status (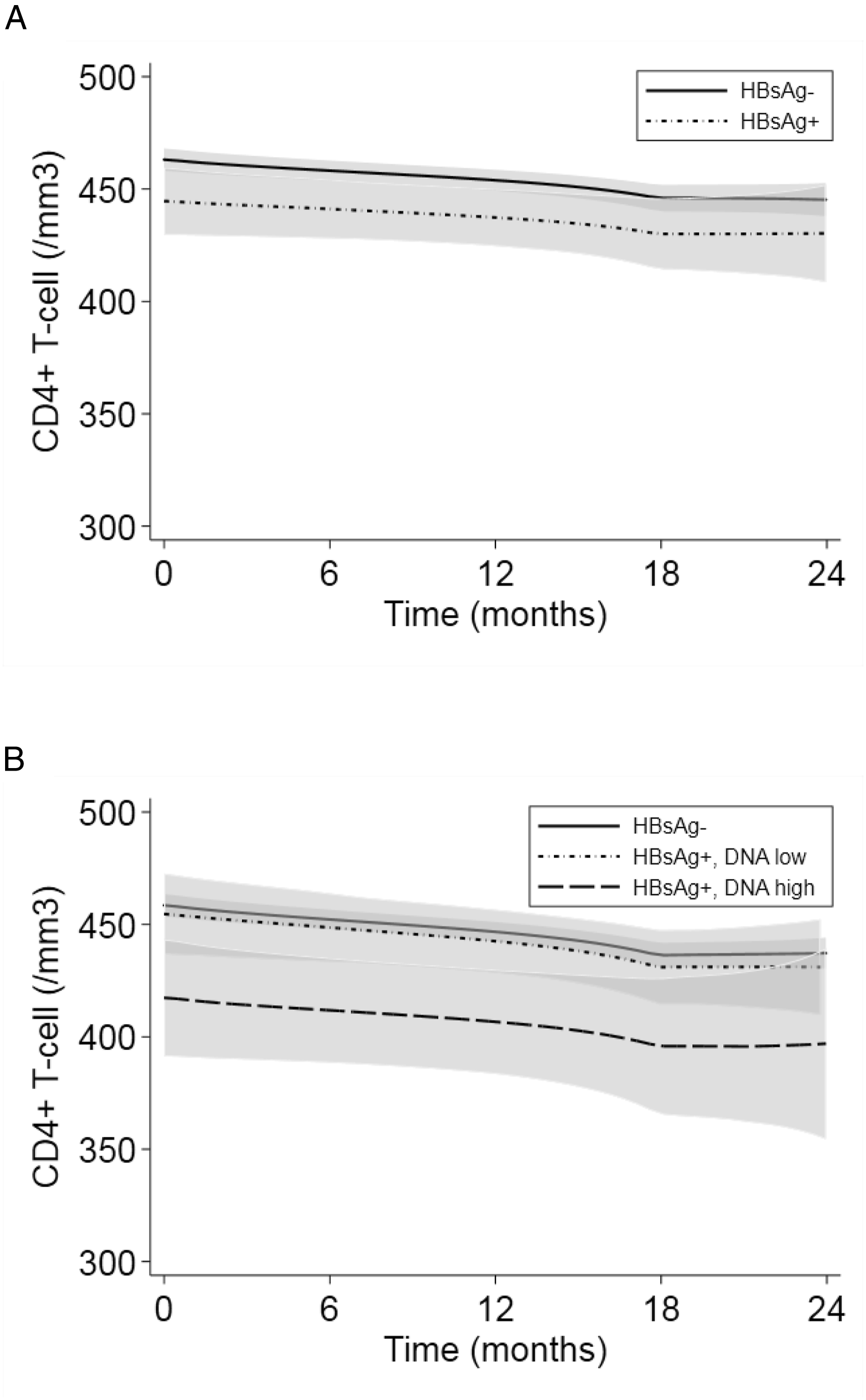
When separating HBsAg-positive patients based on HBV DNA viral load <2000 IU/mL and ≥2000 IU/mL, respectively, mean CD4+ count was 460/mm3 (95% CI = 424–496) and 435/mm3 (95% CI = 376–493) at baseline (n = 54 and n = 27), decreasing to 400/mm3 (95% CI = 361–439) and 380/mm3 (95% CI = 330–431) at 12 months without ART (n = 47 and n = 18), and 351/mm3 (95% CI = 310–391) and 378/mm3 (95% CI = 231–525) at the end of off-ART follow-up (n = 37 and n = 13). There were no significant differences in CD4+ cell decrease between HBsAg-negative patients and HBsAg-positive patients with HBV DNA viral load <2000 IU/mL (unadjusted p for interaction = 0.24 and adjusted p for interaction = 0.38) or with HBV DNA viral load ≥2000 IU/mL (unadjusted p for interaction = 0.71 and adjusted p for interaction = 0.49) (Figure 1(B)).
On-ART
1729 patients initiated ART and had at least two measures of CD4+ cell counts during ART. On-ART median follow-up was 55 months (IQR = 37–61) in this group. In HBsAg-negative patients, mean CD4+ count was 405/mm3 (95% CI = 397–413) at ART initiation (n = 1564) increasing to 588/mm3 (95% CI = 578–599) after 12 months of ART (n = 1404) and 694/mm3 (95% CI = 679–709) at the end of on-ART follow-up (n = 1371). In HBsAg-positive patients, mean CD4+ count was 392/mm3 (95% CI = 369–414) at ART initiation (n = 165) and similarly increased to 577/mm3 (95% CI = 542–612) after 12 months of ART (n = 142) and 673/mm3 (95% CI = 630–715) at the end of on-ART follow-up (n = 139). Based on the mixed-effect non-linear regression model, there were no significant differences in CD4+ cell increase during treatment between HBsAg-negative and HBsAg-positive patients, both unadjusted (p for interaction = 0.33) and adjusted for baseline CD4+ level, HIV RNA level, age, sex, and early versus deferred ART (p for interaction = 0.30) (Figure 2(A)). Modeled CD4+ T cell count increase after initiating antiretroviral therapy (ART). Mean CD4+ T cell counts estimated as root-square increase on ART over time (lines) along with their 95% confidence intervals (shaded) using a mixed-effect regression model. Estimates are adjusted for baseline CD4+ level, HIV RNA level, age, and sex. Curves are stratified by hepatitis B surface antigen (HBsAg)-status (
When separating HBsAg-positive patients based on HBV DNA viral load <2000 IU/mL and ≥2000 IU/mL, respectively, mean CD4+ count was 401/mm3 (95% CI = 373–429) and 369/mm3 (95% CI = 329–408) at ART initiation (n = 117 and n = 48), increasing to 577/mm3 (95% CI = 536–619) and 576/mm3 (95% CI = 506–647) after 12 months of ART (n = 99 and n = 43), and 682/mm3 (95% CI = 633–731) and 650/mm3 (95% CI = 562–737) at the end of on-ART follow-up (n = 99 and n = 40). There were no significant differences in CD4+ cell increase between HBsAg-negative patients and HBsAg-positive patients with HBV DNA viral load <2000 IU/mL (unadjusted p for interaction = 0.17 and adjusted p for interaction = 0.20) or with HBV DNA viral load ≥2000 IU/mL (unadjusted p for interaction = 0.76 and adjusted p for interaction = 0.78) (Figure 2(B)).
Higher incidence of all-cause mortality in HIV-HBV co-infected patients with high HBV DNA levels is only observed at CD4+ below 500/mm3
Incidence of death according to hepatitis B virus infection status within strata of CD4+ cell count.
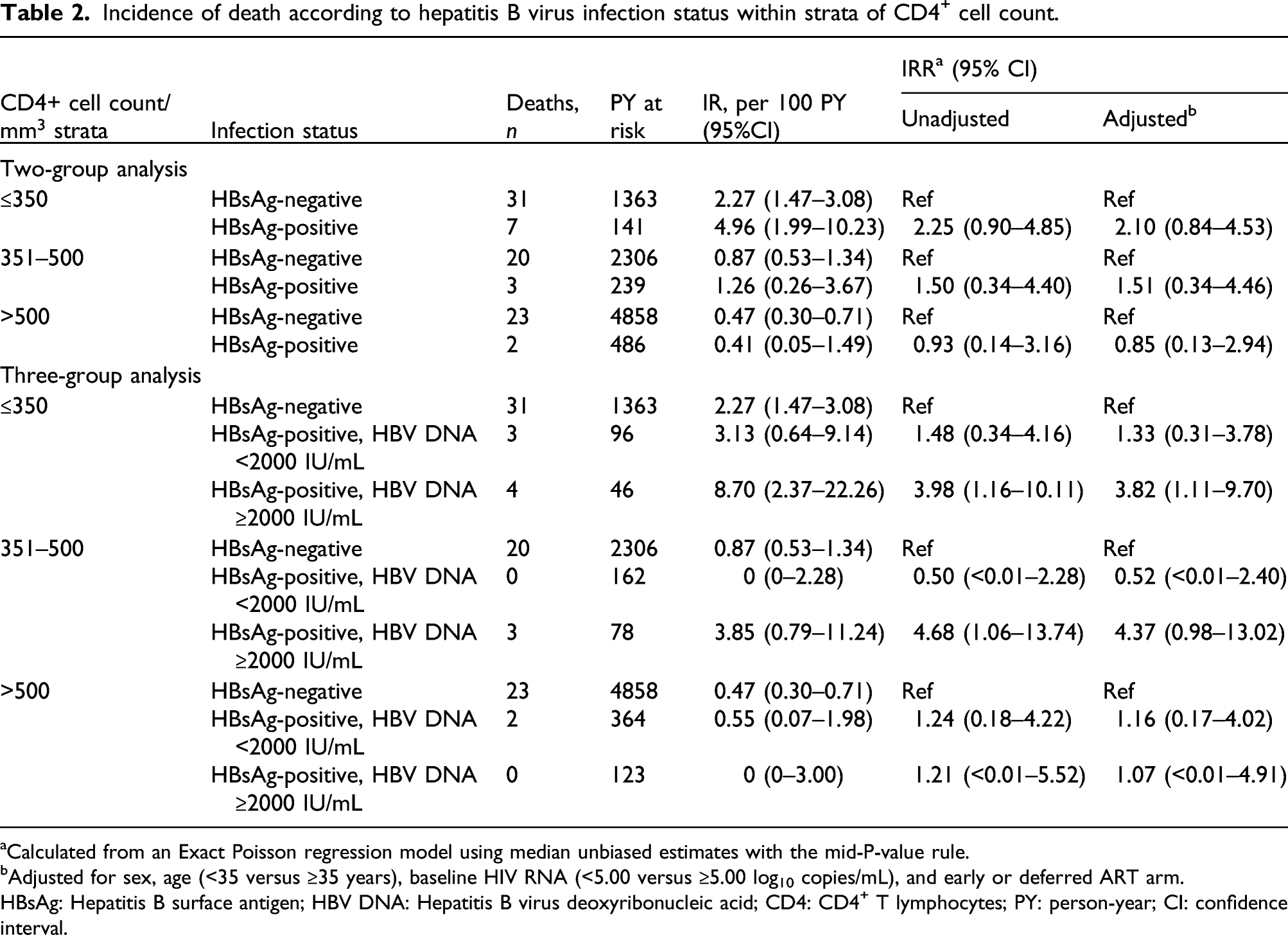
aCalculated from an Exact Poisson regression model using median unbiased estimates with the mid-P-value rule.
bAdjusted for sex, age (<35 versus ≥35 years), baseline HIV RNA (<5.00 versus ≥5.00 log10 copies/mL), and early or deferred ART arm.
HBsAg: Hepatitis B surface antigen; HBV DNA: Hepatitis B virus deoxyribonucleic acid; CD4: CD4+ T lymphocytes; PY: person-year; CI: confidence interval.
Discussion
Using data from a randomized clinical trial, we did not demonstrate any difference in decrease in CD4+ cell count while not receiving ART or in immunorecovery after ART initiation between HIV–HBV co-infection and HIV mono-infection. Even when HBV DNA levels were above 2000 IU/mL in co-infected patients, for whom risk of AIDS-related morbidity and mortality is particularly high,13,18 changes in CD4+ cell counts were no different compared to HIV mono-infected patients. These data provide reassuring information on at least the immunologic outcomes of HIV-HBV co-infected individuals with relatively mild immunosuppression.
HIV-HBV co-infected individuals with higher HBV DNA levels at baseline began follow-up with slightly lower CD4+ cell counts compared to HIV-monoinfected individuals. 13 Estimates from the mixed-effects model additionally showed that while not receiving ART, the declines in CD4+ count ran parallel between HBV-infection groups. Chronic HBV-infection does contribute to immunodysfunction, specifically with regards to depletion of HBV-specific cytotoxic lymphocytes and impaired proliferative capacity of B-cells, similar to most chronic viral infections.19,20 However, it is uncertain whether these HBV-induced immune deficiencies would have any influence on CD4+ T cell counts. Our results would suggest that HBV-infection does not affect CD4+ cell counts in untreated HIV–HBV co-infected patients, at least not when CD4+ levels are above 250/mm3.
During ART, CD4+ increases were rapid in the first 6 months and began to wane in the months thereafter, as expected, 8 while these increases were also parallel between co-infection groups. This finding was also observed in a previous analysis of the Trivacan study, 21 as well as other studies from Europe and South Africa.4,22,23 In contrast, poorer immunorecovery has been observed for HIV-HBV co-infected versus HIV mono-infected patients in studies from Switzerland and Nigeria.5,6 The discrepancy between studies could be partly explained by the fact that most have included individuals with severe immunosuppression and higher prevalence of liver disease, both of which are associated with reduced CD4+ cell counts during ART.8,24 In our study, all patients had a substantially higher CD4+ cell count and, per exclusion criterion, did not have liver impairment as established by transaminase levels. In addition, almost all patients in our study received ART containing tenofovir, which has potent anti-HBV activity. 25 Any purported effect of HBV on immunorecovery would have been effectively reduced.
In a previous analysis, we observed a higher mortality rate in HIV-HBV co-infected individuals with high HBV DNA. 13 In our multivariable analysis, we additionally found that HIV-HBV co-infected individuals with high HBV DNA viral are at significantly higher risk of all-cause mortality, compared to others, when their CD4+ counts are ≤350/mm3. This excess risk is still high as CD4+ levels reach between 351–500/mm3 and is no longer apparent as levels reach above 500/mm3. Given that HIV–HBV co-infected individuals do not have accelerated declines in CD4+ cell counts while not receiving ART and that they are able to exhibit immunorecovery comparably as quick as in those with HIV mono-infection, these data would not support the hypothesis that more severe immunosuppression over time in HIV-HBV co-infected individuals with high HBV DNA levels was responsible for a higher risk in mortality. Rather, these individuals simply have a more pronounced susceptibility to mortality when CD4+ levels are below 500/mm3. Our observations stress the need to immediately treat co-infected patients with high HBV replication and to closely monitor them until CD4+ levels return above 500/mm3.
The underlying explanation for these findings is somewhat unclear. Most deaths in the Temprano study were due to infectious diseases. 14 Co-infected patients in sub-Saharan Africa with elevated HBV replication at ART initiation are more susceptible to HIV-related morbidity, especially invasive bacterial diseases.13,18,26 These causes of morbidity are in contrast to other studies in European settings, where liver-related disease appears to bear more influence on mortality rates in HIV-HBV co-infected individuals.27–29 The liver is known to carry an important immunological function, orchestrating immune responses against pathogens entering via gastrointestinal tract.30,31 Perhaps co-infected individuals with high levels of HBV DNA had more liver damage and as a result, were unable to clear HIV-related infections as CD4+ cell counts were lower. Information on liver fibrosis would be needed to support this hypothesis and were unfortunately not collected in this study.
Interestingly, these data bring support for a previous modeling study calibrated to sub-Saharan Africa, in which a more deleterious impact on mortality would be observed in HIV–HBV co-infected patients if ART were delayed until CD4+ <500. 32 However, this model was based on assumptions that progression of liver fibrosis would lead to death in these individuals and that this progression would become faster as CD4+ counts decreased. Considering its cause for concern in the HIV-HBV co-infected population of sub-Saharan Africa, future models should consider the role of infections on mortality.
Other limitations of our study need to be addressed. First, data were derived from a clinical trial designed to assess the benefits of early treatment in HIV-positive individuals. There could be a potential selection bias for individuals randomized to deferred ART who initiated ART later (i.e., at lower CD4+ cell counts). Second, despite the large numbers of individuals recruited in the Temprano study, the HIV-HBV co-infected subgroups included small sample sizes. Third, individuals with grade 3–4 liver enzyme elevations and severe liver disease were excluded. These exclusion criteria made it difficult to recognize hepatic flares or decompensation following immune reconstitution in those with liver cirrhosis, which could contribute to mortality. Fourth, we did not have any data on HDV serostatus or HDV-RNA replication at baseline, while HDV infection is known to be fairly prevalent among HBsAg-positive individuals in sub-Saharan Africa and contribute to an excess in mortality. 33 Finally, as TDF-containing ART was given to the vast majority of participants, HBV DNA replication was likely controlled in >95% of co-infected individuals, as shown in a previous sub-analysis of the Temprano study, 14 and HBeAg- and HBsAg-seroclearance were rather rare. 34 As such, we were unable to assess whether changes in CD4+ count were associated with persistent HBV replication or seroconversion. In addition, some of the agents used in this study are no longer part of first-line ART regimens and immunorestoration could be different with other current first-line antiretroviral agents, such as integrase strand transfer inhibitors.
In conclusion, we observed that HIV–HBV co-infected and HIV mono-infected patients did not have differences in CD4+ decline while not receiving ART or in immunorestoration after initiating ART. Importantly, the risk of mortality associated with HIV–HBV co-infection at high HBV DNA levels was only evident when CD4+ counts were below 500/mm3. In order to improve the quality of care for HIV–HBV co-infected individuals in sub-Saharan Africa, a deeper understanding of the causes of morbidity/mortality is still needed in this population.
Footnotes
Acknowledgements
We thank all patients who participated in this trial; members of SMIT, CeDReS, CEPREF, USAC, CIRBA, CNTS, La Pierre Angulaire, Hopital General Abobo, Formation Sanitaire Anonkoua Koute, Center de sante El Rapha, the Program PACCI team, and INSERM U1219 IDLIC teams for their valuable contributions; Gilead Sciences, for the donation of Truvada; and Merck Sharp & Dohme, for the donation of Stocrin.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Agence Nationale de Rercheches sur le SIDA et les hépatites virales (ANRS), Paris, France. GMK also received doctoral funding from the ANRS.
