Abstract
This study aimed to identify psychosocial adaptation profiles among patients with myeloproliferative neoplasms (MPN), a rare hematologic cancer, to ultimately better support those struggling most. In a longitudinal study of 338 Dutch MPN patients, hierarchical and K-means cluster analyses were conducted based on core psychosocial adaptation variables coping, resilience, and illness identity (i.e. acceptance, enrichment, rejection, and engulfment). Chi-square and ANOVA tests assessed differences in sociodemographic variables, disease characteristics, self-management, and quality of life between clusters, with Delta scores examining stability 6 months later. Three distinct profiles emerged that differed significantly in the aforementioned variables: ‘low adaptation’ (high disease burden, lower education), ‘high adaptation’ (longest disease duration, strong self-management), and ‘active distance’ (high rejection, low illness-identity, oldest patients). Identifying these profiles derived from cluster analysis can help tailor care, especially for low adapting patients with the highest burden, underscoring the importance of targeted psychosocial interventions to improve their well-being.
Keywords
Introduction
Living with myeloproliferative neoplasm (MPN), a rare type of cancer that arises in the bone marrow and leads to abnormalities in blood cells (Barbui et al., 2013), can be quite challenging. This is partly due to the elevated risk of complications, such as thrombosis (Barbui et al., 2013), as well as increased inflammatory signaling leading to constitutional symptoms (Soyfer and Fleischman, 2023).
Another salient factor is that the symptom burden can be substantial: Previous research has shown that an MPN can have a negative impact on different areas of quality of life (QoL; Harrison et al., 2017; Mesa et al., 2016), as it causes fatigue (as one of the most common and severe symptoms; Harrison et al., 2017), cognitive problems, reduced productivity, social inactivity, and impaired work life. Problems in one area of life often have cascading effects on others, making interventions more complex.
MPN is incurable, and most patients have a stable disease course for years or even decades (Szuber et al., 2018). However, over time, MPN will ultimately progress to more severe stages or variants in most patients (Barbui et al., 2013). This creates persistent uncertainty regarding physical decline and life expectancy. It may also trigger chronic stress responses. The absence of hope for a complete cure and risk of progression complicates acceptance of a new patient identity. This tension between stability and irreversibility challenges the adaptation process. At the same time, it should be noted that patients exhibit heterogeneity in the extent to which they are affected by their MPN (Geyer et al., 2014; Tremblay and Mesa, 2022), sometimes regardless of subtype (Eppingbroek et al., 2024; Geyer et al., 2014).
It is of great importance for patients with a chronic illness such as MPN to gain and maintain the capacity to effectively manage their condition throughout their lifespan. In particular, because psychological adjustment to chronic disease, defined as ‘psychological processes in response to chronic disease and associated treatment’ (Dekker and de Groot, 2018) can have a major impact on health outcomes, wellbeing and QoL (Allart et al., 2013; Dekker and de Groot, 2018; Englbrecht et al., 2012; Möller et al., 2021).
The Common Sense Model (CSM; Leventhal et al., 2001) and the Adaptation Model of Chronic Illness (Whittemore et al., 2010) are both frameworks that describe psychosocial adaptation to illness, providing a theoretical foundation for understanding these processes in MPN. They consider the importance of multiple interrelated factors and the impact of psychosocial adaptation variables on QoL. The Common Sense Model of Disease Self-Regulation postulates how lay perceptions of health threats affect coping and health outcomes (e.g. perceiving MPN as manageable rather than catastrophic is associated with greater coping flexibility) whilst the Adaptation Model underscores that chronic illness management requires addressing both internal (cognitive/emotional) and external (environmental) determinants of adaptation; for example, education level or adjustment correlating with adherence and QoL in MPNs. By targeting modifiable elements like coping or self-management, maladaptive cycles can be disrupted and adaptation fostered.
The little available research in this domain in MPN has demonstrated that the majority of MPN patients, as well as their caregivers, experienced difficulties with adjustment to the life MPN forced upon them, resulting in an impaired QoL (How et al., 2024; Rossau et al., 2022). A small study of newly diagnosed MPN patients (How et al., 2024) revealed that a notable proportion of patients experience psychological distress, perceive their illness as moderately to severely threatening, and frequently rely on avoidance coping strategies.
Furthermore, most research in this area has been focused on the predictive value of individual factors, potentially neglecting meaningful patterns of co-occurring adaptation processes in which factors jointly cluster. As rare chronic illnesses such as MPN frequently entail nonlinear trajectories and idiosyncratic symptom burdens, cluster analysis has the potential to address both its theoretical and practical issues (Clatworthy et al., 2005). Theoretically, it facilitates the processing of relatively homogeneous groups from a heterogeneous sample. Practically, the labeling of these groups, derived from cluster analysis, facilitates the identification of groups that are most in need of intervention (Clatworthy et al., 2005). The CSM and the Adaptation Model both support conceptualizing adaptation as arising from the interplay of factors in a person-oriented approach, making cluster analysis theoretically appropriate. Cluster analysis was found to be valuable in understanding disease heterogeneity, particularly in cases that challenge conventional distinctions between physical and psychological etiology, and to optimize treatments (Windgassen et al., 2018). Clustering might also be useful to better understand differences in perceived QoL and wellbeing that cannot be fully explained by studying individual factors, such as coping strategy. Research on chronic illness and disability (Livneh et al., 2004) highlights that psychosocial adaptability varies significantly across individuals, leading to distinct adaptation profiles. These profiles showed differences in daily functioning, QoL, and health outcomes. Patients in the adaptive cluster experienced lower psychological distress, higher QoL, and less functional limitations than patients in the nonadaptive cluster (Livneh et al., 2004). Another cluster analysis study identified two groups of patients based on their adaptation to chronic illness. One group demonstrated poor adjustment in terms of physical functioning and depression, while the other showed better adjustment, manifesting in more beneficial beliefs (i.e. control), and effective coping (i.e. task persistence, exercising; Rubio Fidel et al., 2022). A study by Trask and Griffith (2004) found comparable results, it identified distinct cancer subgroups based on psychosocial variables. The clusters identified showed significant differences in distress and general health.
The aim of this study is to explore whether unique psychosocial adaptation profiles can be established for MPN patients and whether these profiles are associated with differences in the impact of MPN on daily life, adding to our understanding of the heterogeneous phenotype of MPN. Identification of potentially different patient profiles empowers health professionals to provide individualized and comprehensive support, tailored to each patient’s needs in order to make living with MPN more tolerable. Regular symptom assessment, lifestyle optimization, and access to psychosocial support are critical to reducing the physical and emotional burden of the disease. In addition, it would be valuable for patients to gain insight into habitual patterns, as this can help them understand how their coping mechanisms, emotional resilience and illness identity affect their QoL.
Important psychosocial adaptation variables in this context are coping, resilience, and illness identity, because of their demonstrated impact on well-being in comparable diseases. Coping, which refers to the strategies patients use to manage their disease (Siegel et al., 2016), plays an important role in psychosocial adaptation and impacts QoL (Hill and Frost, 2022; Siegel et al., 2016). Resilience, defined as ‘the ability to maintain or regain mental health, despite experiencing adversity’ (Herrman et al., 2011), has also been shown to improve QoL (Macía et al., 2020). It also decreases the prevalence of disability and somatization, and reduces the incidence of depressive and anxiety disorders (Cal et al., 2015). In addition, low levels of resilience have been linked to an increased risk on disease progression (Cal et al., 2015). Finally, the concept of illness identity, which can be described as the extent to which having a chronic illness is or is not integrated into an individual’s self-perception or identity (Oris et al., 2016), has been shown to influence overall well-being (Rassart et al., 2023). Engulfment, the extent to which illness dominates a person’s identity and daily life, is a key contributor to reduced QoL (Rassart et al., 2023). This finding was also reported in our earlier study on MPN (Eppingbroek et al., 2025).
The present study sought to identify distinct clusters of psychosocial adaptation based on coping, resilience, and illness identity using a person-centered cluster analytic approach. To capture the complexity of psychosocial adaptation without conflating it with contextual characteristics, clusters were derived exclusively from adaptation variables. Subsequently, differences in sociodemographic and disease-related characteristics at baseline (t1), and in self-management, MPN symptom burden, and (subdomains of) QoL 6 months later (t2), were studied to examine what externally characterize these clusters. As these variables are observable in clinical practice, they may facilitate the identification of patients who benefit most from targeted interventions.
The importance of these outcome variables regarding the daily functioning of MPN patients have been demonstrated in previous studies (Eppingbroek et al., 2024; Harrison et al., 2017). Furthermore, self-management will be included in cluster comparisons, as prior research in chronic diseases links it significantly to QoL (Benzo et al., 2016; Musekamp et al., 2016), which can serve as a fundamental starting point for enhancing patients’ well-being. In our study self-management refers to ‘the capacity of an individual to deal with the symptoms, treatment, physical and psychosocial consequences and lifestyle changes inherent in living with a chronic condition’ (Barlow et al., 2002). Finally, this study will test whether the revealed clusters remain stable over a period of 6 months. The interval is chosen for its widespread use in longitudinal (health) psychology studies (e.g. Sirois and Hirsch, 2017) and has been shown to be sufficient to detect meaningful change while minimizing participant burden and reactivity.
Methods
Design, participants and procedure
This is a longitudinal study among 338 MPN patients in the Netherlands. They were recruited through collaborations with the Dutch patient organization and two Dutch private online social platforms for MPN patients. Participants were asked to complete an online questionnaire anonymously twice after providing informed consent thus creating a dataset based on self-reports. Six months after completing the baseline questionnaire (T1), they were asked to complete the second questionnaire (T2). Both questionnaires were identical, with the exception of the sociodemographic and disease-related items (only measured at T1). Inclusion criteria were: living in the Netherlands, being ⩾18 years old, and being diagnosed with an MPN. Participants who completed the questionnaire only once (N = 117) were excluded from the main analyses because the study focused on within-person change over time, requiring repeated measurements. Except from educational level (dropout was significantly higher among patients with lower education level compared to those with middle and high education levels), baseline comparisons indicated no substantial differences between completers and dropouts, suggesting minimal attrition bias.
Measures
Variables in the cluster analysis
Coping
Coping is operationalized by social problem-solving abilities; meaning an individual’s efforts to cultivate effective coping mechanisms to address specific challenging circumstances encountered in daily life (D’Zurilla et al., 2002). This was assessed using the short 10-item version of the Social Problem Solving Inventory-Revised (SPSI-R; Dreer et al., 2009; D’Zurilla et al., 2002). Participants were asked to rate each statement on a five-point scale from 0 ‘not applicable to me’ to 4 ‘totally applicable to me’. Total scores were calculated by summing all 10 items (after reversing the maladaptive components) ranging from 0 to 40. Higher scores indicate greater use of social problem-solving abilities (Willems et al., 2017). Scale reliability was α = 0.71 (T1) and α = 0.69 (T2; one item was omitted due to low reliability).
Resilience
The Dutch version of the Brief Resilience Scale (BRS; Smith et al., 2008) was used to measure resilience. Respondents were asked to indicate their level of agreement with six statements on a five-point Likert scale ranging from 1 ‘strongly disagree’ to 5 ‘strongly agree’. For example, ‘I tend to bounce back quickly after hard times’. The calculated mean score ranges from 1 to 5. A higher total score indicates greater resilience, considered to be beneficial for disease adaptation. The reliability of this scale was α = 0.79 (T1) and α = 0.80 (T2).
Illness identity
We used the Illness Identity Questionnaire (IIQ; Oris et al., 2016) that measures four illness identity states in chronically ill people. For 25 statements, participants were asked to indicate their level of agreement on a five-point Likert scale ranging from 1 ‘totally not agree’ to 5 ‘totally agree’. By means of the IIQ patients are scored on four different illness identity states: A high score for an identity state indicates that it is highly present. Acceptance (items 6–10, example: ‘I accept being a person with MPN’) and enrichment (item 19–25, example: ‘Because of my MPN, I realize what is really important in life’) are considered to be beneficial, engulfment (item 11–18, example: ‘My MPN dominates my life’) and rejection (item 1–5, example: ‘I just avoid thinking about my MPN’) detrimental to disease adaption. The reliability of the four scales ranged from α = 0.83 to α = 0.92 (at T1) and α = 0.80 to α = 0.93 (at T2).
Variables on which the derived clusters have been compared
Sociodemographic and disease-related variables
Sociodemographic variables that were measured: age, gender, level of education (low, middle, high), civil state (having a partner and living together or apart, being single). Disease-related factors that were measured: type of MPN (essential thrombocythemia (ET), polycythemia vera (PV), and myelofibrosis (MF)), duration of the disease (in years), treatment options (wait-and-see, phlebotomy, phlebotomy combined with cytoreduction or cytoreduction only), possible side-effects (none, a little, a lot), complications (no or yes), and comorbidities (no or yes).
Self-management
Self-management was assessed with the Dutch version of the Patient Activation Measure® (PAM®-13 NL; Hibbard et al., 2005) and offers insight into the knowledge, skills and confidence in being able to manage one’s own health or illness. Thirteen statements could be rated on a scale of 1 (‘totally not agree’) to 4 (‘totally agree’) or 5 (‘not applicable’). For example, ‘I am confident I can help prevent or reduce problems associated with my health’. The total score is obtained by assigning points according to a predefined system and then calculating the sum of these points, resulting in a score ranging from 0 to 100. The higher the score, the better people self-manage. The reliability of this scale was α = 83 (both at T1 and T2).
MPN symptom burden
The 10 most specific symptoms related to MPN were measured with the MPN Symptom Assessment Form Total Symptom Score (MPN-SAF TSS; Emanuel et al., 2012). Symptoms, for example ‘bone pain’, could be rated on a scale of 0 (‘absent/not such issue’) to 10 (‘worst imaginable/as bad as it can be’). The Total Symptom Score (TSS) is calculated by summing the individual symptom scores, with a range of 0–100. A higher score indicates greater disease burden, hereafter referred to as MPN symptom burden. The reliability of the scales was α = 0.84 (at T1) and α = 0.86 (at T2).
QoL
To measure QoL and its subdomains, the EORTC-QLQ-C30 was used, a 30-item questionnaire that measures different aspects of QoL in cancer patients. This instrument consists of a global health status/overall QoL scale, five functional scales, three symptom scales, and six single items (Aaronson et al., 1993). We only included the global health status (hereafter referred to as QoL), role, cognitive, emotional and social functioning scales, the symptoms fatigue (scale) and insomnia (single item) in the analyses, based on their earlier found effects in MPN (Eppingbroek et al., 2024). QoL answers ranged from 1 (not at all) to 7 (very much); the other measured items had a range from 1 (not at all) to 4 (very much). The answers were converted to scales and single scores ranging from 0 to 100. A higher score on the QoL and functional scales reflects better health/well-being; a higher score on the symptom scales or single items represents a more severe condition. One example of an item regarding cognitive functioning is: ‘Have you had difficulty in concentrating on things, like reading a newspaper or watching television?’. The reliability of the subscales ranged from r = 0.57 (both T1 and T2) to α = 0.87 (T1) and α = 0.88 (T2; p < 001).
The authors of the Dutch translations of the used questionnaires and psychometrics are listed in Supplemental Appendix 1.
Statistical analyses
Descriptive statistical analyses were used to describe the characteristics of the sample.
A cluster analysis was performed to group MPN patients with similar psychosocial adaptation profiles. The variables used for this classification included baseline mean scores (T1) of coping, resilience and the illness identity subscales rejection, acceptance, engulfment and enrichment. We used the hierarchical cluster analysis technique, standardized scores, Euclidean distance measure and Ward method providing a robust framework for uncovering latent psychosocial profiles in MPN patients. The cluster profiles were visually inspected with a dendrogram. Finally, K-means analysis was performed, using standardized z-scores, algorithm Hartigan-Way and a number of 10 starting values to examine the stability and validity of the clusters created. The default convergence was used.
After clustering, chi-square tests were performed to identify differences between the derived cluster profiles with respect to gender, marital state, education level, MPN subtype, type of treatment, comorbidities, complications, disease course, side effects and impact on working life (all measured at T1).
One-way ANOVA tests identified differences between the cluster profiles and age (T1), time since diagnosis (T1), self-management (T2), MPN symptom burden (T2), role, social and emotional functioning (T2), cognitive functioning (T2), fatigue (T2), and insomnia (T2). Finally, to obtain an indication on the stability of the clusters over time, it was assessed whether self-management, symptom burden and (aspects of) QoL changed over time. For this purpose, we computed and examined the Delta scores between T1 and T2 with regard to these variables. The stability of the clusters was also assessed by testing the Delta scores of each of the cluster variables (T2 – T1).
Analyzes were performed using Jamovi (version2.3.28).
Results
Characteristics of patient population
The majority of participants were female (63.6%) and the mean age was 62.8 years, SD 10.9 (Table 1). Most of the participants were higher educated (57.4%) and in a relationship (84.3%). MPN subtype PV was most frequently reported (45.3%) and the most frequently cited treatment was cytoreduction (60.9%). A lot of participants experienced side-effects (68.5%) and/or comorbidities (53.8%). Complications were less common (24.3%). In the majority of cases, the course of disease remained stable (55.2%). In almost half of the cases, MPN had an impact on the patient’s ability to work.
Sociodemographic and disease-related characteristics of MPN patients (N = 338 a ).
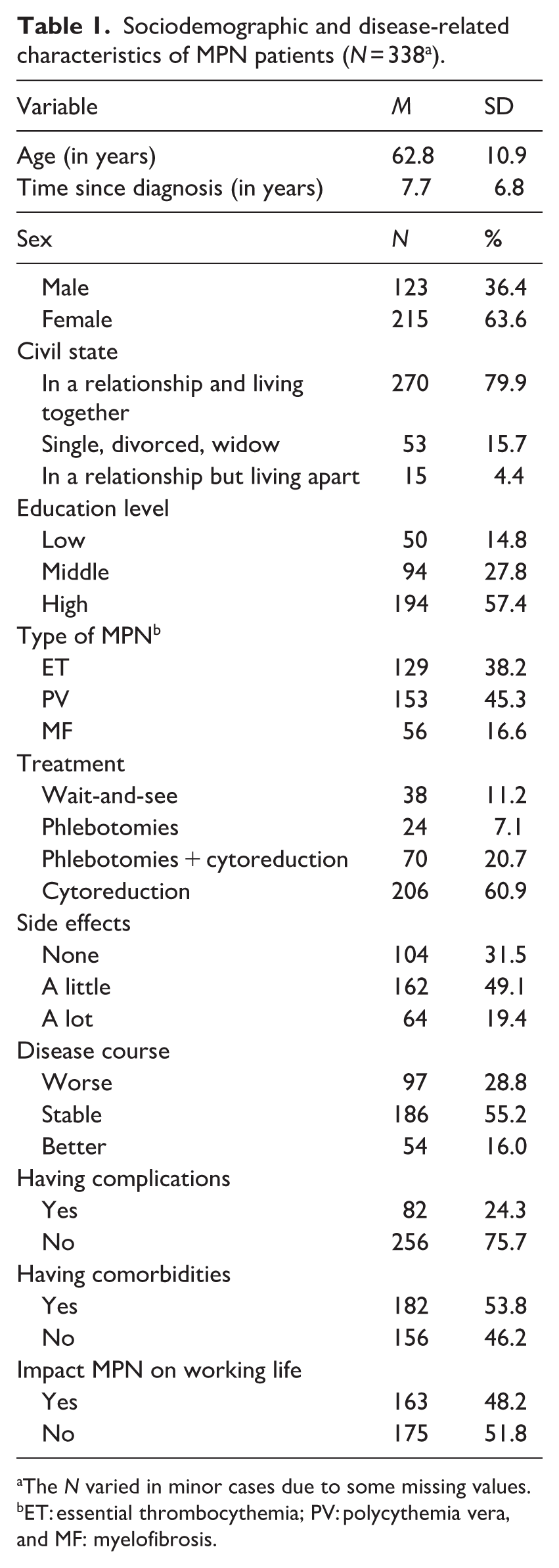
The N varied in minor cases due to some missing values.
ET: essential thrombocythemia; PV: polycythemia vera, and MF: myelofibrosis.
Clusters of psychosocial adaptation
The dendrogram of the hierarchical cluster analysis (Supplemental Appendix 2) showed that the interrelated psychosocial adaptation variables reflected three subgroups of patients that differed on their adaptation profile. This 3-cluster solution has been confirmed by the K-means analysis and the scree plot (Figure 1).
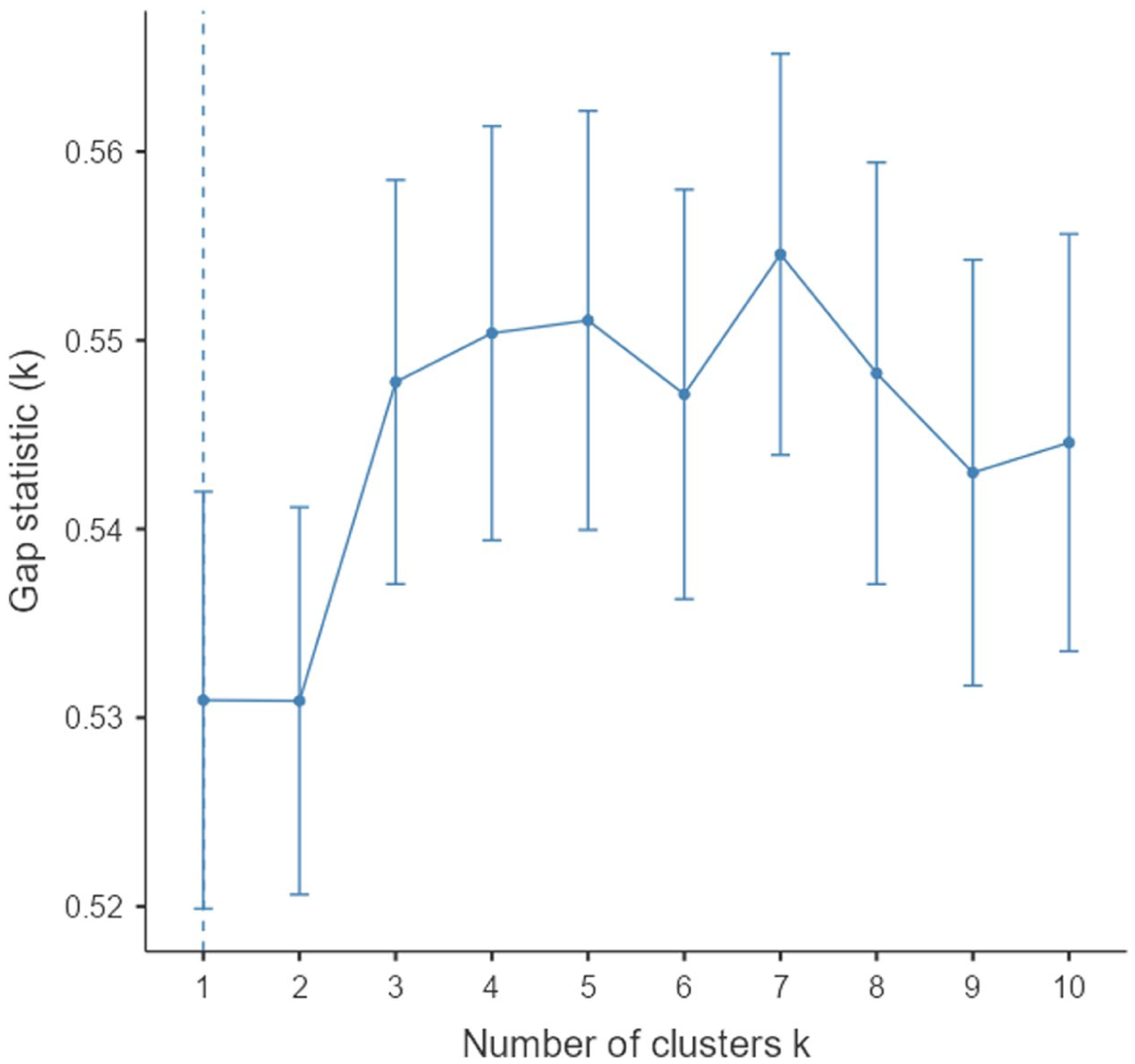
Optimal number of clusters.
Characteristics of the different clusters
The differences between the 3 clusters with regard to psychosocial adaptation are illustrated in Figure 2 and Table 2.
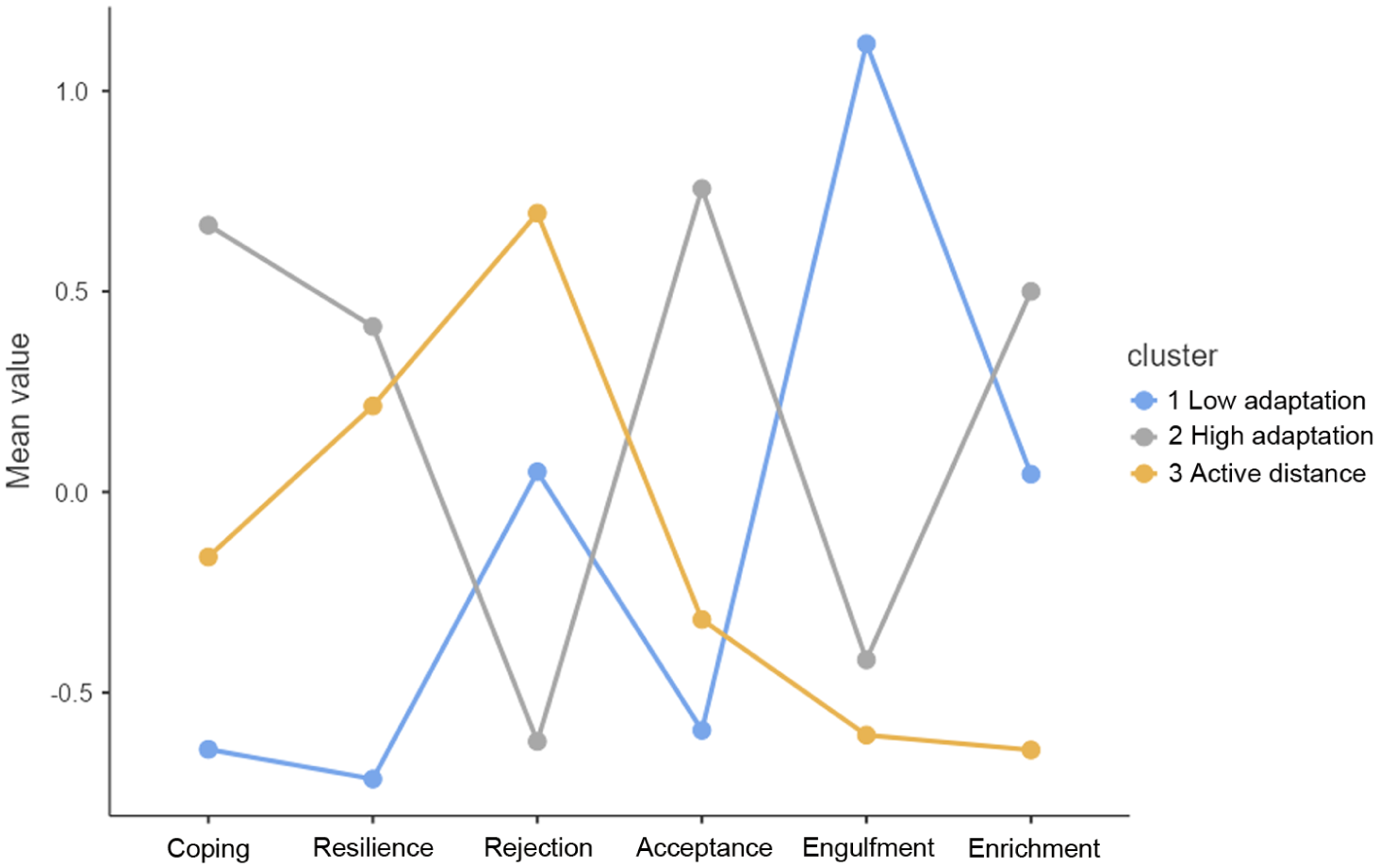
Plot of means of the psychosocial adaption variables for the three clusters.
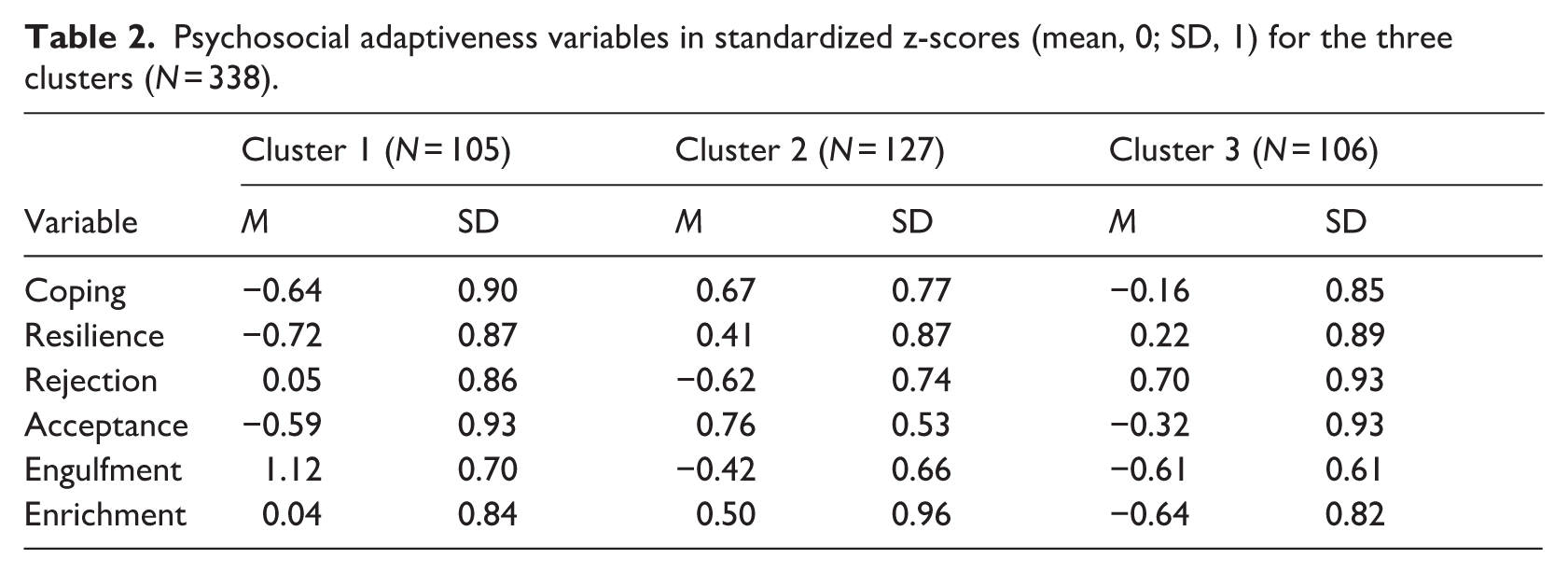
Psychosocial adaptiveness variables in standardized z-scores (mean, 0; SD, 1) for the three clusters (N = 338).
Cluster 1 (C1) ‘Low Adaptation’, (N = 105)
Participants in this group display a low psychosocial adaptation pattern, indicated by their scoring on psychosocial skills: they showed the lowest scores on coping (Z M = −0.64, SD = 0.90) and resilience (Z M = −0.72, SD = 0.87) and the highest on engulfment (Z M = 1.12, SD = 0.70) compared to the other clusters. Also, their acceptance score (Z M = −0.59, SD = 0.93) is below average.
Cluster 2 (C2): ’High Adaptation’ (N = 127)
This cluster is distinguished by its high level of psychosocial adaptation skills. Participants in this group tend to adapt successfully to their condition, indicated by high scores on coping (Z M = 0.67, SD = 0.77), resilience (Z M = 0.41, SD = 0.87), acceptance (Z M = 0.76, SD = 0.53) and enrichment (Z M = 0.50, SD = 0.96). Moreover, engulfment (Z M = −0.42, SD = 0.66) and rejection (Z M = −0.62, SD = 0.74) were significantly lower than the mean scores of the other two clusters.
Cluster 3 (C3): ‘Active Distance’, (N = 106)
This group showed a high score on the IIQ subscale reject (Z M = 0.70, SD = 0.93), indicating active distancing from the illness as part of their identity (e.g. ‘I refuse to let my condition define me’) and also the lowest on engulfment (minimal identification with MPN; Z M = −0.61, SD = 0.61) as well as on enrichment (limited perception of positive growth or benefits from the illness experience; Z M = −0.64, SD = 0.82) compared to the other clusters. This cluster seems to reflect a self-protective stance, avoiding the extremes. In terms of coping and resilience, they fall between cluster 1 (‘low adaptation’) and cluster 2 (‘high adaptation’).
Differences between the clusters regarding sociodemographic and disease related variables
Chi-square and one-way ANOVA tests revealed significant differences between the clusters of psychosocial adaptation, as outlined in Tables 3 and 4. Regarding socio-demographic characteristics, the ’low adaptation’ cluster (C1) had a significantly higher percentage of a low level of education (21%) and was also the youngest group (M = 61.12, SD = 11.07). However, only the ‘active distance’ group (C3) had a significantly higher mean age (M = 65.55, SD = 10.36).
Chi square tests on sociodemographic and disease-related differences regarding the three clusters.
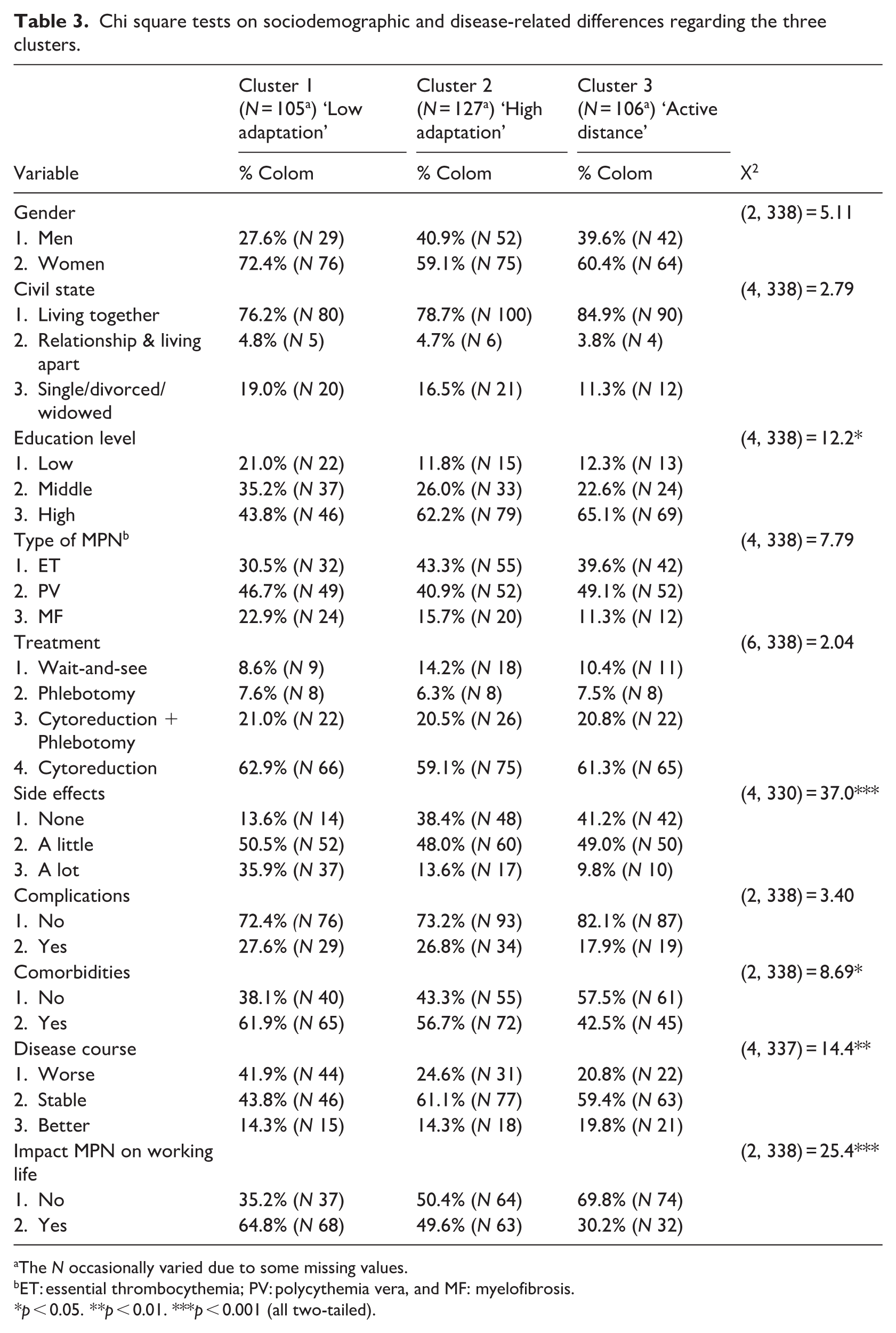
The N occasionally varied due to some missing values.
ET: essential thrombocythemia; PV: polycythemia vera, and MF: myelofibrosis.
p < 0.05. **p < 0.01. ***p < 0.001 (all two-tailed).
ANOVA tests showing sociodemographic and disease-related differences regarding the three clusters.
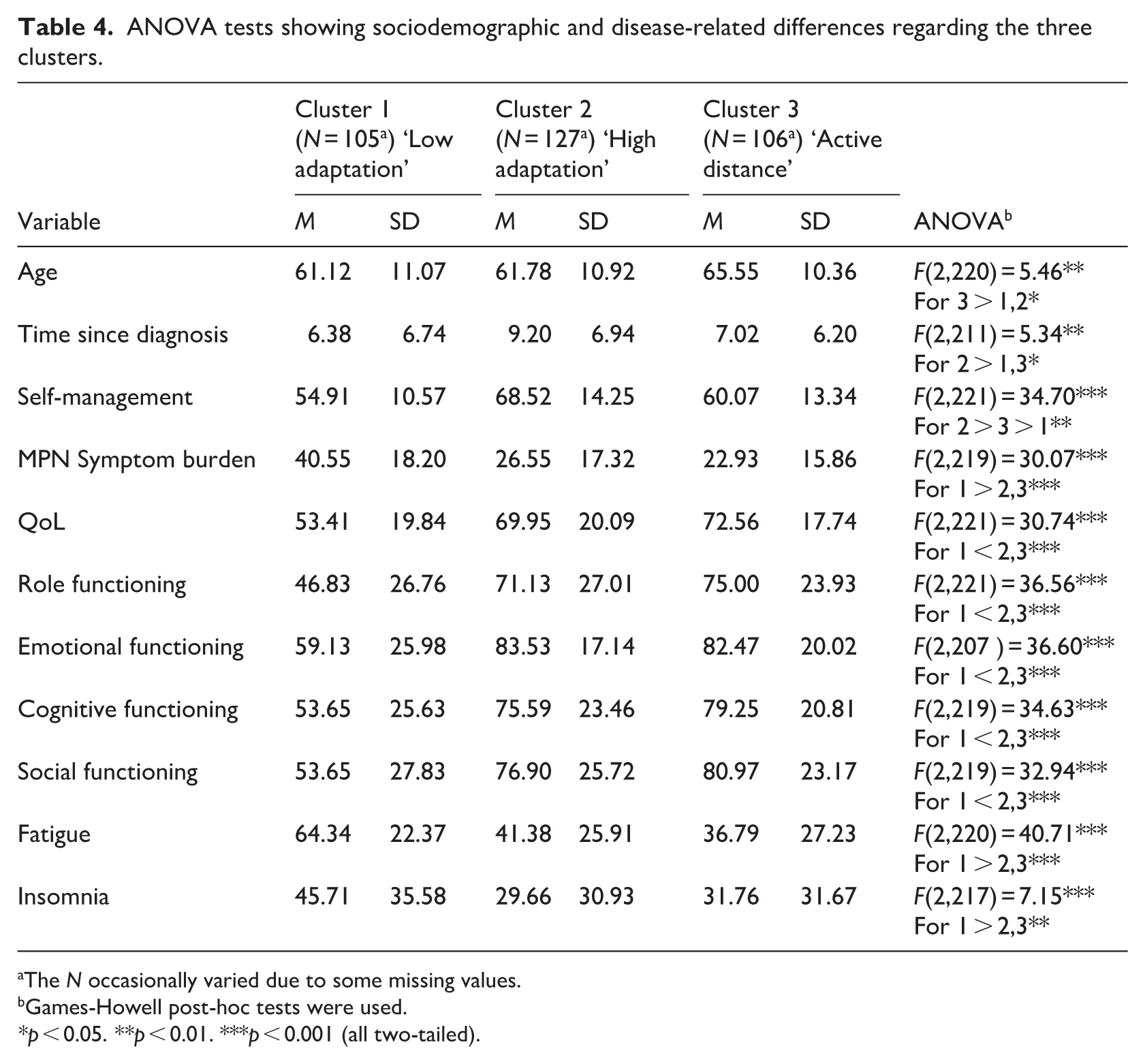
The N occasionally varied due to some missing values.
Games-Howell post-hoc tests were used.
p < 0.05. **p < 0.01. ***p < 0.001 (all two-tailed).
The ‘low adaptation’ cluster (C1) showed the highest percentage of participants whose MPN impacted work life (64.8%), while the ‘active distance’ group (C3) had the highest percentage reporting no impact (69.8%).
Looking at the disease-related variables the ‘low adaptation’ cluster (C1) had significantly highest percentages of participants with a lot of side-effects (35.9%), comorbidities (61.9%) and disease deterioration (41.9%), whilst the ‘active distance’ group (C3) significantly showed the highest percentage of having no side-effects (41.2%), no comorbidities (57.5%) and disease improvement (19.8%). The ’high adaptation’ group (C2) exhibited a significantly longer disease duration (M = 9.20, SD = 6.94) and highest self-management scores (M = 68.52, SD = 14.25), which was the lowest with the ‘low adaptation’ group (C1; M = 54.91, SD = 10.57). Furthermore, the ‘low adaptation’ group (C1) exhibited significantly the most pronounced MPN symptom burden, fatigue, and insomnia. With respect to functional scales, namely role, emotional, cognitive, and social functioning, this cluster (C1) demonstrated significantly lower scores compared to C2 and C3.
Differences between clusters in predictor and outcome variables over time
The Delta scores of the outcome variables (i.e. self-management, MPN symptom burden, QoL, role, emotional, cognitive, and social functioning, fatigue, and insomnia) revealed slight fluctuations over the 6-month period (see Table 5). ANOVA tests showed no significant differences.
ANOVA tests showing delta scores (T2 – T1) on outcome variables regarding the clusters of psychosocial adaptation.
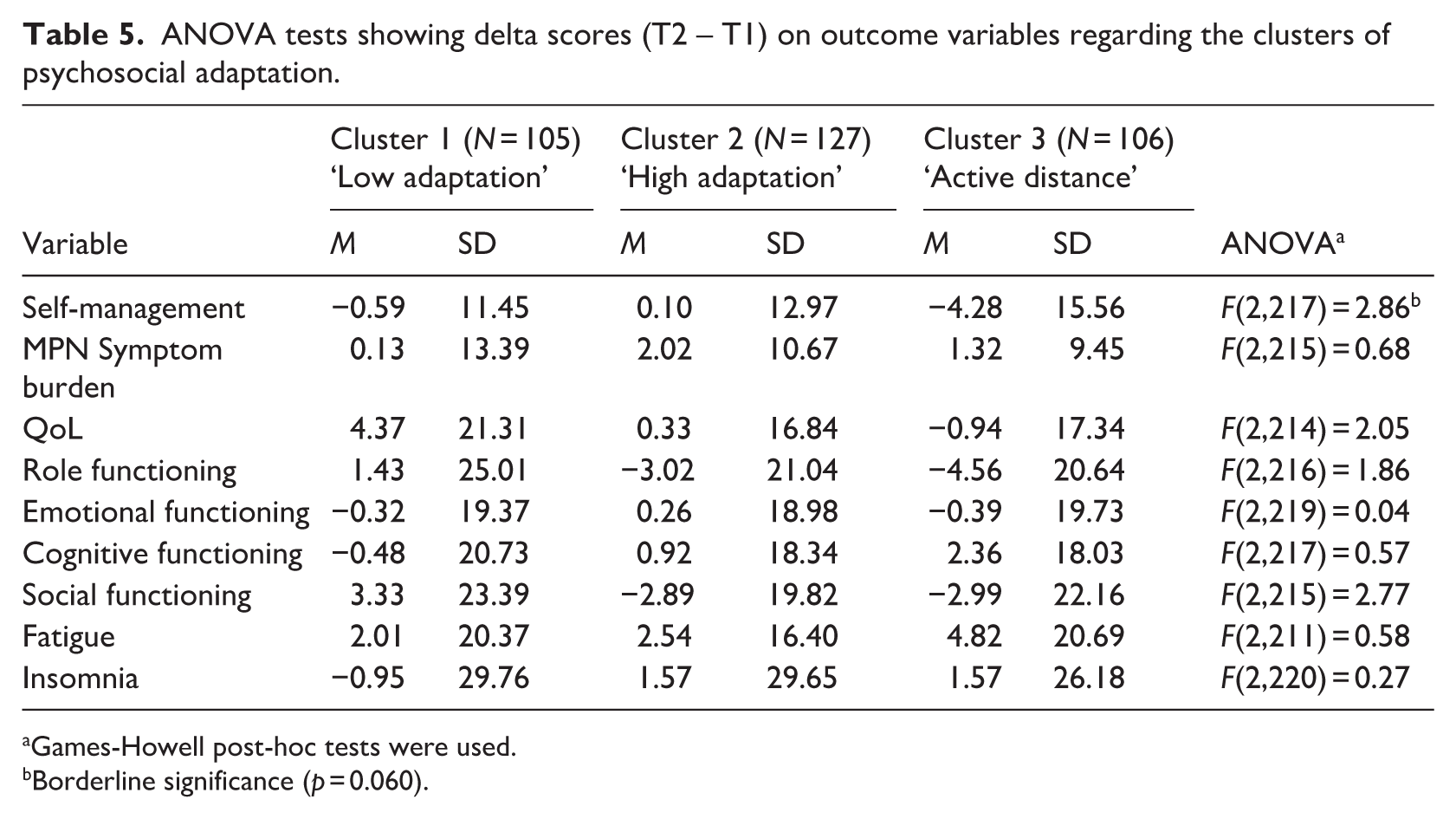
Games-Howell post-hoc tests were used.
Borderline significance (p = 0.060).
Delta scores on the cluster variables (i.e. coping, resilience, rejection, acceptance, engulfment, and enrichment) did illustrate significant differences over time (see Table 6). More specifically, the ‘low adaptation’ group (C1) showed significant increase in problem-solving coping (M = 0.96, SD = 5.37) and acceptance (M = 0.08, SD = 0.87) compared to the ‘high adaptation’ group (C2). The ‘low adaptation’ group (C1) also showed a significant decline in engulfment compared to the other two clusters (M = −0.15, SD = 0.71). Finally, the ‘active distance’ cluster (C3) showed less rejection over time, while the rejection score increased in the ‘high adaptation’ (C2) group (M = 0.24, SD = 0.75).
ANOVA tests showing delta scores (T2 – T1) on cluster variables included in cluster analysis.
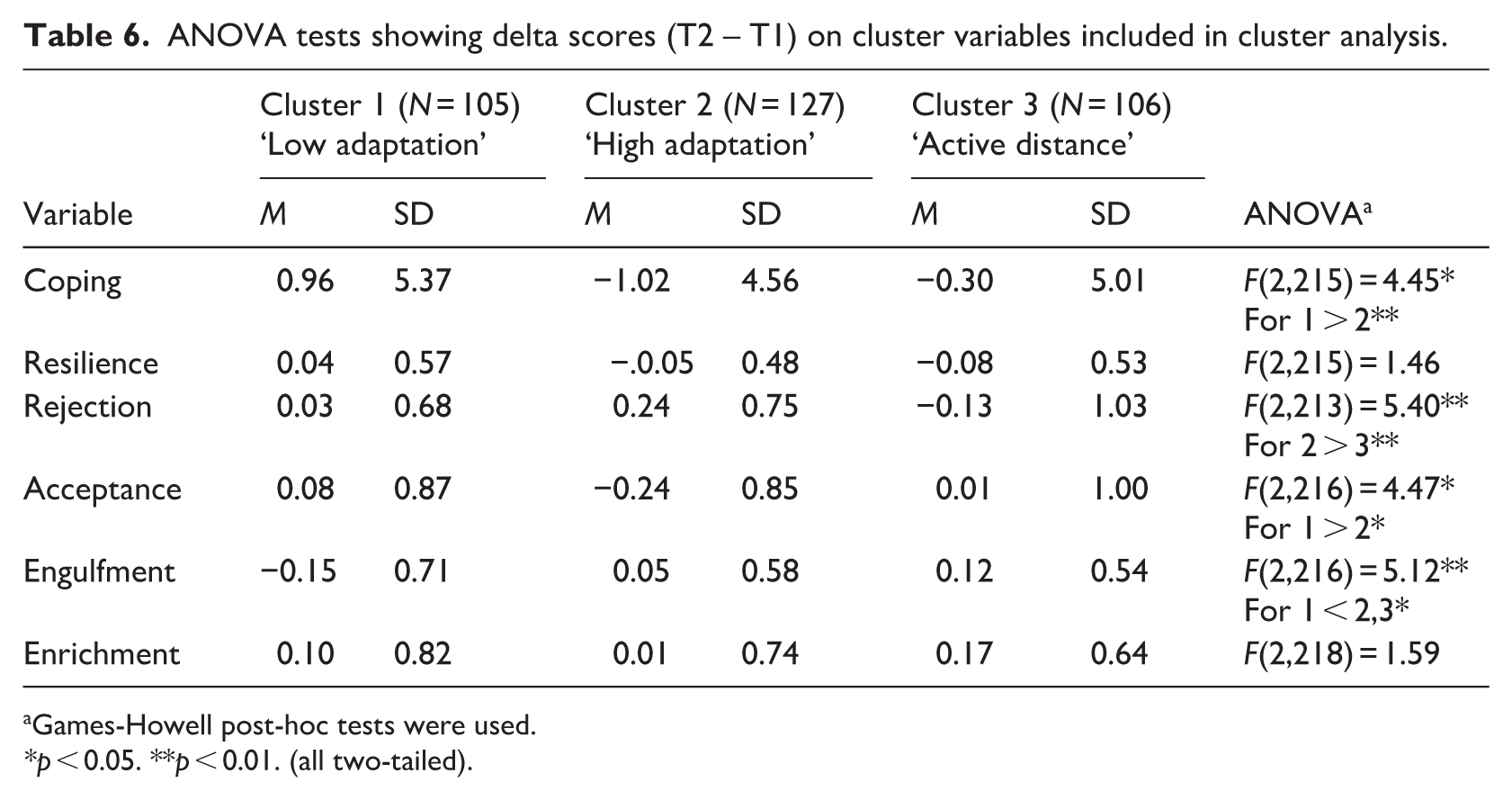
Games-Howell post-hoc tests were used.
p < 0.05. **p < 0.01. (all two-tailed).
Discussion
The present study uniquely employs cluster analysis to explore psychosocial adaptation in MPN patients, thereby enhancing comprehension of the disease and improving support for this lifelong condition. Three distinct profiles concerning psychosocial adaptation in MPN patients were revealed. These groups significantly differed with respect to the sociodemographic and disease-related variables studied. The first group, labeled as ‘low adaptation’ (C1), is characterized by a relatively lower degree of education and the highest level of suffering and impact in various areas of life. Participants in the second group, the ‘high adaptation’ cluster (C2), scored higher on self-management, showed the longest disease duration, and suffered slightly more than the third, ‘active distance’, group (C3), but much less than the ‘low adaptation’ group (C1). Finally, participants in the third cluster, characterized by ‘active distance’ (C3), were the oldest group and seemed to suffer the least from their MPN. This key finding offers a comprehensive explanation for the heterogeneity observed in MPN, as reported in previous studies (Geyer et al., 2014; Tremblay and Mesa, 2022).
It is evident that participants with MPN who exhibit lowest psychosocial adaptation scores (C1) experience the most pronounced suffering and consequences. These findings align with Livneh et al. (2004), who demonstrated that individuals in the adaptive cluster had better QoL and fewer functional limitations than those in the non-adaptive cluster. Similarly, our high adaptation-scoring group (C2) suffered significantly less from MPN than the low-adapting group (C1). However, the ‘high adaptation’ group in our study (C2) did not differ significantly from the ‘active distance’ group (C3) regarding (aspects of) QoL and symptom burden. Given that high psychosocial adaptation and self-management are often found to be associated with improved QoL and health outcomes (Benzo et al., 2016; Musekamp et al., 2016), it is surprising that the disease impact in clusters 2 and 3 are quite similar. One explanation could be that strong adaptation and self-management in C2 mitigate the potential severity of the disease. However, we did not measure this directly, and outcome variables remained stable over time (representing a trait rather than a state). Alternatively, a ceiling effect, indicating that the disease burden is unlikely to improve further, might occur. However, our results showed some significant differences in the Delta scores of the cluster variables (coping, rejection, acceptance and engulfment), indicating that the clusters may be dynamic over time. Thus, even though the clusters were initially formed based on certain psychological traits, the fact that there were significant differences in how these traits changed over time suggests that the clusters themselves are not completely fixed. People within these clusters may shift in their coping, acceptance, level of distancing, tendency to reject and/or feelings of being overwhelmed by illness. This implies that psychosocial adaptation can change and potentially improve over time.
In terms of illness identity participants in the ‘active distance’ group (C3) appear to be less concerned with the illness, given the high rejection score and even lower scores on other subscales (such as engulfment). Although research has shown that patients with high rejection scores were having worse symptoms (Van Bulck et al., 2021), that’s not the case in our study.
It is possible that in our MPN population, rejection may have a beneficial impact on the perceived symptoms, potentially serving as a form of self-preservation or self-protection. This form of psychological distancing or detachment from the illness may serve as a coping strategy. No statistical evidence supported this assumption due to our study design. However, a separate study found positive results for self-protection strategies in preventing high levels of systemic inflammation (Castonguay et al., 2014).
Scores in this ‘active distance’ group (C3) may also relate to the way this group of patients feels about life and ability to put things into perspective, as a study in chronic disease showed that optimism is associated with better QoL (Hurt et al., 2014). However, we haven’t measured optimism nor cognitive emotion regulation, so this cannot be tested. Another explanation could be that the high rejection score is related to the age of this group (C3), which was significantly higher compared to the other clusters. In another study, high age was associated with higher rejection scores (Van Bulck et al., 2021). This could also apply in our study. Conversely, this group (C3) experienced less negative impact from MPN on work life, side effects, comorbidities, and disease progression. This may indicate that the disease is less intertwined with their identity. Though, these interpretations are just hypothetical and require empirical validation. It may be worth exploring whether less preoccupation with MPNs could have a positive effect on its impact. Furthermore, there may be other factors that contribute to this patient profile, such as emotional venting or social support, as described in the model of Leventhal et al. (2001). Nevertheless, these factors fall outside the scope of this study.
Study limitations
This study is self-reported and was conducted in the Dutch context, which may not be representative of international practices.
The study sample overrepresented higher educated participants (57.4% highly educated; 14.8% low education), which limits generalizability of the results. This can be partly explained by the fact that relatively more lower-educated dropped out. It might also have been caused by the recruitment method; a patient organization and two online social platforms. Members of online support groups tend to be more highly educated (Hillen et al., 2014). It is important to include and retain less educated individuals in future research, and this should already be considered in the recruitment method.
This study measured social problem-solving abilities, limiting the analysis of other coping strategies. Future research should use more comprehensive instruments, incorporating emotion-focused coping, a key pathway to adjustment and wellbeing (Lazarus and Folkman, 1984).
Of the three distinct patient profiles, it is evident that the ‘low adaptation’ group (C1) experiences the most significant challenges. Nevertheless, the predictive value of the combined adaptation factors toward self-management, symptom burden, and (aspects of) QoL, as well as the stability of the clusters over a longer period of time, remains to be examined. An earlier study demonstrated that high engulfment exhibited a negative correlation with QoL in MPN, other psychosocial adjustment predictors were not significant (Eppingbroek et al., 2025). It is crucial to investigate whether the combination of these factors can predict suffering in MPN or if it is the other way around. This principle also applies to the other clusters, as it could provide a significant starting point for treatment.
Finally, changes in cluster variables over time suggest that clusters may not be fully stable. Rigid clustering can oversimplify subgroups, whereas dynamic or context-dependent clusters may better capture overlapping traits such as comorbidities and treatment responses. Recognizing possible transitions between these categories enables more targeted interventions across different stages of a patient’s disease. The dynamic nature of these clusters should be interpreted cautiously, and longer-term longitudinal investigations are recommended to confirm their persistence.
Clinical implications
Derived patient adaptation profiles provide care professionals with crucial insights for tailoring interventions. It is essential that they understand these profiles and promptly recognize any issues that need attention.
Those in the ‘low adaptation’ profile (C1) are most affected by MPN. Their treatment should address not only symptom burden, but also improve psychosocial adaptation by strengthening resilience, improving problem-based coping, and mitigating feelings of being overwhelmed. A modest enhancement in psychosocial adaptation was observed, indicating potential for further development. Since 21% of this group was low-educated, treatment should be tailored to their level of cognitive understanding. To illustrate, people in vulnerable social positions, like those with limited education or literacy, may experience health disparities, self-management challenges, poorer health, and higher healthcare consumption (De Raad voor Volksgezondheid & Samenleving, 2021; Heide and Rademakers, 2015).
Although the ‘active distance’ profile (C3) has a disease burden comparable to the ‘high adaptation’ profile (C2), health care providers should be particularly aware of this patient group. Because of their older age and high rejection score, these patients may be less adherent to treatment (Smaje et al., 2018) or to a healthy lifestyle and therefore at greater risk for complications. It is possible that the burden of disease in this group can be improved by increasing psychosocial adaptation and self-management skills, although the predictive value of these factors has not been investigated in this study. Nevertheless, it is recommended that this patient group be made more aware of the risks associated with their illness and the significance of adhering to treatment regimens.
Conversely, we may learn from this patient group. It is plausible that their high rejection and other low illness-identity scores reflect a positive approach to dealing with their MPN, resulting in a lower burden of disease. This approach, if beneficial, may offer valuable insights for other patients as well. For example, they could learn to manage their MPN with a more positive mindset and develop effective self-protection coping strategies. However, further investigation is necessary to fully explore these possibilities.
Conclusion
This study revealed that three distinct MPN patient profiles can be identified based on specific clusters of psychosocial adaptation. These groups differed in sociodemographic and disease-related characteristics, self-management, and (aspects of) QoL. Notably, patients with low psychosocial adaptation-scores are particularly negatively affected by their MPN. These findings provide important starting points for a more targeted and comprehensive psychosocial support and management of MPNs. Future research is recommended to study how MPN patient profiles evolve over time in the long run.
Supplemental Material
sj-docx-1-hpq-10.1177_13591053261438037 – Supplemental material for Heterogeneity among patients with myeloproliferative neoplasms: Identifying distinct profiles through cluster analysis
Supplemental material, sj-docx-1-hpq-10.1177_13591053261438037 for Heterogeneity among patients with myeloproliferative neoplasms: Identifying distinct profiles through cluster analysis by Annemiek A. M. Eppingbroek, Lilian Lechner, Moniek A. de Witte, Marjan D. Nijkamp, Peter Verboon and Catherine A. W. Bolman in Journal of Health Psychology
Footnotes
Acknowledgements
We would like to thank all MPN patients who participated in this study. Their cooperation and willingness to share their experiences were invaluable to our research.
Ethical considerations
The study is approved by the Ethics Committee of the Faculty of Psychology of the Open Universiteit in the Netherlands (11-10-2022; nr file no. of approvement letter: U202208062).
Pre-registration
This study and is preregistered on the Open Science Framework.
Consent to participate
All participants signed a digital informed consent form before participating. Consent was obtained electronically via a secure online platform that complies with data protection and privacy regulations.
Consent for publication
Consent for publication is not applicable to this article as it does not contain any identifiable data
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: One of the authors was a board member of the Dutch MPN patient organization.
Data availability statement
As part of the agreement on using the EORTC and PAM®-13 NL questionnaires, the respective institutions (the EORTC Quality of Life Group and Insignia Health) will receive anonymized data from these questionnaires. In accordance with the FAIR Data Principles, the research group will consider requests for access to the data after publication of the scientific articles. Requests should be sent to the first author, Annemiek Eppingbroek.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
