Abstract
Young adults with type 1 diabetes face challenges managing their condition due to numerous lifestyle factors. Insulin pumps can improve glucose control during this period, but are not used by most adults. Automated insulin delivery systems can further help young adults manage their HbA1c levels. The study examined associations between perceived readiness to use new technology, insulin pump use, and automated insulin delivery system. Higher technology readiness was associated with 9% higher time spent in automated insulin delivery mode [β = 8.99 (0.50, 17.48)] and each higher percent was associated with lower HbA1c [β = −0.01 (−0.02, −0.01)] in a large, national sample of young adults with type 1 diabetes. Findings suggest that readiness to use innovative technology may be associated with the use of insulin pumps and a willingness to rely on automated insulin delivery systems. ClinicalTrials.gov NCT04646473.
Introduction
Only 21% of young adults with Type 1 diabetes (T1D) in the United States meet the American Diabetes Association standards of an HbA1c of below 7.0% (53 mmol/mol; Khadilkar and Oza, 2022). In fact, among all ages with T1D, young adults face unique challenges in managing their chronic illness due to numerous concurrent lifestyle changes (Baudino et al., 2025). Chronically out-of-range blood glucose can lead to significant short and long-term medical complications (Syed, 2022). Short-term risks include severe hypoglycemia and diabetic ketoacidosis, while long-term risks include cardiovascular and renal disease (Syed, 2022).
Despite these barriers, young adults have benefited from recent health-technology advancements (Karges et al., 2017). Insulin pumps streamline T1D management by providing insulin boluses either by connecting to a continuous glucose monitoring device (CGM) or manually programed delivered through direct contact with the body (Karges et al., 2017). Therefore, when used correctly, insulin pumps lower the risk of long-term complications and maintain better HbA1c levels than multiple daily insulin injections (Karges et al., 2017). Recent studies suggest that insulin pumps can reduce HbA1c levels, lower hypoglycemia rates, and reduce glycemic variability, potentially lowering emergency healthcare utilization costs (Karges et al., 2017; Parker et al., 2024).
Additionally, automated insulin delivery (AID) systems also improve HbA1c among young adults (Bassi et al., 2023). AID systems, also known as closed-loop systems, integrate data from continuous glucose monitors through an algorithm that signals insulin delivery from an insulin pump to maintain blood glucose in a predefined target range, reducing the decision-making strain (Renard, 2023). However, they do come with some additional barriers, such as accessibility from medical insurance providers and physician education (Messer et al., 2025). Additionally, certain psychological factors, such as the concern for overreliance, deskilling of traditional insulin management techniques, and the added stress of managing an additional technology, are barriers to AID system adoption (Nefs, 2022).
To our knowledge, recent data on pump use among U.S. young adults are limited. The most recent high-quality study found that about 60% of adults ages 18 years or older across Canada used an insulin pump, where insulin pump use was associated with older age (Soliman et al., 2024). Similarly, a 2022 U.S. study of over 48,000 adults reported a 62% pump-use rate (Ebekozien et al., 2023). Typically, young adults have high technology usage, as over 98% own a smartphone and access the internet regularly (Sheet, 2018). While prior studies have examined physical barriers to insulin pump use, the relationships between perceived readiness to use new technology and using an insulin pump or AID system have not been examined, particularly in young adults managing T1D (Pauley et al., 2021).
Perceived readiness is often measured using the Technology Readiness Index (TRI), a scale used to understand individuals’ dispositions toward adopting new technologies, comprised of four dimensions: optimism, innovativeness, discomfort, and insecurity (Parasuraman and Colby, 2015). Optimism and innovativeness are considered enablers, while discomfort and insecurity are regarded as inhibitors. Individuals who score high on the enabler measures are more willing to experiment with new diabetes technology. On the other hand, high scorers on the inhibitor measures might be reluctant to try new devices. This measure overall serves as a tool to help determine preferences for technology use and integration (Blut and Wang, 2020).
This study examined associations between people’s propensity to embrace new technology and their insulin pump/AID use in a national, cross-sectional sample of 388 young adults with T1D and above target HbA1c. All participants were recruited for a randomized clinical trial of a digital health intervention, and data for this analysis were collected at baseline. In this large, national sample of young adults with T1D and above-target HbA1c, we hypothesized that greater perceived readiness to use new technology would be related to using a pump (vs multiple daily injections) and more time spent in automated mode among users of AID systems.
Methods
Study design
This secondary analysis examined cross-sectional baseline data from 2021 to 2024 from a national randomized clinical trial (NCT04646473) that tested the efficacy of a digital health intervention in improving self-management and blood sugar control in young adults with T1D. This study was approved by the Dartmouth College Committee for Protection of Human Subjects (IRB #23559), and all participants completed informed written consent prior to completion of the survey.
Recruitment and procedures
Full inclusion and exclusion criteria were included in the trial protocol paper (Stanger et al., 2021). In short, participants included young adults ages 19–29 years old with T1D and a recent above-target self-reported HbA1c ≥ 7.5% (Table 1; Khadilkar and Oza 2022). All participants included in this secondary analysis completed a web-based survey assessing sociodemographics, technology readiness, and their primary insulin delivery methods (N = 388). A supplemental subgroup analysis was conducted among AID users (n = 239; Figure 1).
Participant characteristics.
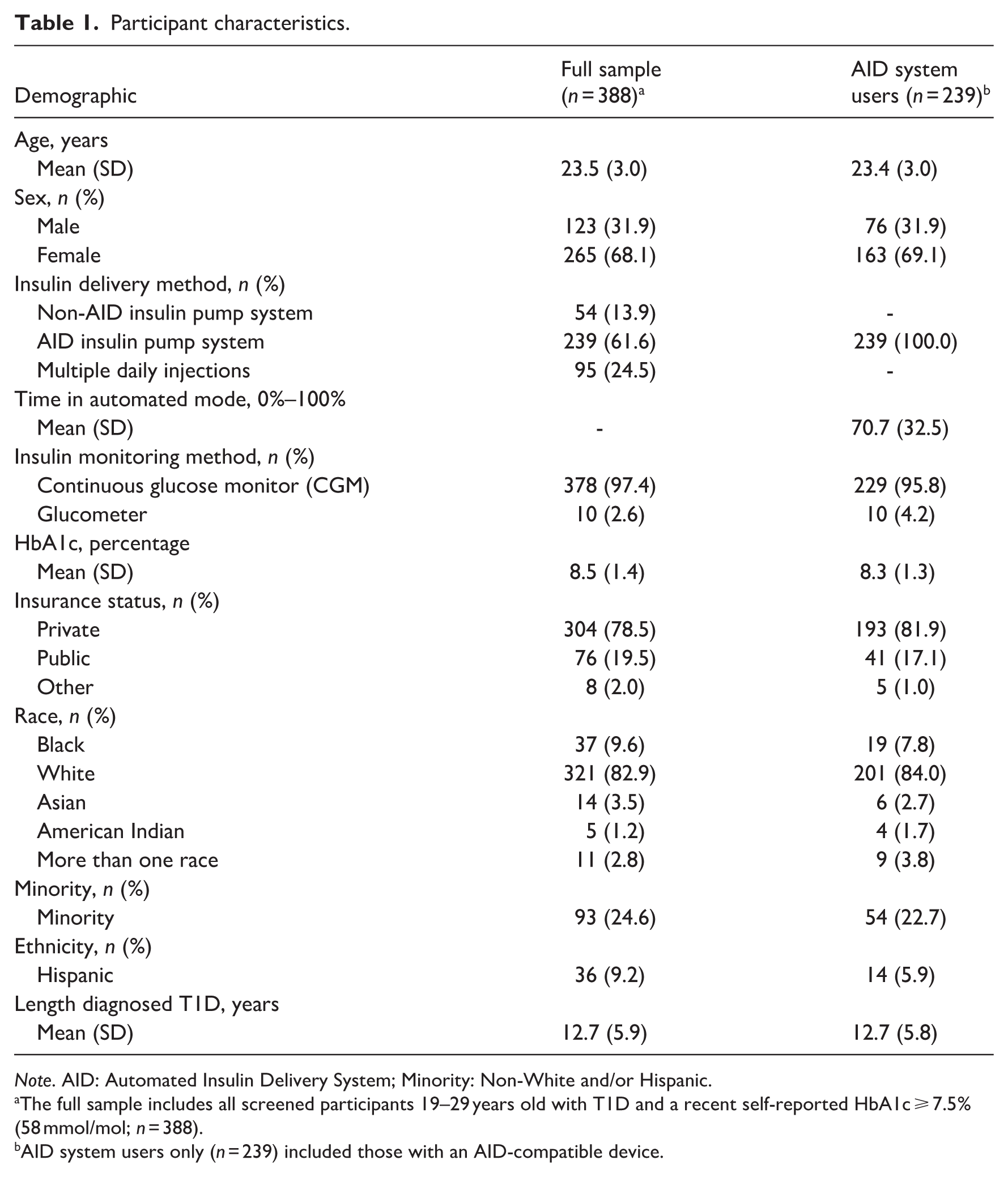
Note. AID: Automated Insulin Delivery System; Minority: Non-White and/or Hispanic.
The full sample includes all screened participants 19–29 years old with T1D and a recent self-reported HbA1c ≥ 7.5% (58 mmol/mol; n = 388).
AID system users only (n = 239) included those with an AID-compatible device.
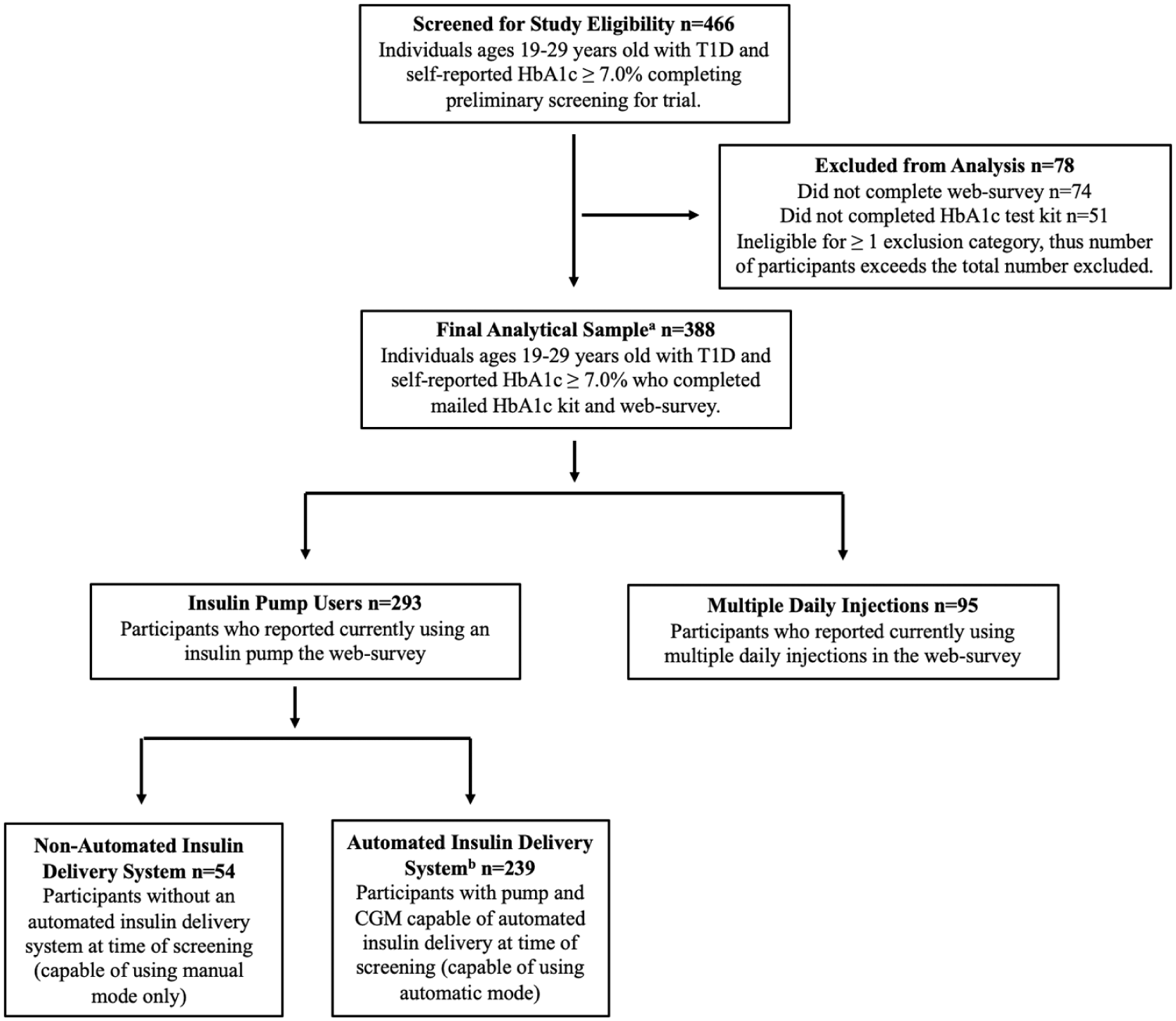
Participant flowchart.
Measures
Technology readiness
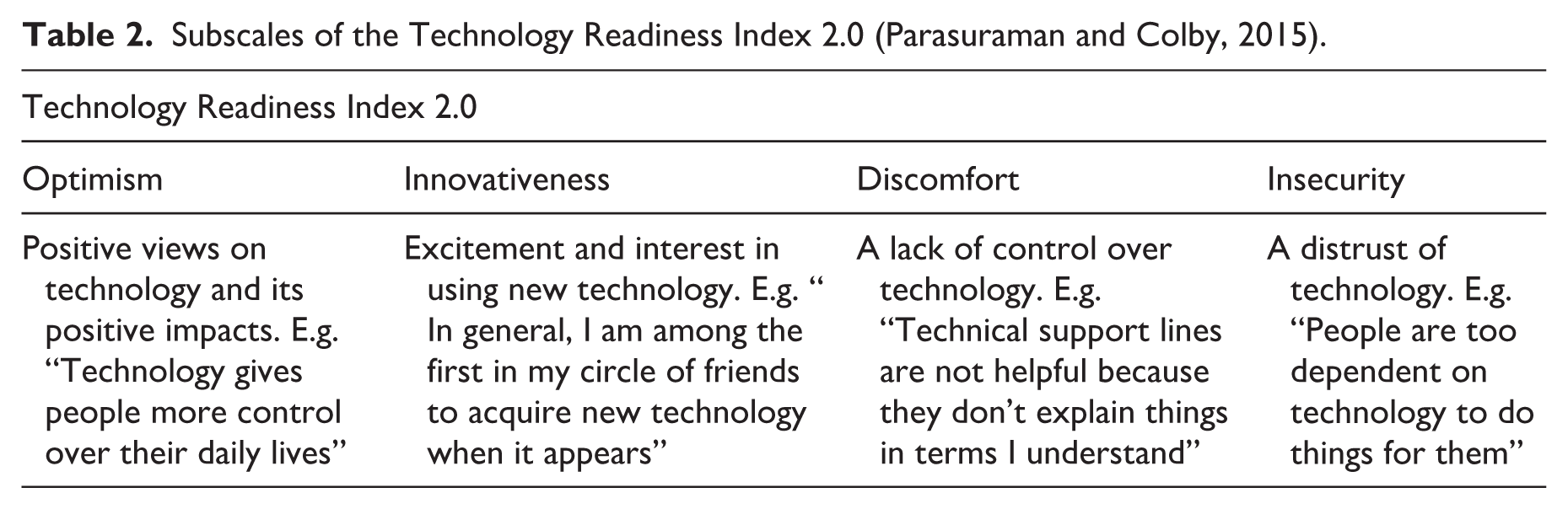
The Technology Readiness Index (TRI) 2.0 questionnaire measured perceived willingness and barriers to using and integrating new technology into daily lives (Parasuraman and Colby, 2015). TRI 2.0 has four different subscales: Optimism (positive views on technology and its impacts), Innovativeness (interest in using new technology), Discomfort (concerns about lack of control over technology), and Insecurity (distrust of technology; Table 2). TRI 2.0 subscale scores are the average of their subscale items. Participants scored each item on a 1 (“strongly disagree”) to 5 (“strongly agree”) point Likert scale, and items were then averaged to calculate mean individual subscale scores. Internal consistency for the four TRI 2.0 subscales was assessed using Cronbach’s alpha (α). Reliability is as follows: Optimism = 0.80, Innovativeness = 0.81, Insecurity = 0.71, and Discomfort = 0.67. These values are considered to be acceptable to good internal consistency across TRI subscales (Taber, 2018).
Subscales of the Technology Readiness Index 2.0 (Parasuraman and Colby, 2015).
Insulin delivery method
During the Glooko upload process, participant insulin delivery method was coded as either pump (n = 293, 75%) or multiple daily injections (MDI). Among pump users, there were 239 participants (81.6%) with an AID-compatible system. For those AID users, we downloaded from Glooko the percentage of time the insulin pump was in automated mode in the 14 days before the trial screening date. There were 29 participants (12%) who had AID-compatible devices at screening but spent 0% of their time in AID mode (100% manual mode), while all other AID users (n = 210, 88%) had ≥1% time in AID mode.
HbA1c blood value
HbA1c was objectively measured through a self-administered, mailed HbA1c blood test (AccuBase kit, DTI Laboratories), which has been used in other T1D studies (Grando et al., 2019). The values used for this study were HbA1c values obtained at the time of study registration.
Statistical analysis
Statistical analyses were conducted in STATA v.18.0 (StataCorp LLC., College Station, TX, USA). Multivariate logistic regression models examined relationships between mean overall TRI (Table 3) and TRI subscales (Table 4) on any insulin pump use (n = 388). Multivariate linear regression models examined the relations between TRI scores and time spent in AID mode (%) in the 14-day period prior to screening (n = 239). All models controlled for current HbA1c, age, insurance status (private insurance vs other), sex (female vs other), time since T1D diagnosis in years, and minority status (white, non-Hispanic vs other).
Technology readiness total score associations with insulin pump use, AID system use, and HbA1c in young adults with type 1 diabetes.
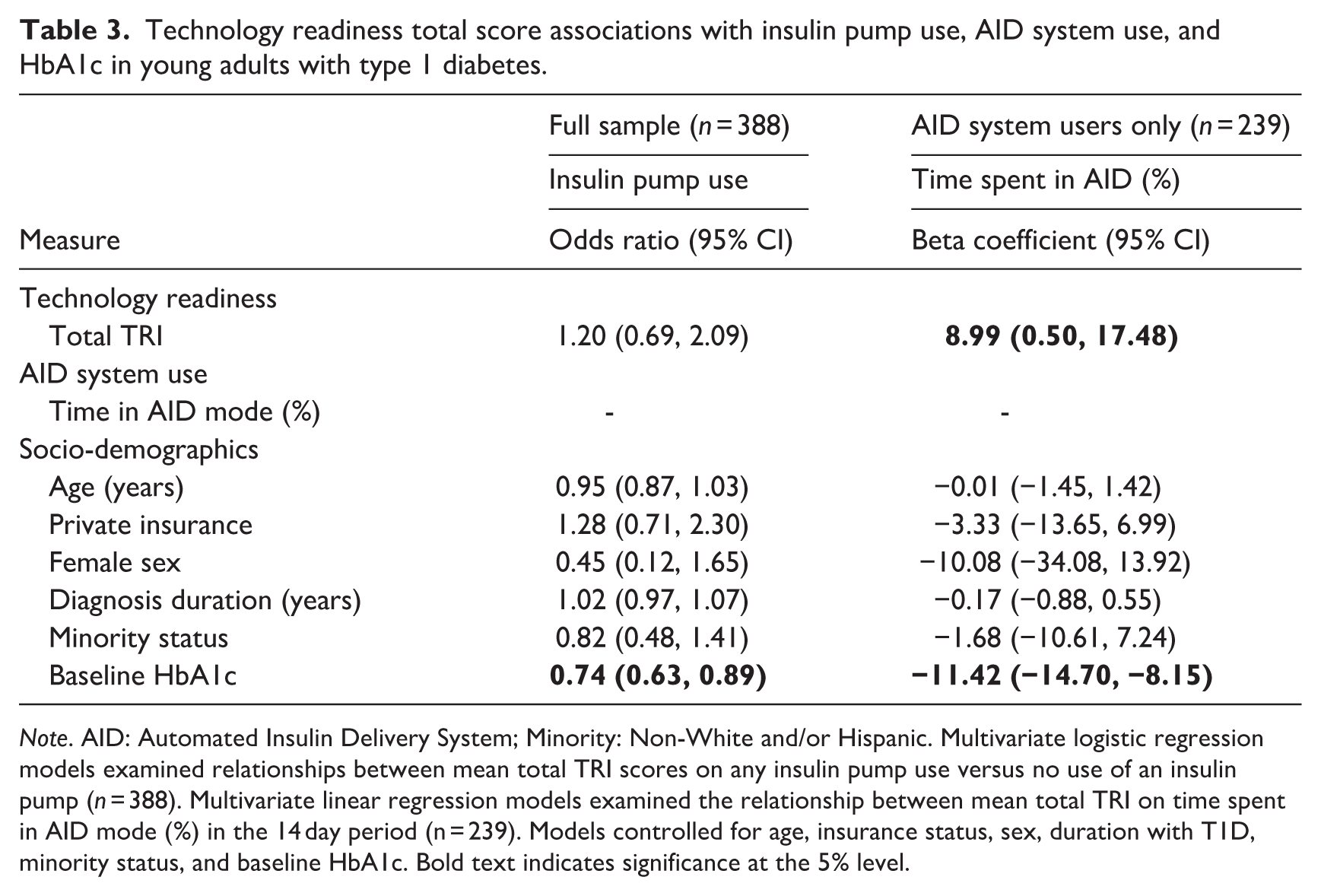
Note. AID: Automated Insulin Delivery System; Minority: Non-White and/or Hispanic. Multivariate logistic regression models examined relationships between mean total TRI scores on any insulin pump use versus no use of an insulin pump (n = 388). Multivariate linear regression models examined the relationship between mean total TRI on time spent in AID mode (%) in the 14 day period (n = 239). Models controlled for age, insurance status, sex, duration with T1D, minority status, and baseline HbA1c. Bold text indicates significance at the 5% level.
Technology readiness subscore associations with insulin pump use, AID system use, and HbA1c in young adults with type 1 diabetes.
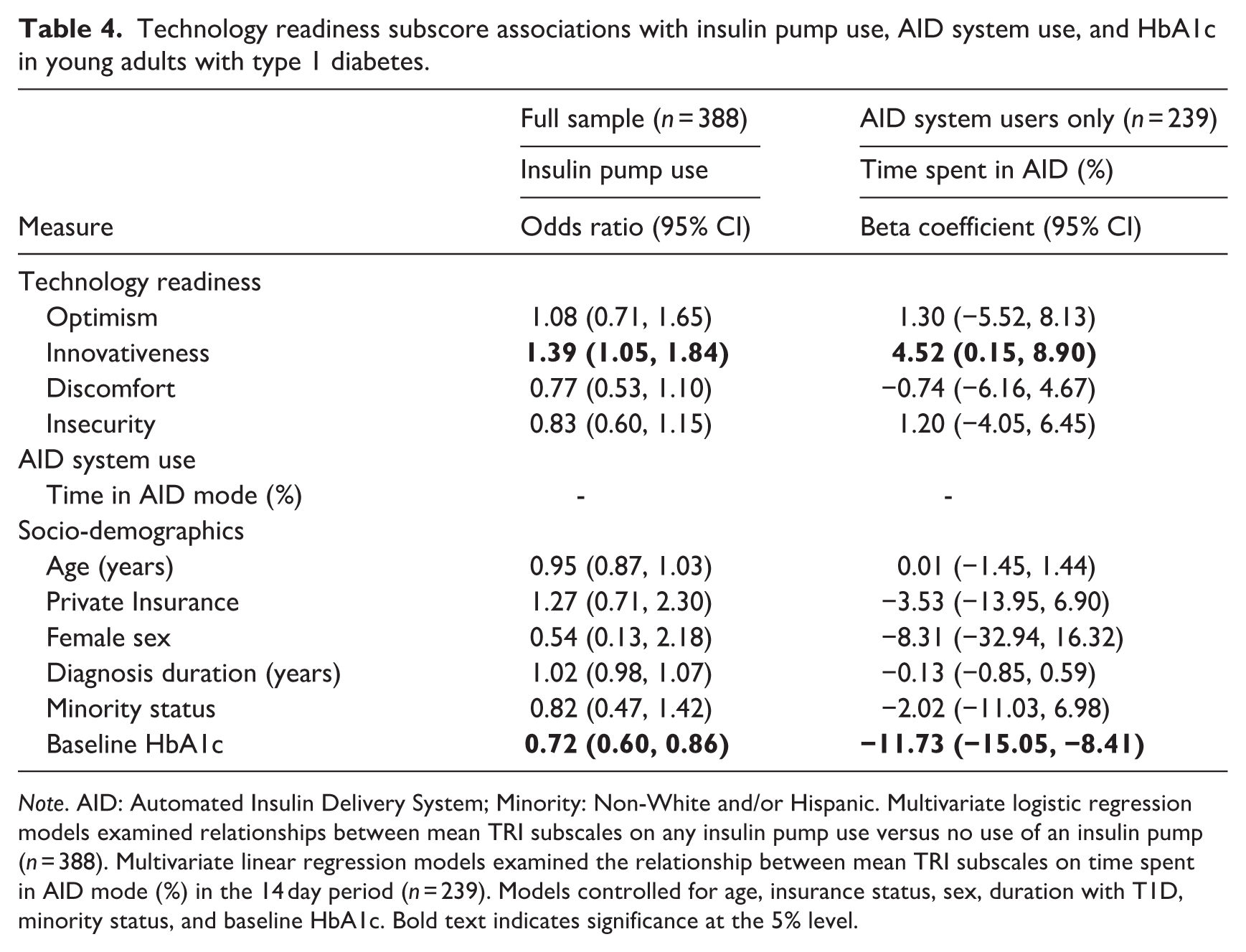
Note. AID: Automated Insulin Delivery System; Minority: Non-White and/or Hispanic. Multivariate logistic regression models examined relationships between mean TRI subscales on any insulin pump use versus no use of an insulin pump (n = 388). Multivariate linear regression models examined the relationship between mean TRI subscales on time spent in AID mode (%) in the 14 day period (n = 239). Models controlled for age, insurance status, sex, duration with T1D, minority status, and baseline HbA1c. Bold text indicates significance at the 5% level.
Results
Sample Demographics
The overall cohort included 388 young adults with T1D and a recent self-reported HbA1c ≥ 7.5% (Table 1). The mean age was 23.5 years (SD = 3.0 years), and the average duration of T1D was 12.7 years (SD = 5.9 years, Range = 2–29 years). The average HbA1c levels were 8.4% (SD = 1.4%). The majority were female (68.1%), White race (82.9%), and 24.6% self-identified as a minority. Most participants had private insurance (78.5%) and primarily used a continuous glucose monitor (97.4%). The majority of participants used an AID insulin pump system (61.6%), with the remainder using a non-AID insulin pump system (13.9%) or multiple daily injections (24.5%). These proportionate values for demographics were also very similar among the AID system user sample (n = 239).
TRI measure analysis
To assess potential multicollinearity among TRI subscales and covariates, we examined Variance Inflation Factors (VIFs). All VIF values for the subscales were below 2.5 (Mean VIF = 1.20), indicating no concern for multicollinearity (Kim, 2019).
Predictors of insulin pump versus MDI
The first multivariate regression model explored overall technology readiness as a predictor of insulin pump usage versus MDI usage. Overall, technology readiness was not significantly associated with an increased likelihood of using an insulin pump (Table 3). However, higher baseline HbA1c was significantly associated with a lower chance of using an insulin pump over MDI [OR = 0.74, 95% CI (0.63, 0.89)]. In the second multivariate model, the four TRI subscales were tested as predictors of insulin pump use. A 1-point increase on the innovativeness subscale (range 1–5) was significantly associated with a 39% increased likelihood of using an insulin pump over multiple daily injections [OR = 1.39, 95% CI (1.05, 1.84)] (Table 4). The other subscale scores were not significantly associated with insulin pump use. In this model, a 1% higher baseline HbA1c was also significantly associated with a 28% decreased chance of using an insulin pump over MDI [OR = 0.72, 95% CI (0.60, 0.86)].
Predictors of AID automated mode usage versus manual mode usage
The first multivariate regression model explored overall technology readiness as a predictor of time in automatic mode (%). Among participants with an AID-compatible system, a 1-point increase in total technology readiness was significantly associated with a 9.0 percentage point higher time in AID mode [β = 8.99 (0.50, 17.48)] (Table 3). Additionally, 1% lower baseline HbA1c was significantly associated with an 11.42 percentage point increase in time spent in automatic mode [β = −11.42 (−14.70, −8.15)]. In the second multivariate model, the four TRI subscales were tested as predictors of time in automatic mode (%). A 1-point increase on the innovativeness subscale was significantly associated with higher time in AID mode (Table 4). In this model as well, lower baseline HbA1c was significantly associated with higher time spent in automatic mode.
Discussion
To our knowledge, this was the first study to examine how perceived technology readiness related to AID use and HbA1c in a large, national sample of young adults with T1D. Our findings suggest that more positive attitudes toward technology innovation, characterized by early technology adoption and finding new technology stimulating were associated with a higher likelihood of currently using an insulin pump over multiple daily injections (Table 4; Parasuraman and Colby, 2015). A 1-point increase in the innovativeness subscale was correlated with a 39% higher likelihood of using an insulin pump over multiple daily injections. This relationship is both statistically and clinically significant, as insulin pump use is associated with important clinical outcomes such as a 32% lower rate of severe hypoglycemia per 100 patient-years and 80–90 additional minutes per day in time-in-range (Ekhlaspour et al., 2023; Karges et al., 2017). These large effect sizes suggest that individuals with lower technology innovation might benefit from targeted encouragement and education to improve outcomes, while minimizing stress and discomfort (Kuo et al., 2013).
We also found that overall technology readiness was related to spending significantly more time in AID mode. Although statistically significant, the effect size (~9% increase) is small and may not translate into clinically meaningful improvements. Prior work shows that simply using AID mode at all, compared to never, is associated with only a 4%–5% reduction in time out of range, indicating that increases in AID use may have limited clinical impact (Renard et al., 2023). Therefore, while technology readiness may meaningfully contribute to AID engagement, it is unlikely to be the primary driver of clinical outcomes.
Prior studies examining technology readiness scores and health technology use is scarce. However, a prior study in a general sample of adults found that higher TRI innovation was associated with higher perceived likelihood to use “digital pills” for future healthcare management (Elvis and Kim, 2022). These digital pills, which monitor and report health data, show similar relationships between innovativeness and device adoption as seen with insulin pumps and AID systems. Across different medical technologies, higher innovativeness appears linked to increased willingness to use devices that support chronic disease management (Elvis and Kim, 2022).
One limitation of this study is that these results may not generalize to all young adults with T1D, as the sample included young adults with above-target HbA1c who were interested in a digital intervention and might have been more technologically inclined. Another limitation of this cross-sectional study is that the true direction of the relationships between technology readiness, HbA1c, and insulin pump use is unknown and may reflect a reciprocal relationship or unmeasured variables. Since all members of this study have already adopted insulin pumps, longitudinal research is needed to examine whether higher technology readiness predicts future adoption of an insulin pump and/or time in automated mode. Additionally, AID engagement was measured over 14 days, which may not reflect typical use given fluctuations due to illness, travel, or routine variability.
Future studies should examine other factors influencing relations between technology readiness, insulin pump use, and use of automated insulin delivery systems that go beyond the sociodemographic variables examined in this analysis. These could include the influence of medical providers’ attitudes toward using an insulin pump or an AID system, including their knowledge and experience with these systems (Rimon et al., 2024). This could also include other psychological variables such as depressive symptoms, anxiety, or self-efficacy that might impact technology adoption for T1D patients (Franklin, 2016).
Conclusions
Overall, this cross-sectional study found that higher technology innovation scores were statistically significantly associated with a greater likelihood of insulin pump use and increased time spent in AID mode among young adults with T1D and above-target HbA1c. These constructs of technology readiness, including early technology adoption, finding new technology stimulating, and independently figuring out how to use new technology, could be used in future clinical practice to guide patients with T1D in better adopting new T1D technology. In particular, interventions can be developed and tested to promote positive attitudes toward and effective use of technology to manage T1D. Increased adoption and effective use of these novel technologies would likely also result in improved HbA1c levels in this vulnerable group.
Footnotes
Acknowledgements
We would like to acknowledge Devan Jones and Miranda Signorelli for assistance with data collection and Scott Gerlach for assistance processing the data.
Ethical considerations
This study was approved by the Dartmouth College Committee for Protection of Human Subjects (IRB #23559) and all procedures were performed in accordance with the ethical standards provided.
Consent to participate
All participants provided written consent to participate in the study through the SweetGoals app.
Consent for publication
Consent for publication is not applicable to this article as it does not contain any identifiable data.
Author contributions
Harshith Yallampalli: Conceptualization, Methodology, Software, Analysis, Investigation, Writing - Original Draft, Writing - Review & Editing, Visualization. Enzo G. Plaitano: Conceptualization, Methodology, Writing - Original Draft, Writing - Review & Editing, Supervision, Funding acquisition. Catherine Stanger: Supervision, Methodology, Investigation, Project administration, Funding acquisition, Writing - Review & Editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Diabetes and Digestive and Kidney Diseases (R01DK124428), a National Institute on Drug Abuse center grant (P30DA029926), a National Institute on Drug Abuse training grant (T32DA037202) and research assistance stipend support from the Undergraduate Advising and Research Program at Dartmouth College.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the present findings are available from the corresponding author upon reasonable request.
