Abstract
With a global annual cumulative incidence of depression at 4.5% in community-dwelling older adults, understanding non-pharmacological interventions is essential. This narrative review explores the neuroprotective mechanisms of physical activity (PA) on brain function and mental health in individuals aged 60 and older. We conducted a search across multiple databases (MEDLINE, PsycINFO, EMBASE) using keywords related to aging, cognition, and physical activity. Our analysis of relevant studies shows that PA benefits the brain through several pathways. Early findings focused on improved cerebral blood flow and glucose utilization. More recent evidence highlights that PA increases neurotrophic factors like BDNF and IGF-1, enhances mood-regulating neurotransmitters, and promotes structural adaptations in key brain regions. These findings suggest that PA is a cost-effective, multi-domain intervention. This review provides healthcare professionals with actionable evidence to incorporate PA into clinical practice for older adults.
Introduction
Physical activity (PA) is a simple, accessible, and cost-effective intervention with significant benefits, particularly for the aging population as a non-pharmacological treatment. However, more than 50% of Canadian adults fail to meet the recommended weekly PA guidelines (Colley et al., 2023; Thornton et al., 2016; Tremblay et al., 2011). PA has long been suggested to provide a range of health benefits beyond enhancing cardiovascular capacity, including improvements in mental health symptoms (Gujral et al., 2017; Kandola et al., 2019) and cognition (Chapman et al., 2013). A well-established mechanism of PA involves increasing heart rate, thus enhancing cerebral blood flow and augmenting neural metabolism (Chapman et al., 2013; Murrell et al., 2013). In addition, PA significantly reduces cardiovascular risk factors such as diabetes, hyperlipidemia, and hypertension, further contributing to the prevention of cerebrovascular disease (Aune et al., 2015; Baker et al., 2010; Carvalho and Cardoso, 2021; Kanaley et al., 2022). PA is thought to initiate a cascade of neuroprotective mechanisms essential for promoting and maintaining neuroplasticity and neuronal survival (Mahalakshmi et al., 2020).
Given the growing body of evidence on the neurobiology of psychiatric diseases, research on PA in psychiatric comorbidities highlights its increasing therapeutic potential. A recent network meta-analysis of randomized controlled trials demonstrated that PA is an effective intervention for depression, showing comparable benefits regardless of comorbid conditions or initial depression severity (Noetel et al., 2024). Globally, the annual cumulative incidence of depression among community-dwelling adults aged 60 years and older is estimated at 4.5% (Brasileiro et al., 2024). The most recent available estimate of life-time prevalence of depression in Canada is approximately 11.3% in the adult general population (Patten et al., 2015). As no newer national data are available, the most recent estimate of depression prevalence among older adults (⩾65 years) remains 6.6% (Østbye et al., 2005). Depression is a leading cause of disability, imposing a substantial economic burden, estimated at over $12 billion annually on direct healthcare costs in Canada (Tanner et al., 2020). The high prevalence of depression and cognitive decline leads to significant psychosocial burden and high healthcare costs necessitating preventative strategies (2021; Kessler, 2012). Multiple international guidelines suggest that older adults who meet the recommended physical activity thresholds experience a 20% reduction in the risk of cognitive decline and dementia (Bull et al., 2020; Piercy et al., 2018; Tremblay et al., 2011). A meta-analysis of prospective interventional studies showed that participants who engaged in high levels of physical activity had significant protection against cognitive decline during follow-up (HR: 0.62, 95% CI: 0.54–0.70; p < 0.00001). Moreover, even low-to-moderate levels of exercise provided significant protection against cognitive impairment (HR: 0.65, 95% CI: 0.57–0.75; p < 0.00001; Sofi et al., 2011). Postponing cognitive decline by 5 years yields significant economic savings (2021) and extends valuable time of preserved cognition and quality of life for patients and their families. Therefore, in this narrative review, we synthesize evidence on the biopsychosocial benefits of PA for brain and mental health, emphasizing its neuroprotective mechanisms, non-pharmacological treatment potential, and psychological and social advantages.
Methods
Defining physical activity
Research on PA frequently uses the terms “physical activity” and “exercise” interchangeably. To clarify, we define PA as any movement that engages muscles and expends energy, irrespective of intensity or specific intent. PA includes daily activities with low or minimal effort, such as standing, sitting, and driving (Bull et al., 2020; Thornton et al., 2016). Exercise is a specific type of PA, which is structured and intended to improve health or fitness. Exercise is typically categorized by intensity levels: low, moderate, and high (Bull et al., 2020).
Multiple factors were integrated to establish specific PA health recommendations (Garber et al., 2011; Piercy et al., 2018; Thornton et al., 2016; Tremblay et al., 2011). The “dose” of PA required to achieve health benefits includes four components: (1) type of activity, (2) level of intensity, (3) duration per session, and (4) frequency of sessions per week. PA health recommendations typically follow the guidelines established by the American College of Sports Medicine (ACSM) and the World Health Organization (WHO): aerobic type PA at moderate-vigorous intensity, for 30–60 minutes per day, five times per week, for a total of 150–300 minutes per week, plus 2 additional weekly sessions of anaerobic (resistance type) PA. This dose of PA is recommended for adults aged 18–65 years, and for older adults aged 65 years and over, with an additional recommendation of balance exercise to the latter (Bull et al., 2020; Garber et al., 2011; Medicine and Science in Sports and Exercise, 1998).
Literature search
We conducted a comprehensive search of MEDLINE, MEDLINE RCTs, PsycINFO, and EMBASE from January 2000 to July 2025, using keywords such as physical activity or exercise combined with cognition, cognitive decline, dementia, neuroplasticity, neuroanatomy, brain health, depression, mood, anxiety, aging, older adults, late-life, or older adults. Representative search strings combined MeSH and free-text terms using Boolean operators (e.g. “physical activity” AND “cognition” OR “neuroplasticity”). Additionally, we included terms related to an active lifestyle to capture broader aspects of PA impact on brain function. A manual search of reference lists from relevant studies was also performed to identify supplementary sources. We included peer-reviewed original studies, meta-analyses, and reviews examining associations between PA and brain structure, function, or cognition in adults aged ⩾ 60 years. Studies limited to populations with severe neurological disorders were excluded. Titles and abstracts were screened for relevance, followed by full-text evaluation of eligible articles. Approximately 420 records were identified, with roughly 300 screened after deduplication, and about 100 meeting inclusion criteria for synthesis. This process was guided by the principles of the Scale for the Assessment of Narrative Review Articles (SANRA; Baethge et al., 2019), to ensure transparency, methodological rigor, and reproducibility in narrative synthesis. Eligible publications were reviewed and analyzed for their contributions to understanding the effects of PA on brain health. Ethics approval was not required for this narrative literature review.
Results
Physical activity and neural mechanisms
Multiple neurophysiological mechanisms by which PA impacts the brain have been identified. Increased heart rate and cardiovascular effort have been suggested to augment peripheral blood flow, leading to a subsequent increase in cerebral blood flow (Chapman et al., 2013; Murrell et al., 2013). This enhanced perfusion promotes cerebral metabolism by improving nutrient distribution, waste removal, oxygen extraction and glucose utilization (Churchill et al., 2002; Lojovich, 2010).
Upregulation of neurotrophic factors has been shown to support neuronal survival, enhance synaptic plasticity, promote neurogenesis, and facilitate structural and functional brain adaptations (Alfonsetti et al., 2023). PA promotes the secretion of brain-derived neurotrophic factor (BDNF), a key mediator of structural brain changes, neuronal development, synaptogenesis, dendritic branching, and neuroprotection (Cotman et al., 2007; Cotman and Berchtold, 2002; Ferris et al., 2007). BDNF is also a primary factor impacting secretion of additional neurotrophic factors, playing a key role in attention, memory, and mood regulation (Erickson et al., 2012; Maass et al., 2016). Recent randomized controlled trials (RCTs) demonstrate increased levels of serum and plasma BDNF in response to exercise interventions (Leckie et al., 2014; Vaughan et al., 2014). However, other RCTs found no change in serum or plasma BDNF levels following exercise interventions in the oldest-old population (⩾80 years old; Hvid et al., 2017; Kim et al., 2015; Ruiz et al., 2015).
Another neurotrophic factor enhanced by PA is the insulin-like growth factor-1 (IGF-1) which supports brain metabolism, neuronal transmission, and neuroplasticity (Dyer et al., 2016; Ferris et al., 2007). Resistance exercise training has been shown to increase in serum IGF-1 levels in several clinical trials (Cassilhas et al., 2007; Miranda et al., 2009; Tsai et al., 2015). However, some studies suggest that older age may affect the responsiveness of serum IGF-1 levels. For example, an RCT by Hofmann et al (Hofmann et al., 2016) exploring resistance-exercise training effect on serum IGF-1 levels in individuals (mean age: 83 years) showed no significant changes. These studies suggest reduced effectiveness of exercise in enhancing BDNF and IGF-1 levels with advanced age, possibly due to a reduced capacity at older age (>80 years) to reach the required exercise intensity for the secretion of these neurotrophic factors. Evidence suggests a dose-response relationship between PA and cognitive benefits, with specific intensity and duration threshold required to maximize neuroprotective effects (Vidoni et al., 2015). In older adults with cognitive impairment, exercise programs featuring shorter session duration and higher frequency may provide optimal cognitive benefits (Sanders et al., 2019). In terms of exercise modalities, while aerobic exercise is the most extensively studied, resistance training also plays a role in modulating neurotrophic factors and supporting cognitive functions (Azevedo et al., 2023).
Physical activity and neuroanatomical changes
Neuro-anatomical changes associated with PA have been demonstrated in multiple imaging studies (reviewed by Dishman et al., 2006; Kramer et al., 2006). Thomas et al., showed increased metabolism and volume of the hippocampus in sedentary adults following a 6-week aerobic training regimen (30 minutes/day, 5 days/week; Thomas et al., 2016). The enlarged anterior hippocampal volume has been attributed to improved myelination, possibly mediated by neurotrophic factors, such as BDNF and IGF-1 (Dishman et al., 2006; Erickson et al., 2011; Thomas et al., 2016). Niemann et al., implemented a coordination-based exercise program and reported an increase in basal ganglia volume, a neuroanatomical structure involved in motor control and cognitive processes (Niemann et al., 2014). In addition, preclinical as well as neuroimaging studies in humans have shown changes to various brain structures, such as increase in white matter and gray matter volume (Matura et al., 2017; Sexton et al., 2016), reduction in white matter hyperintensities (Vesperman et al., 2018), and increased cortical thickness and hippocampal volume following an exercise program (Cotman et al., 2007; Firth et al., 2018; Reiter et al., 2015).
Prospective research is underway to investigate how aerobic exercise induces structural and functional changes in cortico-hippocampal connectivity. One example, the Brain-EXTEND trial, utilizes advanced neuroimaging and molecular analyses to examine exercise-induced neuroplasticity, focusing on both immediate and long-term adaptations in cortico-hippocampal networks. By integrating physiological, genetic, and molecular markers, the study aims to uncover the mechanisms through which aerobic exercise enhances memory and potentially mitigates Alzheimer’s disease risk (Voss et al., 2024).
Physical activity for depressive and anxiety symptoms
PA has been shown to be as effective as antidepressant medications or behavioral therapy in the treatment of depression (Gujral et al., 2017; Kandola et al., 2019), and is part of the standard treatment recommendations for mild-moderate depression (Davidson, 2010). Importantly, PA lacks common adverse effects associated with psychotropic medications (e.g. apathy, weight-gain and sexual dysfunction) that frequently interfere with adherence to treatment. Moreover, some side-effects of psychotropic medications, such as sedation, may increase the risk for falls in older adults. Pharmaceutical treatment for depression must also consider potential negative outcomes of drug-interactions and drug metabolism (Kok and Reynolds, 2017). Therefore, PA may serve as a non-pharmacological therapeutic approach for older adults when evaluating treatment options (Kok and Reynolds, 2017).
PA has been proved beneficial in reducing anxiety symptomatology and is part of the standard treatment for mild-to-moderate generalized anxiety (Gordon et al., 2017). PA enhances release of serotonin, norepinephrine, and dopamine, neurotransmitters involved in mood regulation. These neurotransmitters are the primary targets of many antidepressant and anxiolytic medications, which are postulated to mediate their effects through these pathways (Blier, 2016; Gordon et al., 2017; Kandola et al., 2019; Kraus et al., 2017; Xie et al., 2021). PA reduces stress by modulating the hypothalamic-pituitary-adrenal (HPA) axis, influencing cortisol secretion to regulate stress responses. Recent studies suggest that PA facilitates HPA axis homeostasis, optimizing its effects on the hippocampus, amygdala, and prefrontal cortex, which in turn enhances attention, learning, and memory while mitigating stress (Drogos et al., 2019; Duclos and Tabarin, 2016; Heaney et al., 2014).
Vascular risk factors and cognitive decline
Modifiable vascular risk factors such as hypertension, diabetes, hyperlipidemia, smoking and physical inactivity are strongly associated with cognitive decline, vascular cognitive impairment (VCI) and vascular dementia (Graff-Radford, 2019). Non-modifiable risk factors, such as genetic predisposition are also considered to play a role.
PA is well regarded as an effective strategy to improve multiple modifiable vascular risk factors, beyond physical inactivity. PA has been shown to regulate blood pressure, reduce hyperlipidemia, and mitigate the risk of cerebrovascular diseases (Carvalho and Cardoso, 2021). In addition, PA has been shown in multiple studies to improve glucose metabolism and prevent insulin resistance, and is considered a first line recommendation in the treatment of type 2 diabetes (Aune et al., 2015; Kanaley et al., 2022). Moreover, insulin resistance has been associated with oxidative stress and neuroinflammation and therefore carries additional risk for cerebrovascular disease and cognitive decline (Carvalho and Cardoso, 2021).
PA has been suggested as a useful method in prevention and management of cerebrovascular risk factors and in reducing risk for VCI (Stephen et al., 2020; Zhao and Xu, 2021). An RCT by Liu-Ambrose et al., evaluated cognitive function among older adults with VCI following a 6-month aerobic exercise program with increasing intensity compared with a control group who received usual care and education (Liu-Ambrose et al., 2016). Findings from this study showed significant improvements in cognitive scores among participants in the aerobic intervention group compared with controls. Dougherty et al., demonstrated improved glucose metabolism and neurovascular function among middle-older age adults following a physical activity program (Dougherty et al., 2017). Robinson et al., showed improved brain glucose metabolism following a 12-week high-intensity exercise intervention in adults (Robinson et al., 2018). In addition, Fisher et al., demonstrated increased cerebral perfusion and cerebral oxygenation during exercising in various intensities (Fisher et al., 2013).
Physical activity and cognitive function
In recent years there has been growing interest in studying the potential of PA in preserving cognition, and particularly as a protective factor against cognitive decline and development of dementia. Recent studies show a positive association between PA and improved cognitive functions among older adults (Blumenthal et al., 2020; Buchman et al., 2019). A recently published longitudinal population study (Najar et al., 2019) examined PA levels, cognitive and neuropsychiatric assessments among 899 women over 44 years and showed that continuous PA in mid-life was associated with reduced risk for dementia in later-life and a potentially protective effect on cognition. In addition, Buchman et al. (2019) demonstrated an association between physical activity and motor abilities with improved cognition and reduced risk for all-cause dementia. A meta-analysis by Colcombe and Kramer, included 18 RCTs which explored the impact of various PA programs on cognitive functions in sedentary older adults without dementia (Colcombe and Kramer, 2003). Results suggested that higher fitness levels were consistently associated with the greatest improvements in executive functions. Pentikäinen et al. and the Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability study group (Pentikäinen et al., 2019), reported an association between the cardio-respiratory fitness and improvement of executive functions, processing speed, and overall cognitive function, in a 2-year follow up study of older adults. The Finland, Italy, and the Netherlands Elderly (FINE) study group suggested that maintaining daily PA of mild-moderate intensity may postpone cognitive decline (van Gelder et al., 2004). In addition, Stern et al. showed improved cognitive functions in adults who participated in an aerobic exercise program 4 days a week compared with controls (Stern et al., 2019). Specifically, executive functions showed the most improvement following the aerobic exercise intervention among older age. Laurin et al., presented more than 40% reduced risk for cognitive impairment and Alzheimer’s disease in older adults who completed three or more walks per week (Laurin et al., 2001). Additionally, a review of observational studies with minimum follow-up of 5 years showed a positive association between being physically active and a lower risk for cognitive impairment, cognitive decline, and dementia, compared with sedentary persons (Rockwood and Middleton, 2007). A clinical trial with 1740 older adult participants demonstrated a direct association between regular aerobic exercise and a reduced risk for Alzheimer’s disease (Larson et al., 2006). In older adults with MCI at risk for Alzheimer’s disease, a randomized trial demonstrated cognitive improvements following an exercise program (Lautenschlager et al., 2008). Moreover, a year-long PA program in sedentary adults with MCI revealed improved cerebral vasomotor reactivity and cognitive function (Tomoto et al., 2021).
Furthermore, cognitive impairment and depressive symptoms often overlap (Starkstein et al., 2005), imposing a diagnostic challenge. A subtype of cognitive decline presented mostly by impairment of executive function may resemble the cognitive profile of depression (Shimada et al., 2014), while late-life depression commonly presents as cognitive decline, mostly of impaired attention and executive functions. However, given the benefits of PA both on depressive symptoms and cognition, engagement in PA could potentially improve both entities regardless of the underlying diagnosis.
Psychological, social, and non-pharmacological benefits of physical activity
PA facilitates a supportive and interactive social environment and reduces loneliness. PA instructor-led programs commonly provide positive reinforcement and focus on individuals’ abilities and strengths, thereby improving the participants’ self-esteem. Participants in a geriatric-psychiatry exercise program reported strong feelings of belonging, meaning, and improved self-esteem after participating in the 12-week program (Sivan et al., 2022). Continuous positive reinforcement provided by an instructor further improved mood and motivation and reduced anxiety symptoms. PA carries a positive perception of health and capabilities, which may further enhance the individual’s self-esteem (McAuley et al., 2000; Opdenacker et al., 2009). As a non-pharmacological treatment modality, PA holds additional potential benefits in later-life, including a safer adverse effect profile, as changes in drug metabolism are common. PA is a low-cost treatment option, requiring no financial investment for activities like walking, which can be personalized in pace, duration, setting, and companionship. This flexibility underscores the feasibility of PA guidelines, as 30 minutes of daily leisure activity meets recommended health benefits (Bull et al., 2020).
Discussion
Physical inactivity is a modifiable and preventable risk factor for numerous health conditions (Strain et al., 2024). This narrative review integrates evidence on the diverse benefits of PA for brain health, spanning neurobiological, physiological, and psychosocial dimensions (Figure 1). These findings underscore the importance of incorporating PA health recommendations into clinical practice to support brain health and overall well-being. Moreover, PA contributes to overall health by improving sleep quality, thereby aiding memory consolidation and brain plasticity (Pickersgill et al., 2022). This cycle reinforces cognitive function and may protect against neurodegeneration, highlighting the importance of adopting a holistic approach to brain health. Effective implementation of the PA health guidelines is essential, as adherence to PA recommendations remains low, especially among older adults (Collado-Mateo et al., 2021; Lau et al., 2021). Healthcare providers can promote PA by offering updated PA health-recommendations and leveraging support programs such as Active Living Every Day (ALED; Baruth et al., 2011), designed to maintain routine PA. Most programs focus on behavioral change and motivational interviewing but often show only short-term success (Dixon-Ibarra et al., 2017; Glasgow et al., 2019; Guthold et al., 2018; Healthy Living Workshop | Active Aging Canada, n.d.). To sustain long-term engagement, clinicians should provide ongoing follow-up, prescribe individualized exercise routines, and regularly collaborate with recreational therapists and exercise professionals to effectively integrate PA into patient care. A limitation of this narrative review is the lack of a formal quality appraisal of included studies, which may influence the generalizability of the findings. Future meta-analytic evaluations could further refine and strengthen evidence-based clinical guidelines.
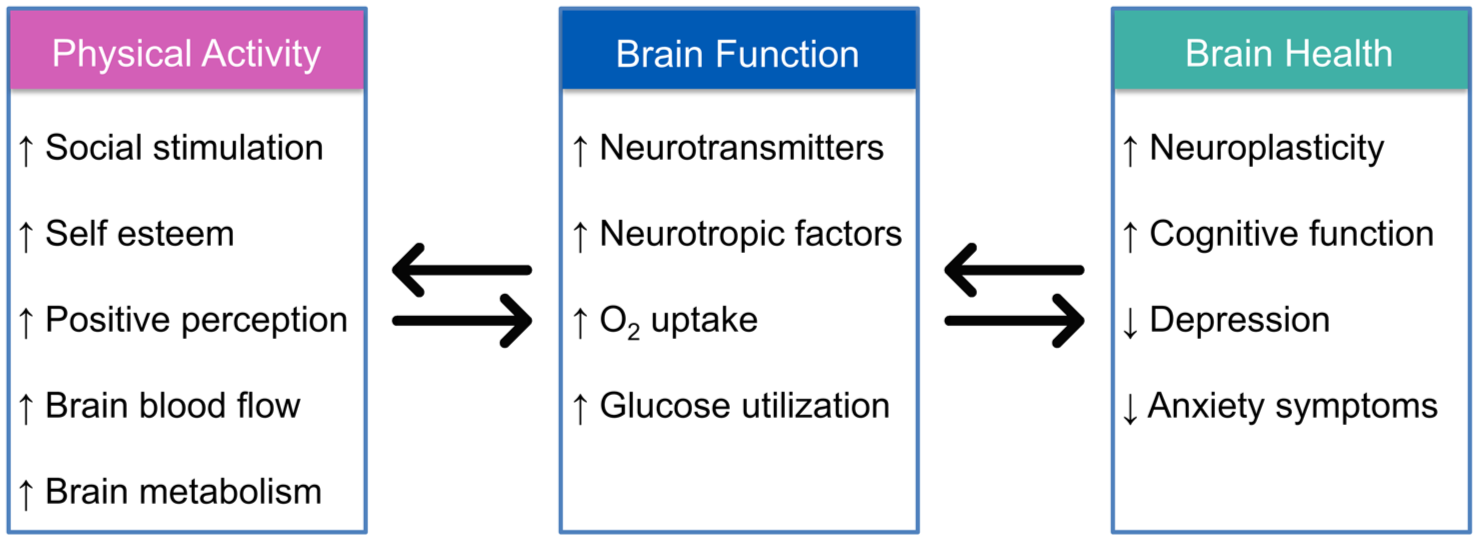
Bidirectional interactions between physical activity, brain function, and well being. Physical activity (PA) triggers a cascade of physiological and psychosocial processes that influence brain function and overall health. Increased cerebral blood flow and metabolism enhance neurotransmitter release, neurotrophic factor expression, oxygen uptake, and glucose utilization, supporting neuroplasticity and cognitive function. These mechanisms may serve as protective factors, mitigating depression and anxiety symptoms while promoting long term brain health. Additionally, the psychosocial benefits of PA, including improved self-esteem, social engagement, and positive self-perception, further contribute to well-being. The bidirectional interactions among these factors highlight the dynamic relationship between PA and neurological health.
Implications for practice
Prescribe safe and effective exercise interventions
A tailored PA regimen should be prescribed based on the participant’s capacity to carry out exercise routines, health status, training responses, and stated goals. For substantial health benefits, adults should engage in 150–300 minutes per week of moderate-intensity aerobic PA, or 75–150 minutes of vigorous-intensity aerobic PA, or an equivalent combination of both, typically achieved through 30–60 minute sessions on 5 days per week. Adults should also incorporate muscle-strengthening activities at moderate or greater intensity involving all major muscle groups on two or more days per week, with balance exercises particularly important for older adults. For novice to intermediate exercisers, an intensity of 60%–70% of the one-repetition maximal (1RM) effort is recommended to improve strength, while older adults beginning resistance training may start at a lower intensity (40%–50% of 1RM). Emphasize that PA accumulated through daily life, including activities at work, leisure, home, or during transportation, contributes to meeting these recommendations (Bull et al., 2020; Garber et al., 2011).
Integrate PA counseling in visits using motivational interviewing
Integrating PA counseling into clinical visits represents a critical opportunity for healthcare professionals, to play an important role in disseminating recommendations. Counseling becomes more effective when practitioners utilize valid behavioral change approaches, such as motivational interviewing, which is an effective method to raise the possibility of behavioral intervention. Given the time constraints of clinical practice, evidence suggests that clinicians should aim for a brief intervention, as a 2–4 minute intervention in primary care effectively promotes PA. This type of counseling is proven effective at improving PA levels for at least 12 months after the intervention and should incorporate an individual assessment of the patient’s needs, motivation, habits, preferences, and barriers. Programs focused primarily on behavioral change and motivational interviewing may have only short-term success and therefore, follow-up is essential to convey the clinician’s confidence, determine the patient’s progress, troubleshoot problems, fine-tune the PA dose, and revise goals. For planning and evaluating the success of these programs, particularly related to external validity and population impact, clinicians and researchers can use the RE-AIM planning and evaluation framework. This framework, which has been applied extensively in public health and health behavior change research, focuses on five dimensions: Reach, Effectiveness, Adoption, Implementation, and Maintenance (Glasgow et al., 2019; Thornton et al., 2016).
Collaborate with allied professionals for safe PA implementation
Safe and effective engagement in PA frequently requires coordinated input from allied health and community exercise professionals. Clinicians who do not routinely provide detailed exercise prescriptions may refer patients to physiotherapists, clinical exercise physiologists, kinesiologists, or certified fitness trainers for assessment and program development. According to recommendations from the ACSM, individualized exercise plans should be reviewed by a qualified health professional, particularly for adults managing chronic diseases or functional impairments. Involvement of trained exercise personnel can enhance initiation, progression, and adherence to prescribed activity, while ensuring appropriate monitoring of physiological responses and safety. Referral to a credentialed exercise specialist is especially warranted for patients with elevated medical risk, such as those with cardiovascular disease or type 2 diabetes. These professionals support optimal dose, progression, and adaptation based on the patient’s capabilities. Clinicians should also identify relevant community programs, including recreation centers and structured activity groups, to establish reliable referral pathways that promote long-term participation in PA (Garber et al., 2011; Thornton et al., 2016).
Monitor and adjust PA plans based on adherence, mood, and cognition regularly
Ongoing follow-up should include systematic evaluation of cognitive performance, with attention to domains such as executive function, memory, and language measures. Evidence from trials such as ENLIGHTEN indicates that individuals assigned to structured exercise interventions maintain superior executive functioning even 1 year after the intervention has ended, despite subsequent declines in their self-reported PA levels. Assessment should also encompass psychosocial well-being and overall Quality of Life (QoL). Individuals who engage in exercise programs routinely demonstrate higher QoL scores compared with those who remain inactive. Because pharmacologic treatments in older adults can lead to adverse effects including apathy, weight gain, and increased fall risk related to sedation, risks that are often amplified by age-related changes in drug metabolism, PA-based approaches may offer a more favorable safety profile (Blumenthal et al., 2020; Kok and Reynolds, 2017).
Connect participants with community programs to foster support and adherence
Recommend participation in structured community programs that support sustained engagement in physical activity outside the clinical setting. Programs such as ALED, adapted for senior centers and other community venues, have demonstrated meaningful improvements in physical functioning across varied older adult populations. ALED delivers a 20-week group-based curriculum that builds behavioral and problem-solving skills essential for initiating and maintaining regular activity, regardless of body mass index, race or ethnicity, or baseline functional capacity. Additional resources, including the Healthy Living Workshop and its Tool Kit, enable trained community leaders to deliver 6-week programs that facilitate lifestyle change. Participants use tools such as the Personal Passport to Healthy Living to monitor activities, document progress, and understand how daily habits influence chronic disease risk. Connecting patients with these programs offers continued support and accountability, enhances adherence to activity goals, and promotes long-term maintenance of PA (Baruth et al., 2011; Healthy Living Workshop | Active Aging Canada, n.d.).
Conclusion
This narrative review synthesizes evidence indicating that PA is a cost-effective, multi-domain intervention that may confer benefits for brain function and mental health in older adults through complementary biological, cognitive, and psychosocial pathways. First, PA appears to enhance cerebral perfusion and metabolic efficiency through increased blood flow, oxygen extraction, and glucose utilization. Second, PA has been associated with neuroplastic adaptations, in part through the upregulation of neurotrophic factors such as BDNF and IGF-1, which may support neuronal survival. These factors are thought to contribute to structural benefits, including increased hippocampal volume and the maintenance of white and gray matter integrity. Third, PA may serve as an effective non-pharmacological strategy for depressive and anxiety symptoms, with several studies reporting outcomes comparable with standard treatments. These effects are postulated to occur via modulation of mood-regulating neurotransmitters and regulation of the HPA axis. PA also offers a potentially safer adverse-effect profile than psychotropic medications, which can pose risks such as apathy, weight gain, sexual dysfunction, or sedation. Fourth, PA helps reduce modifiable vascular risk factors such as hypertension, diabetes, and hyperlipidemia. Improvements in glucose regulation and insulin sensitivity suggest that PA may mitigate risk for cerebrovascular disease and is often recommended as a first-line lifestyle intervention for type 2 diabetes. Finally, evidence from longitudinal and interventional studies suggests that regular PA is associated with slower cognitive decline. Older adults who meet recommended PA thresholds may experience a 20% reduction in the risk of cognitive decline and dementia, with executive functions showing the most consistent improvement following aerobic interventions. Collectively, these findings support PA as an evidence-informed approach that may promote healthy cognitive aging. The literature reviewed emphasizes the value of incorporating PA recommendations into routine clinical practice to help support late-life brain health and functional well-being.
Footnotes
Ethical considerations
As this manuscript is a narrative review of previously published literature, it did not require ethical approval.
Consent to participate
As this manuscript is a narrative review of previously published literature, consent to participate was not required.
Consent for publication
Consent for publication is not applicable to this review article as no identifiable participant data are included.
Author contributions
Conceptualization: SKD, MJ, YS; Data Curation: SKD, YS; Methodology: SKD, YS; Supervision: SKD, YS; Validation: SKD, MJ, YS; Visualization: SKD, YS; Writing – Original Draft: SKD; Writing – Review and Editing: SKD, MJ, YS.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SKD and YS report no conflicts of interest. MJ reports grants from the Northern Ontario Academic Medicine Association; honoraria for lectures from the Parry Sound Friendship Center; and is co-founder and co-owner of Blue Life Rx Community Corp. None of these activities influenced the design, analysis, or reporting of this manuscript.
Data availability statement
All data for this narrative review were obtained from publicly available, open-access sources and do not include any new, unpublished data.
