Abstract
Fatigue has been proposed as a potential factor influencing the perception of pain, which could be pertinent for enhancing chronic pain treatment. Exploring the relationship between pain and fatigue during a 3-week inpatient interdisciplinary multimodal pain therapy program, we evaluated children and adolescents (11–18 years) who reported significant chronic pain. Assessment tools included the Multidimensional Fatigue Inventory and other clinical questionnaires. The participants had higher fatigue scores than healthy subjects, general fatigue was found to be associated with the perception of pain in a random-effects model. The connection between fatigue and pain in adolescents with chronic pain suggests that the degree of fatigue and the changes in fatigue were associated with the intensity of their pain. The participants’ (N = 94) dimensions of fatigue and the intensity of their pain were reduced. We discuss implications for fatigue-reducing treatments in chronic pain management to reduce pain and augment pain management skills.
Introduction
Chronic pain is a growing health concern that profoundly affects individuals’ mental well-being, daily functioning, and occupational capacity. Chronic pain has been recognized as having a particularly detrimental impact on mental well-being during childhood and adolescence, often manifesting as mental stress, sleep disturbances, diminished school attendance, and reduced engagement in daily life (Haraldstad et al., 2011; Huguet and Miró, 2008; Hunfeld et al., 2001; Lambarth et al., 2023; Miró et al., 2023; Murray et al., 2020; Wager et al., 2020; Wrona et al., 2021). Despite its essential physiological role, pain can transform from being a protective mechanism to a dysfunctional and persistent condition. There is a consensus that a multidimensional therapeutic strategy grounded in a biopsychosocial model is effective for managing chronic pain disorders (Yazdani and Zeltzer, 2013). Our research focuses on the roles of fatigue and a multimodal treatment approach in addressing chronic pain (Fishbain et al., 2003; Navratilova et al., 2015).
Fatigue is one of the most common symptoms in patients experiencing chronic pain (Creavin et al., 2010). However, although fatigue plays a significant role in current pain research, its precise definition remains elusive, and it is often described phenomenologically. Van Damme et al. (2018) proposed characterizing fatigue as an aversive motivational state. They suggest that a cost–benefits analysis of goal-directed behavior determines whether fatigue or drive is generated (see Figure 1). This analysis is influenced by reward, (expected) pain, and effort. Chronic pain biases this trade-off toward fatigue by affecting all three components. Based on this cost-benefit analysis, pain expectations make it more difficult to maintain goal-directed behavior, which in combination with other motivational signals often leads to fatigue. Since chronic pain negatively affects all motivational signals, pain reduction should shift the balance away from fatigue (Van Damme et al., 2018). They further proposed that goal-persistent behavior may yield analgesic effects but at the expense of increased effort. Fatigue causes behavior that is directed toward a specific goal (e.g. going to school) to be more strenuous. The characteristic feeling of exhaustion that is associated with fatigue may lead to a worsened perception of pain. Conversely, a reduction in fatigue during a 3-week inpatient interdisciplinary multimodal pain therapy (IMPT) may lead to increased engagement in goal-directed activities and a reduced perception of pain.
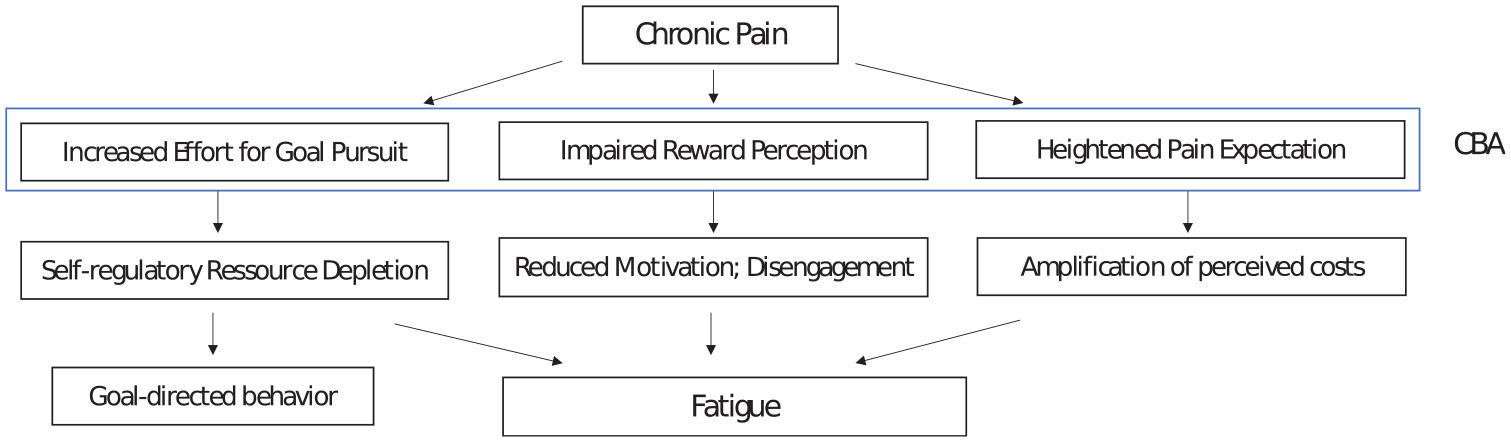
Chronic pain and its impact on fatigue. The cost-benefit analysis (CBA) involves increased effort for goal pursuit, impaired reward perception, and heightened pain expectations. Chronic pain shifts this balance toward fatigue by influencing all three components and makes it more demanding to sustain goaldirected behavior which, combined with other motivational signals, often leads to fatigue.
Building on Van Damme et al.’s fatigue model, we hypothesized that a reduction in overall pain experience following 3-weeks of IMPT therapy could lead to reduced fatigue: In the cost-benefit analysis, chronic pain hinders goal-oriented behavior and increases fatigue, leading to the hypothesis that reducing pain should also reduce fatigue. Hence, we investigated whether the duration of chronic pain had an impact on the level of fatigue that was experienced at the beginning of the IMPT, and whether pain intensity decreased by the end of the IMPT. Additionally, we analyzed whether the dimensions of the Multidimensional Fatigue Inventory (MFI-20) improved during the therapy and whether this correlated with a decrease in pain. We also compared whether the children and adolescents with chronic pain in our study had higher fatigue scores than the young people in the healthy normative group (16–24 years) for the MFI-20, as well as the 16–18 year subgroup of the healthy norm group (for comparability).
Methods
Setting
At the Center for Young People’s Pain Therapy, a specialized children’s hospital in Germany, approximately 500–600 patients with diverse pain syndromes receive inpatient treatment annually. The treatment regimen typically involves a 3-week inpatient IMPT approach, which has proven effective in addressing chronic pain among children and adolescents (Claus et al., 2022). The primary goal of this therapy is to help these individuals lead functional lives, even in the presence of persistent pain, rather than focusing solely on pain reduction. The treatment, which is tailored to each individual, includes medical, psychological support, physical therapies such as physiotherapy and massage, occupational therapy, social work and nursing services, along with an in-clinic schooling program (Hoefel et al., 2016). In the first phase of the treatment, both the patients and their parents are introduced to the biopsychosocial model of pain. This education focuses on the psychological and physical factors that contribute to the development of acute and chronic pain. It is important that the patients understand that despite many differences between acute and chronic pain, both are “real” and can be explained physiologically and psychologically.
In the medical domain, once a diagnosis of a chronic pain disorder is confirmed, ongoing drug treatment is typically tapered, including controlled opiate withdrawal if deemed necessary. Psychotherapists play a crucial role in helping children and adolescents adopt a less pain-focused perception of their bodies and to rebuild confidence through targeted physical activities. Psychological interventions include both individual and group discussions that address various factors (such as stress, anxiety, and social exclusion) known to trigger and modulate the pain disorder. Therapists also offer training in relaxation techniques and coping strategies. Social workers complement these efforts by providing recreational programs and offering support to both parents and patients, with a particular emphasis on school, education, and the management of daily life challenges. The collaborative efforts and communication among professionals from multiple disciplines lay the groundwork for effective individualized therapies, and the development of tailored interdisciplinary treatment plans.
Participants
This study was assessed positively by the Helmut Schmidt University’s ethics committee (5th March 2019). For inclusion in this single-center study, patients had to meet the following criteria: (1) experience of chronic pain for more than 3 months, (2) being between 11 and 18 years old, (3) having received an inpatient treatment at the Center for Young People’s Pain Therapy for a 3-week period between 5 February and 25 June 2019, and (4) having a signed declaration of consent from parents or legal guardians. A total of 156 adolescents met the inclusion criteria. Sixty-two of these patients had to be excluded from the analyses due to either failure to complete a consent form or an incomplete data set. The final data set was therefore based on N = 94 (84 female, M = 14.7, SD = 1.65 (11–18 years)) patients. The mean duration of pain was 42.60 months (SD = 36.57), with an overall range between 3 and 195 months.
Materials
Multidimensional Fatigue Inventory (MFI-20)
The Multidimensional Fatigue Inventory (MFI-20) is a self-assessment questionnaire comprising 20 items (Smets et al., 1995). It includes the dimensions general fatigue, physical fatigue, mental fatigue, reduced motivation, and reduced activity (Smets et al., 1995). Items are answered on a 5-point scale ranging from applies to does not apply at all. To evaluate the MFI-20, the items are added to subscales, for which some items must be reversed. Higher scores indicate a higher level of fatigue. The MFI-total has an adequate internal consistency and test–retest reliability (Elbers et al., 2012; Westenberger et al., 2022). The Cronbach’s alpha for the MFI on general fatigue, physical fatigue, and mental fatigue is good (α > 0.80), and it is adequate for reduced activity and reduced motivation (α > 0.65; Smets et al., 1995). To the best of our knowledge, there were no available questionnaires for measuring fatigue in children and adolescents in German at the time of our study, except in oncology. We used the German version of the MFI-20 and adapted the MFI-20 instructions for children and adolescents (Binz et al., 2022; Brähler et al., 2001; Schwarz et al., 2003).
Depression Inventory for Children and Adolescents (DIKJ)
The Depression Inventory for Children and Adolescents (German: Depressionsinventar für Kinder und Jugendliche–DIKJ), which comprises 29 items, is a screening instrument that measures the severity of depressive symptoms in children and adolescents aged 8–16 years (Keller, 2016). The internal consistency of the questionnaire is satisfactory, with Cronbach’s alpha = 0.87, as well as good discriminatory power between children and adolescents with and without psychopathological issues (Keller, 2016). The inventory handbook states that the questionnaire can be applied to adolescents over the age 16 if they still attend school and have a lifestyle comparable to that of 16-year-olds.
German Pain Questionnaire for Children and Adolescents (DSF-KJ)
The German Pain Questionnaire for Children and Adolescents (German: Deutscher Schmerzfragebogen für Kinder und Jugendliche–DSF-KJ; see Table S6) records patients’ descriptions of their pain. Three different versions (1: initial contact, 2: follow-up questionnaire, 3: follow-up visit) are available for children, adolescents, and parents (Schroeder et al., 2010). In this study, we analyzed the data for the average pain level and the strongest pain level experienced within the last 7 days prior to T1 (before treatment) and T2 (end of treatment). The DSF–KJ has demonstrated good content validity and reliability when tested with more than 3000 patients (Nagel et al., 2002).
Short evaluation
We also used an additional short evaluation, an inpatient questionnaire consisting of items from the Pediatric Pain Disability Index (P-PDI; Hübner et al., 2009), a German adaptation of the American Chronic Pain Association (ACPA) questionnaire, and an evaluation questionnaire from the Center for Rheumatology, Orthopedics and Pain Therapy at the Oberammergau Clinic (see Table S7). A team of doctors, psychologists, and physiotherapists from the Center for Young People’s Pain Therapy developed the short evaluation using a ranking procedure. The questionnaire includes eight items with an 11-point numeric rating scale (0–10); the items reflect pain intensity, activity, social well-being, level of stress, physical fitness, daily impairment, self-confidence regarding the individual’s strength, and sleep.
Procedure
Prior to hospitalization (prior to T1), the MFI-20, DSF–KJ, and DIKJ were collected. The inpatient short evaluation was filled out at the beginning of the inpatient treatment (T1). At the end of the inpatient treatment (T2), the MFI-20, a shortened version of the DSF–KJ, and the short evaluation were collected. The data set was completed using data from the patients’ charts, including age, diagnoses according to ICD-10, the self-reported start date of the pain experience, the results of the DIKJ, and pain intensity and average pain duration reported on the German Pain Questionnaire for Children and Adolescents at times T1 and T2. We conducted an a priori power analysis using G*Power (Kang and Huh, 2021), with the power (1− β) set at 0.90; α = 0.05 one-tailed. Given the absence of prior studies investigating the association between fatigue and chronic pain in children and adolescents at the time, we relied on evidence from a structured review indicating an association between fatigue and pain (Fishbain et al., 2003). However, whether fatigue changes with pain reduction remained unexplored. Therefore, we hypothesized a medium effect size (ρ = 0.4) and calculated that the minimum total sample size was required to be 47 participants.
Statistical analyses
The data were processed using SPSS (64-bit version Build 1.0.0.1298) and StataIC (16-bit version). We calculated linear regressions to examine whether the participants’ degree of fatigue was associated with the duration of their disease. The prerequisites were interval-scaled dependent and independent variables, as well as a linear relationship between the dependent and independent variables (Field, 2018). No outliers were found in the first analysis of the data or the analysis of the residuals.
We verified the changes in the MFI categories as well as the pain intensity using a Wilcoxon sign rank test for dependent samples, owing to the lack of a normal distribution. A median test of the dimensions of the MFI at the beginning and the end of the therapy was necessary for the sign rank test.
An independent samples t-test was conducted to compare children and adolescents with chronic pain to the standard values for a 16–24 age group (N = 229) and an extracted 16–18 age group (N = 70) at the beginning (T1) and end (T2) of the therapy. The Levene test was used to test the variances for both samples.
To test whether the experience of pain decreased when fatigue was reduced, we converted the SPSS data set to StataIC, defining it as a panel data set. The panel design was necessary for the analysis because the design of this study was longitudinal, with two measurement time points, T1 and T2 (Giesselmann and Windzio, 2012). Additionally, we calculated the mean differences for the dimensions of the MFI between T1 and T2 by first calculating a fixed-effects model and then a random-effects model. Due to a non-significant Hausman specification test (X2 = 0.94), the random-effects model is the statistically better choice for testing the hypotheses.
Results
We calculated a linear regression model for each dimension of the MFI to test whether longer durations of pain corresponded to higher levels of fatigue at the beginning of the therapy. Based on the analysis, this hypothesis must be rejected as no correlation was found across any dimensions of the MFI (see Table S1).
Regarding the influence of the therapy on fatigue (see Table 1), the dimensions general fatigue, physical activity, and reduced motivation were significantly improved at the end of the therapy (T2). The dimensions general fatigue, physical fatigue, and reduced activity showed a medium effect size, while the dimension reduced motivation had a small effect size (Cohen, 1988). No significant difference between the beginning and the end of therapy was found for mental fatigue. Pain intensity decreased with a small effect size (Cohen, 1988).
MFI fatigue subscale and pain intensity changes between the beginning and the end of therapy.
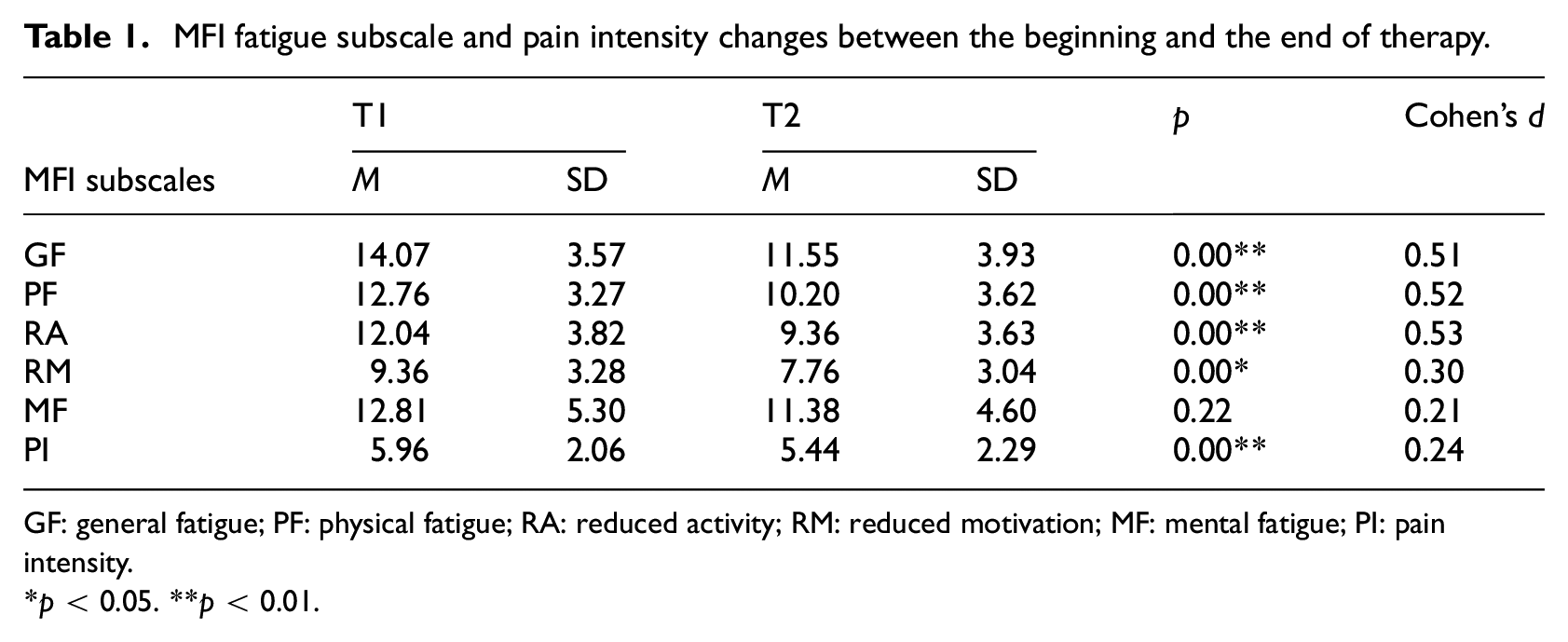
GF: general fatigue; PF: physical fatigue; RA: reduced activity; RM: reduced motivation; MF: mental fatigue; PI: pain intensity.
*p < 0.05. **p < 0.01.
The means of the five dimensions for the participants differ significantly from those for the norm sample (see Tables S2–S5). The differences are significant for both age groups at T1 and T2 (16–24 years and 16–18 years).
The random-effects model considered the intra-individual differences of the patients at time T1 versus T2, without assuming fixed variances (see Table 2). We used this model to check whether the depressive symptoms or the duration of the pain also influenced pain perception. Neither coefficient was significant. The difference in general fatigue was found to be statistically significant and correlated with pain intensity, with each unit of change in general fatigue resulting in a decrease of 0.238 units on a scale of 0–10 in pain intensity. This implies that a reduction in general fatigue followed a reduction in pain intensity in the period between T1 and T2. The degree of general fatigue and the change in general fatigue both correlated with the level of pain at T2.
Random-effects model.
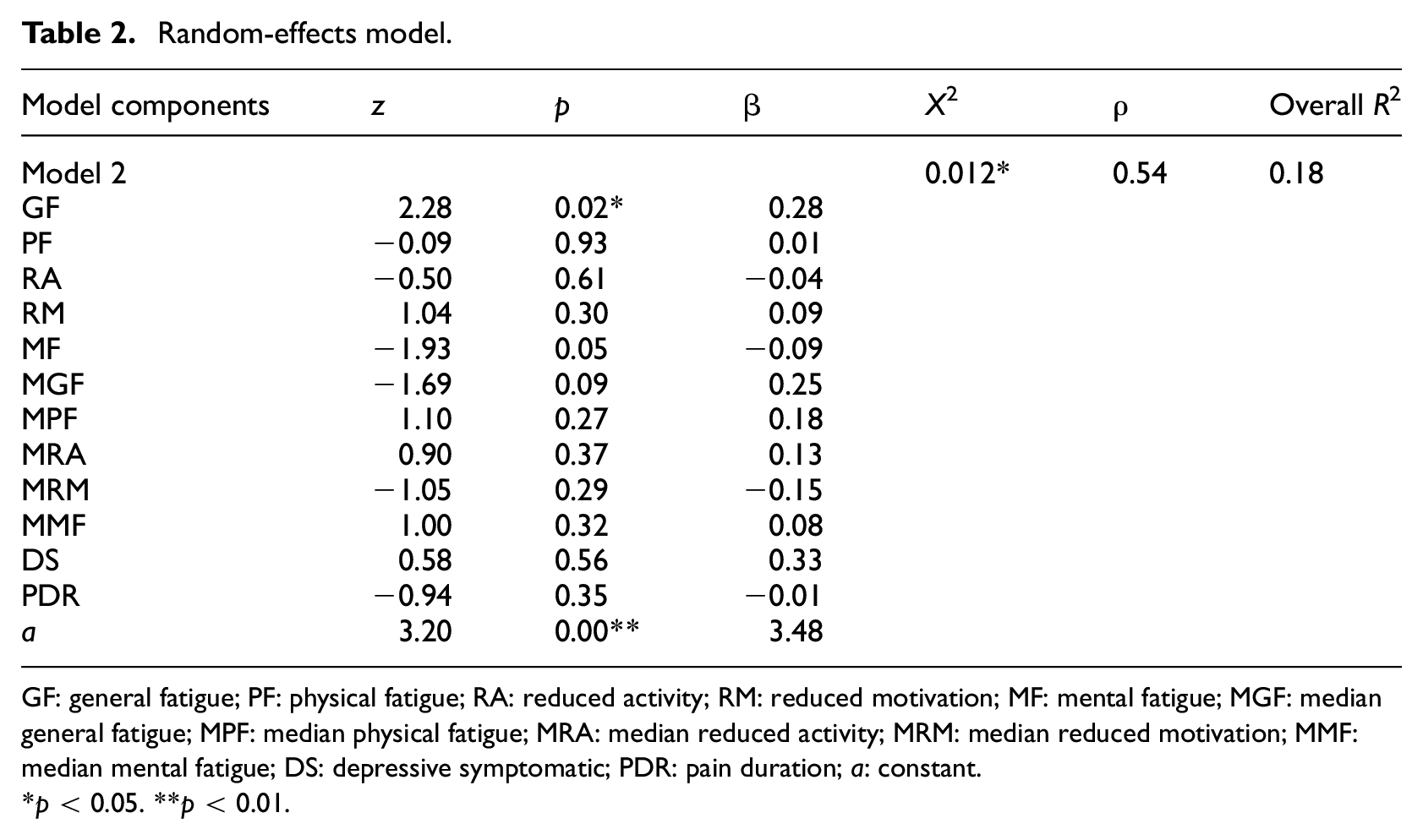
GF: general fatigue; PF: physical fatigue; RA: reduced activity; RM: reduced motivation; MF: mental fatigue; MGF: median general fatigue; MPF: median physical fatigue; MRA: median reduced activity; MRM: median reduced motivation; MMF: median mental fatigue; DS: depressive symptomatic; PDR: pain duration; a: constant.
*p < 0.05. **p < 0.01.
Discussion
The present study aimed to investigate the role of fatigue with respect to chronic pain by examining changes in both fatigue and pain intensity over a 3-week in-patient IMPT program. We observed a correlation between general fatigue and pain intensity. Notably, in contrast to mental fatigue, general and physical fatigue appeared to be influenced by short-term therapy and to have a discernible impact on self-reported pain intensity. Consequently, interventions aimed at reducing chronic pain are useful to reduce fatigue. In addition, fatigue-reducing strategies could be added to further reduce fatigue.
The relationship between fatigue and pain in children and adolescents has not been extensively researched, highlighting a critical gap in our understanding of these interrelated dimensions of health. Our study makes a valuable contribution to addressing this gap by exploring the intricate link between pain and various dimensions of fatigue. Pain and fatigue are known to be closely intertwined (Fishbain et al., 2003), with fatigue having been frequently observed as a symptom of chronic pain (Hunfeld et al., 2001; Yazdani and Zeltzer, 2013). Based on the chronicity of both pain and fatigue, it is plausible that reducing one might influence the other. Although our findings do not confirm that reducing fatigue directly causes a reduction in pain, the significant association between general fatigue and pain observed in the random-effects model constitutes a key step in this direction. This model accounts for variability between individuals and offers robust insights into the broader patterns linking fatigue and pain.
By deepening our understanding of these connections, our results set the stage for more targeted interventions aimed at managing fatigue as a part of comprehensive pain treatment strategies. While the specific mechanisms behind this interaction require further exploration, our study provides a strong foundation for future research, suggesting that interventions focusing on reducing general fatigue may be particularly beneficial for enhancing the overall health outcomes of children and adolescents facing chronic pain. Van Damme’s Fatigue Model (2018) shows that fatigue, reward, and pain interact. Chronic pain exerts a reinforcing influence on expected pain, so that goal-directed efforts are more taxing, which can lead to heightened fatigue and a decrease in drive. This creates a reinforcing loop: as pain (expectations) increase, so does the subjective cost of sustaining effort, further contributing to fatigue. Therefore, high levels of fatigue are closely intertwined with the experience of chronic pain, as the persistent pain signals add to the perceived effort required, which is why we expected an interdependence between fatigue and pain intensity.
After the IMPT treatment, there was a notable decrease in pain intensity. Pain intensity was evaluated using an inpatient short evaluation tool that, although it is not a validated instrument, serves as a quality measurement tool within the clinic. However, it is essential to note that the assessment of pain intensity was based on the German Pain Questionnaire for Children and Adolescents, utilizing the same scale (see Table S7) and it is commonly used in the assessment of pediatric pain (Birnie et al., 2019). It is worth repeating that the primary objective of IMPT treatment extends beyond pain alleviation. The overarching goal includes enhancing the overall quality of life and facilitating participation in age-appropriate activities. While the reduction of pain intensity is a notable outcome, it is only one aspect of a comprehensive approach that aims to improve both the well-being and the functional capabilities of patients. The therapeutic objective of chronic pain treatment also encompasses enhancing patients’ strategies for pain management and coping with daily life challenges. Given that all dimensions of the MFI except for mental fatigue were significantly influenced during the therapy, alternative evaluation approaches may be more appropriate. These could include the now available PROMIS Fatigue Scale (Feagan et al., 2023) alongside pain intensity assessments, rather than focusing solely on pain intensity.
As an apparent effect of the treatment, perceived fatigue decreased, except for mental fatigue, which showed no significant changes. Over the 3-week inpatient treatment, medium effect sizes were found for general fatigue, physical fatigue, and reduced activity, along with a small effect size for reduced motivation. Adolescents experiencing chronic pain typically spend less time in school and more time recovering from pain during a prolonged in-patient treatment. Consequently, one might expect that reducing cognitive effort and stress during inpatient treatment should reduce mental fatigue, which was not observed. It is possible that this type of change requires a longer treatment period, and this could be the subject of further research. Also, it is possible that the adolescents’ reduced cognitive effort limited the relevance of mental fatigue. Moreover, individualized support provided within the clinical school setting may have contributed to a reduction in stress.
There was no significant correlation between the duration of pain and perceived fatigue. Patients showed a wide variation in the duration of their pain experience. It might be hypothesized that there may not be a linear relationship, but rather a curvilinear interrelationship.
In chronic pain management, incorporating strategies that address fatigue through a balance of activation and rest might be beneficial. Many of these strategies are foundational in fatigue management for other chronic conditions, such as rheumatic diseases or oncology, and may also complement pain management efforts. For instance, structured weekly activity plans, pacing techniques that balance exertion with recovery, and supportive interventions like circulatory-stabilizing practices and balanced nutrition can help patients maintain stable activity levels and reduce the risk of fatigue exacerbation (Neill et al., 2006; Vroegindeweij et al., 2024).
Further, educating patients about fatigue and fostering awareness of their individual fatigue levels within chronic pain management could empower them to manage energy more effectively, potentially alleviating some aspects of pain. By integrating these fatigue management strategies, which are typically used in other chronic conditions, chronic pain management could be enhanced, making these approaches a valuable addition to an IMPT framework. Other areas of significant interest include evaluating strategies that target fatigue in therapy and promote increased goal-related behavior, as well as exploring the analgesic effects of rewards in reducing perceived pain.
Limitations
With a total of 94 patients, our sample size was sufficiently powered in terms of statistical accuracy. However, given the wide age range of the participants, this is an inter-individually heterogeneous sample. It should be kept in mind that this age range includes many milestones in the physiological and psychological development of children and adolescents, which may have affected the collected data. Generalization might also be further limited by the fact that the diagnostic examinations as well as the treatment were performed by the same team. Even though it might be advantageous if the team that performs diagnostic examinations is the same as the team that treats the patient, it might also be less objective. Despite following standardized procedures and employing an IMTP, the individual treatments are tailored to the single patient’s needs. Thus, the diagnostic and especially the therapeutic processes may not be entirely objective. One could argue that a separation of diagnostic and therapeutic procedures might thus be preferable for avoiding presumptions. However, it is essential to balance the benefits of continuity and personalized care against the need for objectivity and unbiased evaluation. Future studies could explore ways to address these concerns while maintaining the integrity and effectiveness of the diagnostic and treatment processes in chronic pain management.
In general, German-language multimodal fatigue questionnaires for children and adolescents were lacking at the time of our study, apart from questionnaires used almost exclusively in the field of pediatric oncology—although the German version of the PROMIS® Pediatric Short Form v2.0-Fatigue 10a has since been validated for the assessment of fatigue in chronic pain and should be included in further research (Sommer et al., 2022). Nevertheless, the MFI questionnaire used in this study was not specifically designed for children and adolescents. There are no norms for children and adolescents, but only for young adults aged 16–24 (Westenberger et al., 2022). The children and adolescents with chronic pain in our study reported significantly higher levels of fatigue than young adults aged 16–24 years in the norm group. We extracted the 16–18 year group from the 16–24 year norm group to further eliminate age-related differences. However, both age groups have relatively small sample sizes. Further research should examine norms for children and adolescents aged 12–16 years to replicate and support our findings.
The MFI questionnaire shows different internal consistencies for different samples. The overall internal consistency has a minimum Cronbach’s alpha of 0.89 (Hinz et al., 2020). The dimension of reduced motivation showed insufficient Cronbach’s alpha values in a study with critically ill patients. Despite criticisms regarding the scale structure of the questionnaire, subsequent reviews with seven different samples have advocated for retaining the scale structure (Hinz et al., 2020). The values for reduced motivation should still be viewed critically. Despite a high level of visual validity and clear parallels to child-oriented fatigue questionnaires in English, the results of the present work can only be considered valid based on further research.
Conclusions
Our study highlights the potential relationship between fatigue and pain in the context of a 3-week inpatient treatment. Given that fatigue can be reduced in the context of an IMPT and appears to be a factor influencing pain perception, further research on fatigue-reducing treatments is needed.
Further empirical research in this domain could offer complementary therapeutic approaches for alleviating the experience of pain. By addressing fatigue as an integral component of chronic pain management, clinicians may enhance treatment outcomes and improve the overall well-being of patients. Thus, ongoing exploration in this area has the potential to expand the repertoire of therapeutic measures available for pain reduction.
Supplemental Material
sj-docx-1-hpq-10.1177_13591053251324703 – Supplemental material for The role of fatigue in adolescents’ chronic pain: A study on facets of fatigue in young patients with a chronic pain disorder
Supplemental material, sj-docx-1-hpq-10.1177_13591053251324703 for The role of fatigue in adolescents’ chronic pain: A study on facets of fatigue in young patients with a chronic pain disorder by Vivien Manteufel, Lea Höfel, Johannes-Peter Haas and Thomas Jacobsen in Journal of Health Psychology
Footnotes
Acknowledgements
It is a pleasure to thank Mrs. Christine Lindner for registering the questionnaires and all the patients who took part in the study. We are grateful to Merrie Bergmann for copyediting.
Data sharing statement
The dataset generated and analyzed in the present study is not publicly available because permission has not been granted from the participants or the ethics committee. All participants provided written consent to use their data in a research paper but not to publish their data set in an anonymized form. To publish such anonymized data would violate the written consent obtained from the participants and thus would not be in accordance with European General Data Protection Regulation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Prior to being conducted, the present study was approved by the local ethics committee of the Helmut Schmidt University/University of the Federal Armed Forces Hamburg (03/05/19/VM).
Informed consent
All participants and their parents gave written informed consent before collecting data.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
