Abstract
Epidermolysis bullosa (EB) is a rare disease characterised by skin fragility and a wide variety of symptoms. The Quality of Life in Epidermolysis Bullosa (QOLEB) score is an English 17-item EB-specific validated measurement tool with two dimensions: functioning and emotions. The aim of this cross-sectional study was to develop and validate a culturally adapted German QOLEB. The following steps were carried out: translation, expert evaluation, back translation, linguistic and cultural adaptation, sample-based psychometric testing and evaluation. Data analysis was performed with n = 46 patients across all EB types. The reliability and internal consistency of the translated German QOLEB were excellent (α = 0.901). Regarding convergent validity, the QOLEB correlated highly with the iscorEB (r = 0.879; p < 0.001). Structural similarity with the English original version was confirmed through exploratory factor analysis. In conclusion, the German QOLEB demonstrates internal reliability and construct validity and is suitable to assess the quality of life in German-speaking EB patients.
Keywords
Introduction
Epidermolysis bullosa (EB) is a rare genetic skin fragility disorder contributing to and promoting potentially severe morbidity and mortality, with an estimated prevalence of 1:90,000 worldwide and 1:22,000–40,000 in Germany (Bardhan et al., 2020; Has, 2020; Has et al., 2023; Reimer et al., 2018). Spontaneous or mechanically induced blistering and wound formation is a characteristic feature of EB (Bardhan et al., 2020). To date, 20 different genes have been identified to cause structural and functional impairment of the epidermis and dermo-epidermal basement membrane zone. The level of detachment differentiates four major EB types: EB simplex (EBS), dystrophic EB (DEB), junctional EB (JEB) and Kindler EB (KEB), which leads to a large phenotypic variability with possible organ involvement and complications (Has et al., 2020). The clinical presentation encompasses psychological and social limitations affected by a complex interplay of internal and external factors, such as the socio-economic situation of the person affected (Fine, 2013). The high inter-individual heterogeneity of physical, psychological and social limitations leads to a wide variety of individual challenges and issues for those affected (Sangha et al., 2021; van Scheppingen et al., 2008; Williams et al., 2011) as well as for their relatives (de Azevedo Silva et al., 2020; Ireland et al., 2021). For this reason, a patient-based and holistic approach is fundamental in the context of EB (Butterworth et al., 2019).
One approach that can help to take such a perspective is the construct of quality of life, especially if standardised parameters can be derived from it. When assessing quality of life, the focus is set on the subjective experience of individual life circumstances (Fayers and Machin, 2015). The WHO defines quality of life as ‘an individual’s perception of their position in life in the context of the culture and value systems in which they live and in relation to their goals, expectations, standards and concerns’ (Harper and Power, 1998). Consequently, quality of life is recognised as an individual perspective on the combination of physical, psychological and social factors.
There are many different instruments available to measure quality of life (Cooke et al., 2016). In addition to generic tests, such as the World Health Organisation’s Quality of life instrument (WHOQOL) or the Quality of Life Scale (QOLS), there are more specific instruments for dermatological conditions, such as the Dermatology Life Quality Index (DLQI) or the Skindex (Burckhardt and Anderson, 2003; Chren et al., 1996; Finlay and Khan, 1994; Whoqol Group, 1998). Furthermore, specific instruments are being developed focusing on a group of diseases or on a single disease. These can be used to better capture the various symptoms and their impact on the quality of life in the context of a disease and to help evaluate interventions and treatments (Augustin et al., 2012; Chernyshov, 2019). In the case of lifelong disease, as seen in chronic or genetic diseases, the impact of subjectively experienced disease-related circumstances and challenges require considerable attention (Bhatti et al., 2011).
Accordingly, disease-specific measurement instruments are particularly useful in chronic, multi-systemic diseases with highly variable phenotype, such as EB, as they can best reflect the diversity of different disease courses (Choi et al., 2017). Moreover, from a statistical point of view, disease-specific instruments are also more advantageous for EB, since with the large heterogeneity of the disease, both existing generic and skin-specific measurement instruments usually feature high ceiling effects (Frew et al., 2009) and lead to problems regarding validity and reliability (Frew et al., 2017; Sebaratnam et al., 2012).
In previously published studies, physical factors, such as pain, itch or wound size, significantly reduced quality of life in EB patients (Brun et al., 2017; Eng et al., 2021; Fortuna et al., 2017; Snauwaert et al., 2014), with more severe EB subtypes and patients with higher morbidity reporting a lower quality of life (Jeon et al., 2016). At the same time, psychosocial factors such as religion, culture, environment, education and financial resources have also been shown to influence the quality of life (Adni et al., 2012), with social support in particular having a positive impact on the quality of life of those affected (Butterworth et al., 2019). Furthermore, there is a difference in the quality of life reported depending on age and gender, with children tending to score lower than adults and women reporting a lower quality of life than men (Tabolli et al., 2009). In this context, a new quality of life measurement instrument for EB was developed.
‘Quality of Life in Epidermolysis Bullosa’ (QOLEB)
The ‘Quality of Life in Epidermolysis Bullosa’ (QOLEB), authored by Frew and Murrell (Frew et al., 2009), shows reliable results concerning validity and time-efficient handling and has been the most widely used validated EB-specific measurement instrument to assess quality of life so far (Togo et al., 2020). It consists of 17 items, which can be rated on a four-point Likert scale, ranging from a minimum to a maximum impact on different aspects of quality of life. By forming summative scores, the two dimensions functioning and emotions can be distinguished. For the evaluation and clinical assessment of quality of life, a total score of all items is formed, ranging from 0 to 51 points. This way, the impact of EB on the quality of life can be classified into clinical bands from ‘very mild’ to ‘very severe’ (Frew and Murrell, 2012). The QOLEB is used internationally and has been translated to several languages, with validated versions for Brazilian Portuguese, Dutch, Farsi, Hindi, Romanian and Spanish (Bishnoi et al., 2022; Cestari et al., 2016; Dănescu et al., 2019; Frew et al., 2013; Villar Hernández et al., 2022; Yazdanshenas et al., 2020; Yuen et al., 2014).
The objective of this study is a German equivalent to the original English version of the QOLEB. A detailed qualitative pre-study had already been performed on a non-validated German QOLEB translation (Keller, 2013). The instrument’s comprehensibility and applicability were evaluated in nine semi-structured interviews with EB patients (5 DEB, 3 JEB, 1 EBS; age range from 18 to 64 years; sex and severity were balanced). Several recommendations for revision of the German version were expressed. However, a validated German version is currently missing, which would allow assessment of quality of life for EB in clinical research and practice in Austria, Germany and Switzerland. Therefore, this article presents a German QOLEB translation and examines its reliability and validity via sample-based psychometric testing and evaluation.
Materials and methods
The development of the German version of the QOLEB comprised two stages: the translation, cross-cultural adaptation and semantic validation as well as the psychometric validation of the German version. The translation and psychometric testing process combined the recommendations regarding translation, cross-cultural adaptation and validation of medical and psychological scales (Borsa et al., 2012; Epstein et al., 2015; Sousa and Rojjanasrirat, 2011) and was in line with the COSMIN checklist (Mokkink et al., 2019).
Ethical aspects
Ethical approval was granted by both the Medical and the Psychological Ethics Committees of the Sigmund Freud University. For the pre-study and the main study, informed consent or assent was obtained from all participating patients or legal guardians of underage patients, respectively.
Translation of the QOLEB to German
As a first step of the translation process, four independent forward translations were accumulated: A first non-validated German QOLEB version was obtained from the EB House Austria and used for a pre-study with n = 17 EB patients (9 DEB, 4 JEB, 4 EBS; age range from 10 to 49 years; sex and severity were balanced). A second non-validated German QOLEB version by Keller as well as her patient-based recommendations for clarity, wording and sentence-structure from an interview study were taken into account (Keller, 2013). Additionally, forward translation of the QOLEB from the original English version to German was performed by two independent translators. Consensus between those four versions was achieved by comparing discrepancies of words and meanings with the help of a multidisciplinary research team with psychological, medical and linguistic experts. The heterogeneity of the response specifications of the original questionnaire was found to be problematic. To improve the readability of the questionnaire by means of repetition and to highlight the differences between the answer formats, parallel formats across the different questions were used if possible. Nevertheless, the entire translation process focused on the equivalence of the questionnaire’s content.
Content validity was obtained through back translation to English by two different independent translators. Ambiguities and discrepancies between both back translations were resolved by another independent linguist. The original QOLEB and our back translation were presented to the authors of the original questionnaire for approval. Direct feedback on the clarity and comprehensibility of the final German QOLEB was collected from the EB patient organisation DEBRA Austria. A detailed documentation of all steps of the translation and linguistic validation process can be found in Table 1.
Evaluation of the QOLEB translation process in accordance to the COSMIN checklist (Mokkink et al., 2019).

The final German QOLEB can be found in the appendix (see supplementary file S1).
Recruitment and design
The participants of this cross-sectional online study were recruited between April and September 2021 in cooperation with DEBRA International, DEBRA Austria, the EB House Austria and via social media. Inclusion criteria were a diagnosis of any EB type, a minimum age of 16 years and fluency in German. The online survey contained information on design and relevance of the study, questions collecting demographic data, the German QOLEB and the iscorEB Patient Score (Bruckner et al., 2018; Schwieger-Briel et al., 2015). Further scores targeted personal resources and support, namely the Brief Resilience Scale (BRS), the Perceived Social Support Questionnaire (F-SozU) and the Satisfaction with life Scale (SWLS) in their validated German translations (Glaesmer et al., 2011; Kliem et al., 2015; Kunzler et al., 2018). To take into account the unpredictability of EB and to reduce drop-outs for health reasons, the option was offered to pause the questionnaire for a maximum of 7 days and continue at a later date.
Data analysis and psychometric properties
To describe the sample, frequencies, means and standard deviations or medians and percentiles (interquartile range IQR: 25th and 75th) were calculated. Percentage coverage of the EB population was based on epidemiological estimates for Germany and Austria (Has et al., 2023; Pohla-Gubo and Hintner, 2010). Due to the open online survey and the recruitment via stakeholders, no exact participation ratio can be calculated.
Data inspection was done both on a general level to analyse the distribution of the data (histogram, Kolmogorov-Smirnov-test, skewness statistics) and on an individual item level for floor and ceiling effects. In the case of missing data, additionally to the QOLEB total sum score, an average total score was calculated to include as much collected data as possible. For group comparisons, Mann-Whitney U-test and Kruskal-Wallis-test followed by post-hoc U-test with Bonferroni correction were performed.
In order to assess the construct validity of the data, an exploratory factor analysis (principal component analysis, varimax rotation) was calculated and compared to the original factor solution of the QOLEB. Convergent validity was confirmed by calculating Pearson correlations with established tests that are based on a similar construct: medical state via iscorEB (Bruckner et al., 2018; Schwieger-Briel et al., 2015), satisfaction with life via SWLS (Glaesmer et al., 2011; Hinz et al., 2018) and resilience via the BRS (Kunzler et al., 2018). A correlation coefficient of r > 0.5 was considered as excellent (Cohen, 1988). Divergent validity, on the other hand, distinguishes between two different constructs. Therefore, a low correlation coefficient between the comparing measure for social support via the F-SozU (Kliem et al., 2015; Lin et al., 2019) is desirable. The reliability and internal consistency of the German QOLEB was addressed by the Cronbach’s alpha. An α of ⩾0.7 was considered as acceptable, ⩾0.8 as good, and ⩾0.9 as excellent (Nunnally, 1994). A significance level of 5% was assumed for the entire statistical analyses, which were performed by the use of the software IBM SPSS Statistics, version 27.
Results
Study population
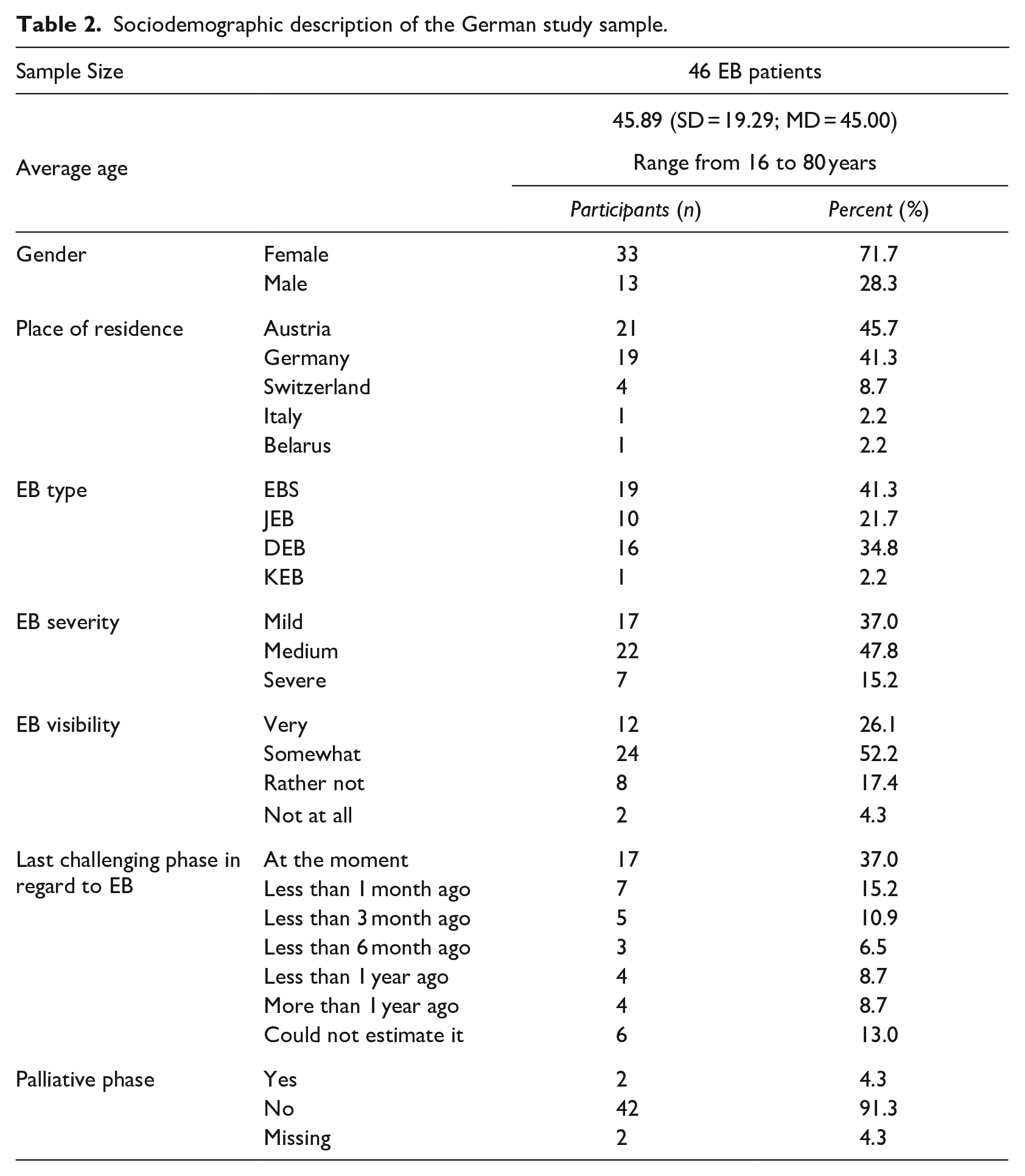
In the current study, a total of n = 46 German speaking EB patients completed the QOLEB. The average age of the sample was 45.89 years (SD = 19.29) and over two-thirds were female (n = 33, 71.7%). The majority of the participants were living either in Austria (n = 21, 45.7%) or Germany (n = 19, 41.3%), which corresponds to 1.0% of the German and Austrian EB population; the rest in Switzerland (n = 4, 8.7%), Italy (n = 1, 2.2%) and Belarus (n = 1, 2.2%). Regarding the language level of the study sample, 91.1% were native speakers and the rest understood German very well.
With regard to the distribution of the EB types within the sample, 19 (41.3%) were diagnosed with EBS, 16 (34.8%) with DEB, 10 (21.7%) with JEB and 1 (2.2%) with KEB. The participants were asked to rate the severity as well as the visibility of their EB. The specified severity ranged from mild (37%) to medium (47.8%) and to severe (15.2%). Concerning the visibility, 26.1% of the EB patients stated that their EB was very, 52.2% somewhat, 17.4% rather not and 4.3% not at all visible. Considering the fluctuation of a chronic disease, the patients were asked when they experienced their last challenging phase with EB. More than a third (37%) indicated that they were going through an acute challenging phase, whereof two patients (4.3%) reported to be in their palliative phase. An overview of the demographic data described above can be found in Table 2.
Sociodemographic description of the German study sample.
QOLEB
Regarding the 17 items of the scale, neither ceiling nor floor effects were found and the vast majority of the EB patients completed the questionnaire (1% missing data). In order to include as much collected data as possible, additionally to the QOLEB total sum score (QOLEB score), a total average score (QOLEB TA) was calculated. The QOLEB TA was normally distributed with a mean of 1.82 (SD = 0.51; n = 46), whereas the distribution of the QOLEB score was not (MD = 28; IQR 23–40; n = 43). The mean time taken to complete the questionnaire was 2.7 minutes (MD = 2.1; range 1–7 minutes).
Women showed a significantly higher impact on their perceived quality of life than men (QOLEB score: U = 90.00, p = 0.009; TA: U = 112.00, p = 0.012). The median QOLEB score of the female participants was 29 (IQR 24–41) and of the male participants 22 (IQR 21.3–31.5). With regard to the two QOLEB subscales, there were significant differences between women and men on the functioning scale (QOLEB score: U = 113, p = 0.022; TA: U = 116.00, p = 0.016) but not on the emotions scale (QOLEB score: U = 151.50, p = 0.282; TA: U = 179.00, p = 0.383). The median functioning scale score for women was 22 (IQR 17–31) and for men 17 (IQR 14–22). On the emotions scale the medium score for female participants was 9 (IQR 7–10) and for male 7 (IQR 6–10.8).
With regard to the QOLEB score and the severity of EB, significant differences between mild and moderate (p = 0.010) and mild and severe (p = 0.003) patient-reported severity were found (QOLEB score: H(2) = 13.72, p = 0.001; mild: MD = 24 (IQR 22–28), moderate: MD = 30 (IQR 26–41), severe: MD = 41 (IQR 40–43); TA: H(2) = 15.30, p < 0.000). On the two subscales of the QOLEB, significant differences between the reported severity groups were found on the functioning (score: H(2) = 16.39, p < 0.000; mild: MD = 15 (IQR 14–19.50), moderate: MD = 23 (IQR 17–28.75), severe: MD = 31 (IQR 31–33); TA: H(2) = 17.54, p < 0.000) but not on the emotions scale. On the functioning scale, the QOLEB scores significantly differed between the mild and moderate (p = 0.003) and the mild and severe (p = 0.001) patient-reported severity.
The QOLEB scores showed no significant differences concerning EB types, patient-reported visibility and acute challenging phase, neither for the QOLEB score nor on the functioning or the emotions subscale (p > 0.05 each; see supplementary file S2).
The QOLEB score distribution and its interpretation into clinical bands as suggested by the original authors (Frew and Murrell, 2012) can be found in Table 3.
Description of the study sample in relation to the QOLEB scores.
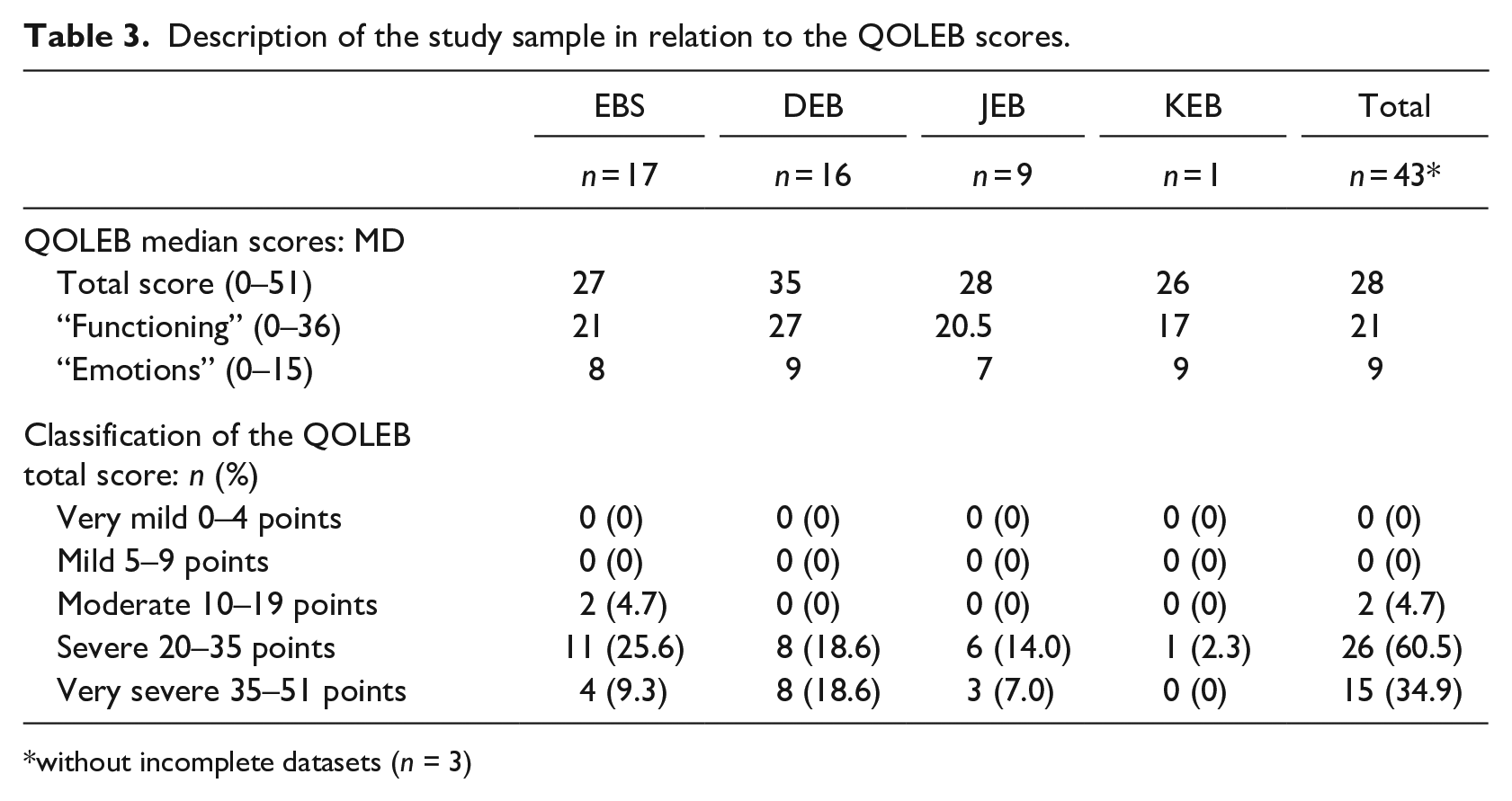
without incomplete datasets (n = 3)
Psychometric properties
The reliability and internal consistency of the German version of the QOLEB with a Cronbach’s Alpha of 0.901 is excellent.
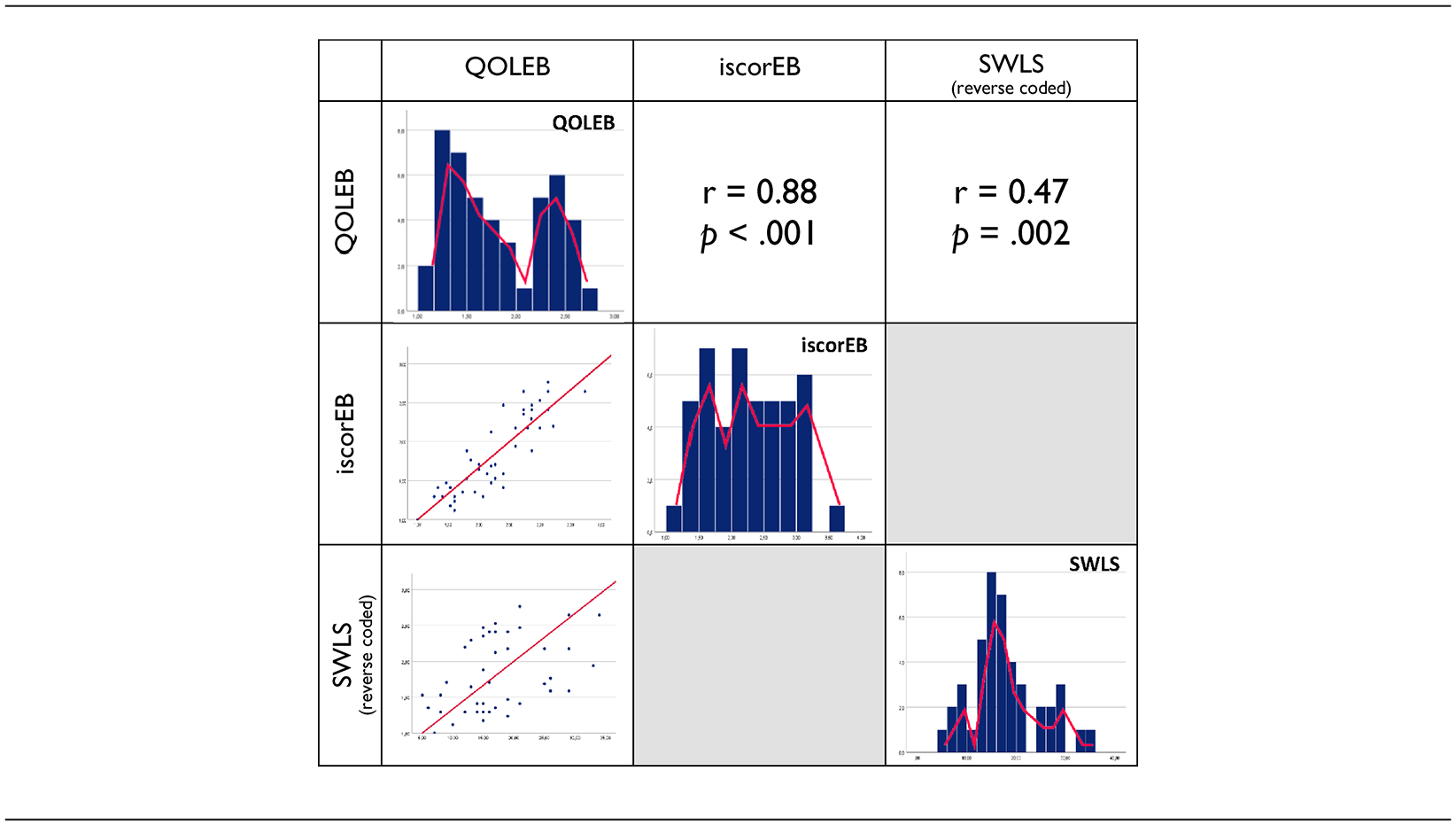
Regarding the convergent validity (Table 4), the German version of the QOLEB correlated highly with the medical state, as assessed by the iscorEB average score (r = 0.879, p < 0.001), and moderately with the satisfaction with life, as measured by the SWLS average score (r = −0.469, p = 0.002), and with resilience, as assessed by the BRS average score (r = −0.312, p = 0.045).
Convergent validity of the German QOLEB.
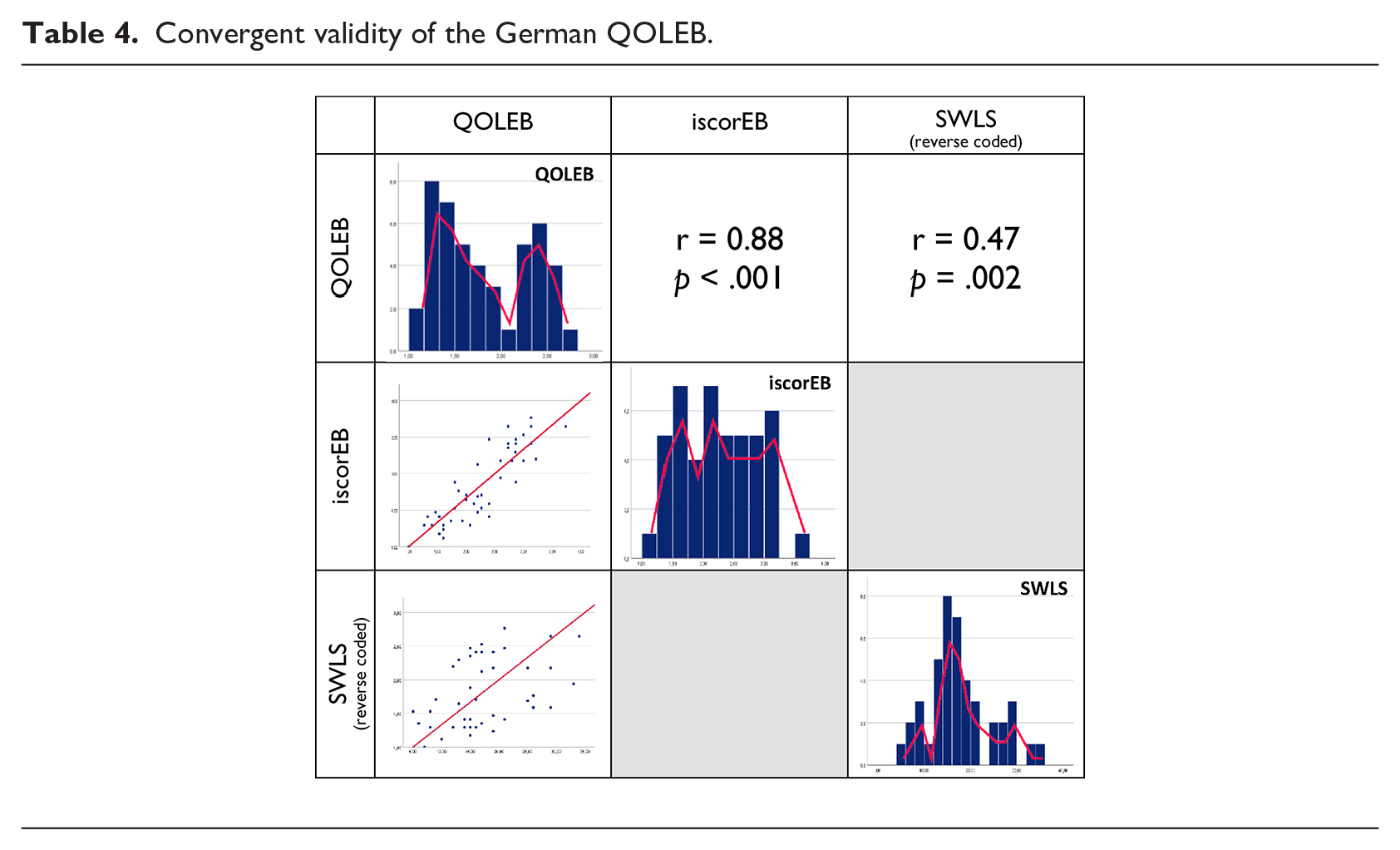
Regarding divergent validity, the QOLEB did not correlate significantly with social support, as measured by the F-SozU average score (ρ = −0.276, p = 0.074).
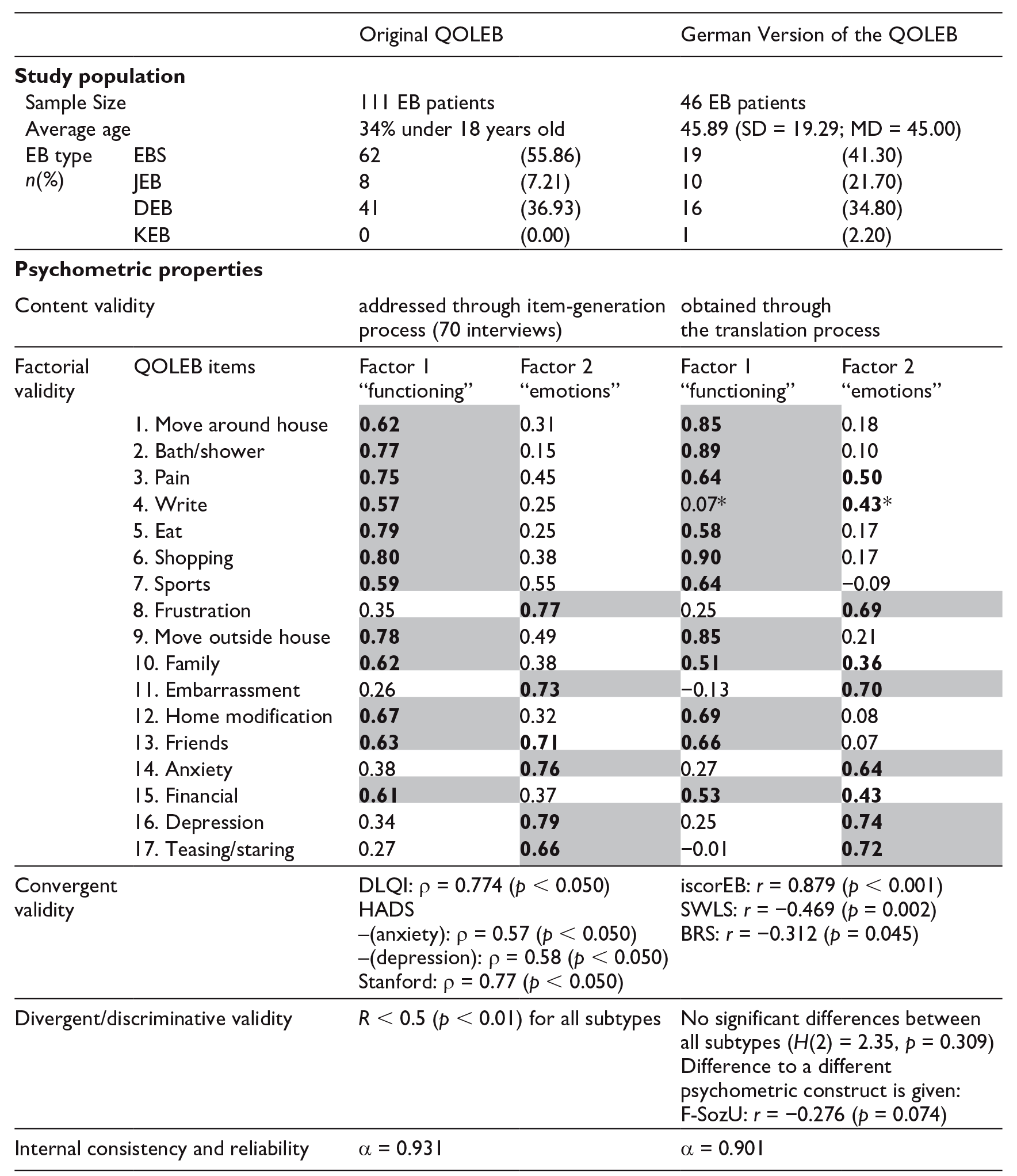
To evaluate the structure of the German version of the QOLEB, an exploratory factor analysis was performed. The Bartlett test (χ²(136) = 420.65, p < 0.001) and Kaiser-Meyer-Olkin Measure of Sampling Adequacy (KMO = 0.783) indicated that the data was suitable for a factor analysis. A principal component analysis with a varimax rotation was performed, yielding a four-factor solution, which accounted for 68.1% of the total variance. For comparability reasons to the original model, a reduction to a two-factor solution led to 54.3% of the total variance.
The psychometric properties and its comparison to the original version can be found in Table 5.
Comparison of the psychometric properties between the original QOLEB (Frew et al., 2009) and the German version.
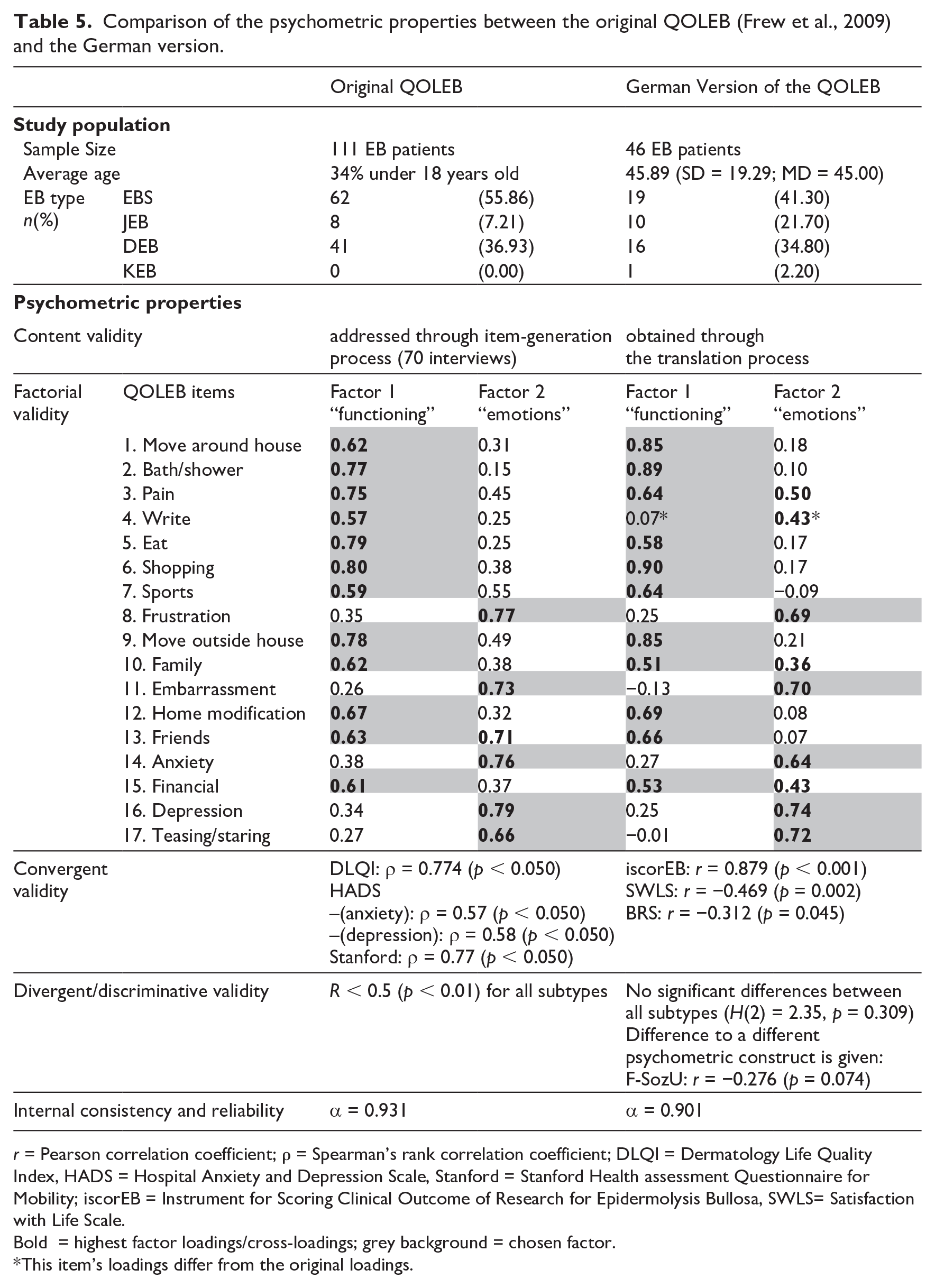
r = Pearson correlation coefficient; ρ = Spearman’s rank correlation coefficient; DLQI = Dermatology Life Quality Index, HADS = Hospital Anxiety and Depression Scale, Stanford = Stanford Health assessment Questionnaire for Mobility; iscorEB = Instrument for Scoring Clinical Outcome of Research for Epidermolysis Bullosa, SWLS= Satisfaction with Life Scale.
Bold = highest factor loadings/cross-loadings; grey background = chosen factor.
This item’s loadings differ from the original loadings.
Discussion
The aim of the current study was to develop and validate a culturally adapted German version of the QOLEB.
Regarding the sample and its characteristics, the sample size in this study is small compared to general requirements for validation studies. However, due to the rarity of EB, the sample size is quite adequate (Ragni et al., 2012) and also in line with previous QOLEB translation studies (Bishnoi et al., 2022; Cestari et al., 2016; Dănescu et al., 2019; Frew et al., 2013; Villar Hernández et al., 2022; Yazdanshenas et al., 2020; Yuen et al., 2014).
The distribution of EB types within the sample is approximately equivalent to the distribution in clinical practice (Bardhan et al., 2020), with EBS as the most frequent and Kindler as the rarest EB type. Many participants could not specify their EB subtype, therefore, no further subdivision can be provided. To account for the variety of clinical status (Has et al., 2020), a patient-reported degree of severity is used as a proxy in this context. The reduction in quality of life with an increase in disease severity has previously been confirmed in the literature (Jeon et al., 2016) and was also significant in our sample. No significant difference was found between EB types in the present sample, which is consistent with reports for the Brazilian Portuguese and the Farsi translation of the QOLEB (Cestari et al., 2016; Yazdanshenas et al., 2020).
Female participants, in comparison to male, are overrepresented within our sample, which might be related to higher responsiveness towards surveys (Curtin et al., 2000). Gender seems to have a significant impact on the QOLEB results according to our sample, which corresponds with previous findings (Tabolli et al., 2009). The age distribution in our sample is higher than in the original QOLEB score due to our minimum age of 16. Therefore, data was collected exclusively through self-reporting, which prevents the bias of proxy-report (Meyer et al., 2016).
The inclusion of participants from most countries with German as their main language allows to take cultural and language variety into account, which leads to higher validity of the data.
Content validity was obtained through the forward-backward translation of the QOLEB, as well as through the comparison of the principal component analysis of the original QOLEB and the German version. The factor analysis shows a high similarity between the original and the German QOLEB. Even though a four-factor solution could explain 68.1% of the total variance, we opted for the two-factor solution, which is very similar to the original version in all items (except the fourth item ‘write’) and in regards to overall variance (54.3%, original version 55.6%). The differing item ‘write’ may be explained with a technology-induced change of habits in everyday writing (Mangen, 2018).
Hereby, the German questionnaire has nearly the same psychometric properties as the original version (Epstein et al., 2015), which confirms internal consistency.
The combination of physical and psychosocial aspects of quality of life was taken into account in convergent validity, which was confirmed via comparison to the iscorEB for physical aspects and the SWLS and the BRS for psychosocial aspects. Divergent validity was confirmed by differentiation to another psychological construct, the F-SozU for social support.
Accordingly, the original construct can be confirmed for the German translation and can be validated as such.
Conclusions and outlook
Overall, the comparison with the original data shows that the German QOLEB is a valid and reliable survey instrument for the quality of life of EB patients. Therefore, we recommend the inclusion of the German QOLEB as a standard in clinical settings and research in all German-speaking countries to establish a holistic and patient-based perspective.
Supplemental Material
sj-docx-1-hpq-10.1177_13591053231221369 – Supplemental material for Translation, cultural adaptation and validation of the German Quality of life in Epidermolysis Bullosa (QOLEB) questionnaire
Supplemental material, sj-docx-1-hpq-10.1177_13591053231221369 for Translation, cultural adaptation and validation of the German Quality of life in Epidermolysis Bullosa (QOLEB) questionnaire by Gudrun Salamon, Sophie Strobl, Ursula Field-Werners, Tobias Welponer, Dédée F Murrell and Anja Diem in Journal of Health Psychology
Supplemental Material
sj-pdf-2-hpq-10.1177_13591053231221369 – Supplemental material for Translation, cultural adaptation and validation of the German Quality of life in Epidermolysis Bullosa (QOLEB) questionnaire
Supplemental material, sj-pdf-2-hpq-10.1177_13591053231221369 for Translation, cultural adaptation and validation of the German Quality of life in Epidermolysis Bullosa (QOLEB) questionnaire by Gudrun Salamon, Sophie Strobl, Ursula Field-Werners, Tobias Welponer, Dédée F Murrell and Anja Diem in Journal of Health Psychology
Footnotes
Acknowledgements
Our biggest thanks go to our study participants: thank you for sharing your experience with us. We thank everybody who contributed to the translation process, especially to Layla Alsalih, Nina Dominik, Vinzenz Hübl, Viktoria Kaczor, Elisabeth Schneider and Teresa Schneiderbauer. We want to thank Leena Bruckner-Tuderman, Cristina Has, Anita Keller and Sophie Kitzmüller for giving us access to previous translation versions. The proprietary of the QOLEB utilised in the study were made possible through the contribution of Prof. Dedee Murrell, Department of Dermatology, University of New South Wales - St George Hospital and the Australasian Blistering Diseases Foundation. Thank you to DEBRA Austria, DEBRA International and to the EB House Austria team for your invaluable support during the data collection phase. Thanks to Jan Aden and David Seistock for sharing your statistical expertise and to Natalie Rodax for your valuable feedback during the writing process.
Author contributions
The study conception, design, material preparation and data collection were performed by Gudrun Salamon and Sophie Strobl. Dédée Murrell contributed with her insights into the original test design. Data preparation and analysis were conducted by Ursula Field-Werners and Sophie Strobl. The first draft of the manuscript was written by Gudrun Salamon, Sophie Strobl and Ursula Field-Werners. Tobias Welponer contributed with a fundamental revision process. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. The whole process was supervised by Gudrun Salamon and consulted by Anja Diem.
Data sharing statement
The data generated during and/or analysed during the current study are not publicly available nor are they available on request due to the rarity of EB, which limits anonymity even with pseudonymisation or exclusion of personal data.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported and funded by DEBRA Austria.
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Ethical approval was granted by both the Medical and the Psychological Ethics Committees of the Sigmund Freud University Vienna (LBVCR72KAUBQ6188306).
Informed consent
Informed consent or assent was obtained from all participating patients or legal guardians of underage patients.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.