Abstract
The current study-within-a-trial explored individuals’ decisions to decline participation in research trialling a chronic illness-focused therapy (i.e. multiple sclerosis). Four themes were identified from seven semi-structured interviews with participation decliners and were confirmed by the host trial’s Patient & Public Involvement (PPI) panel: acknowledgement of the value of research; ‘fit’ of the study; misinterpretation of participant information; and ‘ignorance is bliss’ – discussed in light of theory and research. This study-within-a-trial extends research on trial recruitment and participation decline; while also suggesting that PPI can be utilised in both a practical and impactful manner.
Keywords
Background
Randomised Controlled Trials (RCTs) are often considered the gold standard of clinical trials (Hariton and Locascio, 2018), generally consisting of a systematic research strategy for determining an intervention’s efficacy. However, recruitment of participants to a RCT can be a challenging process; for example, with respect to achieving the pre-determined sample necessary to reliably derive conclusions (e.g. so as to avoid false-negative results in light of statistical non-significance). Thus, the enhancement of recruitment strategies has become an important objective in many RCT designs, as research suggests that poor recruitment rates often result in elevated cost and prolonged use of ineffective or potentially harmful extant interventions (Huang et al., 2018; Hughes-Morley et al., 2016).
One potential method of better understanding strategies for recruitment (and perhaps subsequent retention) is the exploration of the views of those who declined to participate in a RCT. However, this method is often overlooked given the substantial lack of research evaluating the reasoning behind refusal to participate in research and RCTs in particular (e.g. Barnes et al., 2012; Canvin and Jacoby, 2006). Notably, of the few studies that have assessed participation decline/refusal, many have done so quantitatively (e.g. Blanch et al., 2008; Harrison et al., 2016; Langford et al., 2014); however, given that the data to be assessed, in this context, come from individuals who decline to further participate, it is often the case that these data are few and limited – for example, to demographic information only. Thus, a substantial portion of extant research in this area lacks detail regarding the reasons individuals decline to participate, such as that which might be garnered through subsequent qualitative data collection. Nevertheless, the dearth of research that has been conducted suggests that potential participants’ perceptions should be accounted for, including patient perceptions of the intervention’s potential efficacy (Arfken and Balon, 2011).
While some qualitative research has explored more detailed reasons for declining (e.g. negative feelings and experiences influencing perception of the therapy; perceived ineligibility; and misunderstandings about the research (Barnes et al., 2012)), the literature also recommends that more robust research needs to be conducted to further inform the currently lacking evidence base in this particular area (Beijers et al., 2016), which is important considering that the percentage of those who decline – regardless of reason(s) – is not accounted for in terms of accurately representing the population under investigation. Of course, this methodological ‘flaw’ is inherent in all research that studies human participants; however, enhanced efforts to diminish decline and enhance recruitment is not only important for statistical purposes, but also for strengthening the validity of conclusions with respect to implementation in ‘real-world’ scenarios. Thus, the aims of this qualitative study-within-a-trial (SWAT) is to explore individuals’ decisions to decline participation in a research programme trialling a therapy related to their chronic illness (see
Methodology
Study context
The qualitative SWAT took place within the context of a RCT of a Cognitive, Occupation-Based programme for people with Multiple Sclerosis (Dwyer et al., 2020) – from here on referred to as the host trial, which is a single-blind, cluster-randomised controlled feasibility trial, using a two-arm (i.e. treatment and wait-list controls), pre-post study design and two additional follow-up testing times (i.e. 12 weeks and 6 months). Within the host trial, potential participants were recruited through advertisement in relevant newsletters (e.g. monthly
Notably, recruitment was halted after 4 months by arrival of the COVID-19 pandemic. Also noteworthy is that potential participants were provided one of two possible participant information sheets (see Online Supplemental Material), both of which included all information pertinent to study procedures, informed consent and mainly differed with respect to formatting and wording. This design manipulation was implemented consistent with the aims of another one of the host trial’s SWATs, which was to compare the effects of two patient information sheets (PISs): a patient-designed-and-informed PIS and a standard, researcher-designed PIS on: recruitment, decision certainty, retention; understanding, readability, accessibility, likeability and decision to consent (Dwyer et al., 2020). As a result, all individuals expressing interest in the host trial had a 50% chance of receiving the patient-developed PIS or the researcher-developed PIS.
Study design
A series of semi-structured, one-on-one telephone interviews were conducted to explore decliners’ reasons not to participate in the host trial, as well as their perception of the research. An inductive, interpretive qualitative approach was implemented in order to facilitate exploration and a deeper understanding of participants’ perspectives, the identification of clear and relevant themes (Willig and Rogers, 2017) and the gaining of insight into participants’ experiences of little understood topics (Silverman, 2013). Following preliminary analysis of the interview data (Phase 1), a focus group was conducted with the host trial’s
Participants
Participants were recruited from the host trial’s list of individuals who expressed interest in participating, but ultimately declined to take part. Eligibility criteria for this SWAT were: (1) that individuals were eligible to take part in the host trial (see Author, 2020a for these criteria); and (2) these individuals explicitly declined to take part in the host trial (as opposed to ‘not responding’). Recruitment for decliner interviews ceased upon the arrival of the COVID-19 pandemic, given its potential contamination of qualitative data regarding reasons for declining to participate.
Materials
An Olympus Digital Voice Recorder WS-852 was used to record the telephone interviews and focus group. The semi-structured interview guide (see Table 1) was developed in light of the aim of exploring participant reasoning for declining to take part in the host trial and was further informed by: researcher observation during the recruitment process; expert review; and PPI input. Following development, the interview guide was pilot-tested with the research team’s PPI panel. Notably, in an effort to reduce bias, PPI members involved with the Phase 2 focus group were not included in the development or pilot-testing of the interview guide. Coding and analyses were supported by NVivo 11 (2015) software. The COREQ checklist (Tong et al., 2007) provided a framework for study development and guidance for reporting of results.
Semi-structured interview guide.*

The PPI panel was asked to comment, discuss and/or elaborate on the interview responses to these semi-structured questions.
Procedure
Participants were recruited through purposive sampling (i.e. individuals who declined to participate in the host trial) and provided informed consent to participate in an audio-recorded telephone interview. Upon ceasing recruitment for decliner interviews, 228 individuals had expressed interest in the host trial, of whom 110 were both eligible and consenting, with another 64 ineligible. As 37 of the 54 remaining individuals halted correspondence, the researchers were unable to make contact regarding an invitation to participate in an interview exploring reasons for ‘declining’, if applicable. Thus, the 17 individuals who communicated their ‘decline’ were contacted to participate in the interviews. Specifically, the 17 individuals eligible for the SWAT were telephoned by a member of the research team (either a research assistant or post-doctoral researcher) the asked to take part. Those agreeing to participate were sent an information sheet and consent form and returned their informed consent either by post or email. Following recruitment, a series of seven semi-structured interviews were conducted between January and May 2020 (mean duration = 10 m 55 s; ranging between 5 and 19 minutes) by a research assistant or post-doctoral researcher. Both researchers were involved with and knowledgeable of the host trial. Both were also experienced in qualitative research. The audio recordings were transcribed verbatim by two researchers and anonymised. The transcripts were then checked for both accuracy and missing data by both the interview facilitator and another member of the research team, prior to conducting the data analysis.
With respect to the focus group, three of five PPI panel members of the host trial were available to accept invitation to participate in an online focus group. Panel members were first presented and asked the same semi-structured interview questions as the decliners (without amendment and bearing in mind the perspective of interviewees, in light of having only engaged their allocated PIS) as a means of further exploring consistency between what the panel identified as potential reasons for decline and actual Phase 1 responses. The PPI panel was asked these questions prior to being advised of the decliners’ responses in order to limit any potential bias. Notably, PPI focus group members were not previously involved in the development of the PPI-developed PIS. Following discussion of the panel’s responses, PPI members were then presented the decliners’ various reasons for refusing participation; and asked to comment and elaborate, in light of both their perspectives regarding potential reasons for decline (as previously discussed) and their working understanding of the host trial. The resulting audio data from the focus group (duration 1 h 45 m) were again transcribed verbatim and checked for accuracy and missing data by the focus group facilitator (i.e. the post-doctoral researcher), prior to analysing focus group data.
Data analysis
Data were analysed by three members of the research team. Consistent with Braun and Clarke (2020), analysis was conducted by the researchers through: familiarisation with the data; coding and theme identification; and the reviewing and refining of themes. Specifically, familiarisation with the data began during data collection and involved the reading and re-reading of the interview transcripts, accompanied by observational note-taking. Researchers then systematically generated concise, meaningful codes, followed by identification of patterns from/within the data, both of which were discussed by the researchers. Themes were then identified in relation to one another, before completion of the iterative review and refinement of these themes against the transcripts, for the purpose of ensuring their credibility (Mays and Pope, 2000). As such, trustworthiness of the data and credibility of the findings were ensured through a variety of methods, including triangulation (e.g. multiple observers/observations and analysts/analyses), the immersion of researchers into the data for the purpose of ensuring rich descriptions and the consultation of the PPI panel. Notably, member-checking was deemed inappropriate for ensuring trustworthiness during the design of this study in an effort to avoid creating burden for the host trial’s ‘declining’ cohort.
Findings
Though a majority of the 17 ‘decliners’ shared reasons for declining (see Table 2 for reasons provided by the 17 decliners; NB: three individuals provided multiple reasons), only seven (
Reasons for declining.
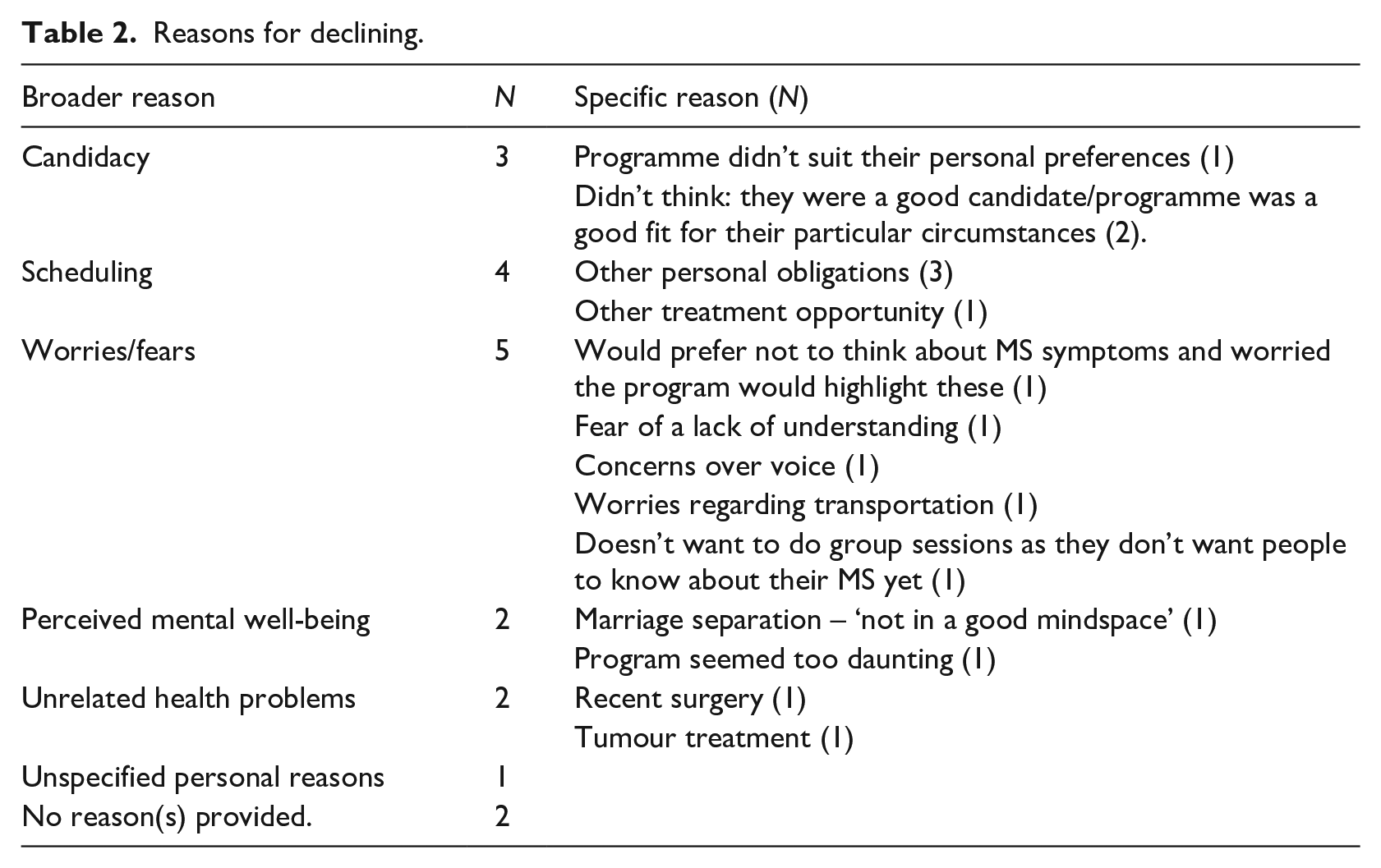
Phase 1
Consistent with the aims of the SWAT, results from a series of seven semi-structured interviews with host trial ‘decliners’ revealed a number of reasons behind the decision to decline participation in the host trial (see Table 3), including mostly personal reasons – as opposed to perceived problems with the program within the host trial. With respect to this thematic analysis, four themes were identified, including: (1) Acknowledgement of the Value of Research; (2) ‘Fit’ of the Study; (3) Misinterpretation of Participant Information; and (4) ‘Ignorance is Bliss’.
Participant characteristics.
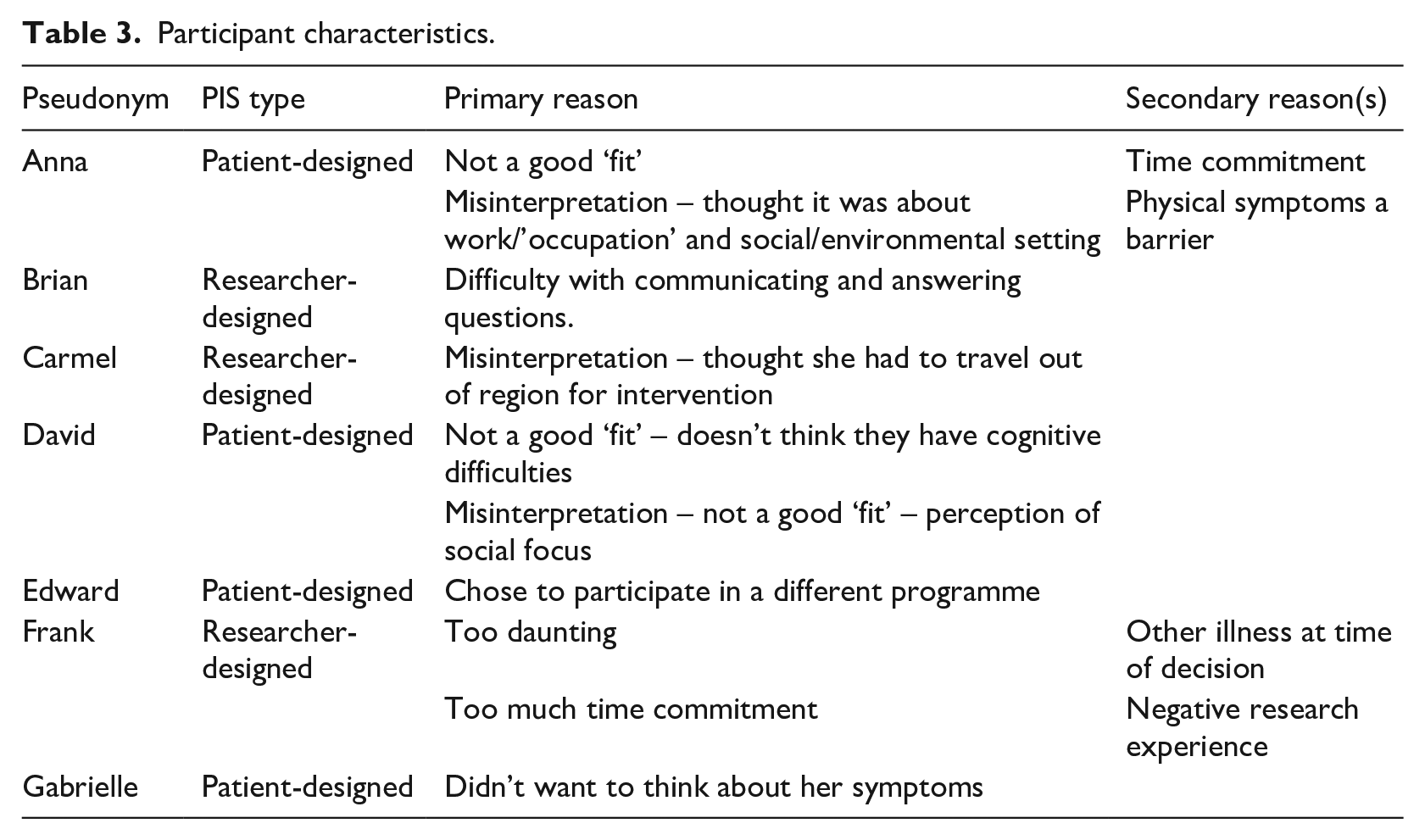
Theme 1: Acknowledgement of the Value of Research
The first theme explains that despite declining to participate, decliners still viewed research, their interest in it and its potential value in a positive light. Given that potential participants for the host trial were required to contact the research team with an expression of interest in order to be sent further information on the trial, it is not surprising that initial thoughts of all seven decliners were positive, though slightly different from one another with respect to the focus of interest. For example, whereas Anna, Frank and Edward were interested specifically in learning about the cognitive aspects of the programme, Brian, David and Gabrielle implied that they were interested because it could be of potential benefit – be it for themselves or others. Carmel was interested in the programme in order to learn more about MS from the standpoint of valuing MS-focused research. That is, though feeling it necessary to decline (as discussed below), a major focus of Carmel’s was the ‘value of research’:
Not only was the value of research acknowledged from an epistemological standpoint, but also from the perspective of being interested in getting actively involved, particularly for the purposes of benefitting others, as well as one’s self. For example,
Theme 2: ‘Fit’ of the study
Whereas Theme 1 focused on decliners’ accounts of why they would have liked to participate in research like the host trial, exposition on both the explicit and more ‘underlying’ reasons why a decision to decline was made begins in the second theme. Specifically, Theme 2 addresses, through three sub-themes (i.e. too much commitment; comorbidity and other symptoms; as well as candidacy), participants’ perceptions of the study’s relevance to them with respect to ‘fit’. With respect to ‘too much commitment’, some of the decliners indicated that the coupling of the study’s duration and time requirements. . .
. . .along with what Frank suggested as the potentially daunting nature of a programme like the host trial, the commitment involved and how it would potentially interfere with life. For example, as Frank put it:
Though no refusal to participate by interviewees was based primarily on ‘comorbidity and other symptoms’ (i.e. the second sub-theme), it was a concern expressed as a secondary reason for declining to take part (see again, Table 3). For example, Anna states:
Brian also emphasised particular symptoms or disease affecting his ability to participate:
The final sub-theme of ‘fit of the study’ referred to decliners’ perceptions regarding their candidacy for the host trial, in light of the participant information sheet and advertisements made available to them. For example, according to Frank:
In David’s case, following review of the participant information sheet, he recognised that the programme wasn’t a good ‘fit’ for him given that he reports not having cognitive difficulties – which is the primary focus of the host trial:
Likewise, Anna also recognised not being a good candidate or ‘fit’ for the programme; however, Anna’s recognition, in this context, was based on a misinterpretation of the trial’s focus:
Anna’s response is particularly notable for multiple reasons. First, of course, it exemplifies the importance of the notion of a study’s relevance and fit with a potential participant, but it also exemplifies both a simplification of decision-making and, to some extent, assuredness in this process, even though the decision is being made from a false – or, more accurately, misinterpreted premise. Such examples suggest not only the importance of potential participants ensuring that they understand the research they choose to participate/not participate in, but also the need for researchers to provide sufficient opportunities to clarify any misinterpretations such individuals may have. In light of this last point, Anna’s quote provides segue to the concept of
Theme 3: Misinterpretation of participant information
Though the host trial does invite people who work to take part, the intervention is not restricted to this demographic and is just as relevant to those who do not work. Though there were indeed three scenarios provided as examples in the patient-designed PIS (see Online Supplemental Material), for how cognitive difficulties might impact on daily functioning, only one addressed ‘work’ (as well as one that addresses socialising, which perhaps account for the misinterpretation in Anna’s previous statement regarding ‘fit’ and ‘operating in groups’). It is also possible that the name of the trial could have facilitated this confusion – a ‘Cognitive Occupation-Based Programme for People with MS’; however, the use of ‘occupation’ in this context refers to discipline of occupational therapy – from which the programme is informed.
Consistent with Anna’s focus on ‘operating in groups’, David may have also misinterpreted the target demographic:
With respect to the sub-theme of misinterpretation of study requirements, Carmel addressed her inability to take part in the host trial as a result of (misinterpreting) the need for lengthy travel. For example:
Another example of the sub-theme of misinterpretation of study requirements is akin to ‘too much commitment’ under the second theme, ‘fit’ – according to Frank:
Notably, whereas the first half of Frank’s response reflects a misinterpretation of what might be required of him (i.e. level of commitment), what follows reflects a position of not wanting to waste resources in light of such misinterpretation, which is also consistent with valuing research, as in the first theme.
Theme 4: ‘Ignorance is Bliss’
The final theme was the concept of
Though David did not decline primarily as a result of this concept (rather, ‘fit’), he does address the notion of ‘
Phase 2
Following responses to the semi-structured interview guide and subsequent discussion of findings from Phase 1, overall, the results of the PPI panel’s focus group were largely consistent with the themes identified within Phase 1. For example, according to Amy, in relation to Phase 1 responses:
The PPI panel also expressed surprise about ‘getting’ as much data as researchers’ collected during Phase 1. For example, Cindy noted that:
With respect to the theme of ‘value of research’, the PPI panel likewise acknowledged the potential benefits of taking part in research, be it for themselves or others. Consistent with Carmel’s perspective in Phase 1, they also noted how some research programmes, such the host trial, have the added value of potentially ‘filling in gaps’ with respect to a lack of services. For example, according to Cindy:
Furthermore, the PPI panel also indicated that one’s valuing of research may result from their background or knowledge of science. For example:
Notably, the PPI panel also identified the concept of knowledge background as an important aspect of ‘misinterpretation of participant information’ (i.e. Theme 3 from Phase 1); for example:
The PPI panel also shed further light on the theme of ‘ignorance is bliss’ through their discussion of various stages of acceptance in living with MS, in which case ‘ignorance is bliss’ was equated with ‘denial’ as a sort of coping mechanism. For example:
In this context, acceptance of MS was also described by the PPI panel as occurring on both an emotional and an intellectual level, in which the relationship between these ‘levels’ may play an important role their decision to decline. For example, though potential participants may have been prepared to engage in the research process on an intellectual level (e.g. finding out more information about their illness), they may subsequently decline as a result of the perceived emotional stress associated with discussing their MS in social settings. According to Amy:
The panel also indicated that the ‘ignorance is bliss’ theme may somewhat, similarly, involve a reluctance to place oneself in situations where they might compare themselves with others who have the same illness, such as the group sessions involved in the host trial’s intervention; wherein decliners may have felt forced to confront their own symptoms. For example, according to Cindy:
Concerns regarding this social-emotional aspect of ‘ignorance is bliss’ also applies to the theme of ‘fit of the study’, in that anxiety over participating in groups may stem from a lack of confidence in social situations, as a result of their MS; thus, being a poor fit for these individuals and, subsequently, influencing their decision to not partake in the research. Notably, this could be of particular significance to Anna’s decision not to participate as a result of her not ‘operating in groups’ (see Phase 1). For example, according to Amy:
Specific unto ‘fit of the study’, the panel indicated that the potential for ‘fit’ could be split into two main categories, including contextual reasons for declining regarding general practicalities around participating (such as the sub-themes from Phase 1 interviews: too much commitment; candidacy; and comorbidity and other symptoms):
. . .as well as reasons for declining as a means of self-preservation and avoiding feelings of being overwhelmed. With respect to this second ‘category’, Cindy described the importance of being mindful of not overworking oneself for fear of her health deteriorating:
She also highlighted the importance of knowing one’s threshold for taking on too much:
. . .and an awareness of not over-burdening herself to the point of being overwhelmed:
Discussion
Interpretation of results
Results from the SWAT revealed a number of reasons behind the decision to decline participation in the host trial, most of which were of a personal nature, rather than perceived problems with the host trial’s intervention (e.g. concerns regarding candidacy, scheduling, mental-well-being, unrelated health problems, as well as other worries and fears). Furthermore, four themes were identified which, cumulatively, suggested that though decliners see the value of research aimed at their particular chronic illness, there remained the potential for declining as a result of either misconstruing the details of the study or perceiving participation as not being a good fit for them. For example, some may already feel at capacity with respect to how they cope with their MS. Thus, not participating may be their way of prioritising their own health (e.g. ‘ignorance is bliss’).
These results are consistent with research by Barnes et al. (2012), who also found that perceptions of candidacy/ineligibility, as well as misinterpretation of study-related information were important reasons for why potential participants often decline. The latter issue is also akin to research findings by Moynihan et al. (2012), regarding confusion about/misinterpretation of trial processes. Likewise, acknowledging the value of research is also consistent with past research (e.g. Canvin and Jacoby, 2006), given that it is a common feature for initially expressing interest in participation. Findings are further consistent with recent research by Mundy et al. (2020), who also found ‘fit’ and ‘time commitment’ (i.e. ‘It’s not for us’) as important (sub)themes regarding participation decline. However, the notion of ‘fit’ and ‘ignorance is bliss’, from the perspective of coping and psychological well-being, appear be to novel – though not entirely surprising – findings that were identified, developed and refined through analysis and discussion as major themes during both Phase 1 and 2 of the SWAT. As indicated, though potential participants may often decline because of misinterpretation or more practical reasons (e.g. scheduling; time commitment; perceptions of candidacy/eligibility; and/or interference from symptoms or comorbid illnesses), they may also decline for more subtle, psychologically-based reasons consistent with their preferred strategies of coping with their illness – be it adaptive (e.g. to avoid overwhelm) or maladaptive (e.g. denial).
Nevertheless, the decision-making process may not always be as automatic or as easy as some participants in this SWAT suggest it to be. According to research by Hughes-Morley et al. (2016), the decision to decline might resemble a four-stage process that is somewhat consistent with many of the themes and sub-themes identified in this SWAT. For example, individuals interested in the research first assess potential participation in the context of their experiences and attitudes. If they remain positive, they then assess their eligibility (e.g. candidacy); thereafter, they assess their level of need for the intervention and its potential benefit (see also Cohen-Mansfield, 2003); and then, finally, they conduct a cost-benefit analysis of participation (e.g. will the potential benefits outweigh the time and effort required to participate). From this perspective, though the decision to decline may not be as automatic or as easy as suggested in this SWAT, it also implies that there are a variety of opportunities to decide to decline before coming to the decision to consent.
Limitations
Though the results of this qualitative SWAT yielded a number of interesting findings, there are multiple limitations that must be considered. For example, Phase 1 interviews were relatively short with respect to what was anticipated by the research team and, similarly, it could be argued that the depth of the data’s qualitative ‘richness’ was lacking – which was addressed by the PPI panel. Though it was anticipated that interviews would be approximately 15–20 minutes each in length, in retrospect, it seems reasonable to suggest that the brevity of the interviews resulted from participants having said all they had to say on the topic. This recommendation is made given: the nature of participants not actually having taken part in the host trial and only being able to report on why they declined (in light of the PIS); and not as a result of the scope of the interview guide (again, see Table 1) or the experience of the researchers. Moreover, despite anticipating longer interviews, the research team was also aware of this potentiality; hence designing the study to incorporate the PPI panel’s focus group, which was conducted in order to elaborate, further explain and interpret Phase 1 responses. Thus, while the length and lack of depth of Phase 1 interviews can be considered a potential limitation of the SWAT, the duration of the shortest interview is consistent with similar, recent research by Mundy et al. (2020) and the addition of a PPI panel focus group succeeded in adding strength to the SWAT by reinforcing efforts to interpret the data.
Another potential limitation that must be considered was the, arguably, small sample size of participants interviewed within Phase 1. As addressed in the
Conclusion
The results of this SWAT are informative with respect to identifying targets of focus in future research aiming to enhance recruitment to RCTs. For example, future research should aim to: (1) design and develop methods of sharing ‘participant information’ that reinforce the value of research, ensuring to make explicit the benefits of the research both to the individual and to others; (2) acknowledge that misinterpretation is likely and so, to avoid this, make greater efforts to ensure clarity and accessibility of information provided to potential participants; and (3) acknowledge that some individuals will not outweigh participation in favour of their own coping strategies – be it avoidance of overwhelm or isolation from large groups. Likewise, the findings also suggest that PPI can be utilised in both a practical and impactful manner, such as in the SWAT, wherein a PPI panel worked with researchers in a focus group to help elaborate on and interpret qualitative data.
Research Data
sj-docx-1-hpq-10.1177_13591053211037736 – for A qualitative investigation of reasoning behind decisions to decline participation in a research intervention: A study-within-a-trial
sj-docx-1-hpq-10.1177_13591053211037736 for A qualitative investigation of reasoning behind decisions to decline participation in a research intervention: A study-within-a-trial by Christopher P Dwyer, Anusha Moses, Fionnuala M Rogers, Dympna Casey, Robert Joyce and Sinéad M Hynes in Journal of Health Psychology
Supplemental Material
sj-pdf-2-hpq-10.1177_13591053211037736 – Supplemental material for A qualitative investigation of reasoning behind decisions to decline participation in a research intervention: A study-within-a-trial
Supplemental material, sj-pdf-2-hpq-10.1177_13591053211037736 for A qualitative investigation of reasoning behind decisions to decline participation in a research intervention: A study-within-a-trial by Christopher P Dwyer, Anusha Moses, Fionnuala M Rogers, Dympna Casey, Robert Joyce and Sinéad M Hynes in Journal of Health Psychology
Supplemental Material
sj-pdf-3-hpq-10.1177_13591053211037736 – Supplemental material for A qualitative investigation of reasoning behind decisions to decline participation in a research intervention: A study-within-a-trial
Supplemental material, sj-pdf-3-hpq-10.1177_13591053211037736 for A qualitative investigation of reasoning behind decisions to decline participation in a research intervention: A study-within-a-trial by Christopher P Dwyer, Anusha Moses, Fionnuala M Rogers, Dympna Casey, Robert Joyce and Sinéad M Hynes in Journal of Health Psychology
Supplemental Material
sj-pdf-4-hpq-10.1177_13591053211037736 – Supplemental material for A qualitative investigation of reasoning behind decisions to decline participation in a research intervention: A study-within-a-trial
Supplemental material, sj-pdf-4-hpq-10.1177_13591053211037736 for A qualitative investigation of reasoning behind decisions to decline participation in a research intervention: A study-within-a-trial by Christopher P Dwyer, Anusha Moses, Fionnuala M Rogers, Dympna Casey, Robert Joyce and Sinéad M Hynes in Journal of Health Psychology
Footnotes
Acknowledgements
The authors would like to acknowledge, with many thanks, the members of the public and patient involvement panel for their involvement panel for their involvement in this research, which greatly facilitated the interpretive aspect of the inferences drawn in the current study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Declarations
Ethics approval was awarded by Galway University Hospitals on 13.08.2019 Ref: C.A 2231 and NUI Galway Research Ethics Committee (19-Oct-10). Participation in this study was based on informed consent. The protocol for this study-within-a-trial (SWAT) was registered with the Northern Ireland Network for Trials Methodology Research (SWAT-139).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study-within-a-trial was conducted within research funded by the Health Research Board (Ireland) under a Definitive Interventions and Feasibility Award (DIFA-FA-2018-027).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
