Abstract
Introduction:
Peripheral artery disease (PAD) is a common manifestation of atherosclerosis. Despite the effectiveness of endovascular therapy (EVT), patients with PAD often face poor prognoses. This study investigates the relationship between specific inflammatory indices and wound healing in patients with Fontaine stage 4 chronic limb-threatening ischemia (CLTI).
Methods:
From June 2021 to January 2024, 168 patients with Fontaine stage 4 CLTI, totaling 185 affected extremities, who underwent successful EVT, were assessed retrospectively. Patients were categorized based on wound healing post-EVT. The Naples prognostic score (NPS), pan-immune inflammation value (PIV), systemic immune-inflammation index (SII), and systemic inflammation response index (SIRI) were calculated. Discrimination and decision curve analyses were used to explore the link between inflammation and wound healing.
Results:
Wound healing was observed in 142 (76.8%) patients post-EVT. Nonhealing patients exhibited higher indices of NPS, PIV, SII, and SIRI. NPS (aOR = 0.381, 95% CI 0.215–0.675, p = 0.001), PIV (aOR = 0.997, 95% CI 0.996–0.999, p < 0.001), SII (aOR = 0.997, 95% CI 0.996–0.997, p < 0.001), and SIRI (aOR = 0.443, 95% CI 0.313–0.625, p < 0.001) were independently predictive of wound healing. SIRI (AUC = 0.840, 95% CI 0.777–0.904) demonstrated superior predictive ability compared to PIV (AUC = 0.799, 95% CI 0.722–0.876, pdif < 0.001), SII (AUC = 0.788, 95% CI 0.712–0.865, pdif < 0.001), and NPS (AUC = 0.760, 95% CI 0.681–0.838, pdif < 0.001). SIRI also showed higher net reclassification improvement over PIV (68.4%, pdif < 0.001), SII (38.5%, pdif = 0.024), and NPS (29.8%, pdif = 0.079). All inflammatory indices, especially SIRI, provided prognostic value in determining wound healing at high treatment thresholds (> 40%).
Conclusion:
In patients with CLTI undergoing EVT, SIRI, SII, PIV, and NPS may help predict the potential for wound healing during in-hospital follow up, with SIRI being the strongest predictor.
Keywords
Introduction
Peripheral artery disease (PAD) is a manifestation of systemic atherosclerosis, commonly affecting the lower extremities. PAD presents a diverse range of clinical symptoms, varying from an asymptomatic condition to chronic limb-threatening ischemia (CLTI), which may require urgent surgical or endovascular interventions. 1 CLTI is a clinical syndrome characterized by symptoms such as rest pain, nonhealing wounds, or tissue necrosis (gangrene), which pose a significant risk of limb loss and cardiovascular events. It has been identified in up to 11% of the more than 200 million patients diagnosed with PAD globally. 2 In addition, up to 10% of individuals diagnosed with PAD may develop CLTI, with up to 25% of this subgroup eventually undergoing a major lower-extremity amputation. 3 In the management of CLTI, primary objectives include alleviating ischemic pain, promoting wound healing, enhancing limb function and mobility, preserving the limb, and extending survival. Endovascular therapy (EVT) and surgical bypass are the primary revascularization approaches for patients with salvageable limbs. EVT can restore blood flow to the distal extremity, offering a minimally invasive option with a shorter hospital stay compared to surgery. Several studies have demonstrated that endovascular procedures are associated with improved amputation-free survival and reduced mortality.4,5 Regrettably, there is a scarcity of literature regarding prognostic factors for life expectancy and limb salvage following EVT in patients with CLTI.
In addition, the progression of PAD is also closely tied to inflammation and nutritional status. Plasma levels of inflammatory and nutritional markers may be used to predict the progression of PAD.6,7 The vascular endothelium regulates crucial processes like coagulation and inflammation, and its dysfunction significantly contributes to atherosclerosis by promoting thrombosis and leukocyte adhesion. In PAD, impaired endothelial function is linked to circulatory failure and elevated inflammatory markers. 8 Key white blood cell components—monocytes, neutrophils, and lymphocytes—serve as biomarkers for systemic inflammation. Monocytes and neutrophils exacerbate atherosclerosis, whereas lymphocytes may modulate it.9,10 Studies show that lymphocyte count and neutrophil-to-lymphocyte ratio (NLR) correlate with survival and limb amputation rates in CLTI.11,12 Platelets promote atherosclerosis by adhering to vessel walls and releasing factors that enhance plaque formation. 13 Wound healing is also influenced by nutritional status.14,15 Serum albumin is widely recognized as a reliable indicator of nutritional status and malnutrition is associated with poorer survival, prolonged hospitalization, and an increased need for amputation.16–19 Previous studies have reported that several scoring systems combining immunological and/or nutritional indices can be prognostically valuable in predicting mortality and related events in patients with CLTI.20,21
The systemic immune-inflammation index (SII), systemic inflammation response index (SIRI), pan-immune inflammation value (PIV), and Naples prognostic score (NPS) are novel indices based on various combinations of peripheral blood cells, platelets, serum albumin, and total cholesterol (TC) (Supplementary Table 1). These indices may hold promise in providing significant insights into systemic inflammation and immune activation. Previous studies have identified NPS, SII, and SIRI as significant prognostic indicators for patients with PAD.22,23 Despite being a relatively recent addition to the literature, PIV has been the subject of limited studies regarding its application in atherosclerotic disease. By incorporating monocytes into the SII, recognized as central players in local inflammation and influential in modulating the inflammatory/antiinflammatory cascade across all phases, PIV emerges as a novel comprehensive inflammatory index, encompassing nearly all blood cell types. PIV has limited research in coronary artery disease (CAD) but is linked to long-term adverse cardiac events in patients with ST-segment elevation myocardial infarction (STEMI). Liu et al. observed a correlation between postprocedural PIV levels in patients with STEMI and the occurrence of long-term major adverse cardiovascular events. 24 NPS is calculated based on serum albumin, NLR, LMR (lymphocyte-to-monocyte ratio), and TC levels. Previous studies have indicated that NPS is a significant prognostic indicator for patients with chronic heart failure, myocardial infarction, and PAD. 23 A recent study highlighted a substantial association between NPS and adverse long-term outcomes among patients with PAD undergoing EVT. Furthermore, NPS emerged as an independent predictor for both all-cause mortality and amputation in this patient population. 23 However, it remains unclear whether the SII, SIRI, PIV, and NPS hold prognostic value in determining patient outcomes following EVT for CLTI. Therefore, in this current study, we aimed to explore the prognostic impact of SII, SIRI, PIV, and NPS, as well as their comparative advantages, on ischemic wound healing in patients with Fontaine stage 4 CLTI who underwent EVT.
Methods
Study design, study population, and definitions
From June 2021 to January 2024, 1034 patients with PAD were examined. Among these, 211 patients who underwent EVT at our department due to Fontaine stage 4 CLTI were evaluated for this retrospective study. Patients with chronic wounds (defined as those persisting for more than 2 weeks) were included in this study. The analysis incorporated both arterial and diabetic wounds (mixed wounds) as well as pure arterial wounds; venous ulcers were excluded from the study. Patients with a history of conditions that could have caused a systemic inflammatory response, including myocardial infarction (n = 4), acute heart failure (n = 3), liver failure (n = 3), stroke or transient ischemic attack (n = 5), or acute limb ischemia (n = 7) within 30 days prior to the procedure, were excluded. Additionally, patients with incomplete laboratory findings, those lost to follow up (n = 10), and those with inflammatory diseases such as immune system disorders or hematological malignancies affecting the immune system (n = 11) were excluded. Ultimately, the study sample comprised 168 patients who underwent successful EVT, encompassing a total of 185 affected extremities. Patients were then divided into two groups based on their wound healing following EVT: group 1 consisted of those with nonhealing wounds and group 2 included those with healing wounds. This study adhered to the principles specified in the Declaration of Helsinki. All participants provided written informed consent for the use of their data. The study protocol received approval from our institution’s ethics committee.
Baseline demographic, clinical, and laboratory characteristics were obtained from patients’ inpatient medical records and telephone interviews. We assessed various baseline characteristics, including age, sex, body mass index (BMI), smoking status, history of congestive heart failure (CHF), CAD, chronic obstructive pulmonary disease (COPD), dyslipidemia, regular hemodialysis, diabetes mellitus (DM), hypertension, atrial fibrillation, and cerebrovascular disease. The criteria for CAD, CHF, hypertension, and dyslipidemia were determined according to the guidelines established by the European Society of Cardiology. The ankle–brachial index (ABI) measurements 25 and findings from duplex ultrasound imaging were recorded. Routine laboratory tests, including white blood cell (WBC), neutrophil, lymphocyte, monocyte, and platelet counts, hemoglobin levels, lipid levels, albumin, C-reactive protein (CRP), and glucose, were analyzed. Preprocedural clinical data and medical history were documented. The left ventricular ejection fraction of each patient, determined using two-dimensional echocardiography, was recorded. The Fontaine classification was used to characterize the severity of clinical symptoms. This classification system delineates four stages of PAD, with Stage 3 denoting ischemic rest pain and Stage 4 signifying ulceration or gangrene. 26 According to the Trans-Atlantic Inter-Society Consensus (TASC) definition, ‘critical limb ischemia’ applies to any patient presenting with chronic ischemic rest pain, ulcers, or gangrene resulting from objectively confirmed arterial occlusive disease. 26 In our study, we enrolled patients classified as Fontaine stage 4.
Treatment protocols
Patients were categorized based on the primary locations of arterial involvement: aorto-iliac disease, femoropopliteal disease, infrapopliteal disease, and multilevel disease. EVT was administered to patients with confirmed angiographic severe stenosis or occlusion in a lower-extremity peripheral artery by experienced interventional cardiologists. The decision between EVT and open-surgery procedures was based on the patient’s overall condition, preoperative comorbidities, ischemic pain, and lesion characteristics, in accordance with the TASC II guidelines. 26 Detailed procedures of EVT have been described previously. 26 The interventional procedures, including balloon angioplasty, stent placement, atherectomy, and procedural complications, were documented. Procedural success was defined as the successful crossing of the culprit lesion with the guidewire and an appropriately sized balloon, followed by the successful completion of the procedure without any immediate technical complications, such as vessel perforation, dissection, thrombus formation, or distal embolization. Conversely, the procedure was considered unsuccessful if the lesion could not be crossed with the guidewire, the balloon could not be delivered to the lesion, or if there was no flow or slow flow after ballooning.27,28 Minor dissections and perforations were managed conservatively with observation and repeat imaging if they did not cause significant hemodynamic compromise. In more severe cases, further intervention was performed, such as additional balloon dilation, stent placement, or surgical repair.
Wound properties and healing
The Society for Vascular Surgery Lower Extremity Threatened Limb Classification System (SVS WIfI) was used to evaluate wound characteristics and infection status. 29 Wound healing was assessed until complete epithelialization was achieved. Complete wound closure was defined as ‘full epithelialization without drainage or no longer requiring additional dressing,’ the most commonly used definition by the Food and Drug Administration for healed wounds. 30 The wound healing interval was defined as the period from the revascularization of the peripheral artery to the complete epithelialization of the wound without evidence of active infection. If there were multiple wounds on a single extremity, complete healing was defined as the healing of all wounds on that extremity. In patients with wounds on both extremities, each extremity was evaluated separately. Major amputations were characterized by below-the-knee or above-the-knee amputations and minor amputations, distal to the ankle joint, were defined as preserving the ankle joint.
Calculation of prognostic scores
Blood samples were collected from all patients before EVT. SIRI and SII were calculated using the following formulas: SIRI = monocyte count × neutrophil/lymphocyte ratio; SII = platelet count × neutrophil/lymphocyte ratio. 22 PIV was calculated as neutrophil × platelet × monocyte / lymphocyte. 31 NPS was calculated based on the following four metric scores: serum albumin (≥ 4 g/dL = 0, < 4 g/dL = 1), TC (> 180 mg/dL = 0, ≤ 180 mg/dL = 1), NLR (≤ 2.96 = 0, > 2.96 = 1), and LMR (> 4.44 = 0, ≤ 4.44 = 1). The NPS was then evaluated as the sum of these scores. 23
Statistical analysis
The statistical analyses were conducted using R statistical software, version 4.3.2 (R Foundation for Statistical Computing, Vienna, Austria). The normality of the variables was assessed through the Kolmogorov–Smirnov test, in combination with the inspection of histograms and probability plots. The continuous variables were expressed as the mean ± SD for the data that were normally distributed, and as the median (IQR25–75) for the data that were not normally distributed. Categorical data were presented as counts and percentages. Group-wise comparisons of categorical variables were conducted using Fisher’s exact test or the χ2 test, as appropriate. Independent Student’s t-test and Mann–Whitney U-tests were applied for comparing continuous variables between groups.
The least absolute shrinkage and selection operator (LASSO) penalized selection method was used to refine variables identified as significant in univariable logistic regression analysis, applying an optimal lambda value to prevent overfitting (Supplementary Figure 1). The objective of the LASSO variable selection method is to shrink the coefficients (beta coefficients) of noncontributory variables to zero, thereby excluding these variables from subsequent analyses. This technique effectively minimizes overfitting and mitigates bias in the variable selection process. The following features were selected for further analysis: age, sex, DM, smoking status, cerebrovascular events/transient ischemic attacks, CAD, creatinine, glucose, hemoglobin, HbA1c (glycated hemoglobin), LDL-C (low-density lipoprotein cholesterol), HDL-C (high-density lipoprotein cholesterol), and CRP. Correlations between hematologic inflammatory indices were analyzed using Kendall’s tau-b coefficient. The optimal cut-off value for inflammatory indices in determining wound healing was identified using X-tile software, version 3.6.1 (Yale University School of Medicine), based on the lowest p-value and highest chi-squared value (Supplementary Figure 2). Odds ratios (OR) and 95% CIs were calculated for all LASSO-derived regression analyses. Multicollinearity was examined using the variance inflation factor, with a threshold of > 3 indicating significant multicollinearity. The Hosmer–Lemeshow test evaluated the goodness-of-fit for logistic regressions.
The performance of the model was evaluated using a number of criteria, including the Akaike information criterion (lower values indicating a better fit), Brier score (lower values indicating better calibration), adjusted R2 (higher values indicating a better fit), and Harrell’s C-statistic (higher values indicating better discrimination). Kaplan–Meier curve analyses and log-rank tests were conducted to elucidate the relationship between biomarkers and wound healing. The accuracy of wound healing determination was evaluated using the receiver operating characteristic (ROC) curve, area under the curve (AUC), continuous net reclassification improvement (NRI), and integrated discrimination improvement (IDI) analyses. The DeLong method was employed to assess the statistical significance of differences in the discriminative ability of inflammatory indices. A decision curve analysis was conducted to ascertain whether indices, including NPS, PIV, SII, and SIRI, provide predictive value compared to all-treatment and no-treatment strategies. All statistical analyses employed two-sided tests with a significance level (alpha) of 0.05.
Results
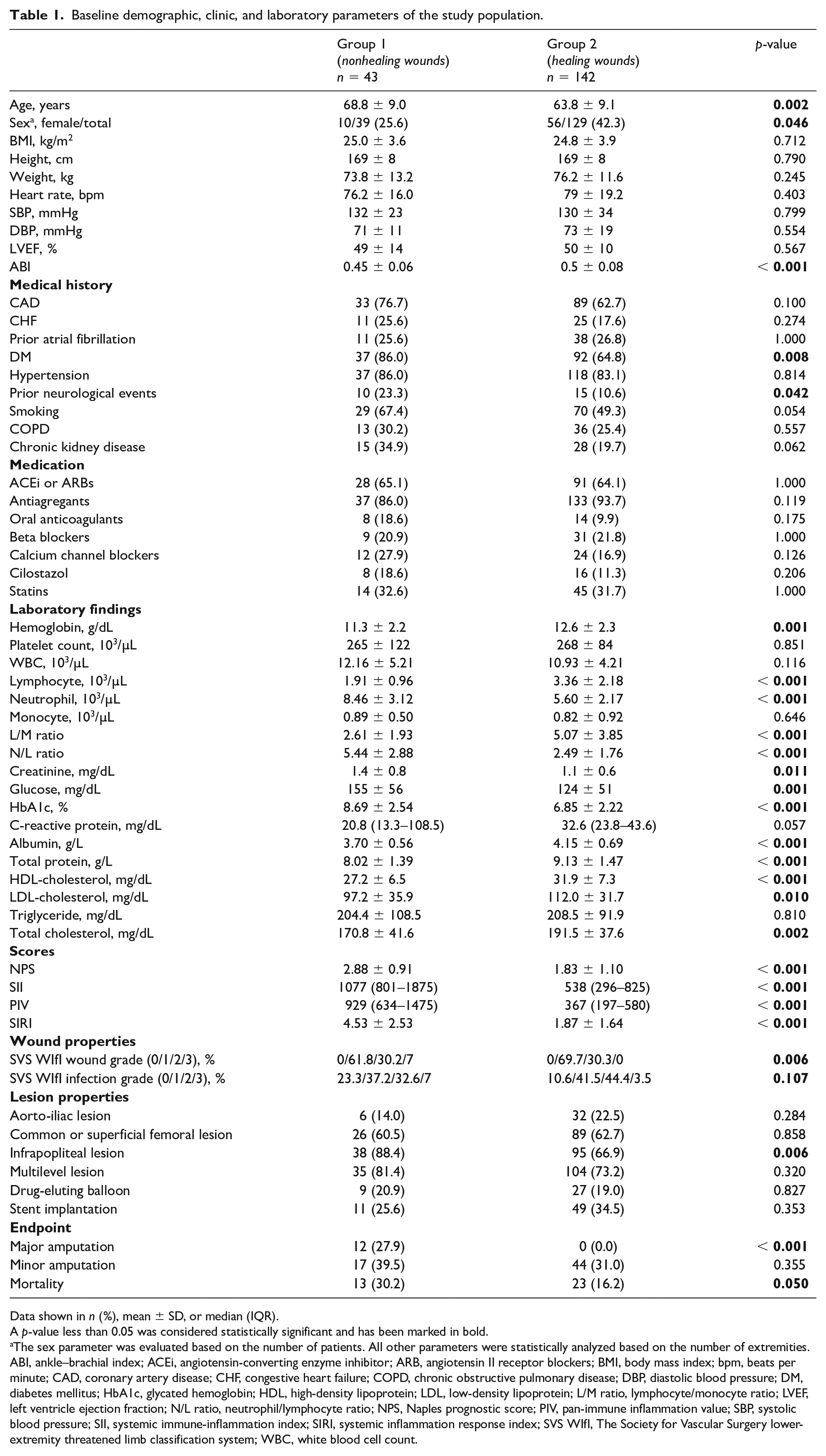
Of the 185 affected extremities in 168 patients with CLTI, the follow-up period was 42 (23–73) weeks. A total of 142 (76.8%) exhibited wound healing in response to EVT, and the healing time for these patients was 9 (5–13) weeks. Patients with healing wounds (group 2) were younger (64 vs 69 years, p = 0.002) and more likely to be women (42.3% vs 25.6%, p = 0.046). Conversely, patients with nonhealing wounds (group 1) were more likely to be diabetic (86.0% vs 64.8%, p < 0.001) and to have a history of cerebrovascular disease (23.3% vs 10.6%, p = 0.042). Other risk factors and vital signs at hospital admission were comparable between the two groups (Table 1).
Baseline demographic, clinic, and laboratory parameters of the study population.
Data shown in n (%), mean ± SD, or median (IQR).
A p-value less than 0.05 was considered statistically significant and has been marked in bold.
The sex parameter was evaluated based on the number of patients. All other parameters were statistically analyzed based on the number of extremities.
ABI, ankle–brachial index; ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin II receptor blockers; BMI, body mass index; bpm, beats per minute; CAD, coronary artery disease; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; DBP, diastolic blood pressure; DM, diabetes mellitus; HbA1c, glycated hemoglobin; HDL, high-density lipoprotein; LDL, low-density lipoprotein; L/M ratio, lymphocyte/monocyte ratio; LVEF, left ventricle ejection fraction; N/L ratio, neutrophil/lymphocyte ratio; NPS, Naples prognostic score; PIV, pan-immune inflammation value; SBP, systolic blood pressure; SII, systemic immune-inflammation index; SIRI, systemic inflammation response index; SVS WIfI, The Society for Vascular Surgery lower-extremity threatened limb classification system; WBC, white blood cell count.
In terms of laboratory parameters, patients with nonhealing wounds had higher serum creatinine, glucose, neutrophil, and HbA1c levels, whereas patients with healing wounds had higher serum HDL-C, LDL-C, total cholesterol, albumin, lymphocyte, and hemoglobin levels (p < 0.05 for all). Medications taken at admission were similar between the groups. The nonhealing group exhibited a higher prevalence of major amputation (27.9% vs 0.0%, p < 0.001) and infrapopliteal lesions (88.4% vs 66.9%, p = 0.006) compared to the healing group. Additionally, the nonhealing group had a higher rate of in-hospital mortality (30.2% vs 16.2%, p = 0.050) compared to the healing group. Patients in the nonhealing group exhibited elevated levels of inflammatory indices, including NPS (2.88 ± 0.91 vs 1.83 ± 1.10, p < 0.001), PIV (929 [634–1475] vs 367 [197–580], p < 0.001), SII (1077 [801–1875] vs 538 [296–825], p < 0.001), and SIRI (4.53 ± 2.53 vs 1.87 ± 1.64, p < 0.001) (Table 1). A significant correlation was observed between all inflammatory indices (Figure 1).
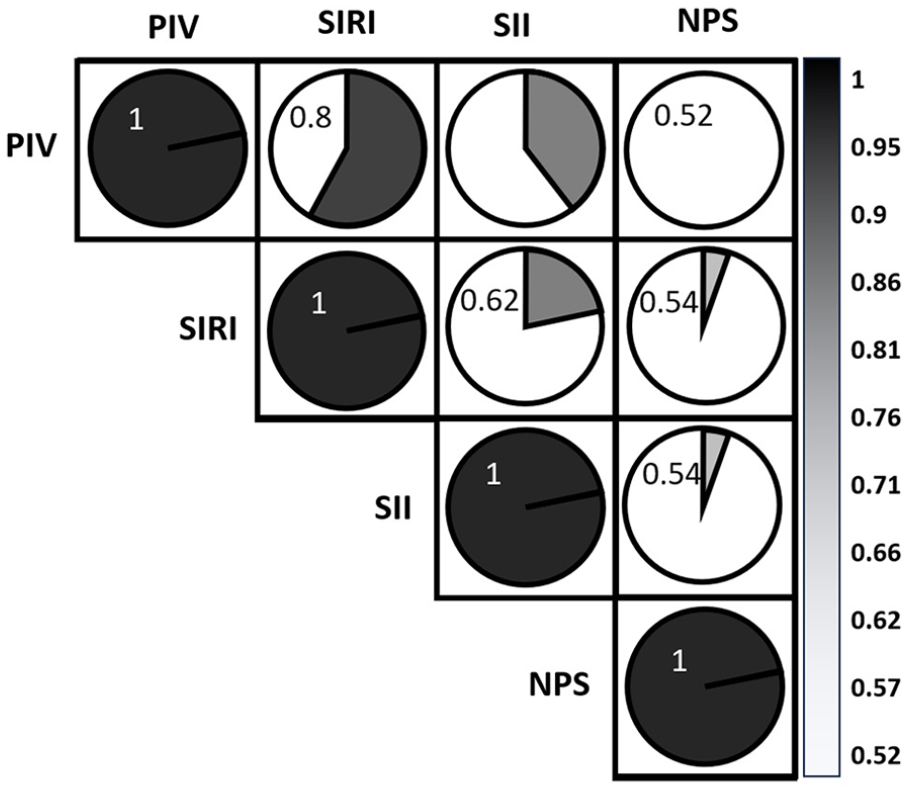
Visualization of the correlation matrix of inflammatory indices.
Among patients undergoing EVT for Fontaine stage 4 CLTI, NPS (adjusted OR [aOR] = 0.381, 95% CI 0.215–0.675, p = 0.001), PIV (aOR = 0.997, 95% CI 0.996–0.999, p < 0.001), SII (aOR = 0.997, 95% CI 0.996–0.997, p < 0.001), and SIRI (aOR = 0.443, 95% CI 0.313–0.625, p < 0.001) were independently associated with wound healing. Conversely, these inflammatory indices were not associated with in-hospital mortality (Table 2). SIRI (AUC = 0.840, 95% CI 0.777–0.904) demonstrated superior capability in determining wound healing compared to PIV (AUC = 0.799, 95% CI 0.722–0.876, pdif < 0.001), SII (AUC = 0.788, 95% CI 0.712–0.865, pdif < 0.001), and NPS (AUC = 0.760, 95% CI 0.681–0.838, pdif < 0.001). Similarly, SIRI exhibited a higher net reclassification improvement compared to PIV (68.4%, pdif < 0.001), SII (38.5%, pdif = 0.024), and NPS (29.8%, pdif = 0.079) (Figure 2, Table 3). Additionally, all inflammatory indices, particularly SIRI, demonstrated prognostic value in determining wound healing at high treatment thresholds (Figure 3).
Multivariable logistic regression analysis showing the association between inflammatory indices and wound healing and mortality.
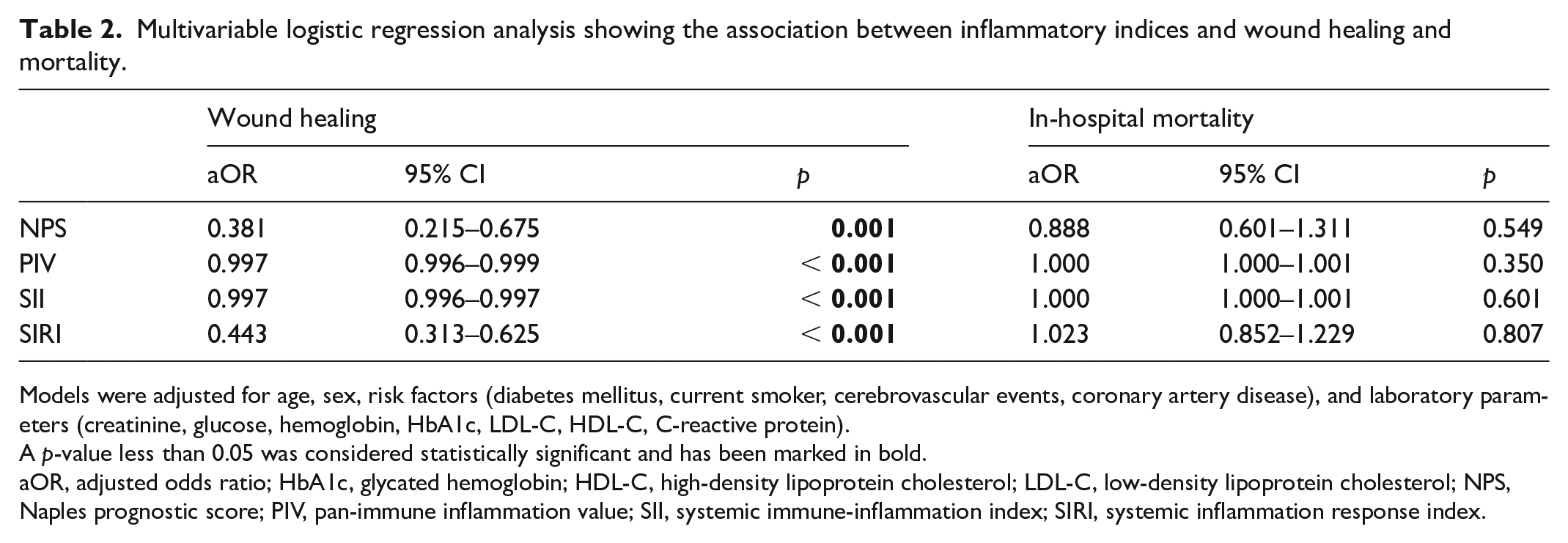
Models were adjusted for age, sex, risk factors (diabetes mellitus, current smoker, cerebrovascular events, coronary artery disease), and laboratory parameters (creatinine, glucose, hemoglobin, HbA1c, LDL-C, HDL-C, C-reactive protein).
A p-value less than 0.05 was considered statistically significant and has been marked in bold.
aOR, adjusted odds ratio; HbA1c, glycated hemoglobin; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; NPS, Naples prognostic score; PIV, pan-immune inflammation value; SII, systemic immune-inflammation index; SIRI, systemic inflammation response index.
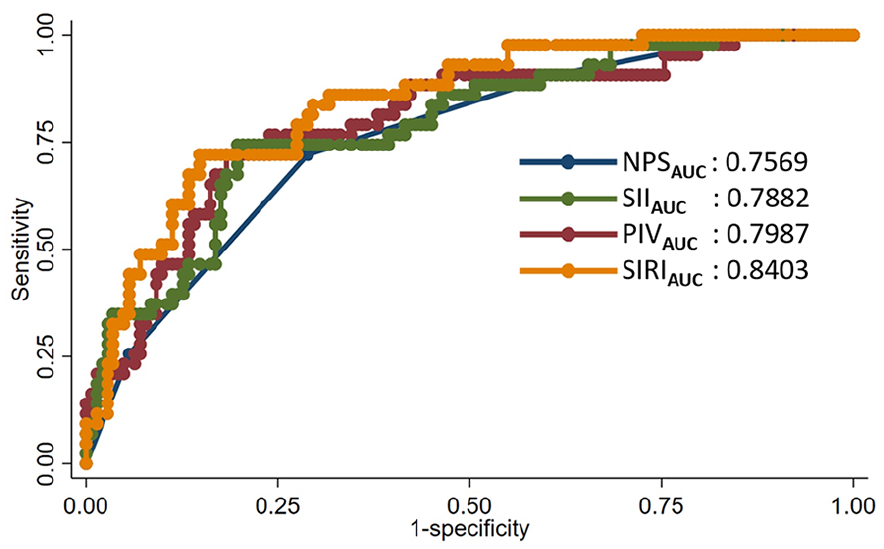
Receiver operating characteristic curve analyses of the inflammatory indices.
Discrimination and reclassification analyses of inflammatory indices for estimating wound healing.
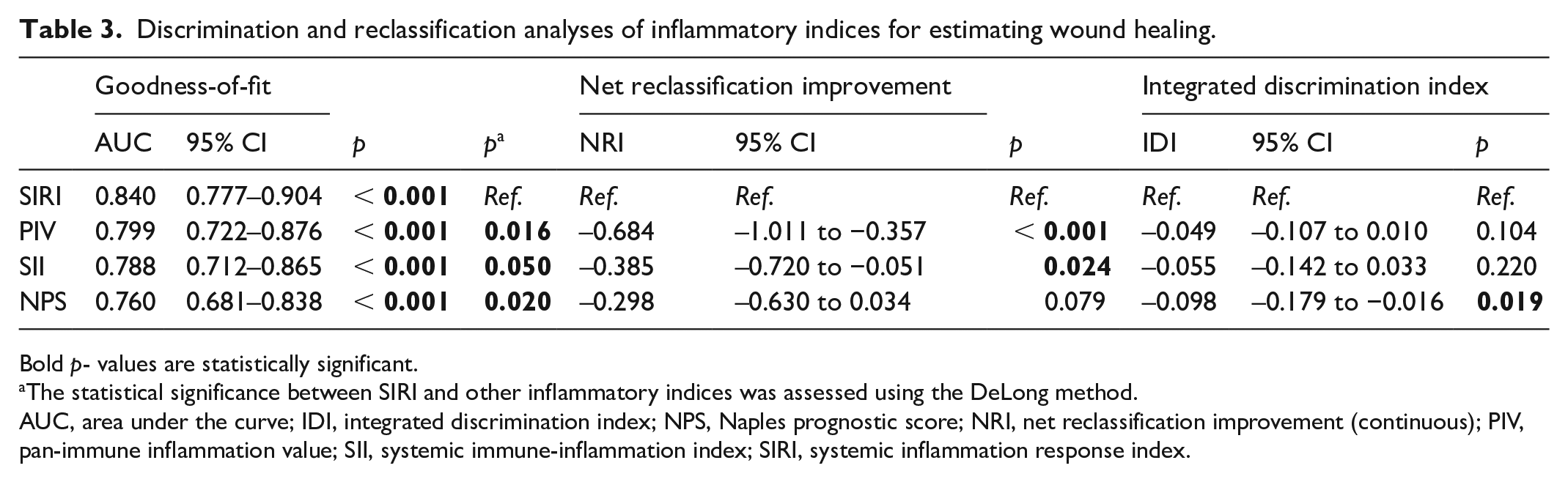
Bold p- values are statistically significant.
The statistical significance between SIRI and other inflammatory indices was assessed using the DeLong method.
AUC, area under the curve; IDI, integrated discrimination index; NPS, Naples prognostic score; NRI, net reclassification improvement (continuous); PIV, pan-immune inflammation value; SII, systemic immune-inflammation index; SIRI, systemic inflammation response index.
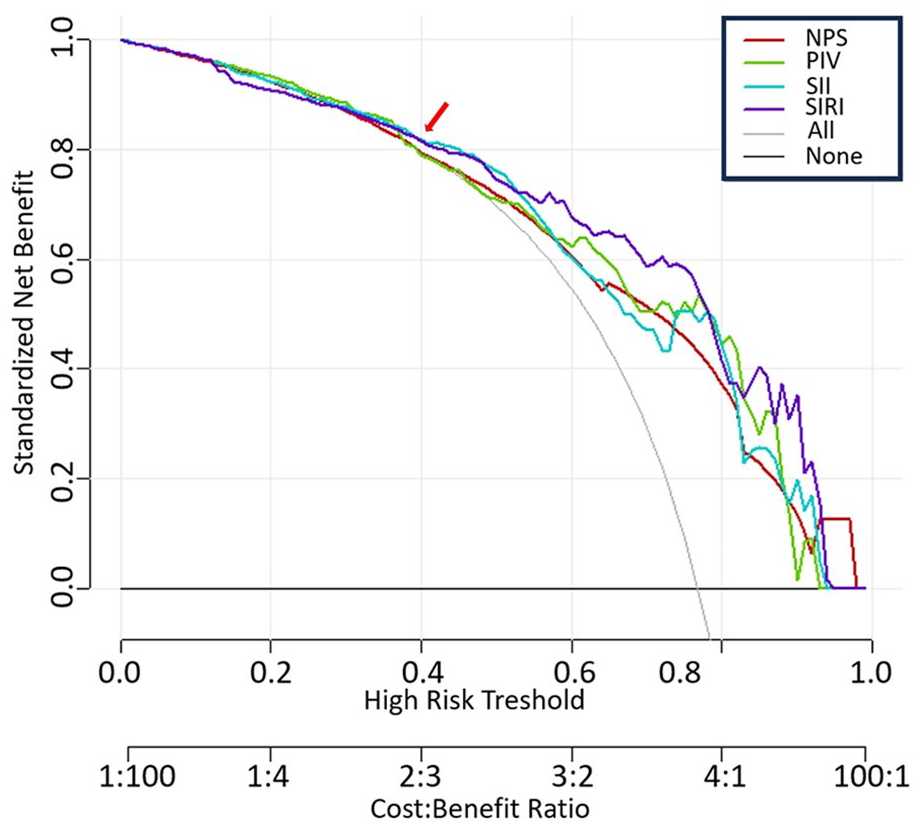
Decision curve analysis demonstrating the prognostic value of inflammatory indices for determining wound healing.
When examining the cumulative wound-healing probabilities during in-hospital follow up based on the inflammatory indices dichotomized according to X-tile analysis, the healing rates were higher in the low categories for PIV (91.5% vs 50.8%, plog-rank < 0.001), SII (91.1% vs 47.5%, plog-rank < 0.001), and SIRI (90.9% vs 41.5%, plog-rank < 0.001). However, for NPS, this difference remained numerical and did not reach statistical significance (80.7% vs 42.1%, plog-rank = 0.243) (Figure 4).

Kaplan–Meier curves illustrating the cumulative probability of wound healing in relation to inflammatory indices.
Discussion
The present study demonstrated that inflammatory indices, including NPS, PIV, SII, and SIRI, predicted wound healing in individuals with Fontaine stage 4 CLTI undergoing EVT. Notably, SIRI exhibited superior discriminatory power in predicting healing outcomes compared to the other indices. Given its predictive capability, the clinical use of especially SIRI may provide a significant advantage in planning the treatment approach and management for this patient group. To the best of our knowledge, this is the first report to comparatively demonstrate the association between inflammation and wound healing in individuals with advanced PAD.
SIRI is derived from the counts of monocytes, neutrophils, and lymphocytes, presenting itself as an innovative and dependable indicator of chronic, low-grade inflammation. Prior investigations have indicated a close correlation between SIRI and the prognosis of various cardiovascular conditions, including ischemic stroke and acute coronary syndrome.32,33 Dziedzic et al. affirmed a substantial link between SIRI and the severity of CAD. Notably, individuals with STEMI exhibited elevated SIRI levels compared to patients with stable CAD, with even higher levels observed in those with three-vessel CAD. 34 Our study further emphasizes the importance of SIRI as a significant predictor of wound healing in patients with Fontaine stage 4 CLTI. With an AUC of 0.840 (95% CI: 0.777–0.904), SIRI demonstrated superior predictive capability compared to PIV (AUC = 0.799), SII (AUC = 0.788), and NPS (AUC = 0.760), indicating its strong association with favorable healing outcomes. The analysis revealed that patients in the low SIRI group had a healing rate of 90.9% compared to only 41.5% in the high SIRI group (plog-rank < 0.001), further supporting SIRI’s potential as a valuable prognostic index in clinical practice. Additionally, SIRI demonstrated superior net reclassification improvement compared to other inflammatory indices, with PIV showing an improvement of 68.4% (pdif < 0.001), SII at 38.5% (pdif = 0.024), and NPS at 29.8% (pdif = 0.079). These findings suggest that SIRI not only reflects inflammatory responses but may also serve as a critical index in the management and treatment of patients with CLTI.
SII is an innovative inflammatory index derived from platelet, neutrophil, and lymphocyte counts, serving as a precise reflection of the inflammatory status. Xu et al. identified a notably stronger positive correlation between SII and total cardiovascular risk in individuals with hypertension or diabetes. 35 In a study aiming to assess the utility of SII in patients with PAD, the complexity of atherosclerotic disease was categorized based on the TASC II classification, and the results revealed a notable correlation between SII and the severity of PAD. 36 In a retrospective study, researchers investigated the relationship between novel inflammatory indicators and PAD in patients with type 2 DM. This study demonstrated a significant association between certain indicators, including SII and SIRI, and a higher prevalence of PAD in individuals with type 2 DM. Moreover, these indices were correlated with the severity of the disease. 22 Our results indicated that SII was independently associated with healing outcomes (aOR = 0.997, p < 0.001) and demonstrated a significant AUC of 0.788 (95% CI: 0.712–0.865), although slightly lower than SIRI (AUC = 0.840). Notably, patients in the low SII category exhibited a healing rate of 91.1% compared to 47.5% in the high SII group (plog-rank < 0.001), highlighting its prognostic value. These findings suggest that incorporating SII into clinical assessments can enhance risk stratification and inform treatment strategies for patients with CLTI, with the ultimate goal of improving wound healing outcomes.
PIV, which delineates immune and inflammatory status by comprising blood components such as neutrophils, lymphocytes, monocytes, and platelets, was initially identified as a significant predictive indicator in the prognosis of patients with cancer. 37 In our study, we found that PIV was an independent predictor of wound healing in patients with Fontaine stage 4 CLTI. PIV demonstrated a significant net reclassification improvement of 68.4% (pdif < 0.001) regarding healing outcomes. The ROC analysis indicated an AUC of 0.799 (95% CI: 0.722–0.876), which, though lower than that of SIRI (AUC = 0.840), still exhibited important predictive performance for wound healing. These findings suggest that PIV reflects inflammatory responses and may play a prognostic role in wound healing, indicating its potential as a valuable index in assessing healing processes in clinical practice.
The NPS is a novel prognostic indicator based on inflammatory and nutritional indices. Initially utilized to assess the prognosis of colorectal cancer, the NPS has since been reported as an independent prognostic factor for various cancers.38–41 Our study found that though a numerical difference existed between high and low NPS categories, its prognostic value appears limited compared to other indices, suggesting that further investigation is needed to better understand its potential role in clinical practice.
Inflammatory and nutritional indices included in the present study have not been previously investigated or compared in patients with Fontaine stage 4 CLTI, who represent the most challenging subset of PAD characterized by the highest rates of amputation and mortality. The findings of this study indicate that, compared to other indices assessed, SIRI emerges as the most potent inflammatory indicator, effectively predicting wound healing following EVT in patients with Fontaine stage 4 CLTI. In this patient cohort, characterized by challenging the predictability of treatment outcomes, we believe the indices investigated in our study may serve as valuable indicators.
The study has several limitations that merit acknowledgment. Firstly, the single-center and retrospective study design, along with the modest number of patients, inherently weakens the ability to generalize the findings. Therefore, further large-scale, prospective, randomized, and multicenter studies are needed. Additionally, the present study lacks data on temporal changes in nutritional status, thus preventing us from determining whether short- and long-term improvements in nutrition led to better clinical prognoses during follow-up. We meticulously excluded patients who could significantly influence the inflammatory indices, such as those with acute myocardial infarction or pneumonia. Nevertheless, certain potential confounding factors, such as undetectable bacterial or viral infections, remain unavoidable, constituting a limitation in our research. A notable limitation of our study is that only 30% of patients with stage 4 CLTI received optimal medical therapy. The lack of consistent medical treatment across the study population may have influenced clinical outcomes and the generalizability of our findings. This limitation should be considered when interpreting the results, particularly in regard to the impact of medical therapy on wound healing and overall prognosis. Additionally, our retrospective design leads to limitations in determining causation and highlights the need for prospective studies. Although we aimed to establish the baseline association between inflammatory indices and wound healing, we did not explore their dynamic changes before and after EVT. Future research could involve a comparative design assessing patients with CLTI at different stages of treatment, evaluating how alterations in inflammatory indices throughout the healing process, including at the time of wound healing, might predict healing outcomes.
Conclusion
The current study has revealed a significant relationship between postprocedure wound healing and scoring systems, including SIRI, SII, PIV, and NPS, with SIRI being the strongest determinant. However, these findings also open new avenues for future research. A promising direction is the dynamic monitoring of these inflammatory indices over time, both before and after EVT, to determine their role in tracking treatment response and guiding real-time clinical decisions. Incorporating nutritional interventions and inflammation-targeted therapies into a longitudinal study design could reveal whether modifying these indices can directly influence wound healing and patient survival. Moreover, future prospective, multicenter trials should investigate whether combining these indices with other clinical factors can lead to the development of a comprehensive risk assessment model. This model could refine patient selection for EVT, optimize treatment protocols, and potentially reduce the rates of major amputation and mortality in CLTI. In summary, SIRI and similar indices hold significant promise as valuable clinical tools, but further research is required to fully integrate these markers into the management of CLTI, with the ultimate goal of improving wound-healing outcomes.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X251320867 – Supplemental material for Predictive value of the inflammatory indices on wound healing in patients with chronic limb-threatening ischemia revascularized via percutaneous intervention
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X251320867 for Predictive value of the inflammatory indices on wound healing in patients with chronic limb-threatening ischemia revascularized via percutaneous intervention by Yeliz Guler, Omer Genc, Furkan Akbas, Abdullah Yildirim, Ilyas Cetin, Aslan Erdogan, Ufuk S Halil, Huseyin Akgun, Berat Erdem, Ahmet Guler and Cevat Kirma in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
