Abstract
Background:
Endovascular abdominal aortic aneurysm repair (EVAR) is a preferred surgery to prevent aneurysm sac enlargement and minimize the risk of life-threatening rupture in patients with AAA. Serious complications of type II endoleaks following EVAR can cause sac expansion and increase rupture risk. This study focused on evaluating clinical and blood characteristics in patients with type II endoleaks to refine our understanding of systemic fluctuations associated with unsuccessful EVAR.
Methods:
This retrospective study included 146 patients with AAA who underwent primary elective endovascular procedures (EVAR/fEVAR) between 2013 and 2021. Clinical characteristics, complete blood count (CBC) and imaging data were analyzed from patients who did and did not develop type II endoleaks.
Results:
Mean platelet volume (MPV) was significantly increased in patients who developed type II endoleaks after EVAR. Receiver operating characteristic analysis showed that MPV has a satisfactory discriminatory performance in distinguishing post-EVAR patients who developed type II endoleaks, yielding an area under the curve (AUC) value of 0.64. A risk stratification panel incorporating MPV, type II diabetes history, and administration of dual antiplatelet therapies yielded an AUC of 0.70 and predicted an endoleak-free survival rate with a hazard ratio of 2.94. A nomogram revealed that MPV had the highest scoring weight among all significant variables.
Conclusion:
Patients with type II endoleaks following EVAR have elevated MPV indicative of different phenotypes of circulating platelets. MPV presents an attractive predictive criteria for assessing the occurrence of type II endoleaks in patients with AAA.
Keywords
Background
Abdominal aortic aneurysm (AAA) is a life-threatening disease that carries a high mortality rate. The latest epidemiological data show that AAA is ranked as the 15th leading cause of overall mortality in the United States and the 10th most lethal disease in men > 55 years old. 1 AAA is characterized by the abnormal bulging of the renal segment of the abdominal aorta (diameter > 3 cm) indicative of a highly degraded aortic wall that is imminent of rupture.2,3 Though most AAA typically remain asymptomatic until the event of rupture – an event that carries a mortality rate of up to 90% 4 – epidemiological studies show that ruptured AAA account for approximately 5% of sudden deaths. 5 Current management to prevent aneurysmal rupture relies on monitoring aortic diameter and surgical intervention, with endovascular abdominal aortic aneurysm repair (EVAR) becoming the preferred treatment modality since its inception over 30 years ago due to its lower perioperative risk compared to open surgical repair.6–9 EVAR offers advantages in reducing short-term complications and death,6–10 but it is associated with a greater long-term risk of secondary interventions, 11 largely due to the development of type II endoleaks, a retrograde flow into the sac from collateral pathways such as the inferior mesenteric artery (IMA) or lumbar arteries, which have the potential to increase aneurysm sac growth and risk of rupture following repair.8,12 Therefore, the identification of factors that contribute to the development of type II endoleaks and their associated complications is in dire need.
Currently, lifelong imaging surveillance is recommended in all patients following endovascular repairs. Computed tomography (CT) and CT angiography (CTA) remain the gold standard technique to examine the stability of the endograft and to measure the aneurysmal sac. 13 Nevertheless, consistently implementing these methods incurs significant expenses 14 and allows repetitive exposure to radiation. 15 As the incidence of AAA cases and the demand for EVAR procedures continue to rise, it becomes imperative to increase our understanding of the mechanisms that cause the failure of the repair surgery which could improve the safety and efficacy of patients with AAA undergoing surgery.
Recent studies have attempted to identify potential circulating biomarkers, such as C-reactive protein and matrix metalloproteinase-9 that may predict aneurysm sac behavior and the development of endoleaks.16–19 However, these studies have either concentrated solely on sac diameter expansion rate, combining all types of endoleaks, or assessed measurements many months following surgery. Interestingly, one clinical study observed a lower platelet count on day 7 after EVAR in patients who further developed malignant type II endoleaks, 20 thus identifying the possible role of platelets in sac enlargement in patients after an endovascular procedure. Other clinical studies have also identified low intramural thrombus burden as a risk factor for postoperative endoleaks and aneurysm sac growth.21–23 Though these studies have informed us of the association between platelet number and platelet tissue activity in endoleak development, they did not assess intrinsic platelet characteristics, such as the size and volume of platelets.
In this study, we analyzed complete blood count (CBC) collected postoperatively in conjunction with demographics, medical history, and anatomic factors in a retrospective cohort of 146 patients undergoing elective endovascular procedures at New York University Langone Medical Center to identify significant clinical and intrinsic blood profiles that are associated with type II endoleaks. We aimed to further develop a risk stratification panel as a readout to distinguish type II endoleaks following endovascular repair.
Methods
Study design
This is a single-center, retrospective cohort study of 146 patients undergoing primary elective endovascular repair, EVAR, or fenestrated EVAR (fEVAR) between September 2013 and July 2021. Patients were selected based on participation in previous or ongoing studies at our institution. 24 Institutional review board approval was obtained and the need for patient consent was waived. A review of institutional medical records was conducted for this study. Of the original cohort selected, which included 184 patients, we excluded 38 patients with missing data for one or more of our predictors and outcomes of interest.
For each patient, CBC data from day one after surgery was recorded. CBC was performed using an automated hematology analyzer on blood samples collected from the patients in EDTA tubes. Likewise, imaging characteristics for operative planning imaging as well as postoperative surveillance imaging up to 5 years following the index operation were collected. CT was used to detect aneurysms sac size. The aneurysm size was determined by measuring the shortest diameter of the largest axial section of the aneurysm. During post-EVAR monitoring, an increase in the aneurysm sac size of 5 mm or more, compared to the preoperative CT scan taken within 6 months before the procedure, was classified as significant enlargement. A type II endoleak was diagnosed by contrast-enhanced CTA followed by duplex ultrasonography. Delayed imaging for CTA assessment was acquired in the venous phase in all patients to distinguish type I versus type II endoleaks. When a type II endoleak was identified on magnetic resonance angiography (MRA), a corroborative duplex was obtained. Patients who developed type I or type III endoleaks were excluded such that only patients who developed type II endoleaks or patients who did not develop endoleaks were included in this study. Patient-level demographics, comorbidities, and postoperative medications were obtained by merging CBC and imaging data with institutional data from the Vascular Quality Initiative (VQI) of the Society for Vascular Surgery (SVS). Details on the VQI data collection and maintenance process have previously been described. 25
Variable definitions of imaging characteristics
Our primary outcome of interest were type II endoleak-free survival post-EVAR or fEVAR. Other imaging characteristics reported include IMA and lumbar artery patency, mural thrombus burden, and cumulative change in aneurysm sac diameter. IMA and lumbar artery patency as well as mural thrombus burden were assessed using CTA or MRA. Maximum aneurysm sac diameter was assessed using noncontrast CT, CTA, noncontrast magnetic resonance imaging (MRI), or MRA. The presence of an endoleak was assessed using CTA, MRA, or duplex ultrasonography. The presence of endoleak is defined as any detectable contrast within the aneurysmal sac on both CTA, MRA, or any flow detected within the sac on the color duplex ultrasound. Delayed imaging in the venous phase was employed for all CTA assessments. Metal artifact reduction techniques were utilized for MRA imaging. Mural thrombus burden was classified qualitatively as minimal, moderate, or severe, with minimal representing little to no mural thrombus present on operative planning imaging, severe representing circumferential mural thrombus occupying > 50% of the aneurysm sac lumen on axial cross-sectional imaging, and moderate representing all cases in between minimal and severe. Cumulative change in sac diameter was calculated by subtracting the maximum AAA diameter measured on each patient’s most recent postoperative CTA or MRA from the maximum AAA diameter on operative planning imaging.
Data analysis
We compared baseline patient characteristics to evaluate type II endoleak outcomes using appropriate statistical tests as follows. Categorical variables were compared using Pearson’s chi-squared test. All continuous variables were tested for normality using the Shapiro–Wilk and Anderson–Darling tests. Normally distributed continuous variables were compared using the two-sided Student’s t-test for unpaired samples, and nonnormally distributed variables were analyzed using the Mann–Whitney U-test. For multiple comparisons, we performed the Kruskal–Wallis test in a one-way ANOVA procedure.
To assess type II endoleak-free survival, we used Cox proportional hazards models. Initially, we conducted a bivariate analysis for demographics, health status, operative planning imaging, postoperative CBC, and discharge medications. Subsequently, variables with p-values less than 0.2 from the bivariate analysis were included in a multivariate model. To avoid false positive associations due to overfitting, we performed an unbiased stepwise backward variable selection process by sequentially eliminating the least statistically significant predictors from each model until the number of covariates was less than the number of outcome events.
The receiver operating characteristic (ROC) curves were plotted to assess the predictive potential of variables from both the bivariate and the multivariate Cox proportional hazards model. One-way logistic regression analysis was used to determine and formulate an endoleak-free risk stratification score model. We dichotomized the risk score model into low and high values according to the formula obtained from the logistic regression ROC curve model. A waterfall plot illustrated the risk score value in the cohort. Kaplan–Meier survival analysis and log-rank tests were used to estimate and compare endoleak-free survival between groups. We established a risk stratification nomogram that includes the significant clinicopathological variables from the previous analysis. 26 All statistical analysis and figures were generated using the RStudio environment (v1.3.959) (R Foundation for Statistical Computing, Vienna, Austria) and GraphPad Prism Version 9.0 (GraphPad Software, San Diego, CA, USA). A p-value less than 0.05 was considered statistically significant.
Results
Clinicopathological characteristics associated with type II endoleaks
We initially compared baseline characteristics, CBC data for patients who underwent elective endovascular procedures (EVAR/fEVAR) resulting in type II endoleaks (n = 56) versus patients who underwent elective endovascular procedures with no subsequent endoleaks (n = 90) (Table 1). Notably, the median number of patent lumbar arteries on operative planning imaging was significantly greater among patients who developed type II endoleaks (6 vs 4.5, p = 0.009). Interestingly, from the CBC analysis, the median MPV was increased among patients who developed type II endoleaks (10.3 vs 10.0 fL, p = 0.045).
Baseline characteristics and postoperative outcomes for patients who underwent an endovascular procedure (EVAR/fEVAR) that resulted in type II endoleaks versus patients who did not develop endoleaks.
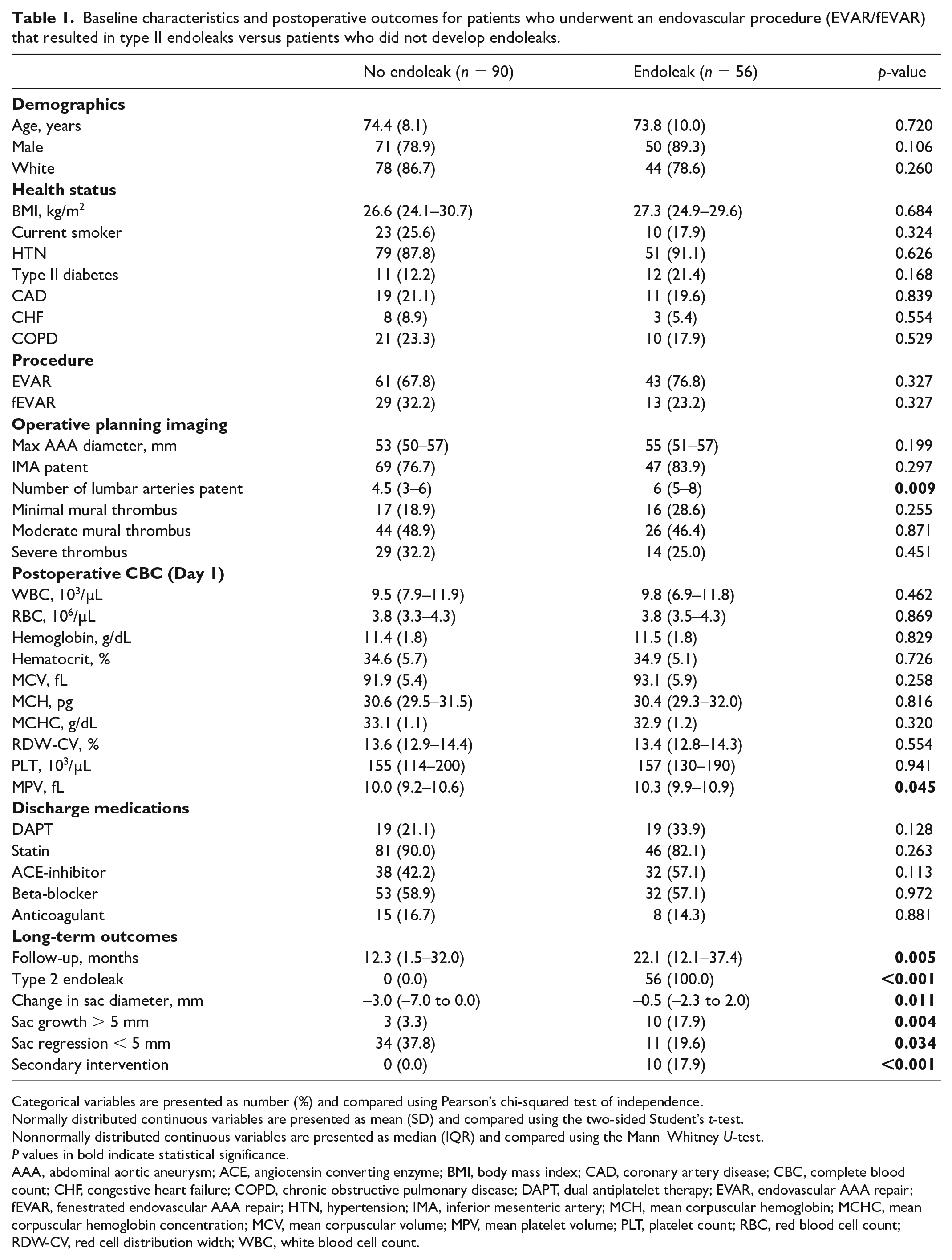
Categorical variables are presented as number (%) and compared using Pearson’s chi-squared test of independence.
Normally distributed continuous variables are presented as mean (SD) and compared using the two-sided Student’s t-test.
Nonnormally distributed continuous variables are presented as median (IQR) and compared using the Mann–Whitney U-test.
P values in bold indicate statistical significance.
AAA, abdominal aortic aneurysm; ACE, angiotensin converting enzyme; BMI, body mass index; CAD, coronary artery disease; CBC, complete blood count; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; DAPT, dual antiplatelet therapy; EVAR, endovascular AAA repair; fEVAR, fenestrated endovascular AAA repair; HTN, hypertension; IMA, inferior mesenteric artery; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; MPV, mean platelet volume; PLT, platelet count; RBC, red blood cell count; RDW-CV, red cell distribution width; WBC, white blood cell count.
According to the long-term outcome, patients who developed type II endoleaks were significantly more likely to experience aneurysm sac growth > 5 mm (17.9% vs 3.3%, p = 0.004), significantly less likely to experience aneurysm sac regression < 5 mm (19.6% vs 37.8%, p = 0.034), and more likely to require a secondary intervention (17.9% vs 0.0%, p < 0.001). Notably, patients who experienced type II endoleaks had significantly longer median follow-up periods than the control group (22.1 vs 12.3 months, p = 0.005).
To address potential bias due to the longer follow-up time among patients who developed an endoleak, we conducted a time-to-event analysis for type II endoleak-free survival using bivariate Cox proportional hazards modeling, incorporating all variables from Table 1 as potential predictors of endoleak-free survival (Table 2). In the bivariate analysis, an increased number of patent lumbar arteries, increased red blood cell distribution width (RDW), higher MPV, and dual antiplatelet therapy (DAPT) on hospital discharge were significantly associated with development of type II endoleaks. In multivariate analysis, MPV (hazard ratio [HR] 1.42; 95% CI 1.11–1.81, p = 0.005), type II diabetes (HR 2.03; 95% CI 1.05–3.92; p = 0.035), and the number of patent lumbar arteries (HR 1.19; 95% CI 1.05–1.36; p = 0.009) emerged as significant variables associated with development of type II endoleaks. Moreover, DAPT treatment on hospital discharge (HR 2.34; 95% CI 1.30–4.21, p = 0.005) was also associated with type II endoleak development, yet no significance was observed with anticoagulant treatment.
Cox proportional hazards analysis of endoleak-free survival among patients who underwent an endovascular procedure (EVAR/fEVAR).
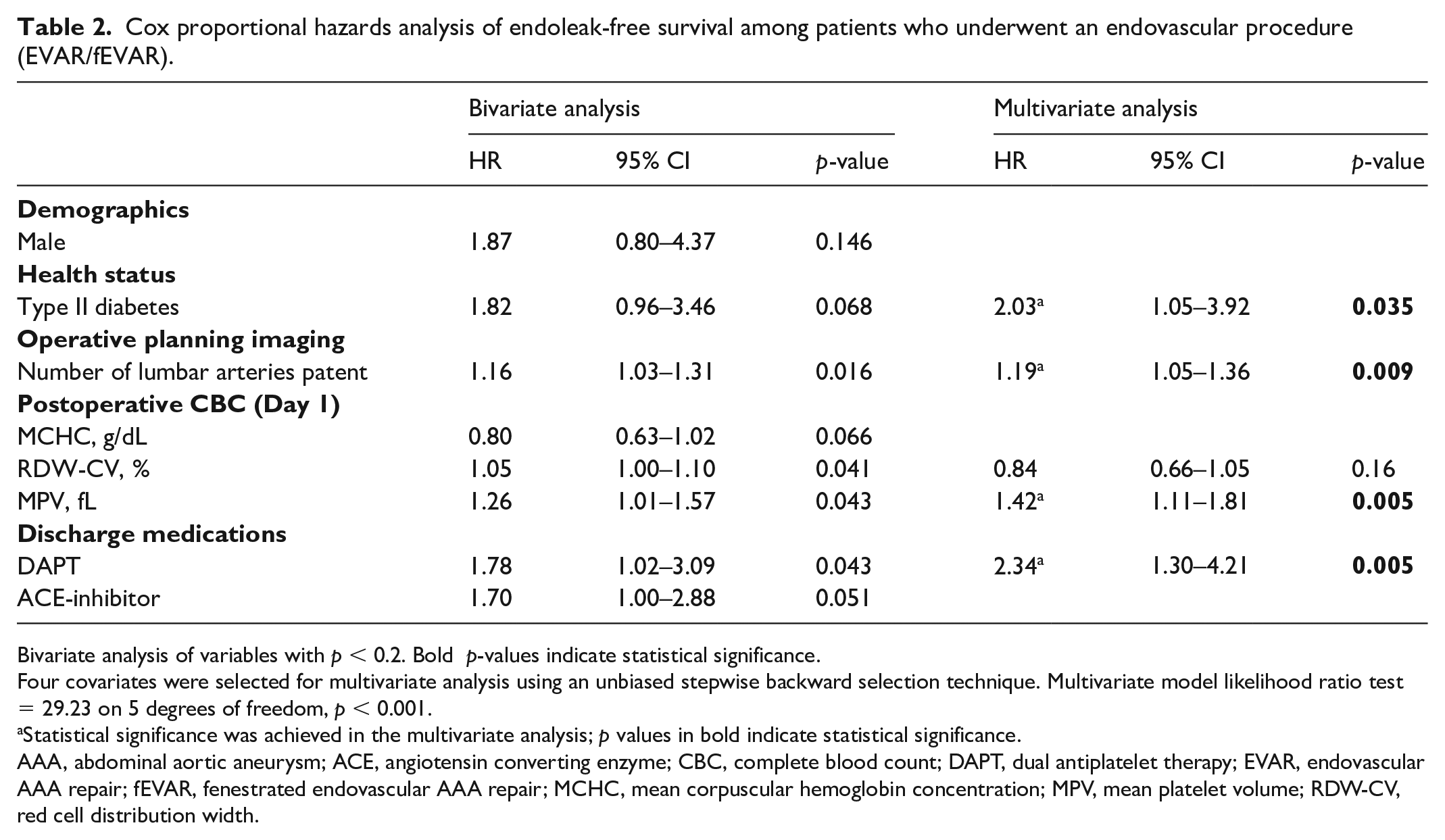
Bivariate analysis of variables with p < 0.2. Bold p-values indicate statistical significance.
Four covariates were selected for multivariate analysis using an unbiased stepwise backward selection technique. Multivariate model likelihood ratio test = 29.23 on 5 degrees of freedom, p < 0.001.
Statistical significance was achieved in the multivariate analysis; p values in bold indicate statistical significance.
AAA, abdominal aortic aneurysm; ACE, angiotensin converting enzyme; CBC, complete blood count; DAPT, dual antiplatelet therapy; EVAR, endovascular AAA repair; fEVAR, fenestrated endovascular AAA repair; MCHC, mean corpuscular hemoglobin concentration; MPV, mean platelet volume; RDW-CV, red cell distribution width.
Mean platelet volume (MPV) is a significant variable that distinguishes type II endoleaks after EVAR
We further analyzed MPV levels in patients based on the type of endovascular procedure: EVAR or fEVAR. According to the distribution of data, we observed significantly higher MPV in EVAR (n = 104) patients who developed type II endoleaks (p = 0.037), though no differences were observed in patients who underwent fEVAR (n = 42) (p = 0.334) (Figure 1A). To further clarify the discriminatory potential of MPV for type II endoleaks after EVAR, we performed an ROC curve analysis and observed that MPV had satisfactory discriminatory power, yielding an AUC value of 0.64 (95% CI 0.53–0.75, sensitivity 63.41% and specificity 58.33% for cut-off value > 10.25 and p = 0.01). We included patent lumbar arteries in our analysis since it was previously reported as a significant predictor of type II endoleaks after EVAR. 27 Interestingly, we observed that MPV reached comparable discriminatory potential with patent lumbar arteries (AUC: 0.67). We also observed that MPV surpassed the other two significant clinical variables: type II diabetes (AUC: 0.60) and DAPT (AUC: 0.57) from the previous multivariate analysis (Figure 1B). By grouping EVAR patients into low (⩽ 10.25) or high MPV (> 10.25) based on the cut-off threshold of the ROC curve analysis, we performed Kaplan–Meier survival analysis which revealed that higher MPV was associated with reduced endoleak-free survival, with a Cox–Mantel HR of 1.96 (95% CI 1.06–3.62, p = 0.03) and a median survival of 407 days (Figure 1C). Taken together, these results suggest that altered platelet maturity as indicated by high MPV is associated with severity and mortality of type II endoleaks.
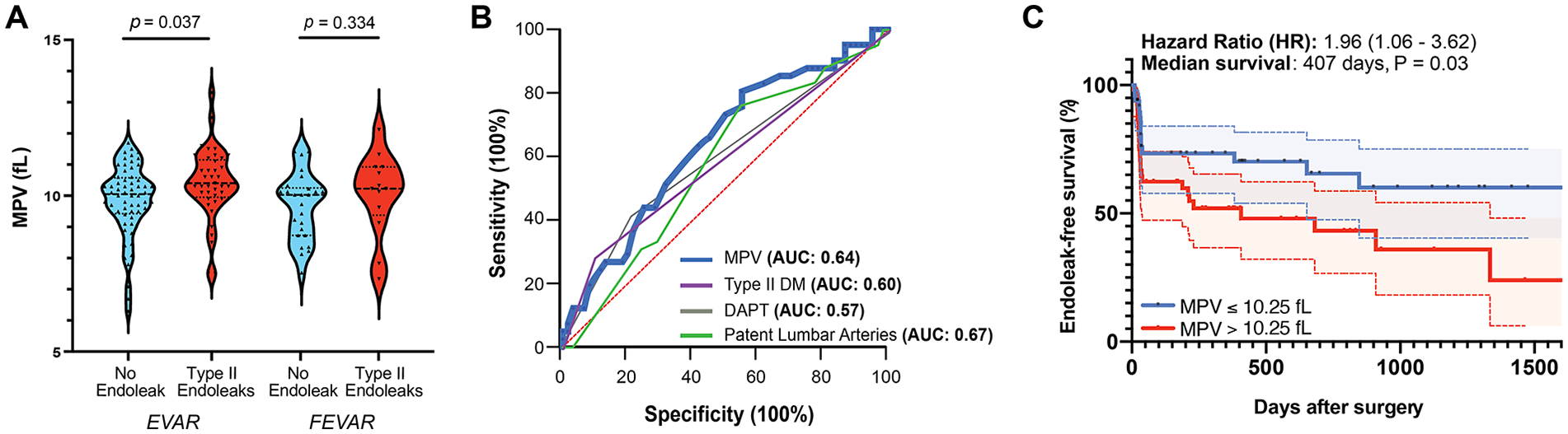
Mean platelet volume (MPV) is increased in patients with type II endoleaks after EVAR.
MPV-based risk stratification panel for type II endoleaks after EVAR
To harness the discriminatory potential of MPV alongside other significant clinical variables, we integrated MPV with type II diabetes (HR = 2.03, p = 0.035) and history of DAPT (HR = 2.34, p = 0.005) from the previous multivariate Cox regression analysis as a risk stratification panel to assess the likelihood of developing type II endoleaks post-EVAR. ROC curve analysis of this panel using a logistic regression model revealed improved discriminatory value on distinguishing post-EVAR patients who developed type II endoleaks, resulting in an AUC value of 0.70 (95% CI 0.58–0.80, sensitivity 62.07%, specificity 65.28% and p = 0.001) (Figure 2A). We calculated the risk probability score using a formula derived from this logistic regression model as follows: –6.1 + [0.50 × MPV (fL)] + [0.647 × type II diabetes (1 = yes, 0 = no)] + [0.43 × DAPT (1 = yes, 0 = no)]. Using this score, patients were then dichotomized into high- and low-risk scores based on the classification cut-off value (0.5) as shown in the waterfall plot (Figure 2B), which further confirmed the potential of this panel. Moreover, patients with high-risk scores exhibited a significantly poorer endoleak-free survival rate after surgery, with HR of 2.94 (95% CI 1.62–5.35, p < 0.0004) and median survival of 230 days (Figure 2C). Taken together, these results highlighted the robustness of the MPV-based panel to discriminate post-EVAR patients who developed type II endoleaks.
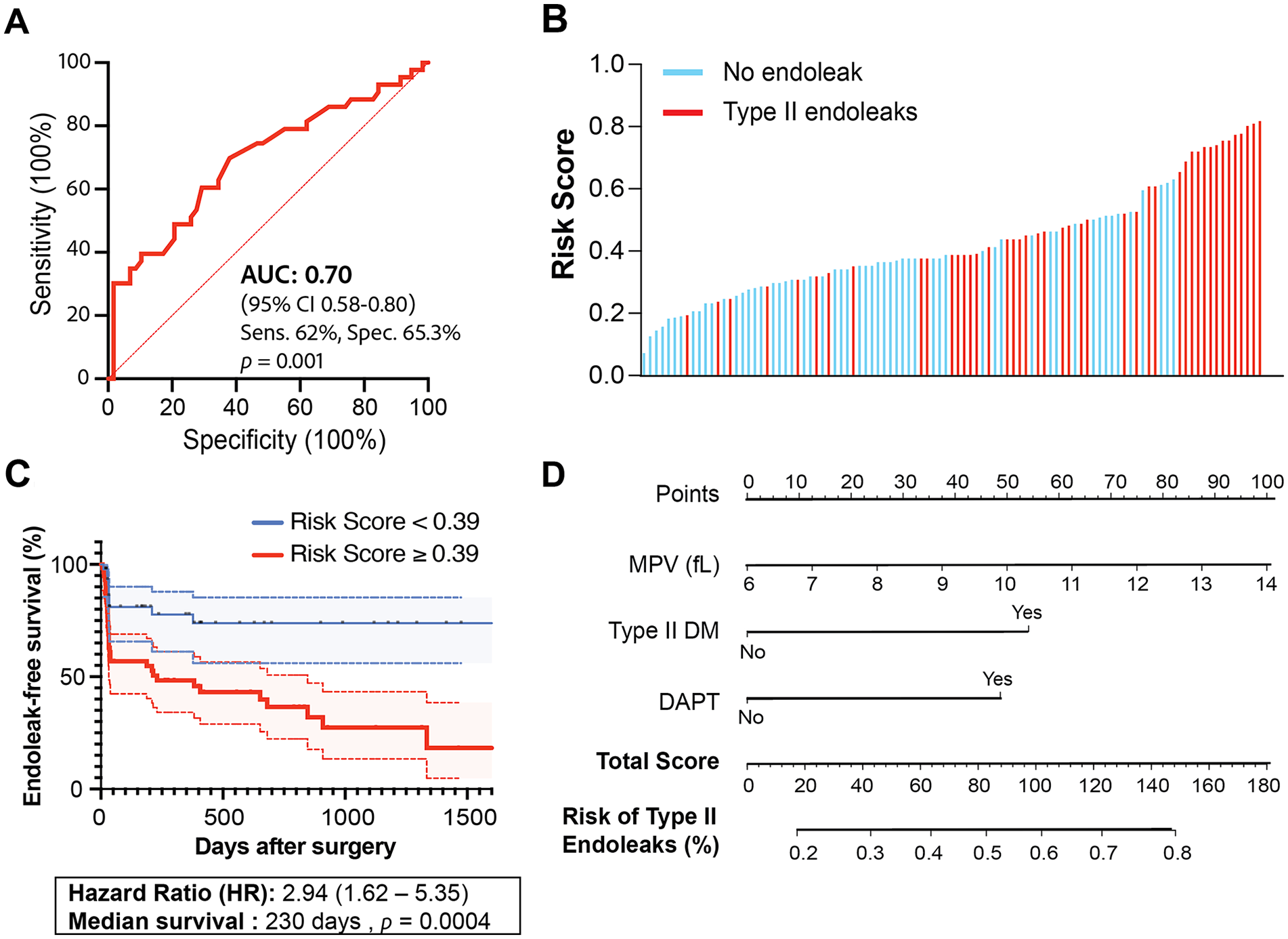
Risk stratification panel to distinguish type II endoleaks after EVAR. (
To test the potential clinical utility of this risk stratification panel, we constructed a user-friendly nomogram as a simple graphical representation that allows clinicians to estimate the likelihood of the occurrence of type II endoleaks post-EVAR procedure (Figure 2D). This nomogram integrates the effect estimator of each significant variable (MPV, type II DM, and DAPT) by formulating a numerical point system (0 to 100) based on each variable’s relative contribution to the risk of type II endoleaks. For instance, DAPT upon hospital discharge (yes/no) and type II DM (yes/no) were positively correlated with type II endoleaks; thus, patients who have type II or are assigned for DAPT treatment will be assigned higher point values, reflecting their increased risk. Interestingly, the nomogram algorithm assigned the highest scoring allocation to MPV, suggesting its strong predictive value in determining type II endoleaks.
We selected two patients with a type II endoleak (patients 1 and 2) and two without (patients 3 and 4) an endoleak to evaluate and verify the nomogram accuracy (Supplementary Figures 1A and 1B). As anticipated, patient 2, who had type II DM, received DAPT, and had an MPV of 11.5 fL, scored 170 on the nomogram, indicating > 80% likelihood of type II endoleaks post-EVAR procedure. True to the nomogram, patient 2 indeed experienced type II endoleaks 85 days following the EVAR procedure. Patient 1, despite exhibiting high MPV levels (MPV of 13.3 fL) and no DAPT usage or type II DM, received a score of 90, indicating a 55% risk of type II endoleaks post-EVAR. Remarkably, despite the moderately elevated risk, the real-life outcome of patient 1 revealed that they developed type II endoleaks just 19 days after surgery, highlighting the potential significance of MPV as an independent indicator of early type II endoleak onset. Additionally, the nomogram correctly predicted patient 3 (MPV = 8.4 fL, no type II DM, no DAPT) and patient 4 (MPV = 8 fL, no type II DM, DAPT upon discharge) to have low risks of type II endoleaks, 25% and 44% respectively, aligning with their actual endoleak outcomes (Supplementary Figure 1C).
Discussion
In this study, we identified that high MPV, indicative of larger and immature platelets,28,29 is elevated in patients who developed type II endoleaks following EVAR procedures. MPV has been described as a morphologic metric of platelet size assessed by mean volume, 30 bearing abundant mRNA, ribosomal content, 31 and more dense granules capable of increasing the storage of mediators of platelet activation. 32 However, though some studies have shown that high MPV is associated with increased platelet reactivity in CAD,33,34 contrasting results debating this association have been described. For example, no relationship between high MPV and platelet activity measured by impedance aggregometry using arachidonic acid was observed by De Luca et al. 35 Notably, high MPV was shown to associate with some (collagen and thrombin receptor activating peptide (TRAP)-induced platelet aggregation), but not all metrics of platelet activity (arachidonic acid and adenosine diphosphate (ADP)-induced platelet aggregation), 36 suggesting a more complex relationship between MPV and platelet activity. Indeed, patients with Bernard–Soulier syndrome (BSS) have excessive bleeding and typically have larger platelets with high MPV. 37 While more studies are needed to define the role of high MPV in type II endoleaks, our data suggests that high MPV can be used as a metric to predict the occurrence of type II endoleaks following EVAR.
Our data also strongly suggest a relationship between altered platelet phenotype and the occurrence of type II endoleaks. Indeed, it has been shown that EVAR significantly impacts platelet behavior, 38 leading to reduced platelet counts and increased prothrombotic activities, likely as a compensatory mechanism to stabilize the graft and prevent endoleaks. We observed that DAPT prescribed upon hospital discharge significantly correlated with type II endoleaks in our cohort. These observations indicate that despite the common clinical practice of DAPT administration to reduce prothrombotic events and endotension, 39 it might be linked to the development of type II endoleaks. Together, this result adds to several other reports linking antiplatelet therapy to an increased risk of type II endoleak post-EVAR.40–42 Conflicting evidence remains to understand the relationship between MPV and DAPT, with some studies reporting increased MPV following antiplatelet use in acute coronary syndrome 35 and clopidogrel nonresponsiveness. 43 Conversely, no effects of aspirin on MPV was observed in a cohort of healthy volunteers. 36 Future studies with larger sample sizes within multicenter patient cohorts are necessary to analyze EVAR patients with or without DAPT administration on hospital discharge and link them to the occurrence of type II endoleaks during follow-up visits.
Emerging studies have provided evidence for the involvement of platelets in the development of AAA. 44 Blocking several surface receptors of platelets such as P-selectin was shown to decrease the risk of AAA rupture in rat and mouse models.45,46 Moreover, intramural thrombus formation has been linked to higher rates of aneurysm sac growth and increased concentrations of reactive oxygen species, matrix metalloproteinases, and inflammatory cytokines, contributing to the weakening of the vessel wall.47–50 In patients undergoing elective EVAR, lower platelet counts after 1 week of surgery were associated with higher rates of persistent type II endoleaks requiring secondary interventions. 17 Our study expands on the role of platelets in the AAA field by identifying that elevated MPV in the immediate operative period may be an important indication of the development of type II endoleaks following elective EVAR. Given that higher MPV is generally associated with greater proportions of large and immature platelets circulating in the bloodstream,28,29 these correlations extend to postsurgical complications given that higher MPV has previously been linked to AAA rupture. 51 Moreover, MPV has been reported as a marker of inflammation in many diseases.28,52,53 Elevated MPV tend to reflect a state of more reactive platelets, 30 which are often found in pro-inflammatory milieu. It is possible that the activation of megakaryocytes by pro-inflammatory cytokines increases the ploidy of the cells 54 and promotes the release of larger, immature and more reactive platelets that amplify the inflammatory response. In cardiovascular diseases, elevated MPV is associated with the incidence of acute myocardial infarction, 33 higher thrombotic risk for elderly patients, 55 and mortality following ischemic heart disease. 56 Though it was technically not possible to capture local inflammation in the aortic tissue in our cohort, we did not detect overt changes in white blood cell count (WBC) between no-endoleak and type-II endoleak patient groups. Based on this evidence, we can speculate that a subset of platelets with high MPV could indicate platelet pools with high inflammatory roles and defective coagulation properties. This could explain the likelihood of endoleaks as deleterious outcomes of post-EVAR procedures.
Our findings indicate that elevated MPV was observed specifically in patients who were operated on using EVAR but not fEVAR. fEVAR is preferred for more complex aneurysms near critical arteries, using custom stents with fenestrations to maintain blood flow to vital organs and offers a minimally invasive alternative to open repair for patients. The infrarenal segment of individuals undergoing fEVAR exhibited much less expansion over time compared to EVAR, indicating that fEVAR may offer enhanced stability of the infrarenal neck contributing to more effective sac remodeling and better long-term durability of the repair. 57 However, a study revealed that a larger (33.1 ± 6.6 mm) seal zone is instead associated with a higher thrombotic event, 58 raising questions about the impact of seal zone size on platelet dynamics post-fEVAR. Based on these results and our data, which indicate no difference in platelet volume in fEVAR, this suggests that endoleaks observed in fEVAR might be due to alternative mechanisms possibly independent of platelet activity. These results also indicate that the EVAR patients with type II endoleaks have an inherently different population of platelets that may remodel the aorta. Confirmatory studies in larger cohorts that analyze and merge platelet behaviors and morphologies with computational fluid dynamics (CFD), and can differentiate EVAR and fEVAR patients in multiple pre- and postsurgical time points of outcome, would be necessary to identify the causality of high MPV in endoleaks.
Notably, type II diabetes emerged as a significant clinical factor associated with type II endoleaks. This may be attributed to the impaired tissue repair, delayed healing and abnormalities in extracellular matrix (ECM) production, and remodeling often observed in patients with type II DM, 59 which could potentially influence the outcome of EVAR. Our findings align with previous studies that have reported a higher risk of adverse outcomes after AAA repair in patients with type II DM. 60 Similarly, increased MPV has been observed in these patients, showing a positive correlation with cardiovascular complications. 61 The relationship between type II diabetes, platelet morphology, and endoleaks warrants further investigation to determine the contribution of each factor to the healing process of the repaired aorta post-EVAR.
No significant differences were observed in other blood parameters between patients with and without endoleaks (Table 1). Specifically, WBC count, a common blood marker for inflammation, showed no significant variation, suggesting that systemic inflammation may not play a dominant role in the development of endoleaks in our cohort. Similarly, the red blood cell (RBC) count profile − including RBC count, hemoglobin, hematocrit, and RDW − was comparable between the two groups. This suggest that there were no major disruptions in erythropoiesis or RBC morphology that could be linked to the presence of endoleaks.
This study has some limitations. It is a single-center retrospective cohort study with a relatively small sample size. Larger multicenter validation cohorts are essential to confirm these findings. Moreover, although we observed relatively few significant differences in baseline characteristics across study groups, follow-up time emerged as an important potential confounder for our outcomes, with patients developing type II endoleaks having longer follow-up periods. We controlled for the bias introduced by differential follow-up time using Cox proportional hazards analysis to model event-free survival for our outcomes of interest before conducting predictive analyses. In this study, the limited follow-up period hindered the identification of aneurysm expansion in the patient cohort.
Conclusion
We report that high MPV is associated with the occurrence of type II endoleaks following EVAR. Further research is warranted to gain insights into this relationship. For example, in future studies, we will conduct a prospective analysis on EVAR patients with or without type II endoleaks to define platelet characteristics (morphology, maturity, and activation) pre- and post-EVAR. By also evaluating the percentage of reticulated platelets using thiazole orange, we will be able to identify the pool of immature platelets pre- and post-EVAR segregated by type II endoleaks. MPV values will be correlated with platelet activation to different agonists, and to aggregation (impedance aggregometer). These new results will offer mechanistic insights into the relationship between MPV, platelet reactivity/maturity, and outcomes following EVAR. MPV level along with clinical parameters can be used to establish a robust panel of risk stratification models for type II endoleaks following EVAR. These results should be validated in a larger multicenter cohort before they could be utilized as a low-cost approach for the surveillance of endoleaks.
Supplemental Material
sj-png-1-vmj-10.1177_1358863X241298926 – Supplemental material for Altered platelet phenotype in patients with type II endoleaks following abdominal aortic aneurysm repair
Supplemental material, sj-png-1-vmj-10.1177_1358863X241298926 for Altered platelet phenotype in patients with type II endoleaks following abdominal aortic aneurysm repair by Muhammad Yogi Pratama, Benjamin King, Katherine A Teter, Christina Bi, Natalie Presedo, Keerthi B Harish, Catarina Santos Giardinetto, Sheehan Belleca, Ethan Chervonski, Thomas S Maldonado and Bhama Ramkhelawon in Vascular Medicine
Footnotes
Acknowledgements
A commentary by Aggarwall et al. accompanies this article. 62
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The B. Ramkhelawon laboratory is supported by the National Institutes of Health (R01 HL146627 and R01HL149927).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
