Abstract
Patients undergoing coronary artery bypass grafting (CABG) are at increased risk of perioperative stroke. Carotid atherosclerosis has been identified as an independent risk factor for stroke during and in the early postoperative period of cardiac surgery. However, the pathogenesis of peri-CABG stroke is multifactorial and frequently involves other noncarotid causes, such as cardiac emboli and aortic atheroma. Therefore, routine population-wide screening of carotid stenosis is not recommended, but target screening of patients at high risk of carotid-related perioperative stroke can have benefits. Carotid duplex sonography is recommended as an initial screening tool. Elimination of carotid stenosis before cardiac surgery is indicated in patients in whom carotid atherosclerosis is suspected to be the primary contributor to perioperative stroke. In patients with advanced carotid atherosclerosis, an individualized revascularization approach, including simultaneous or staged procedures, is advocated. The prevailing consensus is that synchronous surgery is safer than staged procedures. Carotid artery stenting represents a less invasive alternative, but its role in high-risk patients requires further investigation. In conclusion, the risk of perioperative stroke in patients undergoing CABG involves different factors, and carotid artery stenosis is involved in its pathogenesis only in some patients. Therefore, individualized approaches and careful consideration of patient risk factors are essential in determining the need for carotid screening and revascularization before CABG.
Introduction
Atherosclerosis is a systemic condition that can concurrently affect multiple arterial beds. Between 2% and 14% of patients have coexistent coronary (CAD) and carotid artery disease. Of those undergoing coronary artery bypass grafting (CABG), 8% have significant carotid artery stenosis. 1 Conversely, clinically evident CAD is present in nearly one-third of patients with advanced carotid stenosis in whom carotid endarterectomy (CEA) is indicated, and in patients undergoing CEA, the prevalence of CAD is much higher and was found in 40–50%. 2
Stroke remains a major cause of morbidity and mortality after CABG. The period of stroke related to CABG is divided into three categories: intraoperative (detected immediately after extubating the patient), early postoperative (7 days after the operation), and late (beyond 7 days). Intraoperative and early postoperative stroke are also categorized as perioperative stroke. 3 The general risk of perioperative stroke in patients with CABG is between 1.3% and 2.0%, and its etiology differs.4,5 The main causes of stroke during the operative procedures are thromboembolism and hypoperfusion. Most strokes that occur during the first 7 postoperative days are related to arrhythmias and low cardiac output syndrome, but the late stroke is usually thromboembolic and is caused by the progression of cerebrovascular atherosclerosis and atrial fibrillation. 6 Extracranial carotid disease has been shown to be involved in the etiology of perioperative CABG stroke. However, the data indicate that only advanced carotid atherosclerosis – severe unilateral stenosis (> 70%) or occlusion, bilateral stenosis, and unfavorable morphologic characteristics of the carotid lesion (e.g., ulceration) – independently predict an increased risk of stroke during cardiac CABG. 7 Most intraoperative strokes (70–80%) are caused by thromboembolism because of aortic manipulation or atrial fibrillation, and 20–30% follow hypoperfusion secondary to hypotension.6,7 Older age, female sex, diabetes mellitus, hypertension, left main CAD, peripheral vascular disease, advanced atherosclerosis of the aortic arch, and carotid stenosis were identified as important risk factors for perioperative stroke.8,9 Perioperative stroke is related to high mortality. A meta-analysis of 34 articles, which included 174,969 patients with cardiac operations, showed that the pooled event rate for early (intraoperative) stroke was 0.98% (95% CI 0.79 to 1.23) and delayed (postoperative) stroke occurred in 0.93% (95% CI 0.73 to 1.11; p = 0.68). Intraoperative stroke had a 30-day mortality of 29%, those with postoperative stroke 18%, and in patients with no stroke, perioperative mortality was only 2.4%. 3 The incidence of perioperative stroke varies among different types of surgical procedures, and in CABG it accounts for an average of 2%. 10
This review summarizes the current evidence on the pathogenesis of stroke after CABG and suggests strategies for patients in need of investigation of carotid arteries before CABG to reduce the risk of stroke related to carotid lesions.
Methods
This article is a narrative review. A comprehensive search was performed to identify studies examining the incidence and risk of carotid atherosclerosis and the need for carotid revascularization before CABG. The following databases were searched: Ovid Medline, Ovid Embase, and Cochrane Library. The most relevant papers, according to methodological and clinical considerations, were selected.
Causes of stroke related to carotid artery disease during cardiac surgery
Carotid atherosclerosis is involved in the pathogenesis of CABG-associated perioperative stroke only in some patients. Less than 50% of patients suffering from stroke will not have significant carotid disease, and a quarter of strokes result from embolic events related to carotid artery stenosis. Only severe carotid artery stenosis (80–99%) represents the risk of perioperative stroke 11 (Table 1). In a prospective multicenter observational study involving 2813 patients undergoing CABG, asymptomatic carotid stenosis greater than 50% was identified in 26% of the patients, whereas carotid occlusion was found in 1.1% of cases. 12 In patients undergoing CABG without significant carotid lesions, the stroke rate is less than 2%.7,13 The stroke rate increases with the progression of atherosclerosis: in asymptomatic carotid lesions it is about 3% if a patient has unilateral 50–99% carotid stenosis, 5% in those with bilateral 50–99% stenosis, and 7–11% in those with carotid occlusion. 10 This review additionally demonstrated that half of the stroke patients during the perioperative period lacked significant carotid disease, and up to 60% did not exhibit territorial infarctions on autopsy/computed tomography that aligned with carotid disease patterns. However, patients undergoing CABG with prior transient ischemic attack (TIA) or stroke had the highest rate of postoperative stroke. 10 In the study by Chen et al., the incidence of post-CABG stroke was 1.7%: 8% in patients with severe (> 70%) carotid stenosis and 0.0% with nonsevere carotid stenosis. 7 The systemic review of 36 observational studies showed that prior stroke was the most important predictor of perioperative stroke, whereas carotid stenosis was not a statistically significant predictor of stroke. 3 Besides carotid stenosis there is also an interrelationship between other cerebrovascular diseases and perioperative CABG stroke. 14 A retrospective cohort study, which included 20,241 patients undergoing CABG, showed that the risk of post-CABG stroke increased with the degree of carotid stenosis. An almost threefold increased risk of postoperative neurologic events occurred in patients with 80–90% carotid stenosis in comparison to those with 50–79%. 15 These findings indicate that the risk of perioperative CABG stroke increases with the severity of carotid stenosis, but only in advanced forms of carotid atherosclerosis. Different studies otherwise showed that carotid endarterectomy decreases the 5-year risk of ipsilateral stroke in patients with asymptomatic isolated carotid disease. 4 However, the long-term risk reduction of stroke by operative procedure should not be used as a justification for performing carotid revascularization in patients undergoing CABG.
Mechanisms of stroke in patients undergoing CABG.
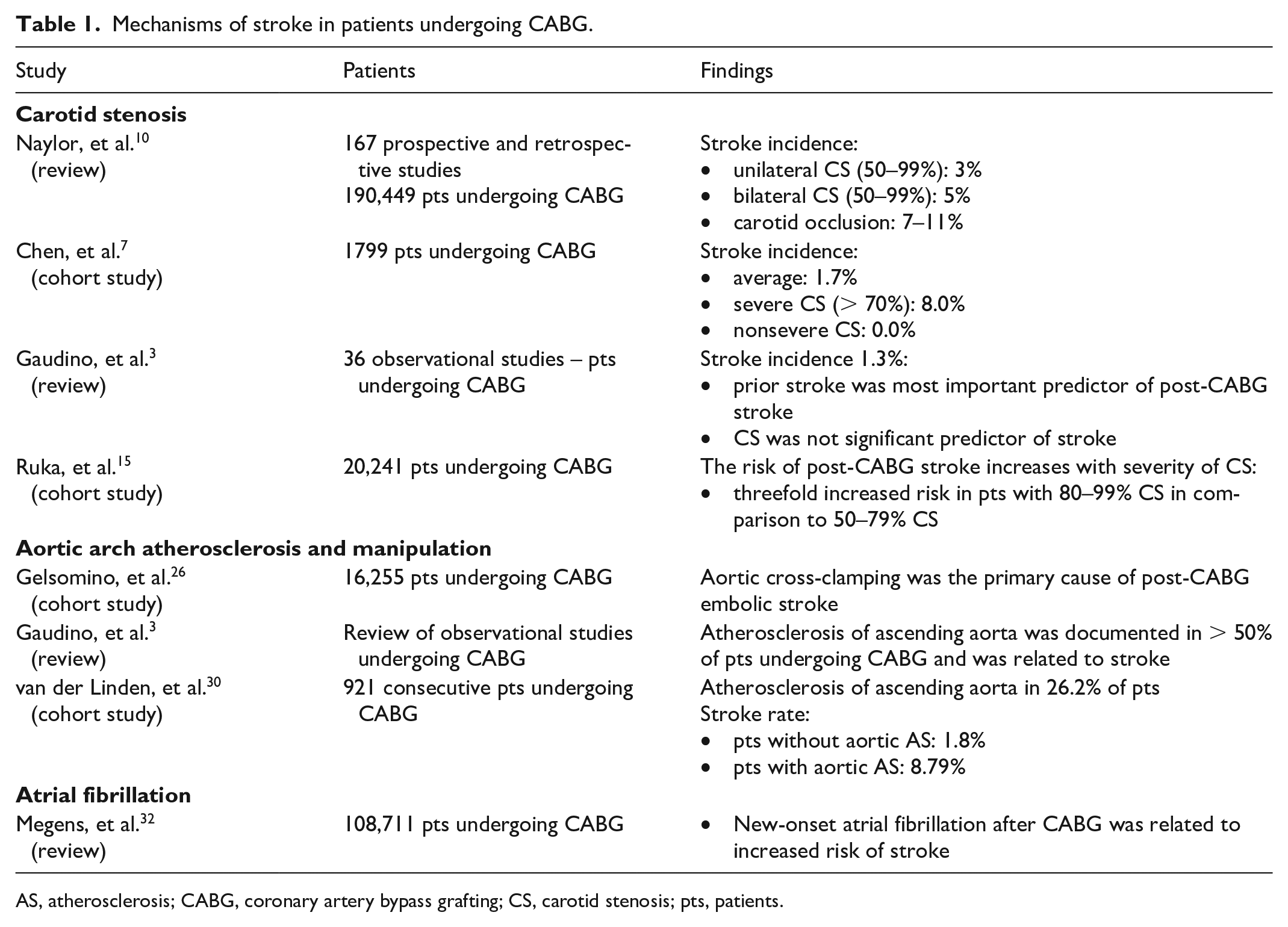
AS, atherosclerosis; CABG, coronary artery bypass grafting; CS, carotid stenosis; pts, patients.
There are different mechanisms by which carotid artery stenosis could lead to perioperative stroke. It was shown that carotid intraplaque hemorrhage can destabilize plaque and cause thromboembolism. Intraplaque hemorrhage is associated with an increased risk of ipsilateral stroke.16–18 Administering anticoagulants during CABG may contribute to an increased risk of carotid intraplaque hemorrhage during operation. 19 At the same time, additional mechanical factors, such as turbulent blood flow and hypertension, can also trigger this event. 20 Further, plaque morphology influences the cardiovascular thromboembolic event rate. It includes lipid reach plaque, extensive plaque, thin fibrous cap, heavily calcified plaque, and stenosis at the bifurcation involving both internal and external carotid arteries. 21 Impaired cerebral autoregulation distal to carotid stenosis is another risk factor of perioperative stroke, which causes cerebral ischemia and is associated with an increased risk of stroke. 22
Noncarotid causes of stroke during cardiac surgery
The cause of peri-CABG stroke is multifactorial and is frequently caused by noncarotid causes. There are no studies that have systematically assessed stroke risk in asymptomatic carotid patients and identified those subjects with asymptomatic carotid stenosis for whom CABG without carotid revascularization poses a greater risk of stroke compared to prior carotid surgery.
Carotid atherosclerosis as a cause of stroke during CABG is probably overestimated. Particularly in patients with asymptomatic carotid stenosis, CABG stroke is more likely to be caused by noncarotid atherosclerosis, including cardiac emboli and atherosclerotic lesions in the ascending aorta. 23
Manipulation of the aorta represents one of the main causes of perioperative CABG stroke 24 (Table 1). In this study, two-thirds of strokes occurred after surgery. This indicates that hemodynamic deterioration during surgical procedures has a less important role in the pathogenesis of perioperative stroke. The studies also showed that 70% of perioperative strokes are bilateral or multiple. This indicates that the sources of emboli are not carotid arteries but proximal territories, including the ascending aorta.3,25
Macroembolization with atherothrombotic material from the atherosclerotic deteriorated aorta is typically related to CABG-provoked postoperative stroke. 26 Further, intracardial thrombus and intermittent clamping of the aorta during surgery are predictors of major perioperative stroke. 27 The dislodgement of emboli from the ascending aorta, potentially triggered by its manipulation during surgery, can provoke embolic stroke. 28 The risk of CABG stroke correlates closely with the presence or absence of extended (> 5 mm into the aorta lumen) ulcerated plaques or the presence of multiple aortic atheromas. Patients with severe aortic disease, represented by porcelain aorta, which is characterized by circumferential calcification throughout the whole perimeter of the aorta, have a 5–19% risk of perioperative stroke compared with 0–2% in patients who do not have a significant aortic atherosclerotic disease. 29 In the study by van der Linden et al., which included 921 consecutive patients undergoing cardiac surgery, 26.2% of patients had atherosclerosis of the ascending aorta. 30 The incidence of stroke was 1.8% in patients without atherosclerotic disease of the ascending aorta and 8.7% in patients with aortic disease. 30 It indicates that evaluation of atherosclerotic deterioration of the ascending aorta may improve risk stratification of stroke prior to CABG. 31 A significant cause of embolic stroke is also atrial fibrillation and mobilization of blood clots from the heart during manipulation by the surgeon.32,33 Also, atherosclerotic lesions of cerebral arteries can cause embolic stroke in patients undergoing CABG. All these factors are important in the pathogenesis of perioperative stroke, as approximately 45–50% of coronary patients also exhibit severe aortic and intracranial atherosclerotic disease. 34 Therefore, perioperative stroke is not merely the consequence of carotid atherosclerosis, but the other noncarotid risk factors must be borne in mind.
In patients undergoing CABG, the risk of stroke is also related to the operative procedure. Off-pump surgery reduces the amount of aortic manipulation by avoiding cannulation and cross-clamping. A meta-analysis found that on-pump CABG was associated with an increased incidence of stroke in comparison to off-pump CABG. 35 Therefore, the preoperative treatment of asymptomatic carotid stenosis cannot completely prevent perioperative strokes. One of the systemic reviews showed that prophylactic carotid revascularization could prevent only 40–50% of strokes in patients undergoing CABG. 10 Particularly in patients with significant cardiac and/or aortic atherosclerotic disease, carotid revascularization would prevent fewer perioperative strokes.
Carotid screening before CABG
Carotid duplex sonography (DUS) is the most widely utilized screening method to assess carotid arteries, particularly the degree of stenosis. This noninvasive technique employs B-mode and Doppler imaging to evaluate blood flow within the carotid arteries. Carotid DUS offers extensive availability and excellent sensitivity (94%). 36 Alternative noninvasive diagnostic test options include computer tomography angiography (CTA) and magnetic resonance angiography (MRA). Despite their advantages, MRA and CTA present limitations. MRA may overestimate the degree of stenosis, whereas the value of CTA’s evaluation of calcified lesions is limited. Additionally, both techniques struggle to distinguish between subtotal and complete arterial occlusion. 37 Catheter-based angiography offers a more detailed anatomical view of arteries than traditional angiography. Because of the invasiveness, it is usually used before therapeutic interventions such as CAS. 38
Besides the degree of stenosis, plaque morphology influences plaque vulnerability (rupture) and is related to thromboembolic events. Identification of vulnerable plaque, irrespective of the degree of stenosis, can help to identify patients who are at the highest risk of thromboembolic carotid-related embolic events during CABG. 21 Therefore, the degree of stenosis alone is not sufficient to decide which patients are at the highest risk of perioperative stroke. Noninvasive imaging and invasive techniques are used to determine plaque morphology. 39 Ultrasound B-mode ultrasonography and contrast-enhanced ultrasonography are the two most widely used techniques for assessing carotid plaque morphology. Further, high-resolution magnetic resonance imaging is currently considered the most competitive imaging method for the examination of the carotid artery wall morphology. 40 Positron emission tomography assesses intraplaque inflammation and angiogenesis. Intravascular ultrasound enables the presentation of two-dimensional images in real time, displaying different components of the vessel wall.41,42
Carotid stenosis is common in patients with CAD, but only a few carotid lesions are related to perioperative stroke. Therefore, the effectiveness of nonselective carotid screening in patients undergoing CABG is unknown. Further, selective or nonselective carotid DUS screening identifies only a low proportion of patients who will develop perioperative stroke. 36 However, despite this finding, most (approximately 90%) of carotid revascularizations are performed on neurologically asymptomatic patients. 43 Also, observational data showed that the risk of stroke in neurologically asymptomatic patients with unilateral 70–99% stenosis undergoing CABG is exceedingly low. 2 Furthermore, with the combined operation of carotid and coronary arteries, an increased risk of morbidity and mortality due to the potential risk of stroke and acute myocardial infarction (AMI) is expected. 44 Therefore, it indicates again that carotid revascularization before or in combination with CABG might not significantly decrease the risk of neurologic complications, particularly in asymptomatic patients. Consequently, prophylactic investigation and revascularization of carotid arteries may be of no significant benefit in preventing perioperative stroke. 45 The significant advantage of carotid revascularization in patients undergoing CABG is expected only in those patients with a history of neurologic events (TIA or stroke) and severe carotid atherosclerotic lesions. 46 Also, recent guidelines do not recommend population-wide screening of asymptomatic carotid stenosis by duplex ultrasound. 47 European Society of Vascular Surgery (ESVS) guidelines concluded that in patients undergoing open heart surgery, routine carotid surgery is not recommended (Class III, Level of evidence: C). 48 The ACC Foundation/AHA Task Force on Practice Guidelines recommend that performing carotid DUS is justifiable in specific patients identified with high-risk factors, including individuals aged over 65 years, those with a carotid bruit, left main coronary artery stenosis, peripheral artery disease, hypertension, a history of smoking, diabetes mellitus, and a past medical history of cerebrovascular disease (stroke or TIA) or cervical carotid disease (Class IIa, Level of evidence: C) 49 (Figure 1). Also, the Society of Vascular and Interventional Neurology suggested screening a specific group of patients with suspected carotid lesions undergoing CABG (grade B recommendation). 50
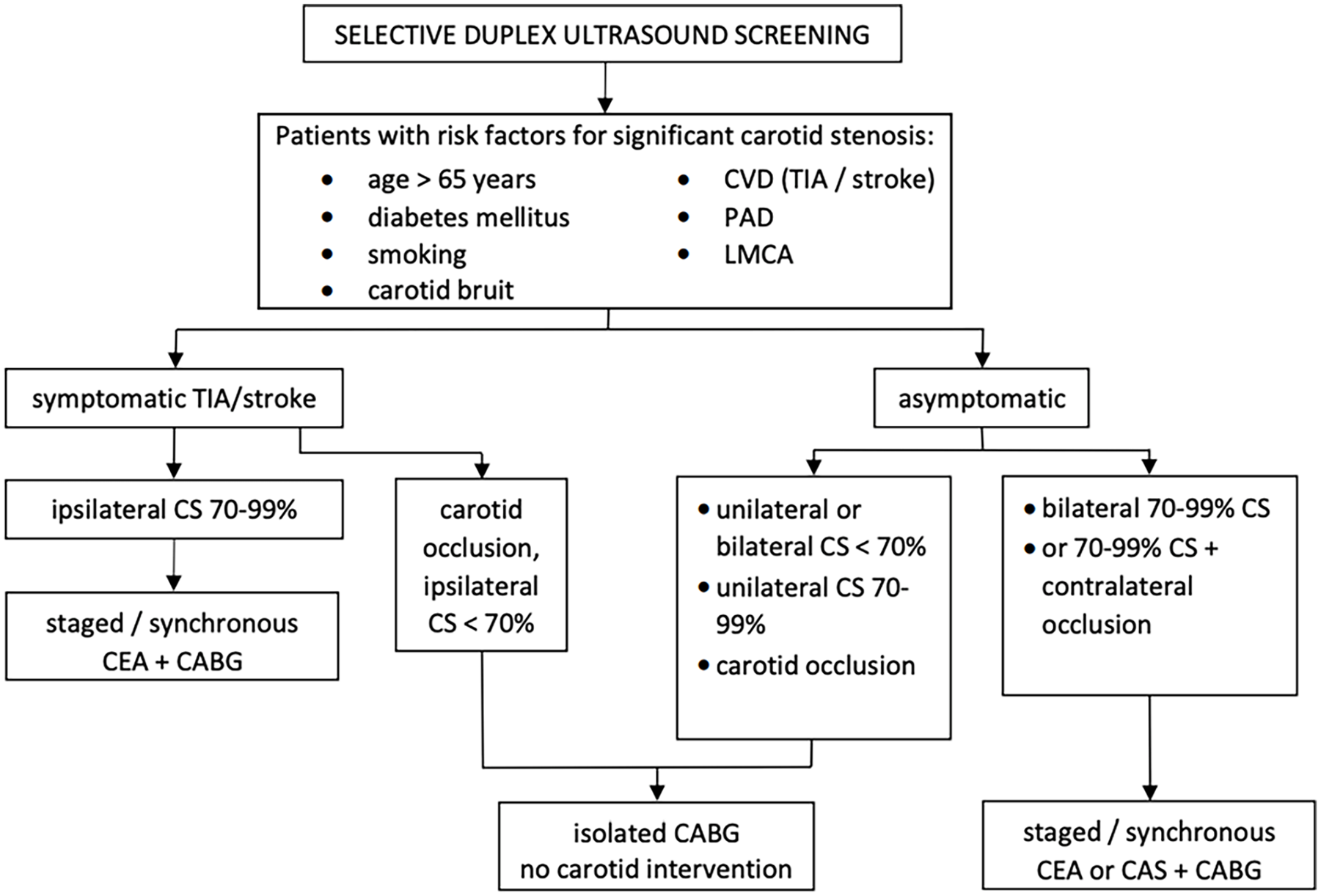
Screening and management of carotid stenosis (CS) in patients undergoing coronary artery bypass grafting (CABG).
Besides detecting carotid atherosclerosis using imaging techniques in subjects undergoing CABG, clinical variables alone can help identify patients with significant carotid artery disease who may need additional investigation. 4
Risk of carotid revascularization procedures in patients undergoing CABG surgery
Carotid revascularization is accepted and well supported by evidence from patients undergoing CABG who have symptomatic carotid disease, bilateral 70–99% carotid stenosis, or unilateral 70–99% stenosis with contralateral carotid occlusion48,51 (Figure 2). Nevertheless, these procedures are mainly carried out preventively in patients with asymptomatic unilateral carotid stenosis. The effectiveness and significance of such preventive measures remain uncertain.52,53 Various findings did not answer the question of the utility of carotid revascularization in asymptomatic patients undergoing CABG due to the absence of a direct relationship between carotid artery atherosclerosis and perioperative ipsilateral stroke. 54
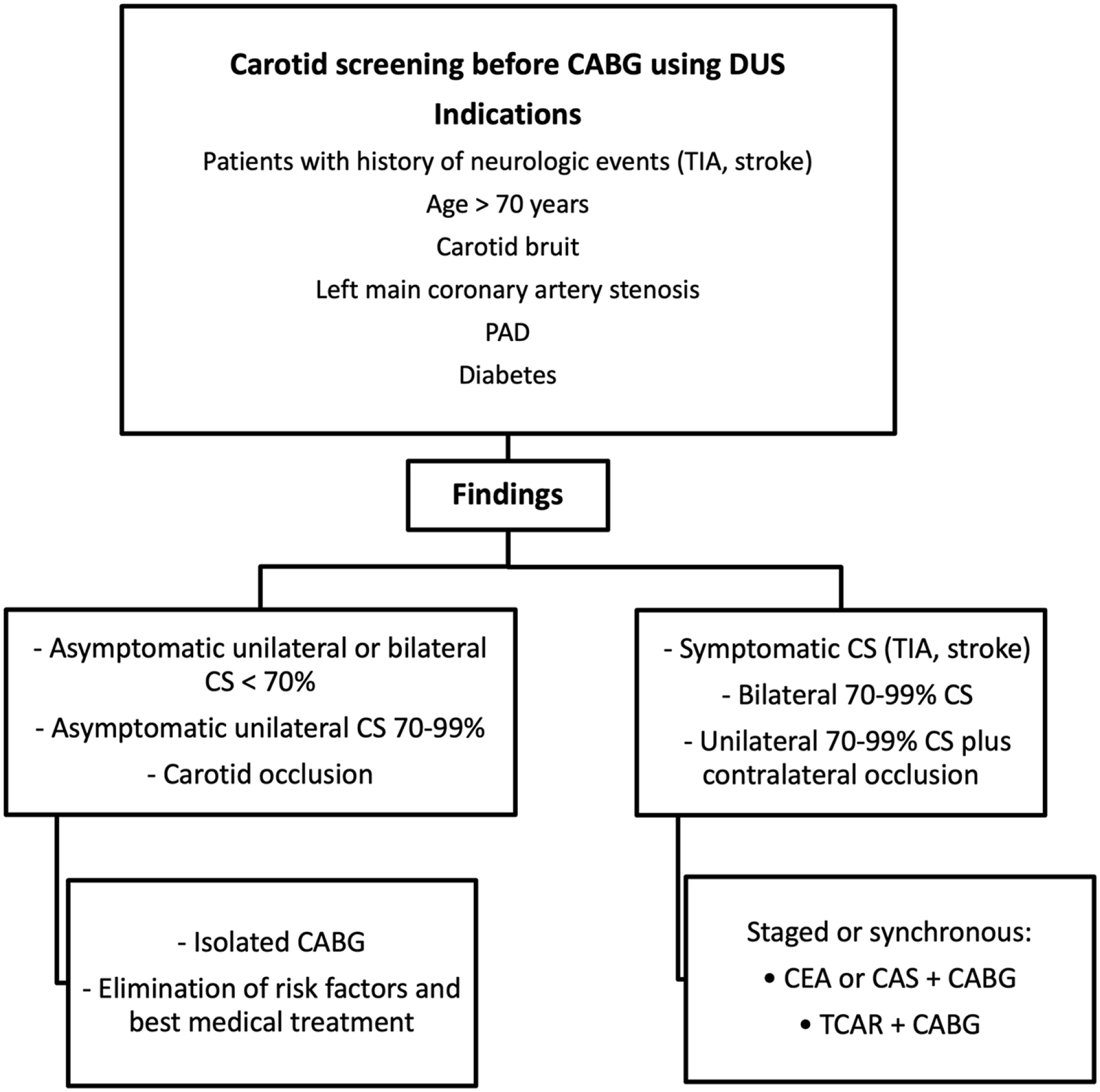
Management of patients with carotid stenosis (CS) undergoing coronary artery bypass grafting (CABG).
The optimal approach for carotid and coronary revascularization remains controversial. Different treatment strategies exist, which include combined or synchronous surgical procedures where CABG and CEA are performed simultaneously; in staged surgical procedures, where CABG is performed with subsequent CEA or CEA with subsequent CABG; and hybrid techniques, which consist of CABG with carotid artery stenting (CAS) or percutaneous coronary intervention (PCI), and percutaneous procedures, which combine PCI with CAS. 2 There is currently no consensus on the temporal sequence of revascularization of carotid and coronary arteries in patients undergoing CABG, with evidence supporting both simultaneous and staged procedures. 55
A systemic review and meta-analysis showed a significantly higher risk of stroke and death, but a lower risk of AMI with the combined compared to the staged procedures. 56 In detail, the synchronous approach is associated with elevated stroke and mortality rates, ranging from 7.4% to 9.4%, 57 which is significantly higher than the risk of each operation alone. Another review of 1160 procedures found an even greater risk of stroke and/or death in patients with synchronous CEA and CABG. Notably, 18.6% of patients in this group experienced these adverse events. 58 A systematic review analyzing 7863 patients with combined CEA and CABG revealed a nonnegligible cardiovascular risk associated with the combined procedure. 59 In this review, a significant reduction in the risk of death/stroke was observed in studies published between 1993 and 2002 compared with those from 1972 to 1992 (7.2% (95% CI 6.5 to 9.1) versus 10.7% (95% CI 8.9 to 12.5), p = 0.03). Notably, similar information was unavailable for a comparable cohort of patients with CABG in whom prophylactic CEA was not performed. Therefore, recently, the need for revascularization of carotid arteries has probably been reduced entirely.
Consistent with the elevated risk observed in synchronous procedures, patients who underwent CEA prior to CABG also faced increased complication rates. The CEA guidelines prepared by the Stroke Council and American Heart Association registered a 16.44% incidence of stroke, AMI, and death in patients with combined CEA and CABG. 60 This risk remained substantial even with staged procedures, with 26.15% for CEA followed by CABG and 16.35% for CABG followed by CEA. According to the ESVS, for patients with concomitant severe coronary and carotid artery disease, an individualized surgical approach is recommended. 48 Owing to the substantial operative complication rate associated with carotid surgery, either as an alone procedure or in conjunction with CABG, the long-term benefits of secondary stroke prevention are expected to be limited to high-risk patient populations.
Strategies for enhanced perioperative neurological safety in CABG procedures
To mitigate the risk of perioperative neurological events in patients undergoing CABG, several strategies can be implemented, including meticulous blood pressure monitoring throughout the procedure and postoperatively. Maintaining normotension is crucial, as both hypertension and hypotension pose significant risks. Hypertension can provoke cerebral bleeding, whereas hypotension can lead to cerebral ischemia, both of which can have detrimental neurological consequences. 61 Intraoperative assessment of the aorta is recommended before clamping, and it should be followed by procedures to decrease the shedding of emboli from the aorta during surgery. 26
No statistically significant difference was observed in the risk of death or stroke when comparing pre- versus on-cardiopulmonary bypass timing of CEA. 59 However, several smaller studies suggested potentially improved outcomes when combining CEA with off-pump cardiopulmonary bypass.62,63
Carotid artery stenting (CAS) in patients undergoing CABG with high surgical risk
CAS has emerged as a promising alternative to CEA for the treatment of carotid artery disease. In the past, several small studies have indicated a significantly lower complication rate among patients undergoing CAS followed by CABG compared to those undergoing combined CEA and CABG. 64 Also, a meta-analysis of outcomes 30 days after the procedure showed that CAS before CABG or CAS plus CABG is an attractive and less invasive procedure than CEA plus CABG. 65 A systemic review, including 23 studies encompassing 873 and 459 patients in the staged and synchronous groups, evaluated the comparative outcomes of synchronous versus staged CAS and CABG in patients with concomitant carotid and CAD. 66 The overall death/stroke/AMI rate was 8.5% (95% CI 7.6 to 9.4) in the staged group and 4.8% (95% CI 3.8 to 5.8) in the synchronous group, suggesting a potential advantage of the simultaneous approach. However, due to the limitations of available data, statistical comparisons between the two groups were not feasible.
Another meta-analysis, which included 80% of patients with asymptomatic carotid stenosis, has shown similar mortality of staged CAS + CABG and synchronous CAS + CABG. However, there was a higher risk of stroke and AMI with staged CAS + CABG. 67 Feldman et al. examined 22,501 patients who had three different revascularization methods, and they found significantly lower rates (1.9%) of in-hospital death in CAS + CABG than in combined CEA + CABG (4.4%) and 3.8% in staged CEA + CABG. 68 The studies also showed that staged or simultaneous CAS and percutaneous coronary procedures have outcomes similar to hybrid surgical interventions. 69 Although the number of reported studies and included patients was small, the data support the validity of hybrid (CABG + CAS) revascularization, particularly in high-risk surgical patients.
Recently, a new, less invasive trans carotid artery revascularization (TCAR) was developed, which provides minimal invasive revascularization with stenting of the stenotic carotid arteries using direct carotid access while simultaneously reversing the flow of blood in the carotid artery aimed at minimizing the risk of stroke during the revascularization process. 70 The TCAR procedure can be performed under local anesthesia, and it minimizes aortic arch manipulation and consequently reduces antegrade embolic stroke rate. The preliminary outcomes of TCAR in high-risk patients seem to be more favorable than those of transfemoral CAS. 71 A retrospective study by Vyas et al. examined 14 patients with either symptomatic carotid artery stenosis greater than 50% or asymptomatic carotid artery stenosis greater than 70% alongside either contralateral internal carotid artery occlusion or widespread cerebral occlusive disease observed on radiographic imaging. These patients underwent staged TCAR before CABG surgery. Over a 12-month follow-up period, there were no reported cases of perioperative stroke, myocardial infarction, or death. 72
Performing TCAR with flow reversal concurrently with CABG has been recently suggested as an alternative approach for carotid artery revascularization in a patient with severe concomitant carotid and CAD. 73 In this case, TCAR is performed through a median sternotomy, which also mitigates stroke risk by avoiding aortic arch manipulation. However, the challenge with this approach is that patients cannot be on dual antiplatelet therapy before surgery, and the loading dose of clopidogrel can only be administered after the CABG procedure. Clopidogrel takes approximately 2 hours to reach its full effect, leaving the stent potentially unprotected during this period.
Conclusions
Patients with advanced carotid atherosclerosis who undergo CABG are at an elevated risk of stroke provoked by surgery. The risk is highest in patients with a history of stroke or TIA. However, beyond carotid atherosclerosis, other risk factors, particularly advanced cardiac and aortic atherosclerotic disease, also contribute to perioperative stroke. Consequently, screening for carotid artery stenosis prior to CABG is only recommended for patients with expected advanced atherosclerotic carotid lesions and concomitant atherosclerosis in other vascular beds. In patients with asymptomatic carotid stenosis, elimination of risk factors and best medical treatment represent basic therapeutic options. Elimination of carotid stenosis before coronary surgery is indicated in patients at high risk of perioperative stroke, where carotid atherosclerosis is suspected to be the primary contributor.
An individualized surgical approach is advocated as the most adequate management strategy in patients with CAD and advanced carotid artery disease. When considering surgical revascularization, choosing between synchronous or staged carotid revascularization and CABG is exacting. The prevailing consensus is that synchronous surgery is safer than staged procedures. For high-risk surgical patients, CAS represents a less invasive alternative prior to CABG.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
*
These authors contributed equally to this work.
