Abstract
Background:
External iliac artery endofibrosis (EIAE) is a rare vascular disease which has been traditionally seen in avid cyclists. The conventional approach has been surgery, although no high-quality evidence suggests superiority of surgery over percutaneous endovascular intervention. There are limited data on the efficacy of stenting in EIAE.
Methods:
Over a 14-year period, we treated 10 patients (13 limbs) with EIAE with stents. These patients had declined surgery. The mean follow up was 8.4 ± 3.3 years. There were eight women. Five patients were competitive runners, three were cyclists, and two were triathletes. The mean age was 40.7 ± 2.9 years and body mass index was 19.46 ± 1.6. Intravascular ultrasound (IVUS) was used in eight limbs.
Results:
Procedural success was achieved in all. The recurrence of symptoms occurred in three patients at a mean of 9.3 ± 2.1 months postindex intervention. The other seven patients remained symptom free. IVUS revealed a pathognomonic finding which we termed ‘perfect circle appearance’. It results from symmetric or asymmetric hypertrophy of one or more layers of the arterial wall leading to negative remodeling, which creates a distinct echo dense structure contrasting itself from the luminal blood’s echoluscent appearance. It is identical to IVUS images of diffuse venous stenosis with important implications in the treatment technique.
Conclusions:
We conclude that stenting in EIAE is safe and effective with a good long-term outcome. It can be an alternative to surgery, particularly in those patients who refuse a surgical approach. The IVUS image is pathognomonic and ‘sine qua non’ of EIAE.
Keywords
Background
External iliac artery endofibrosis (EIAE) is a rare vascular disease affecting young high-performance athletes.1–3 It has been traditionally reported in competitive cyclists, which, if left untreated, can lead to cessation or limitation of their activity.2,4,5 None of the traditional atherosclerotic risk factors for vascular disease are at play in the pathogenesis of EIAE and consequently the diagnosis may be delayed due to lack of awareness of providers. Often their symptoms are dismissed as ‘neuropathy’, ‘arthritis’, or ‘psychiatric’ in nature. The classic diagnostic tools such as ankle–brachial index (ABI) and imaging studies are often interpreted as ‘normal’. The conventional approach to treatment has been surgery.3,6,7 This choice has not been based on good quality evidence but tradition, and the limited benefit of a few endovascular reports which had primarily used balloon angioplasty alone.8–10 These patients are young and very active and usually prefer alternatives to surgery. We herein report the long-term follow up of our experience with 10 patients (13 limbs) with EIAE who underwent percutaneous endovascular stenting.
Methods
Study population
From October 2006 through June 2020, we treated 10 patients with symptomatic EIAE with endovascular stents. There was a total of 13 EIAE as three patients had bilateral disease. They had all refused surgery. This study was approved by the Institutional Review Board of A.T. Still University under a consent exemption decision due to the retrospective nature of the investigation.
Patient selection and outcome measurement
The patients were identified retrospectively from our office computerized database and were followed prospectively for clinical outcomes. The patients’ medical records including clinical histories, angiograms, and available intravascular ultrasound (IVUS) studies were reviewed. Recorded information included: type of exercise, type and duration of symptoms, age at the time of procedure, body mass index (BMI), height, sex, bilaterality of disease, level of exertion to elicit symptoms, type of stents implanted, and restenosis rate. The mean follow up from the index operation was 8.4 ± 3.3 years.
To establish the diagnosis, the patients were to be relatively young athletes with the development of lower-extremity ischemic symptoms at high levels of exertion. They were then screened for evidence of stenosis in the external iliac artery by duplex ultrasound (and computed tomography angiography [CTA] if available). If there was a relative reduction, albeit small, in the diameter of the external iliac artery in comparison to the more distal arterial segments, EIAE became highly suspected. These patients underwent invasive angiography. If any reduction in the angiographic diameter of the external iliac artery was noted as compared to more distal arterial segments, the diagnosis of EIAE was made. No provocative testing was performed during angiography. Some patients underwent IVUS imaging based on availability of IVUS at the time of angiography. The pathognomonic IVUS findings described below would make the diagnosis definitive. Although resting ABI was performed in all patients, its normalcy did not exclude EIAE. Furthermore, normal or mildly abnormal exercise ABI did not exclude the disease.
Percutaneous endovascular intervention was offered to these patients with full disclosure of limited information about the long-term outcome.
Results
Clinical characteristics
The patients’ clinical characteristics are shown in Table 1. They were uniformly slim individuals with athletic build. Five were competitive runners, three were cyclists, and two were triathletes. Although their symptoms would reproduce with high-intensity exertion, at rest and with moderate exercise they had no symptoms. Their resting ABI was normal. Exercise ABI was done in four of the 10 patients, and in two it had dropped to 0.87 and 0.88 on the affected side. The symptoms are shown in Table 1. All had more than one symptom. Two patients had been mistakenly diagnosed with neuropathy and hip arthritis. Their ‘neuropathic and arthritic’ symptoms resolved after intervention. There were minimal traditional risk factors for atherosclerosis in these patients (Table 1).
Clinical characteristics of patients at baseline (N = 10).

Values are mean ± SD or number (%).
BMI, body mass index.
In only three of the 10 patients did the reading radiologist mention some form of stenosis of the external iliac artery on the CTA report. It was probably related to our specific request to look for EIAE, otherwise it would have been missed.
In nine of the 13 limbs, some degree of arterial stenosis was noted on external ultrasound using the curvilinear probe. There was no flow velocity increase on spectral Doppler.
Percutaneous endovascular approach
The procedures were done in a retrograde ipsilateral approach in 10 limbs and in an antegrade contralateral approach in three patients. A 6-8 French sheath was utilized. All received 325 mg of aspirin before the procedure and 80 U/kg of heparin as bolus. Aspirin was at 81 mg daily for 6 months. The mean stent diameter and length were 8.54 ± 1.33 mm and 78.46 ± 17.25 mm, respectively. They were all self-expanding stents (Table 2). All underwent postdilatation with the same size balloons, or 2 mm larger (in later cases). The decision on stent sizing underwent a dynamic change as our experience increased. Initially we used the distal normal segment as the reference and chose a stent with a 1:1 ratio for size selection. After witnessing the initial restenosis cases, we averaged the distal and proximal normal segment diameters and added 2 mm to this. The rationale to this approach is described in the Discussion.
Stents utilized at the index intervention (n = 13).

Absolute: Abbot Cardiovascular, Plymouth, MN; EverFlex and Protégé: Medtronic, Minneapolis, MN; LifeStar: Bard, Tempe, AZ; SMART Control and SMART Flex: Cordis, Santa Clara, CA; Zilver: Cook Medical, Bloomington, IN.
There were two men and eight women. IVUS had been used in eight limbs. There were no immediate complications postprocedurally.
Recurrence of symptoms
There were three patients with recurrence of symptoms at 9.3 ± 2.1 months postindex intervention. The mean stent diameter was 6.7 mm in these patients (one 6 mm and two 7 mm stents). Two of them underwent a redo procedure with IVUS guidance and received a larger stent with high-pressure dilatation. The other patient declined further treatment as she had given up strenuous exercise for personal and lifestyle reasons. These patients had not undergone IVUS assessment at the index procedure. At a follow up of 3 years, no symptoms recurred in the two retreated patients.
At a mean follow up of 8.4 ± 3.3 years, the remaining seven patients had remained entirely asymptomatic. Four of them continued to perform high-level exertion as before and three had reduced their high-intensity activities due to lifestyle changes (and not symptoms), although they were still quite active by normal standards.
All patients had three uniform characteristics on imaging: With angiography, the caliber of the more caudal segment, such as the common femoral artery, was always larger than the external iliac artery. Although this finding was often subtle, it was always present. It was also seen on careful evaluation of ultrasound images. The other findings were the ubiquitously present ‘perfect circle appearance’ on IVUS and the automatic central positioning of the IVUS catheter within the lumen as described below.
There was no death or limb loss at follow up.
Unique IVUS findings
In all eight limbs which underwent IVUS imaging, a clear and nearly perfect circular appearance of the diseased arterial segment was noted. We called this finding the ‘perfect circle appearance’ (Figure 1A). It was noted most visibly in the circle created by the external elastic lamina. In contrast, segments of external iliac artery without endofibrosis did not show this pattern (Figure 1B). The perfect circle appearance was a uniform finding in all eight limbs in this series that had undergone IVUS imaging (Figures 1 and 2). A review of the literature showed an identical pattern in cases who had published IVUS images (Figure 3A) without necessarily elaborating on the features.11,12 In fact, we could not find a published IVUS image of EIAE which did not have this pattern. This difference of appearance was easily distinguishable. Additionally, in EIAE, the IVUS catheter would often automatically acquire a central or near central position in the lumen (no matter how much the operator tried otherwise) as opposed to the normal segments where it would gravitate and stay to one side. Again, this finding was noted in all our cases as well as in all others who had concentric hypertrophy (Figures 1–3). In two case reports with eccentric hypertrophy of the vessel wall this central positioning was not seen.13,14
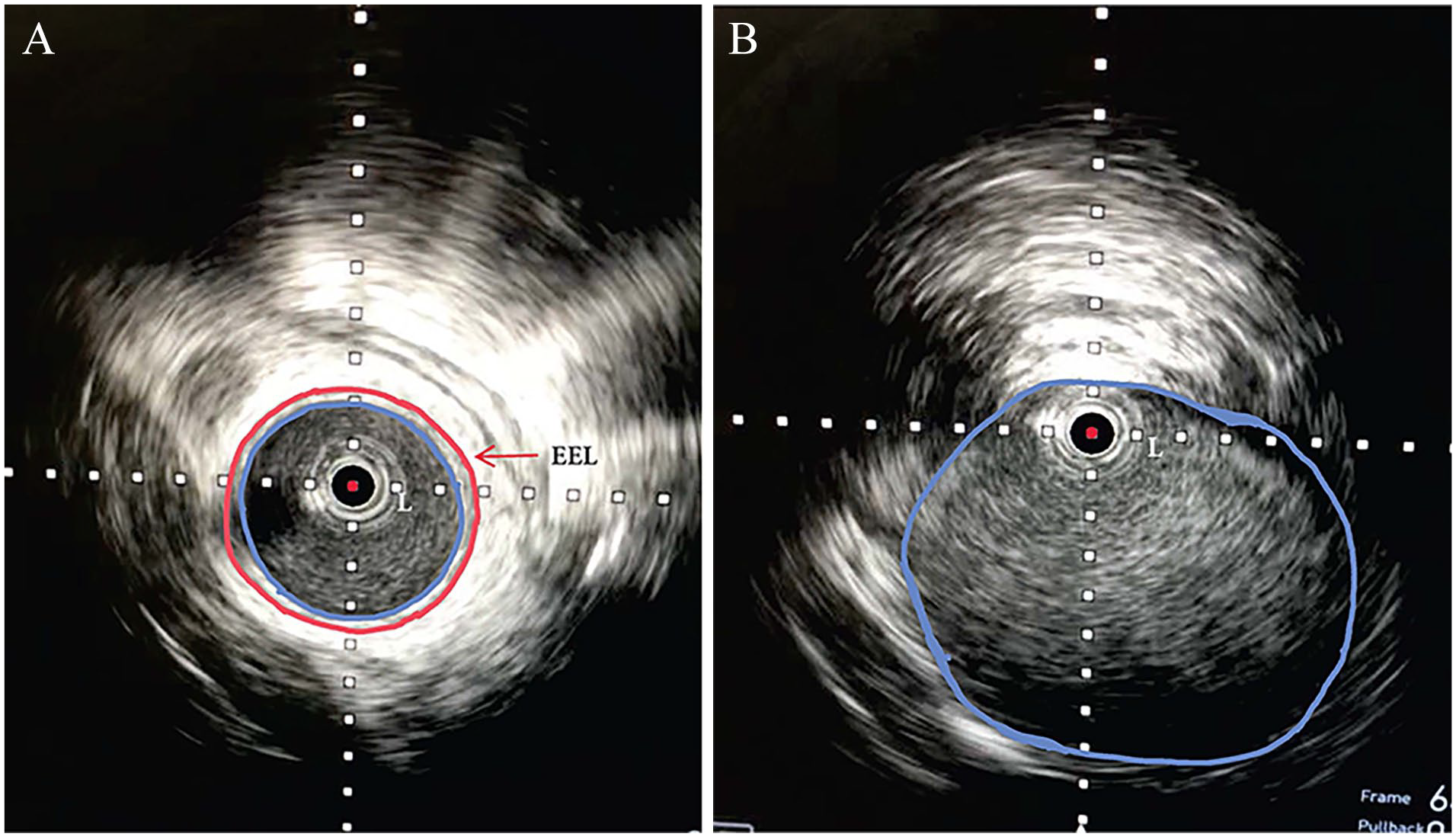
IVUS of a 35-year-old woman and avid runner with external iliac artery endofibrosis. The pathognomonic ‘perfect circle appearance’ is clearly visible.
IVUS showing external iliac artery endofibrosis in a 40-year-old woman who has been a triathlete for over 10 years.
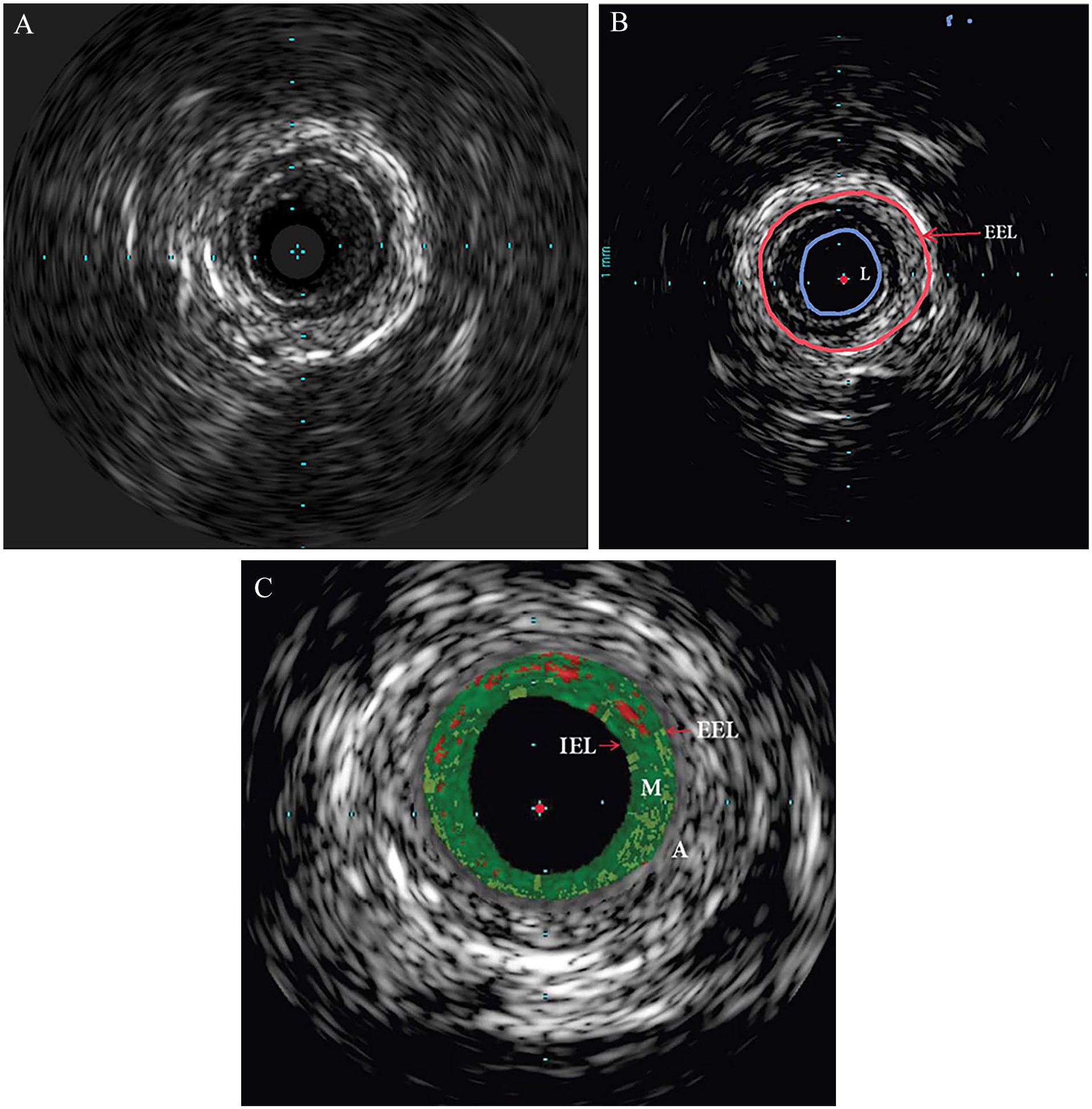
IVUS images of the right
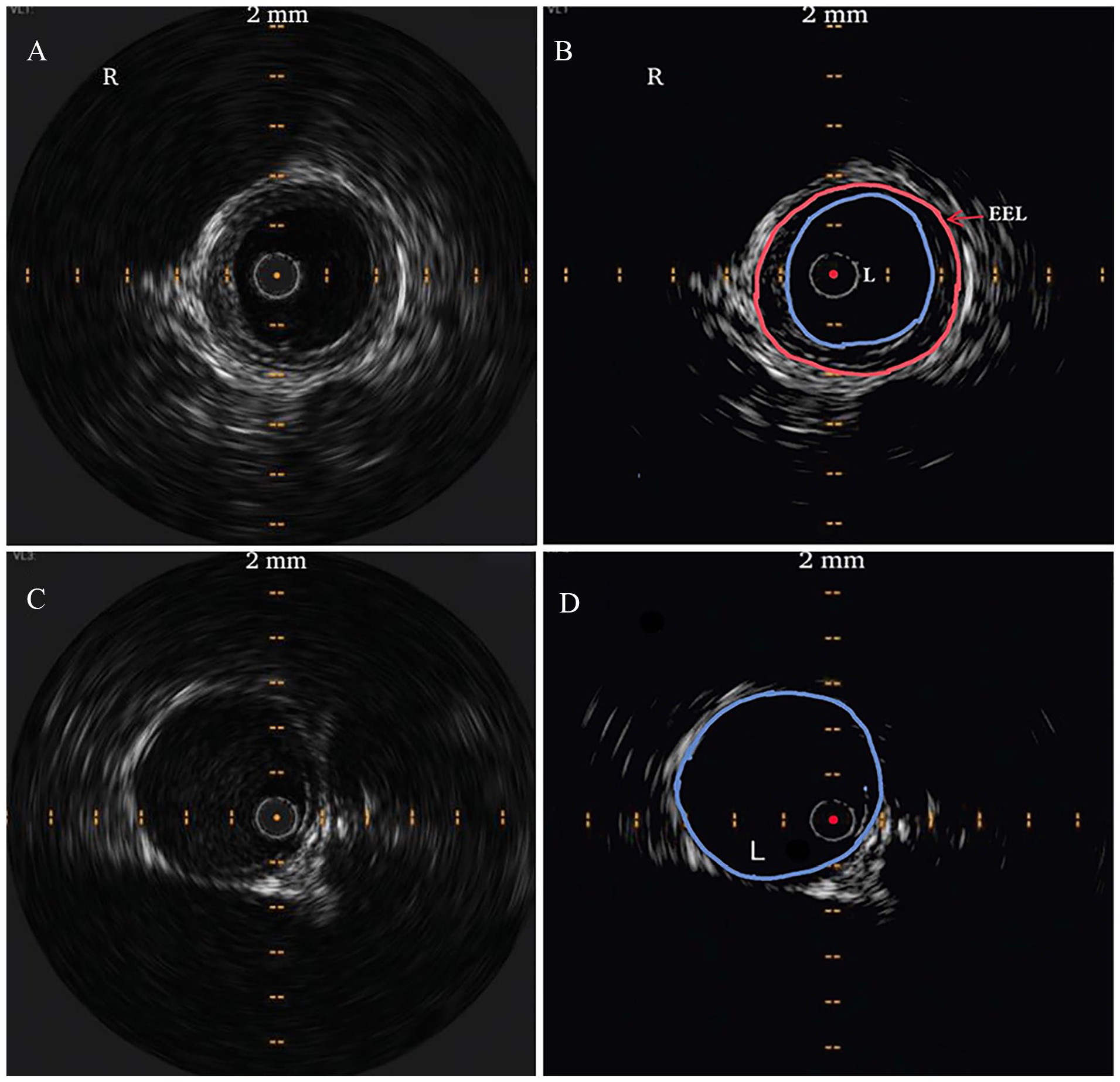
The angiogram of the patient in Figure 1 (IVUS images) is shown in Figure 4. It deserves particular attention. On visual assessment the extent of stenosis is around 60–70%, which may not be considered as flow-limiting in atherosclerotic lesions. Similarly, on IVUS imaging, the extent of stenosis (intimal hyperplasia in Figure 1A) is mild to moderate. Ordinarily this lesion may not be considered for intervention in classic atherosclerotic lesions. However, there is clear ‘negative remodeling’ of the entire vessel wall, which in high-performance athletes would render it highly flow-limiting, as explained in the Discussion.
Angiographic images of a 35-year-old woman and avid runner (same patient as in Figure 1) with left external iliac endofibrosis prior to intervention
Statistical analysis
The reported continuous variables are shown as mean ± SD. Computations were performed using IBM SPSS software, Version 26 (IBM Corp., Armonk, NY, USA).
Discussion
The conventional approach for treatment of EIAE has been surgery.3,15–18 Surgical literature has considered open operative reconstruction as the ‘gold standard’. This designation has been made as no other alternative approaches, such as endovascular intervention, exist to date to compare it to. In a few balloon angioplasty-alone case reports, the short-term outcome was variable, with restenosis occurring early due to the high elastic recoil of the artery.8–10 Stent placement has been more encouraging but the results were based on a handful of case reports with short follow ups.17,18
Therefore, the gold standard designation for surgery has been based on tradition and very limited data on alternative approaches rather than robust evidence and rigorous comparisons.3,15,16 There are no randomized data between percutaneous endovascular intervention and surgery.
Existing surgical approaches
Different surgical techniques have been described. The ‘release only’ suprainguinal surgical approach mobilizes the associated restrictive fibrous tissues and divides the tethering branches. 6 More commonly, end-to-end anastomosis following resection of the stenotic segment is done.3,7 Other methods have included use of prosthetic conduits and application of panel grafts by using the lower-extremity superficial veins.3,15,16 Endofibrosectomy with or without prosthetic or venous patch plasty has also been described.3–5 Surgery is not without its complications. In one large series of surgical intervention in 27 limbs, three patients (11%) had occlusion or severe stenosis of the surgical site at 2.1 years requiring four redo surgical or interventional procedures. 19 Despite surgery, four patients (15%) had no long-term improvement in symptoms. There were three wound infections (11%) and one deep vein thrombosis (4%). Venous patch dilatation was seen in one patient (4%) and five patients (19%) developed paresthesias due to damage to the lateral cutaneous nerve of the thigh. 19 Stated differently, 53% of this surgical cohort had major or minor complications. 19 It is therefore premature, at least in our opinion, to consider surgery as the gold standard. Surgery should be regarded as the traditional treatment modality as there has been no comparison with endovascular approaches.
Characteristic IVUS features
An important finding of this study is the perfect circle appearance of EIAE on IVUS. It was seen in every single case in which IVUS was done. By reviewing the literature and published IVUS images, we have come to believe that this finding in the arterial circulation is both pathognomonic and ‘sine qua non’.11,12,20 In other words, if an external iliac artery segment shows this feature, EIAE is present and if it does not show it, the disease is absent.
The perfect circle appearance can be explained by the histopathologic findings. Wall thickening occurs in all layers usually but not necessarily in a circumferential fashion, although variations in the extent of thickening can occur.15,18 In a series of four patients who underwent surgery, intimal thickening was noted, mostly in a symmetrical fashion. The media and adventitia exhibited both symmetric and asymmetric hypertrophy. 20 Interestingly, no inflammatory process was noted. In the media, smooth muscle cells had proliferated in a circular pattern. 20 This pattern of hypertrophy across all layers remodels the vessel such that the cross-sectional area of the vessel wall on IVUS shows a well circumscribed structure which is highly echo dense, thereby distinctly contrasting itself from the luminal blood’s echoluscent area (Figures 1A, 2, 3A, and 3B). The macroscopic cross-sectional appearance of resected lesions (Figure 5A) has been previously reported. 20 Pan-hypertrophy of the vessel wall with resulting negative remodeling is readily apparent on inspection of these specimens. The rubbery consistency of the endoluminal wall is quite obvious (Figure 5A). A representative, full cross-sectional photomicrograph of the lesions is shown in Figure 5B. All layers are thickened and the central lumen is impinged upon. Variations in the extent of thickening can occur in different parts of the vessel wall. In this specimen (Figure 5B), there is disproportionate and eccentric adventitial thickening. The histopathologic findings are shown in Figure 5C. In this photomicrograph, the adventitia is preferentially infiltrated by smooth muscle cells and extensive fibrosis.
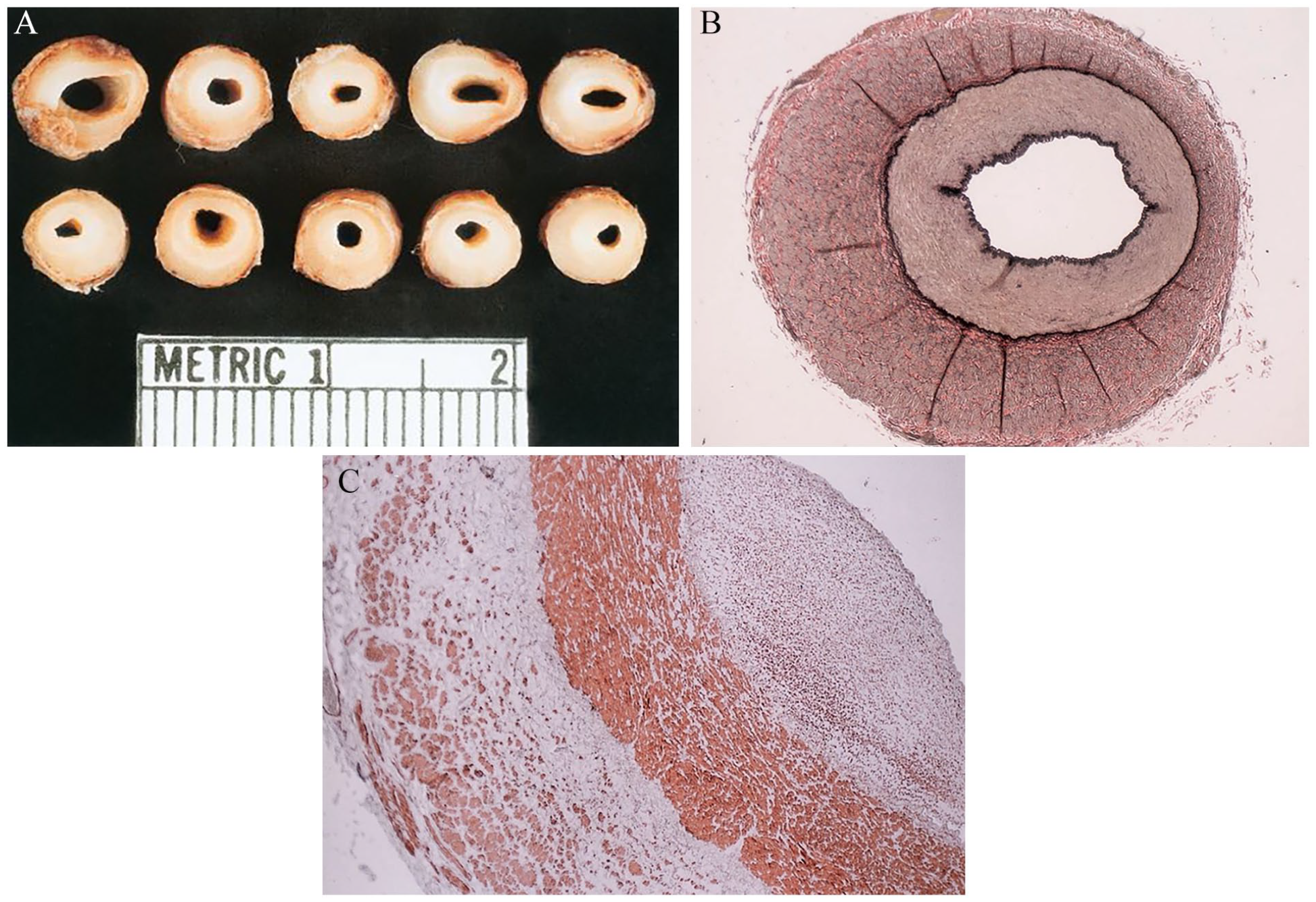
Macroscopic
A consequential feature of concentric wall thickening is that the IVUS catheter frequently stays at or near the center of the lumen whereas in normal segments it usually gravitates to one side of the endoluminal surface (Figures 1–3). No matter how many times the operator purposefully changes the catheter position in the lumen, the released resting place of the IVUS catheter will be in the center. We believe this finding is due to the thickened, noncompliant and ‘rubbery’ wall which consistently deflects the catheter to the center, whereas the normal segment, by being softer, does not bounce the catheter away. In only two reported cases that had eccentric hypertrophy the IVUS catheter was not centrally located.13,14
Principal findings
The recurrence of symptoms was in those with smaller stents, without IVUS guidance and earlier in our experience. High-pressure inflation was not used in these patients. We now recommend at least 2 mm over-sizing of a self-expandable stent followed by high-pressure inflation. Based on the perceived danger of vessel rupture seen in elderly patients with atherosclerosis, we were very conservative in our earlier cases. We learned that this approach is not optimal for EIAE as the pathophysiology and histology are distinctly different than atherosclerotic stenosis. The response of EIAE to treatment appears to be similar to that of iliac venous stenosis. Venous stenosis seldom responds to angioplasty alone and requires stenting, preferably with a strong radial force. The histopathologic appearance of both diseases is very similar. Hyperproliferation of smooth muscle cells within all vessel wall layers and fibrosis is uniformly present. Macroscopic samples of EIAE show circumferential thickening of the vessel wall with a rubbery consistency (Figure 5A). We have noted the same perfect circle appearance in cases of diffuse iliac venous stenosis as well. 21 This has important clinical implications suggesting that the two entities can be similarly treated. Extrapolating our findings from the treatment of venous stenosis, we can postulate that restenosis can be reduced with use of IVUS, a larger stent, and high pressure postdilatation, similar to the techniques used in endovenous intervention. Indeed, we have not had recurrence of symptoms in our more recent cases wherein the above measures were applied. Percutaneous treatment of EIAE is therefore different than that for atherosclerotic arterial stenosis and a larger stent should be considered.
The findings on computerized tomographic and magnetic resonance angiography and external ultrasound may be subtle and not picked up by the reading radiologist. The images should be reviewed by the operator with a high index of suspicion. As a general guideline, if the external iliac artery caliber is smaller than that of the common or proximal superficial femoral artery on any form of imaging in the younger age group, EIAE should be highly suspected. The absence of calcification in the vessel wall is also important. Since symptoms occur at a high level of exertion, resting ABI is of limited value. Even exercise ABI was only mildly positive in 50% of the tested patients. Some case reports have reported on a significant but transient decline in ABI, especially during exertion.4,9,17 These were probably due to the more advanced stage of the disease. ABI should be done at far higher levels of exertion than with a few minutes of walking on a treadmill. In general, we do not believe that a normal resting or exercise ABI would exclude EIAE and the necessity for treatment. Conversely, if ABI is abnormal, it would be an indicator of severity of disease. A limitation of this study was that exercise ABI was not performed in all patients. Consequently, the diagnosis of EIAE and necessity to intervene was not dependent on ABI.
In our series, EIAE was not limited to cyclists but was seen in runners (and triathletes) as well. In general, these patients are slim, of low BMI, and usually have a low normal or shorter height. Women were more affected in this study. This is contrary to some published reports in which men were more affected3,19 and consistent with others showing a higher frequency in women. 20
Negative remodeling and flow limitation
At first glance, the lesions of the representative patients (Figures 1, 2, and 4) may be considered as moderate by our existing frames of reference.
Specifically, examination of the angiogram suggests a 60–70% lesion (Figure 4) and the intimal thickening in Figure 1A is mild to moderate and therefore not necessarily ‘flow-limiting’. However, we believe that the IVUS findings show a highly flow-limiting lesion, which results from the negative remodeling process inherent in EIAE. This concept is elucidated in the following calculation.
The vertical diameter of the luminal area in Figure 1A is about 5.3 mm. In the distal segment (which in normal individuals should be less than the proximal segment) it is about 9 mm (Figure 1B). The corresponding radii will be 2.65 mm and 4.5 mm, respectively. From Poiseuille’s law, it follows that for a given perfusion pressure, any reduction in the radius of the cross-sectional area of a cylinder would lead to a reduction in the flow rate by the 4th power of the radius. In other words, a 50% reduction in the radius would drop the flow by 16-folds. In the instant case, the radius has dropped from approximately 4.5 mm to 2.65 mm (radius reduction to 59% of reference), thereby leading to an approximate 8.3-fold flow reduction. This reduction in overall luminal diameter is due to hyperproliferation of all components of the vessel wall which impinge on the effective luminal cross-sectional area. Thus, the issue is not the extent of intimal thickness but the relative negative remodeling and loss of the effective luminal cross-sectional area as one moves from a normal to an abnormal segment. An 8.3-fold reduction in flow, in a high-performance athlete at peak exercise, is undoubtedly ‘flow-limiting’. In reality, the reduction in flow would be more as one has to compare the common iliac artery diameter (larger, and hence a steeper decline) than that between the external iliac artery and the common femoral artery. This example highlights that our existing frames of reference derived from the treatment of atherosclerotic lesions are not necessarily applicable to EIAE.
Conclusions
We conclude that in patients with symptomatic EIAE, stenting is effective and safe with a good long-term outcome. It can be an alternative to surgery, especially for those reluctant to have an operation. The finding of the perfect circle appearance on IVUS establishes the diagnosis. Centralization of the IVUS catheter would be an additional clue. Beyond 1 year following stenting, symptom recurrence is unusual. The technical considerations of endovascular treatment for EIAE are different than those of atherosclerotic arterial stenosis and are similar to percutaneous endovenous intervention for venous stenosis. A multicenter, randomized clinical trial between surgery and endovascular treatment is required to evaluate the best treatment approach.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
