Abstract
Introduction:
Intracranial aneurysm (IA) is a common vascular enlargement that occurs in the wall of cerebral vessels and frequently leads to fatal subarachnoid hemorrhage. PDZ and LIM domain protein 1 (PDLIM1) is a cytoskeletal protein that functions as a platform for multiple protein complex formation. However, whether PDLIM is involved in the pathogenesis of IA remains poorly understood.
Methods:
Loss-of-function and gain-of-function strategies were employed to determine the in vitro roles of PDLIM1 in vascular endothelial cells (VECs). A rat model of IA was generated to study the role of PDLIM1 in vivo. Gene expression profiling, Western blotting, and dual luciferase reporter assays were performed to uncover the underlying cellular mechanism. Clinical IA samples were used to determine the expression of PDLIM1 and its downstream signaling molecules.
Results:
PDLIM1 expression was reduced in the endothelial cells of IA and was regulated by Yes-associated protein 1 (YAP1). Genetic silencing of PDLIM1 inhibited the viability, migratory ability, and tube formation ability of VECs. Opposite results were obtained by ectopic expression of PDLIM1. Additionally, PDLIM1 overexpression mitigated IA in vivo. Mechanistic investigations revealed that PDLIM1 promoted the transcriptional activity of β-catenin and induced the expression of v-myc myelocytomatosis viral oncogene homolog (MYC) and cyclin D1 (CCND1). In clinical settings, reduced expression of PDLIM1 and β-catenin downstream target genes was observed in human IA samples.
Conclusion:
Our study indicates that YAP1-dependent expression of PDLIM1 can inhibit IA development by modulating the activity of the Wnt/β-catenin signaling pathway and that PDLIM1 deficiency in VECs may represent a potential marker of aggressive disease.
Introduction
Intracranial aneurysm (IA) is a local abnormal expansion of the wall of the cerebral artery. IA is a severe disease with a prevalence of 3–5% in the general public.1–3 IA is a major cause of subarachnoid hemorrhage (SAH) and IA rupture leads to significant consequences, such as death and disability. 4 However, there are no effective treatment protocols for IA patients and only surgical operations such as microsurgical clipping and/or endovascular coiling can be used to treat patients with IA. 5 Considering the high prevalence of IA and poor clinical outcome of SAH, there is an urgent need to develop more effective methods or therapies in the management of IA.
It is generally believed that IA is histopathologically characterized by degenerative changes and inflammatory infiltrates in the arterial walls.6,7 Specifically, the mechanical signal of abnormal blood flow is transmitted to endothelial cells, inducing damage to endothelial cells, causing inflammatory cells to infiltrate the arterial walls and release inflammatory factors.8,9 Disruption of endothelial barrier function is essential for the pathogenesis of IA. Under inflammatory stimulation, smooth muscle cells gradually dedifferentiate and migrate to the endothelium, releasing a variety of active substances, thus activating proteases, destroying the elastic layer, degrading the extracellular matrix, and inducing cell apoptosis. Eventually, the arterial walls become thinner, the elasticity decreases, a bulge is induced, and IA is formed. Interestingly, the development of IA is not a complete positive feedback loop. Cells within the arterial walls can also secrete negative regulators to inhibit the progression of IA. 10 Therefore, deciphering the negative regulators involved in the pathogenesis of IA may represent a novel strategy for developing therapeutic targets in these patients.
PDZ and LIM domain protein 1 (PDLIM1), also known as CLP36, Elfin or CLIM1, is an important cytoskeletal protein that associates with actin filaments and stress fibers. 11 PDLIM1 is a member of the PDZ and LIM protein family that contains a C-terminal PDZ domain and one or more N-terminal LIM domains. PDLIM1 plays diverse roles in physiological processes such as cytoskeleton regulation and synapse formation.12–15 For example, PDLIM1 is a negative cytoskeleton organization regulator involved in spermatid differentiation. 16 Emerging evidence also demonstrates that PDLIM1 is profoundly implicated in many malignant phenotypes of cancer cells.11,17–19 For instance, PDLIM1 knockdown induces epithelial-to-mesenchymal transition (EMT) in hepatocellular carcinoma cells, elevates their invasive capacity, and promotes metastasis by activating Hippo signaling. 18 Yes-associated protein 1 (YAP1) is a major transcriptional effector of the evolutionarily and functionally conserved Hippo pathway. 20 To identify noninvasive biomarkers for the diagnosis and classification of ruptured and unruptured IAs, we recently performed proteomic analysis and found that PDLIM1 is differentially expressed between IAs and matched superficial temporal artery tissues. 21 However, the functional significance, molecular mechanism, and clinical relevance of PDLIM1 in IAs remain to be explored.
Methods
Clinical specimens
The human IA specimens (n = 10; Table 1) and control arterial walls (n = 10) were acquired from the Department of Neurosurgery, Huashan Hospital, Fudan University. Patients who had a history of hypertension, diabetes, or tumors were excluded. IA tissue samples were collected after removal surgery and dissected specimens were fixed in formalin solution and embedded in paraffin. Then, 5-μm thick slices were prepared for hematoxylin–eosin (HE) staining and immunohistochemical analysis. This study was approved by the institutional review board of Huashan Hospital, Fudan University and performed in accordance with the Declaration of Helsinki. All patients enrolled in this study gave written informed consent.
Characteristics of included patients (N = 10).
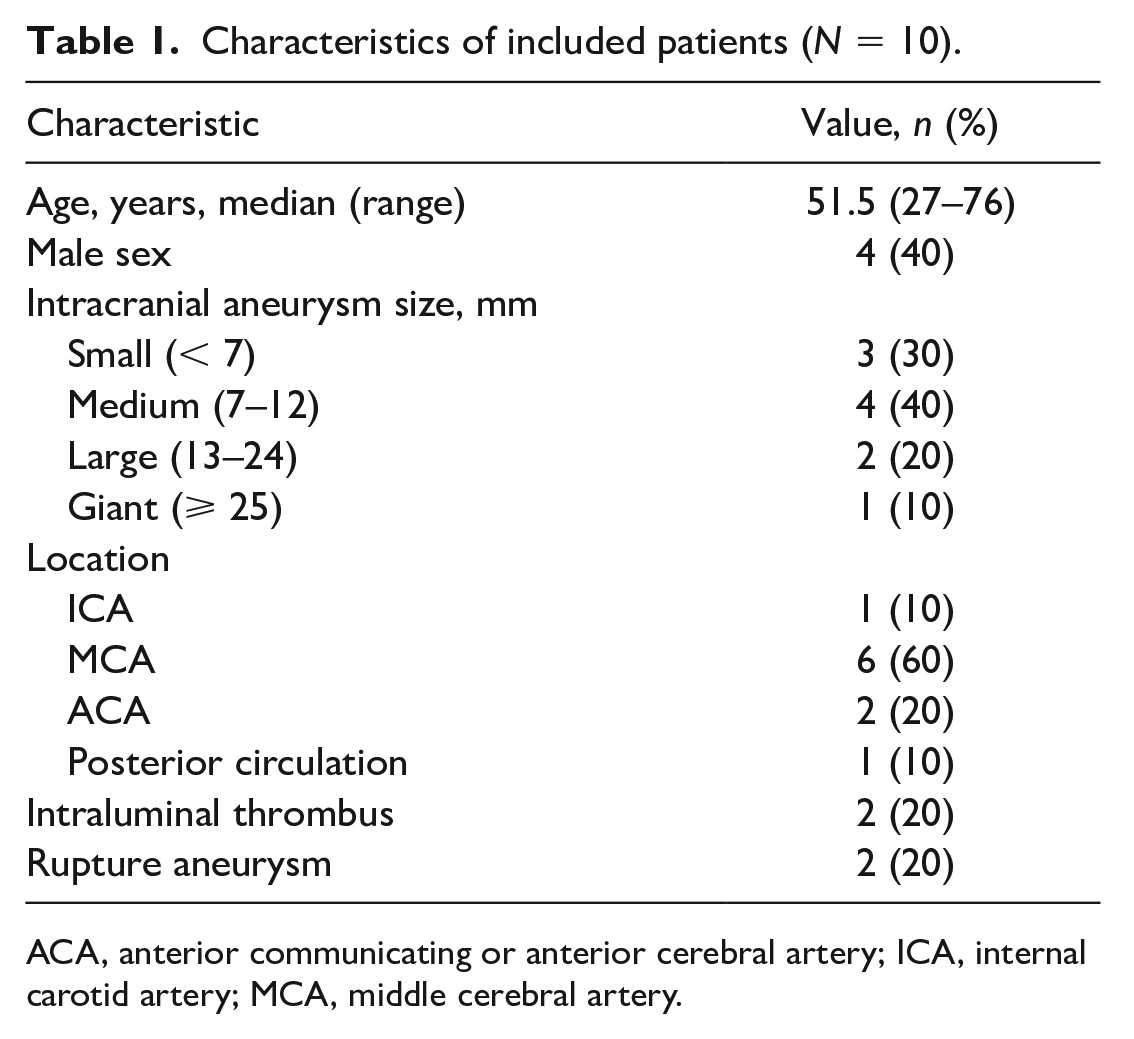
ACA, anterior communicating or anterior cerebral artery; ICA, internal carotid artery; MCA, middle cerebral artery.
Cell culture and reagents
Human umbilical vein endothelial cells (HUVECs) were cultured using Dulbecco’s Modified Eagle’s Medium (DMEM) (Life Technologies, Carlsbad, CA, USA) containing 10% fetal bovine serum (FBS) (Gibco, NY, USA) and 100 U mL−1 penicillin-streptomycin (Invitrogen, Carlsbad, CA, USA). Cells were maintained at 37°C in a humidified incubator under 5% CO2 conditions. The β-catenin inhibitor ICG-001 (S2662) was purchased from Selleck Chemicals (Shanghai, China).
RNA isolation and quantitative real-time polymerase chain reaction
Total RNA was isolated from HUVECs or animal tissues using an RNeasy Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. The purity and concentrations of RNA were determined by using Nanodrop absorbance measurements at 260/280 nm. The process of reverse transcription of RNA into complementary DNA was conducted in accordance with the instructions of the reverse transcription kit (Takara, Tokyo, Japan). Quantitative real-time polymerase chain reaction (qPCR) was conducted using SYBR Green PCR Master Mix (Takara) with Applied Biosystems 7500 System (Life Technologies). All results were normalized to the expression of the housekeeping gene, glyceraldehyde-3-phosphate dehydrogenase (GAPDH). The relative expression levels of target genes were calculated by the 2−△△Ct method.
Cell transfection and grouping
HUVECs in the logarithmic phase were assigned into five groups: overexpression-negative control (OE-NC) group (transfected with OE-NC plasmid), PDLIM1-OE group (transfected with PDLIM1-OE plasmid), NC-siRNA group (transfected with an irrelevant siRNA), PDLIM1-siRNA1 group (transfected with siRNA against PDLIM1), and PDLIM1-siRNA2 group. The above plasmids were synthesized by GenePharma (Shanghai, China). The sequences targeted by PDLIM1 siRNAs were (siRNA1) 5′-GCUCAGAAGUUGCCUAUGUGUTT-3′ and (siRNA2) 5′-GCCUUGUCAUCGACAAAGAAUTT-3′. Cell transfection was performed using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. Two days later, the cells were harvested and used for further analysis.
Luciferase reporter assay
HUVECs with PDLIM1 overexpression or knockdown were cotransfected with the MCAT-Luc YAP1/TEAD responsive reporter plasmid using X-tremeGENE 9 according to the manufacturer’s protocol (Roche, NY, USA). Three days later, the cells were lysed in fresh medium containing 1:10 (v/v) Steady-Glo luciferase assay reagent (Promega, Madison, WI, USA); the luciferase activity was measured according to the manufacturer’s protocol. Each experiment was performed in triplicate.
Online database analysis
IA-related microarray GSE75436 was downloaded from the Gene Expression Omnibus (GEO) dataset to determine the differentially expressed genes (DEGs) involved in IA. The expression microarray data are available at https://www.ncbi.nlm.nih.gov/gds/. For differential analysis, the ‘limma’ package2 in R software was used. Genes with a fold change larger than 2 and a p-value less than 0.05 were considered DEGs.
Western blotting
HUVECs were lysed in lysis buffer (20 mM Tris/HCl at pH 7.8, 150 mmol/L NaCl, 20% glycerol, 50 mmol/L NaF, and proteinase inhibitor), and proteins were clarified by centrifugation (14,000 × g for 30 min at 4°C). The protein concentration was determined using the BCA kit (Pierce Chemical Corp., Dallas, TX, USA). Then, lysates were separated using an SDS-PAGE gel system transferred to a nitrocellulose membrane (Millipore, MA, USA) and subjected to immunoblotting. The following antibodies were used in this study: YAP1 antibody (13584-1-AP, Proteintech, Shanghai, China, diluted at 1:1000), PDLIM1 antibody (11674-1-AP, Proteintech, Shanghai, China, diluted at 1:1000), and GAPDH antibody (ab8245, Abcam, Cambridge, MA, USA, diluted at 1:2000).
Cell viability assay
For detection of cell viability, HUVECs (5 × 103) with the indicated genetic manipulation were seeded into wells of 96-well plates, per well, and cultured overnight. Cell viability was evaluated by Cell Counting Kit-8 (CCK-8; Dojindo, Kumamoto, Japan) according to the manufacturer’s instructions. At 0, 24, 48, and 72 h, 10% CCK-8 solution (v/v) was added and allowed to incubate for 1 h. The absorbance was measured at 450 nm with a multifunctional microplate reader (Tecan, Männedorf, Switzerland). The experiments were repeated three times independently.
Transwell assay
HUVECs (2 × 104) were starved in serum-free media overnight and transferred into the upper chambers of 24-well transwell plates with 8-μm pore diameter (Corning Inc., Corning, NY, USA) in 200 μL of serum-free medium. In the lower chamber, 5% FBS was added as the chemoattractant. After incubation for 10 h, nonmigrating HUVECs in the top chamber were removed with a cotton swab. Migrated cells on the lower surface of the insert membrane were fixed and stained with 0.2% crystal violet (Sangon, Shanghai, China). The number of migrated cells was counted under a microscope and photographed using an Olympus BX51 microscope (Olympus, Tokyo, Japan).
Wound healing assay
HUVECs were placed into 24-well plates. When HUVEC monolayer cultures reached approximately 95–100% confluency, a 200-μL pipette tip was used to scratch the cell monolayer across the center of each well. The plates were washed with endothelium growth medium to remove detached cells. After 10 h, the wound was imaged using an inverted microscope. Wound closure was analyzed using ImageJ, and the results are expressed as wound width.
Tube formation assay
HUVECs (3 × 104) were resuspended in serum-free media and seeded on top of Matrigel (BD Biosciences, NY, USA) in a 24-well plate. Following incubation for 18 h at 37°C in 5% CO2, tubules were imaged in five randomly selected microscopic fields, using a phase-contrast microscope at 10× magnification (Axiovert 25 CFL; Carl Zeiss Microscopy, Jena, Germany).
TOP/FOP Flash Reporter Assay
HUVECs (5 × 104) were seeded into a 24-well plate and TOP/FOP-Flash (Genechem, Shanghai, China) plasmids were cotransfected into HUVECs along with the PDLIM1 silencing or overexpression vector using Lipofectamine 2000 (Invitrogen). The TOP/FOP-Flash values were normalized to the Renilla reniformis (Promega) reading. After transfection for 48 h, the Luciferase Reporter Assay Kit (Promega) was used to detect the activities of both firefly and Renilla luciferase reporters according to the manufacturer’s instructions. Experiments were performed in triplicate.
HE staining
The paraffin sections of human and rat IA tissues were first baked at 60°C for 30 min. Then, the tissue sections were fixed in xylene, dehydrated with gradient absolute alcohol, and cleaned with phosphate-buffered saline (PBS). The tissue slices were dyed with hematoxylin, treated in ammonia for a few seconds, stained with eosin for 2 min, dehydrated, and cleared. Then, the tissue slices were dripped with neutral gum and sealed with cover glass. Finally, a microscope (Nikon, Tokyo, Japan) was used for evaluation.
Establishment of rat models of IA
The IA model was established in male Sprague–Dawley rats (Shanghai JSJ Laboratory Animal Co., Ltd, Shanghai, China). Animals were housed in sterile cages (a maximum of three rats per cage) in an individually ventilated rack system under specific pathogen-free conditions, maintained with standard housing and husbandry conditions and had free access to food and water. In brief, the rats were anesthetized using an intraperitoneal injection of pentobarbital (50 mg/kg). The left carotid artery and the left renal artery were ligated to increase hemodynamic stress. After awakening, the rats were fed 1% saline. Two weeks later, 0.12% 3-aminoacrylonitrile was added to the diet or drinking water to induce systemic hypertension by salt overdose. Moreover, the stereotactic technique was used to inject protease (concentration 10 U/mL) after anesthesia. After the establishment of the IA model, 20 rats were randomly divided into a short hairpin RNA (sh)-negative control group (n = 10, model rats were treated with a stereotactic injection of 100 μL NC-OE packaged virus once a day) and a Pdlim1-OE group (n = 10, model rats were treated with a stereotactic injection of 100 μL Pdlim1-OE packaged virus once a day). The above packaged virus was obtained from Shanghai Genechem Co. Ltd (Shanghai, China). The status of IAs was visualized under a microscope and scored according to the following rules: grade 0, normal artery; grade 1, artery dilation or tortuosity; grade 2, unruptured aneurysm; and grade 3, ruptured aneurysm. Unruptured aneurysms were defined as aneurysms 1.5 times larger in diameter than the parent artery. Scoring was conducted by two researchers in a blinded manner. This in vitro animal study was approved by the Institutional Review Board of Huashan Hospital, Fudan University. All animal experiments were in line with the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health.
Immunohistochemical (IHC) staining
The immunohistochemical (IHC) experiment was routinely performed. The paraffin sections of human and rat IA tissues were first dewaxed and hydrated, followed by incubation with 3% hydrogen peroxide for 15 min to eliminate the activity of endogenous peroxidase. Next, the tissue sections were dripped with blocking solution and incubated with normal goat serum working solution for 45 min at room temperature. After blocking, tissue sections were probed with primary antibodies against YAP1 (13584-1-AP, Proteintech, diluted at 1:200), PDLIM1 (11674-1-AP, Proteintech, diluted at 1:200), β-catenin (ab32572, Abcam, diluted at 1:500), c-Myc (ab32072, Abcam, diluted at 1:100), and CCND1 (26939-1-AP, Proteintech, diluted at 1:200), and incubated at 4°C overnight. The sections were reprobed with horseradish peroxidase-conjugated secondary antibody for 60 min, incubated with newly prepared diaminobenzidine (DAB) solution, counterstained, and blocked. The image was acquired by a microscope (Nikon, Tokyo, Japan).
Statistical analysis
Data are presented as the mean ± SD. Statistical analysis was performed with GraphPad Prism 5 (San Diego, CA, USA) software. Comparisons between two groups were conducted by Student’s t-test, whereas comparisons among multiple groups were assessed by one-way analysis of variance (ANOVA), and pairwise comparisons were implemented by Tukey’s multiple comparisons test. Pearson correlation analysis was employed to analyze the correlation between YAP1 and PDLIM1 expression in patients with IA. Statistical significance was considered to be p < 0.05.
Results
PDLIM1 enhances endothelial cell functions
To characterize the potential roles of PDLIM1 in the development of IA, we employed gain-of-function and loss-of-function studies in HUVECs. The overexpression and knockdown efficiency of PDLIM1 in HUVECs were verified by Western blotting analysis (Figure 1A). After performing a CCK-8 assay, we found that the cell viability of HUVECs on Day 3 was significantly increased by PDLIM1 overexpression, but attenuated by PDLIM1 knockdown (Figure 1B). Next, transwell and wound healing assays were performed to investigate the effects of PDLIM1 on the migratory ability of HUVECs. In the transwell assay, PDLIM1 overexpression led to an approximately 1.6-fold increase in the migratory ability of HUVEC; in contrast, PDLIM1 knockdown resulted in remarkable downregulation in HUVEC migration (Figure 1C). Consistently, the wound healing scratch assay supported the effects of PDLIM1 in promoting the migratory ability of HUVECs (Figure 1D). Specifically, transwell/wound healing assays were performed 10 h after cell incubation. At this point, PDLIM1 knockdown or overexpression did not affect cell proliferation (Figure 1B). Moreover, in vitro angiogenesis was studied using HUVECs for the tube formation assay. As a result, PDLIM1 overexpression promoted tube formation, yet PDLIM1 knockdown decreased the tube formation of HUVECs (Figure 1E). Collectively, these findings indicate that PDLIM1 may induce endothelial cell alterations to influence IA progression.
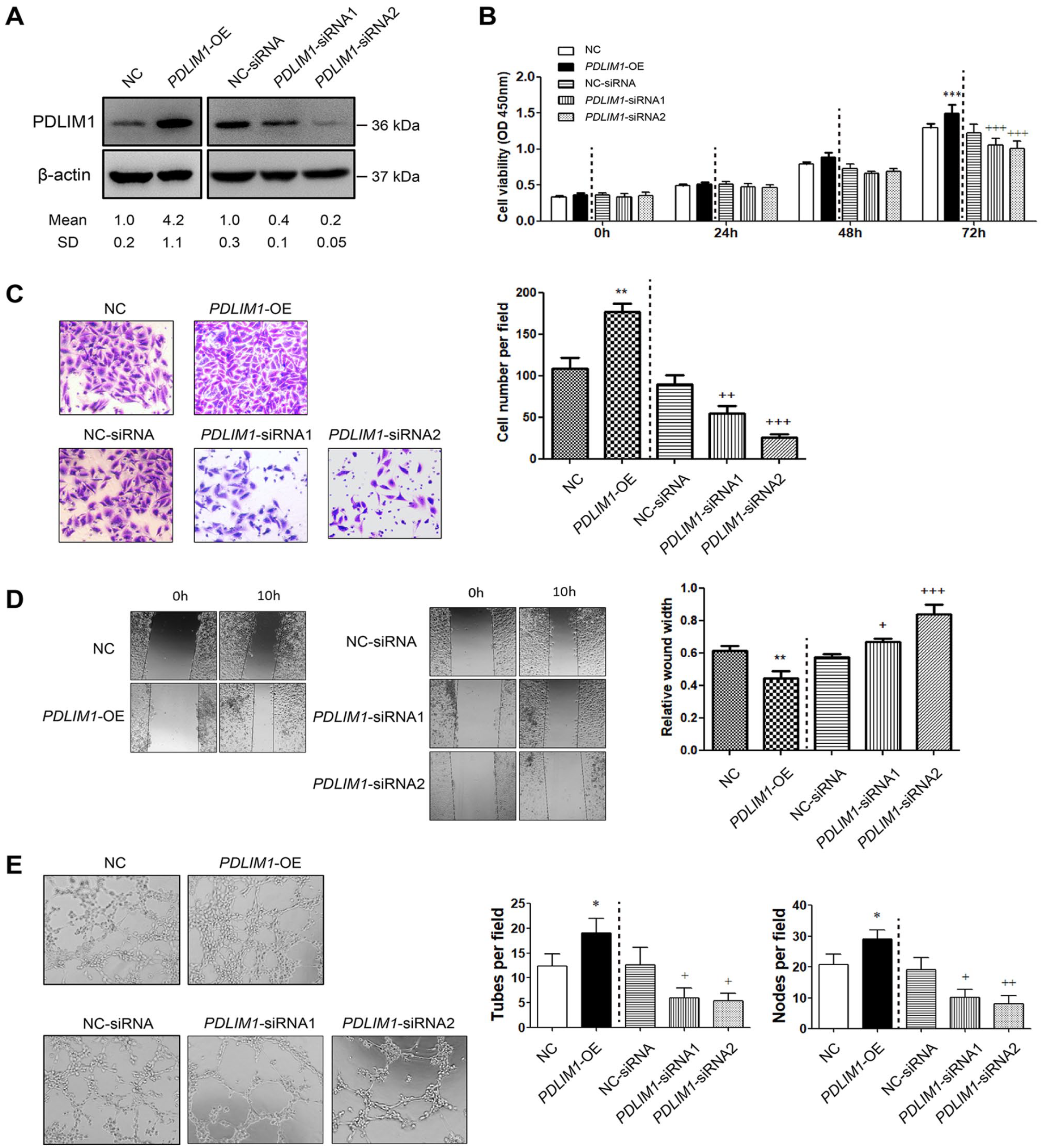
PDLIM1 enhances endothelial cell functions.
PDLIM1 activates β-catenin signaling in endothelial cells
To interrogate the cellular mechanism by which PDLIM1 affects endothelial cell function, we first compared the gene expression profiles between the PDLIM1-low group and PDLIM1-high group by using microarray data from 15 IA tissues (GSE75436). Gene set enrichment analysis (GSEA) showed that PDLIM1 expression was closely associated with the activation of Wnt/β-catenin signaling (Figure 2A). To test this hypothesis, we analyzed Wnt/β-catenin activity using TOP/FOP flash assay. Compared with NC cells, TOP/FOP activity was increased in PDLIM1-OE HUVECs; similarly, HUVECs transfected with siRNAs against PDLIM1 had reduced TOP/FOP activity compared with that of control cells (Figure 2B). V-myc myelocytomatosis viral oncogene homolog (MYC) and cyclin D1 (CCND1) are known downstream targets of the Wnt/β-catenin pathway. Therefore, we detected their expression upon PDLIM1 overexpression or knockdown. As expected, MYC and CCND1 expression was increased in PDLIM1-OE HUVECs, but decreased in PDLIM1-siRNA HUVECs (Figure 2C). Moreover, to confirm whether PDLIM1-induced activation of Wnt/β-catenin signaling is responsible for endothelial cell function, we determined the effects of ICG-001, a specific inhibitor that blocks the binding of CREB-binding protein (CBP) to β-catenin, in NC or PDLIM1-OE HUVECs. Intriguingly, the PDLIM1-dependent increase in cell proliferation, cell migration, and tube formation of HUVECs was largely compromised by ICG-001 (Figure 2D–F), suggesting that PDLIM1 can, at least to some extent, regulate β-catenin signaling to promote endothelial cell functions.
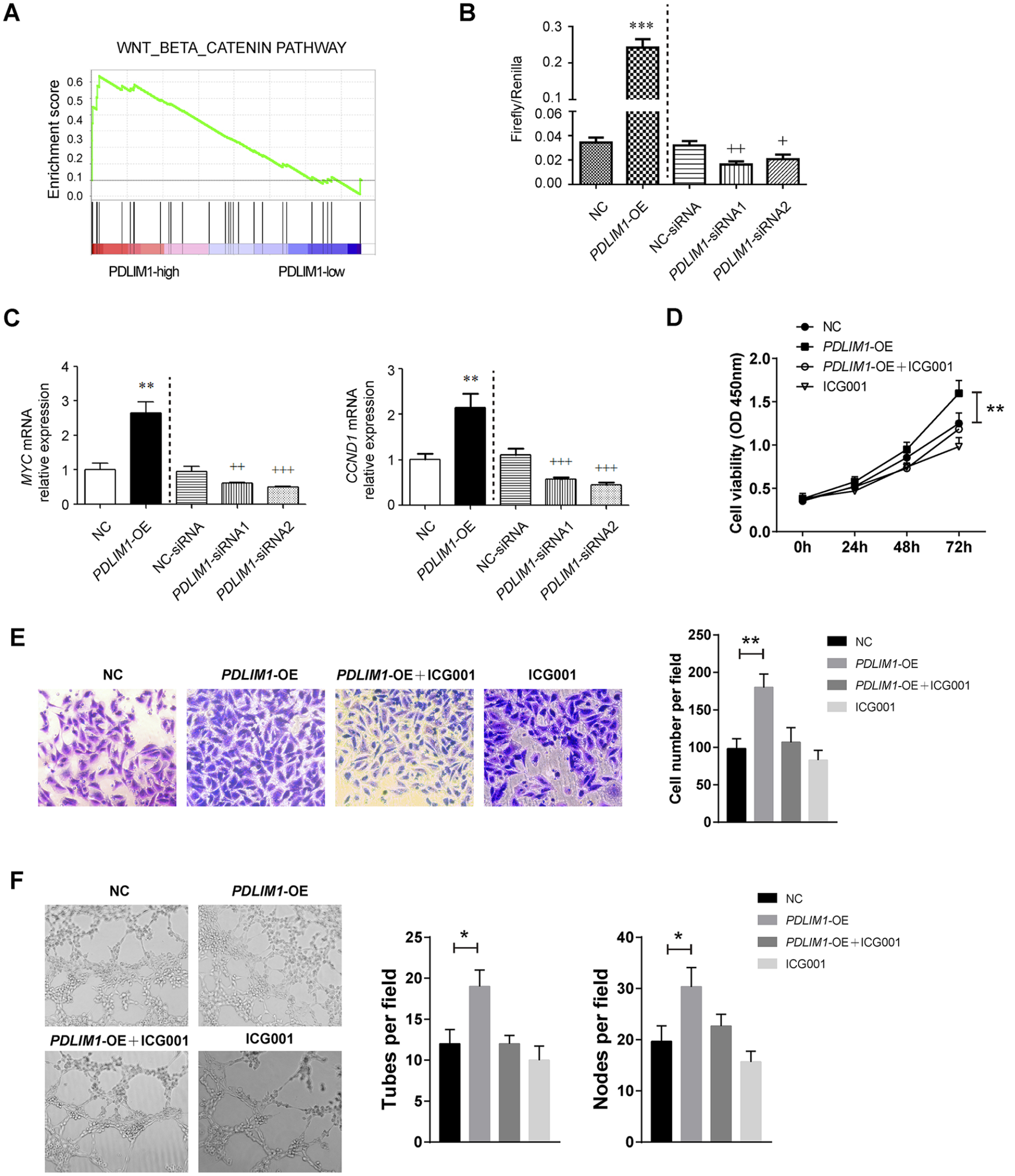
PDLIM1 activates β-catenin signaling in endothelial cells.
PDLIM1 overexpression mitigates IA in a rat model
The in vitro findings hint that genetic manipulation of PDLIM1 could be a therapeutic approach for IA treatment. To test such a hypothesis, we generated a rat IA model and evaluated the effects of PDLIM1 via injection of lentivirus carrying Pdlim1 after surgical induction of IA. First, HE staining showed that the blood vessels in model rats were strikingly deformed and that the vascular lesions were improved in the group with viruses packaged with Pdlim1-OE (Figure 3A). Compared with the IA group, the composition of pathological grades in the IA+Pdlim1-OE group was reduced, but the difference was not significant (Figure 3B). Consistent with histological changes, PDLIM1 was reduced in the endothelial cells collected from IA rats and could be restored by treatment with viruses packaged with Pdlim1-OE (Figure 3C). Compared with those in the sham group (with injection of sh-NC viruses), real-time qPCR analysis also showed that the expression levels of β-catenin, MYC, and CCND1 were downregulated in the IA group; in comparison to those in the IA group, the expression levels of β-catenin and its downstream targets were elevated in the IA + Pdlim1-OE group (Figure 3D). Collectively, PDLIM1 levels in endothelial cells may be essential to hijack the progression of IA in vivo.
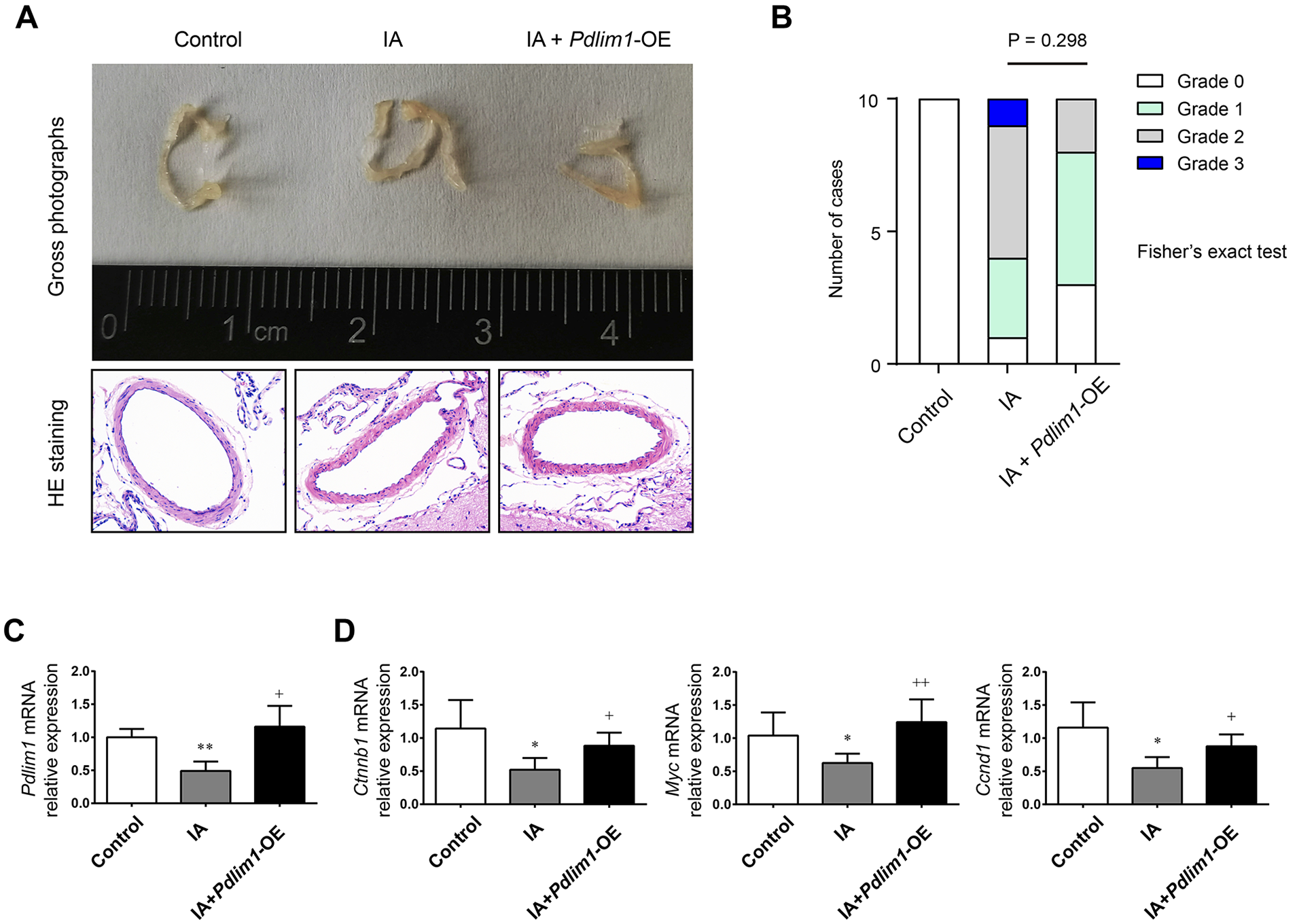
PDLIM1 overexpression mitigates IA in a rat model.
YAP1 induces PDLIM1 expression in endothelial cells
To understand the reason for the dysregulated expression pattern in IA, we further analyzed the GSE75436 dataset, which contains 15 IA tissues and 15 superficial temporal artery tissues. Differential analysis revealed 432 upregulated genes and 458 downregulated genes related to IA (Figure 4A and Supplemental Table S1 [available online]). YAP1 has been reported as a baroreceptor of VECs, which can sense hemodynamic changes and regulate the physiological functions of VECs. 22 When YAP1 in endothelial cells is knocked out, the polarization ability of VECs is reduced, filopodia increase, and differentiation into IA-like endothelial morphology occurs. 23 YAP1 can also regulate the function of VE-cadherin and maintain the adhesion, migration, and other functions of VECs. 24 Interestingly, YAP1 was downregulated in the IA tissues (Figure 4B). To determine how YAP1 regulates endothelial cell function during IA, we focused on the investigation of its downstream target genes. We found that YAP1 expression was closely associated with PDLIM1 expression in IA tissues, suggesting a YAP1-PDLIM1 connection (Figure 4C). To test whether PDLIM1 expression is regulated by YAP1 in IA, we genetically overexpressed and silenced YAP1 expression in HUVECs. Using the muscle-CAT luciferase (MCAT-Luc) YAP1/TEAD responsive reporter, we noticed that YAP1/TEAD activity was increased by YAP1 overexpression, but decreased by YAP1 knockdown (Figure 4D). Using Western blotting analysis, we observed that YAP1 overexpression increased PDLIM1 protein levels in HUVECs; in contrast, YAP1 knockdown reduced PDLIM1 expression (Figure 4E).
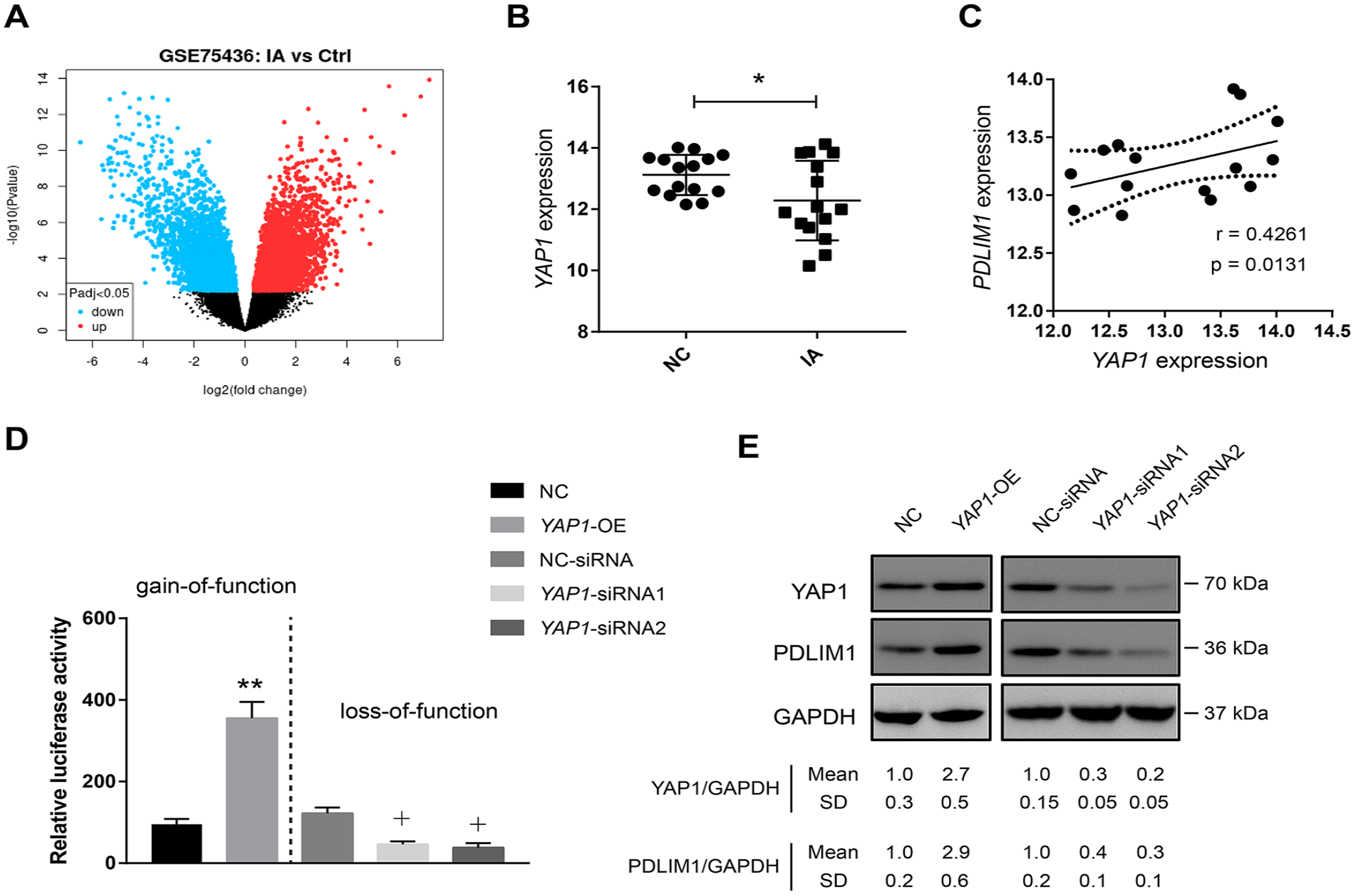
YAP1 is decreased in IA tissues and regulates PDLIM1 expression in the endothelial cells.
Clinical relevance of the YAP1-PDLIM1-β-catenin axis in IA
Finally, we collected human IA tissues (n = 10) and control arterial walls (n = 10) to examine whether PDLIM1 is expressed in endothelial cells of the human intracranial artery and whether PDLIM1 expression correlates with downstream signaling molecules in human IA lesions. Indeed, IHC analysis revealed that PDLIM was significantly reduced in IA tissues compared with that in IA tissues (Figure 5A). Likewise, YAP1 was decreased in IA (Figure 5B). Consistent with the loss of PDLIM1 expression in IA tissues, the expression of β-catenin, MYC, and CCND1 in IA walls was also reduced in these IA lesions (Figure 5C–F). Taken together, the YAP1-PDLIM1-β-catenin axis is also dysregulated in human IA tissues. Targeting this axis may be exploited as a novel therapeutic strategy for IA treatment (Figure 6).
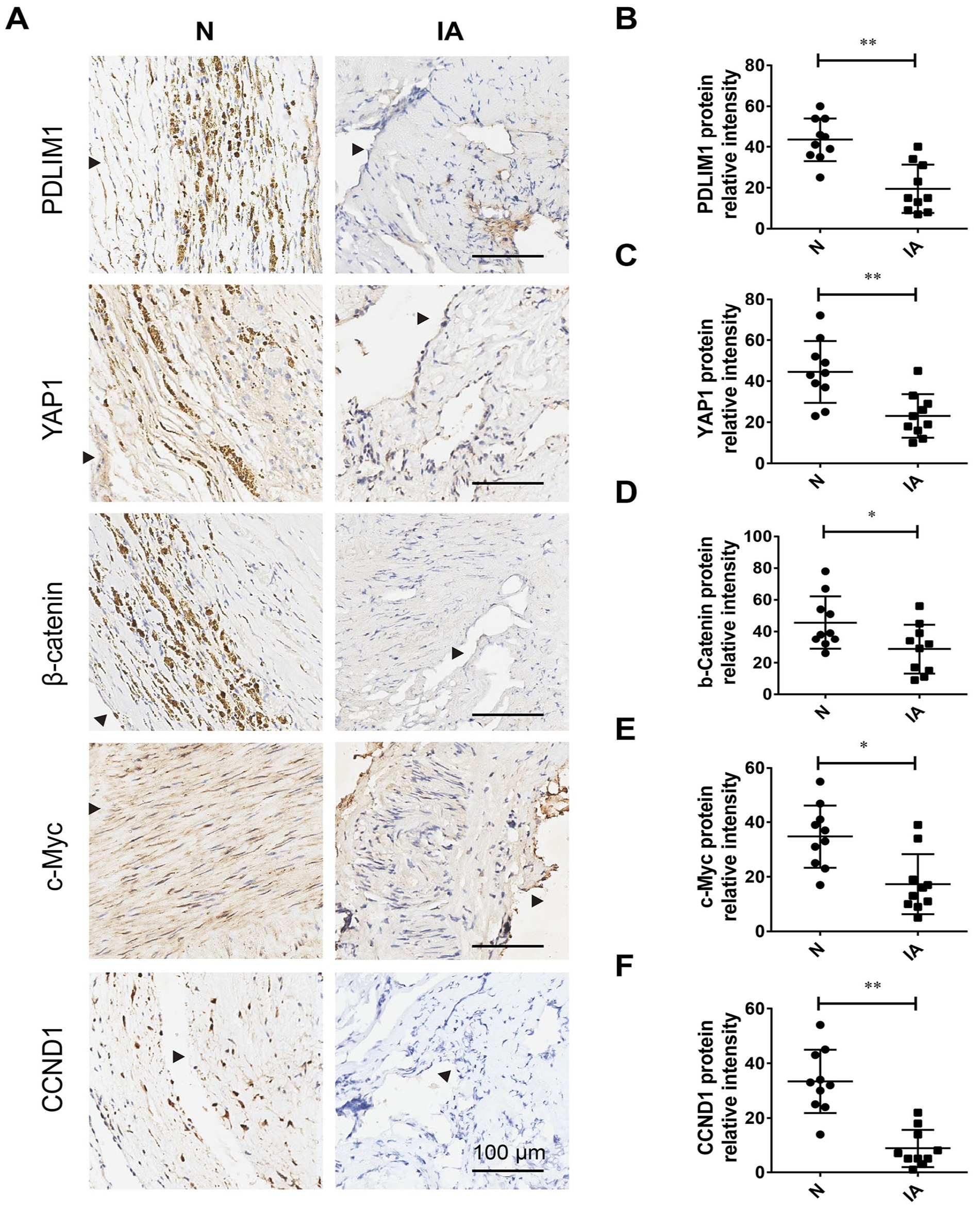
Clinical relevance of PDLIM1-β-catenin axis in IA. Immunohistochemical analysis of the PDLIM1-β-catenin axis
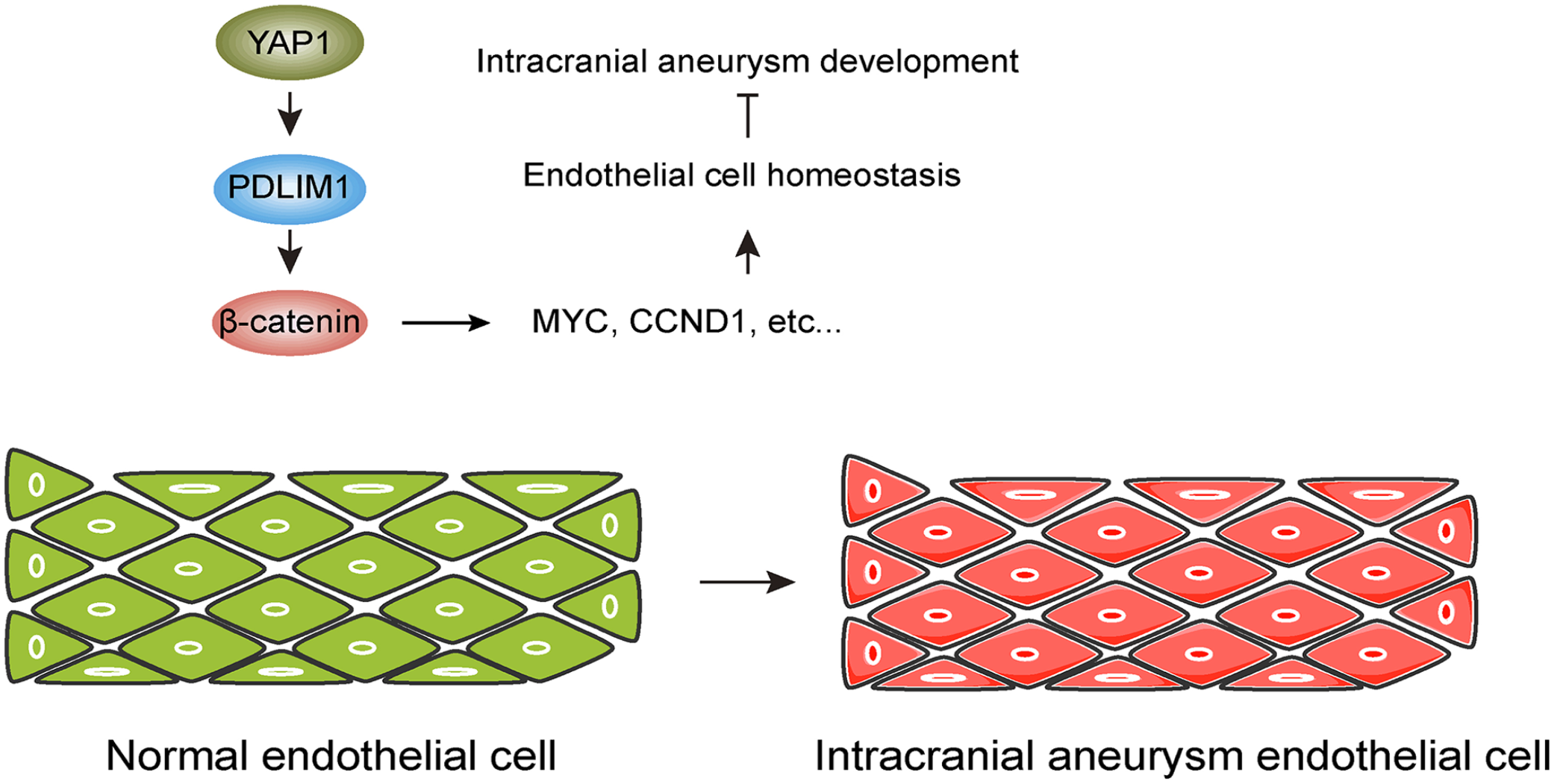
Mechanism model of PDLIM1 in the pathogenesis of intracranial aneurysm (IA). PDLIM1 expression is reduced in endothelial cells of IA tissues. PDLIM1 dysregulation results in inactivation of Wnt/β-catenin signaling and decreased expression of MYC and CCND1, which are essential for the physiological function of endothelial cells. Ultimately, loss of PDLIM1 contributes to the development of IA.
Discussion
The highly progressive nature of cerebral aneurysms renders them incurable by medicines and surgery, thus warranting attention to the molecular mechanisms underlying disease progression. In this study, we uncovered, for the first time, a previous unprecedented role of PDLIM1 in the pathogenesis of IA. Using gain-of-function and loss-of-function approaches, we defined a specific regulatory role of PDLIM1 in VECs. We demonstrated that PDLIM1 is needed for the cell proliferation, cell migration, and tube formation ability of VECs via activation of the Wnt/β-catenin pathway. Moreover, we showed that PDLIM1 is a negative regulator of IA development in vivo.
Previously, accumulated studies have documented many molecules involved in IA, such as long noncoding RNA myocardial infarction associated transcript (MIAT), 25 stromal cell-derived factor-1alpha (SDF-1α), 26 histone deacetylase 9 (HADC9), 27 and coenzyme Q10. 28 Using an IA-related microarray, we provided new insights into YAP1-dependent functions in IA. YAP1 modulates the strength and magnitude of endothelial activation and vascular inflammation; 29 however, its roles in IA remain uncharacterized. YAP and TAZ pair primarily with the TEAD family to elicit gene expression. 20 In the present study, we identified that PDLIM1 is a downstream target gene of YAP1 and is downregulated in IA tissues. Recently, emerging evidence has suggested that PDLIM1 participates in many physiological and pathological processes including but not limited to cytoskeletal organization, organ development, and tumor progression. PDLIM1 can modulate multiple signal transduction pathways to exhibit tissue-specific functions. For instance, PDLIM1 attenuates the NF-κB-mediated inflammatory cascade by sequestering p65 NF-κB in the cytoplasm. 30
In this study, we demonstrated a mechanistic link between PDLIM1 and the Wnt/β-catenin signaling pathway. In normal cells, β-catenin acts as a skeletal protein to maintain cell adhesion. When the signaling molecule Wnt activates the Wnt/β-catenin pathway, β-catenin is activated and accumulates in the nucleus to regulate physiological functions. In VECs, β-catenin can anchor VE-cadherin to maintain its cell adhesion and cell connection functions. 31 When β-catenin is inactivated, the expression of vascular endothelial (VE)-cadherin also decreases. Correspondingly, the connection of VECs is weakened, and the permeability of endothelial cells increases. 32 In contrast, the restoration of Wnt/β-catenin pathway function increases the expression of VE-cadherin, and cell permeability is also restored. The Wnt/β-catenin pathway also affects vascular endothelialization after IA treatment. 33 Recent studies have pointed out that VE-cadherin deficiency in VECs results in polarization disorder, decreased cell migration ability, increased microvascular bleeding, and reduced vascular endothelial permeability.34,35 What is more interesting is that a series of dysfunctions of VECs mentioned above can be alleviated by the uncoupling of VE-cadherin and actin, and actin is one of the important interactors of PDLIM1. Several studies have also confirmed that inhibition of intracellular PDLIM1 can lead to the inactivation of the physiological functions of the Wnt/β-catenin pathway, which in turn leads to weakened cell proliferation and increased apoptosis. 36 In colorectal cancer, PDLIM1 suppresses the EMT phenotype and invasive capacity of cancer cells via stabilization of β-catenin at cell-cell junctions. 17 However, whether pharmacological inhibition of Wnt/β-catenin activity compromises the effect induced by PDLIM1 in a rat model of IA warrants further investigation. Altogether, PDLIM1-dependent regulation of β-catenin activity may be a major mechanism for its role in IA.
Study limitations
There are several limitations in the present study. First, our study mainly focused on the investigation of PDLIM1 in VEC, and whether PDLIM1 is expressed or plays a role in other cells within the arterial walls, such as vascular smooth muscle cells and inflammatory immune cells, warrants further investigation. Second, adequate control tissue, such as superficial temporal artery, is essential for the pathophysiological study of IA. Our study regarding gene expression investigation is limited by not including proper control vessels. Third, our study highlights the biological effect of PDLIM1 on IA development via activation of Wnt/β-catenin signaling. However, we cannot rule out the possibility that PDLIM1 might regulate other signaling cascades to affect IA development.
Conclusion
Our findings demonstrate a protective role of PDLIM1 against intracranial aneurysm via a β-catenin-dependent mechanism. Loss of PDLIM1 in the VECs is YAP1 dependent. PDLIM1 deficiency in IA tissues may contribute to dysregulation of endothelial cell function and is a potential indicator of aggressive disease. Thus, PDLIM1 may be exploited as a biomarker or therapeutic target in the individualized management of patients with intracranial aneurysm.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231218210 – Supplemental material for Decreased PDLIM1 expression in endothelial cells contributes to the development of intracranial aneurysm
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231218210 for Decreased PDLIM1 expression in endothelial cells contributes to the development of intracranial aneurysm by Yan Yan, Xuanfeng Qin, Yongtao Zheng, Tao Jin, Yuanyuan Hu, Qingzhu An and Bing Leng in Vascular Medicine
Footnotes
Availability of data and material
The data used and analyzed during this study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the National Natural Science Foundation of China (82171312).
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
