Abstract
Aortic aneurysm (AA) and aortic dissection (AD) are prevalent severe cardiovascular diseases that result in catastrophic complications and unexpected deaths. Owing to the lack of clinically established and effective medications, the only treatment options are open surgical repair or endovascular therapy. Most researchers have focused on the development of innovative medications or therapeutic targets to slow the progression of AA/AD or lower the risk of malignant consequences. Recent studies have shown that the use of fluoroquinolones (FQs) may increase susceptibility to AA/AD to some extent, especially in patients with aortic dilatation and those at a high risk of AD. Therefore, it is crucial for doctors, particularly those in cardiovascular specialties, to recognize the dangers of FQs and adopt alternatives. In the present review, the main clinical observational studies on the correlation between FQs and AA/AD in recent years are summarized, with an emphasis on the relative physiopathological mechanism incorporating destruction of the extracellular matrix (ECM), phenotypic transformation of vascular smooth muscle cells, and local inflammation. Although additional data are required, it is anticipated that the rational use of FQs will become the standard of care for the treatment of aortic diseases.
Keywords
Introduction
Aortic aneurysm (AA) and aortic dissection (AD) are the most lethal aortic diseases in adults. 1 In Western countries, 1–2% 2 of deaths are caused by AA, which is a persistent localized dilatation of the aorta. As its most serious complication, AA rupture frequently results in deadly hemorrhage, with a prehospital mortality rate of up to 80%. 3 It is reported that 60% of all AAs are abdominal aortic aneurysms (AAAs).4,5 AD is the separation of the aortic media caused by aortic intimal tears or aortic wall hemorrhage. The mortality rate can reach 90% if treatment is delayed. 6 Recent studies have indicated that fluoroquinolones (FQs), in addition to their antibacterial abilities, can cause structural damage to the aorta. The associated mechanisms involve extracellular matrix degradation (ECM) and remodeling, increased phenotypic transformation and loss of vascular smooth muscle cells (VSMCs), inflammatory response, and oxidative stress in the arterial wall. Considering the increasing expansion of FQ use, FQ therapy is anticipated to become a nonnegligible aggravating factor in AA and AD. 7 However, current understanding suggests that the aortic risk of FQs is not frequently recognized. From initial observational studies to subsequent animal and cell tests, research in related fields has increased worldwide in recent years. In this review article, the latest research results have been summarized to expand the clinical pharmacological reference guidance.
Characteristics and clinical observation of fluoroquinolones (FQs)
Overview of FQs
FQs are one of the most widely used antibiotics worldwide owing to their broad antibacterial spectrum, good oral bioavailability, and wide tissue penetration. 7 FQs exert antibacterial effects mainly by targeting bacterial DNA topoisomerase IV and gyrase. 8 The most frequently prescribed FQs are ciprofloxacin, norfloxacin, ofloxacin, levofloxacin, enrofloxacin, danofloxacin, and moxifloxacin. 8 Ciprofloxacin is the most commonly used FQ. 9 Initially, use of FQs was associated with an increased incidence of tendinopathy and retinal detachment. The results showed that FQs enhanced the expression of matrix metalloproteinase (MMP) (Mmp1/13, 10 Mmp2, 11 and Mmp3 12 ) in tendon tissue and Mmp2/9 in corneal tissue. 13 Type I collagen (COL) and β1-integrin production decreased and degradation increased.10,11 This leads to structural damage and reconstruction of ECM. In addition, FQs exacerbate the damage and oxidative stress to the mitochondria and other organelles of tendon cells, resulting in tendon cells malfunctioning and increased apoptosis.10,14 Excessive ECM degradation mediated by MMPs and parenchymal cell death were also closely related to the occurrence and development of AA/AD.15,16
Clinical observation studies on FQs and susceptibility to aortic aneurysum and dissection (AA/AD)
In a nested case–control analysis in Taiwan, Lee et al. discovered a greater than twofold increase in the risk of AA or AD within a 60-day risk period for FQ exposure. This risk was more pronounced in patients older than 70 years and in women, and was substantially related to a longer term of exposure. 17 Previous data have shown that the occurrence of AAAs is two to five times more prevalent in men aged > 65 years than in women. 18 In the International Registry of Acute Aortic Dissection (IRAD) registry, 65% of patients with AD were men. 19 However, the results of this study showed that AD incidence in women (relative risk [RR], 1.83; 95% CI, 1.27–2.64) appeared to be higher than that of men (RR, 1.61; 95% CI, 1.28–2.03); hence, women were more susceptible to FQs.
Another cross-case study involving 1,213 AA/AD patients by the same investigators compared the distribution of FQ exposure in the same patients 60 days (risk period) before the AA/AD event and 60–180 days (reference period) before the AA/AD event. In the main case crossover analysis, exposure to fluoroquinolone was more frequent during the hazard periods than during the referent periods (1.6% vs 0.6%; odds ratio [OR], 2.71; 95% CI, 1.14–6.46). 20 However, antibiotic use is frequently accompanied by infection, which is associated with an increased risk of AA/AD. 21 Pathogenic bacteria can cause mycotic aneurysms (MAs) through the blood stream, spread to adjacent lesions, or direct infection by trauma. 22 Thoracic and abdominal aortic MAs comprise 0.7%–4.5% of all AAs. 23 Gram-positive bacteria (e.g., Staphylococcus, Enterococcus, Streptococcus, and Bacillus cereus) and gram-negative bacilli (including Salmonella and Haemophilus influenzae) are the main pathogenic bacteria that cause aortic infections. Therefore, FQs must be compared with other types of antibiotics (such as penicillin and cephalosporins) in case–control studies to exclude the effect of MA on authenticity as much as possible. Otherwise, the aortic risk associated with FQs may have been overestimated. In another longitudinal observational study conducted in Ontario, Canada, the use of FQs was associated with a higher risk of AA within 30 days of exposure (hazard ratio [HR], 2.24; 95% CI, 2.02–2.49) among 1,744,360 adults over 65 years of age who were tracked. This risk was comparable to that posed by hypertension (HR, 2.20; 95% CI, 2.09–2.31) and remained significant when compared with that of amoxicillin (HR, 1.50; 95% CI, 1.32–1.70). 24 Similarly, a nationwide historical cohort study from Sweden found a 66% greater risk of AA/AD in the aged (> 50 years) during the 60 days following oral FQs compared to that with amoxicillin. 25
These findings also prompted the US Food and Drug Administration (FDA) and European Medicines Agency (EMA) to issue safety warnings regarding FQs and to identify ciprofloxacin, levofloxacin, and moxifloxacin as significantly associated with AA/AD.26–29 Relevant warnings are listed in Table 1. In patients with chronic obstructive pulmonary disease, these medications are not indicated as first-line treatments for acute sinusitis, bacterial infections, or urinary tract infections because the risks outweigh the benefits. 30 A US retrospective analysis found that use of FQs increased the risk of adult aneurysms, iliac artery aneurysms, and other abdominal aneurysms compared to that with multiple antibiotics. 31 The use of FQs was associated with aortic mortality and resulted in a higher risk of death from any cause (HR 1.61; 95% CI, 1.50–1.73). 32 A meta-analysis also showed that compared to amoxicillin, azithromycin, doxycycline, or no antibiotics, FQs were associated with a higher risk of developing AA/AD within 30/60 days. 33 These studies emphasized the correlation between the use of FQs and the occurrence and adverse effects of AA/ADs. Although infection increases the risk of AA/AD, the use of FQs further increases this risk.
US Food and Drug Administration and European Medicines Agency warnings for fluoroquinolones.

EMA, European Medicines Agency; FDA, Food and Drug Administration; FQ, fluoroquinolones.
However, other studies have reported contradictory results. In a recent cohort study based on a Taiwanese population, there was no connection between FQs and an increased incidence of AA/AD in patients with urinary tract infections (UTIs) compared with first- or second-generation cephalosporins. 34 However, this study had several limitations. Patients with UTIs usually do not undergo a comprehensive aortic evaluation, and there were surveillance biases in this study. Additionally, the number of events in the subgroup analysis was relatively limited. Nevertheless, this study also suggests that the major objective for patients with UTIs is the administration of adequate medications and infection management. Dong et al. analyzed 21,651,176 adult patients in the Taiwan Population Health Insurance claims database and found patients with sepsis (OR, 3.16; 95% CI, 2.63–3.78) and abdominal infection (OR, 2.99; 95% CI, 2.45–3.65) had the highest AA/AD risk increase if they received FQs within 60 days. 35 However, FQs did not demonstrate an increased risk of AA/AD when compared with amoxicillin–clavulanic acid or ampicillin–sulbactam (OR, 1.01; 95% CI, 0.82–1.24), and the results were stable when compared with those using extended spectrum cephalosporins (OR, 0.88; 95% CI, 0.70–1.11). Although various corrections have been made for detection and monitoring biases in contemporary studies, no definite causal relationship has been derived from these observational studies. Hence, additional randomized clinical studies with larger sample sizes and stringent control for other factors should be performed. In addition, the long-term use of FQs remains common in patients with multidrug-resistant tuberculosis (MDRTB) and chronic bacterial prostatitis. 36 Future studies should consider the impact of long-term FQ exposure on aortic disorders. Additional study characteristics are presented in Table 2.
Clinical studies of aortic aneurysm or aortic dissection (AA/AD) risk associated with fluoroquinolones (FQs).
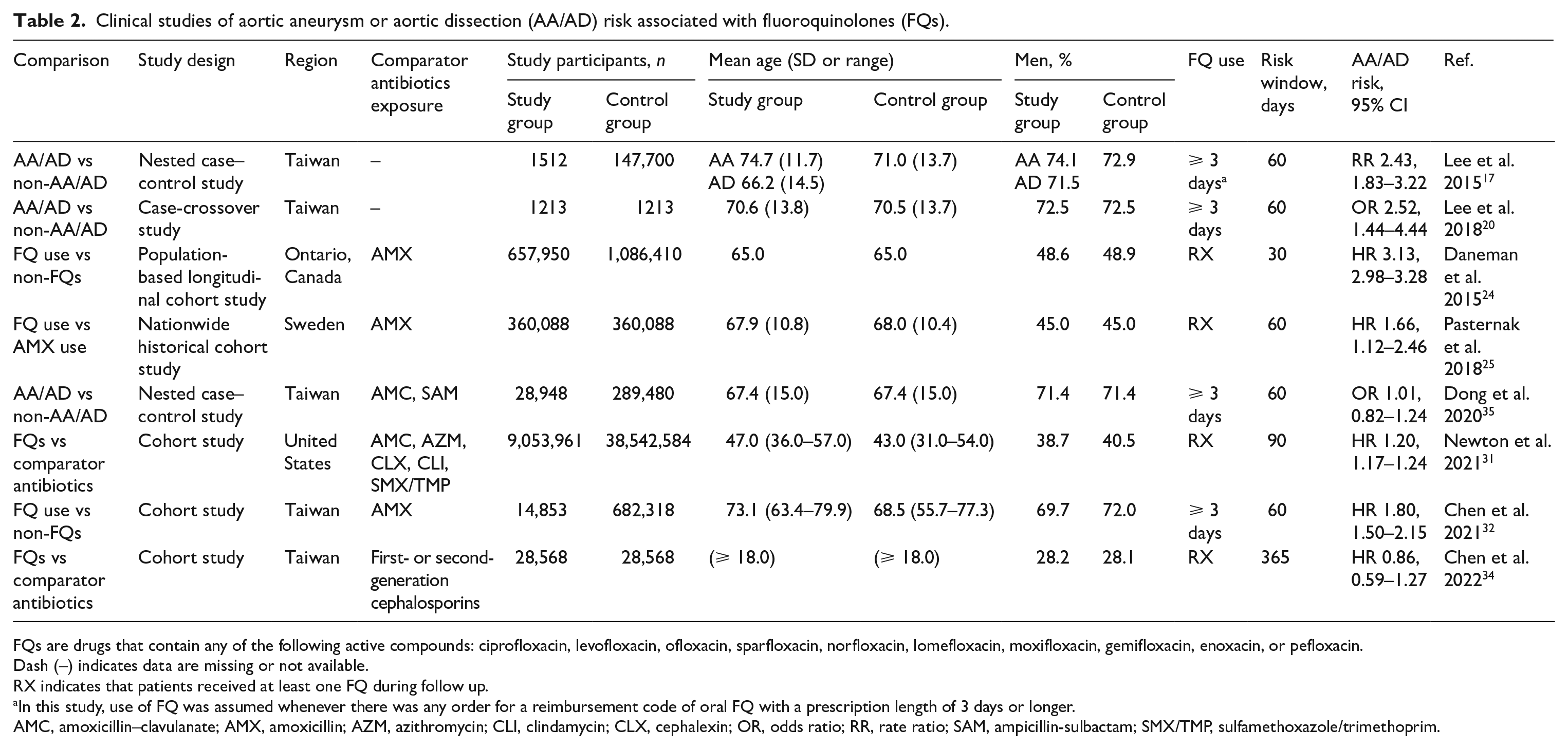
FQs are drugs that contain any of the following active compounds: ciprofloxacin, levofloxacin, ofloxacin, sparfloxacin, norfloxacin, lomefloxacin, moxifloxacin, gemifloxacin, enoxacin, or pefloxacin.
Dash (–) indicates data are missing or not available.
RX indicates that patients received at least one FQ during follow up.
In this study, use of FQ was assumed whenever there was any order for a reimbursement code of oral FQ with a prescription length of 3 days or longer.
AMC, amoxicillin–clavulanate; AMX, amoxicillin; AZM, azithromycin; CLI, clindamycin; CLX, cephalexin; OR, odds ratio; RR, rate ratio; SAM, ampicillin-sulbactam; SMX/TMP, sulfamethoxazole/trimethoprim.
FQ-related structural damage to the aorta
The aorta wall is primarily composed of various types of cells and ECM. 37 Anatomically, the aortic wall can be divided into three layers arranged in concentric circles: the intima, media, and adventitia. 38 The three layers are separated by internal and external elastic lamina. The endothelial cells lining the inner surface of the aortic wall constitute the majority of the cellular composition. 39 The media is the thickest and is predominantly composed of multilayer VSMCs and elastic lamellae. 40 The adventitia comprises adipocytes, fibroblasts, and blood arteries that supply the tissue. 41 The media consists of dozens of annular VSMCs and annular elastic lamellae arranged in a concentric circular structure. 42 The function and number of cells in the aorta wall and the composition of ECMs determine the structure and strength of the aortic wall, which in turn affects the risk of aortic dilation or dissection.37,43 According to recent investigations, FQs can disrupt the aortic ECM, induce VSMC damage and phenotypic alterations, and cause local aortic inflammation (Table 3).
In vitro and animal experiments associated with fluoroquinolone-related aortic aneurysm or aortic dissection.
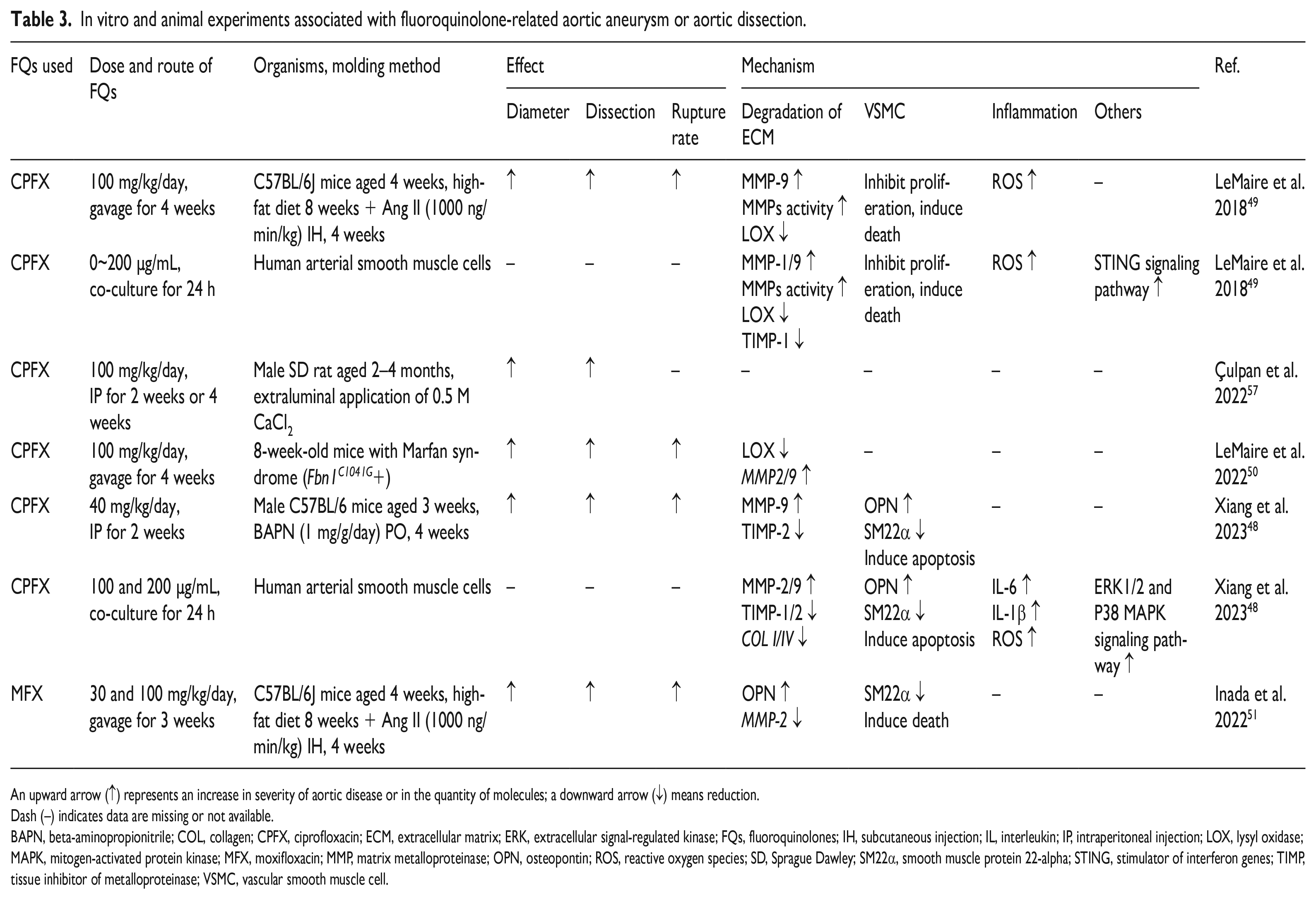
An upward arrow (↑) represents an increase in severity of aortic disease or in the quantity of molecules; a downward arrow (↓) means reduction.
Dash (–) indicates data are missing or not available.
BAPN, beta-aminopropionitrile; COL, collagen; CPFX, ciprofloxacin; ECM, extracellular matrix; ERK, extracellular signal-regulated kinase; FQs, fluoroquinolones; IH, subcutaneous injection; IL, interleukin; IP, intraperitoneal injection; LOX, lysyl oxidase; MAPK, mitogen-activated protein kinase; MFX, moxifloxacin; MMP, matrix metalloproteinase; OPN, osteopontin; ROS, reactive oxygen species; SD, Sprague Dawley; SM22α, smooth muscle protein 22-alpha; STING, stimulator of interferon genes; TIMP, tissue inhibitor of metalloproteinase; VSMC, vascular smooth muscle cell.
Disruption of the ECM
ECM is the predominant noncellular component of the aorta, and its components and structure are in a dynamic balance between constant renewal, degradation, and remodeling. 44 Collagen and elastin are the primary ECM components responsible for maintaining mechanical properties of the aorta wall, accounting for 30% and 35% of its dry weight, respectively. 45 In addition, overexpression of MMP degrades collagen and elastin, resulting in structural damage to the ECM. 46 As shown in Figure 1, when the ECM is damaged or its structure is altered, aortic strength and elasticity are diminished. 47 As a result, the aorta is more susceptible to permanent dilatation or rupture of the lining as it opposes the pulse of blood, with particularly pronounced variations in blood pressure.
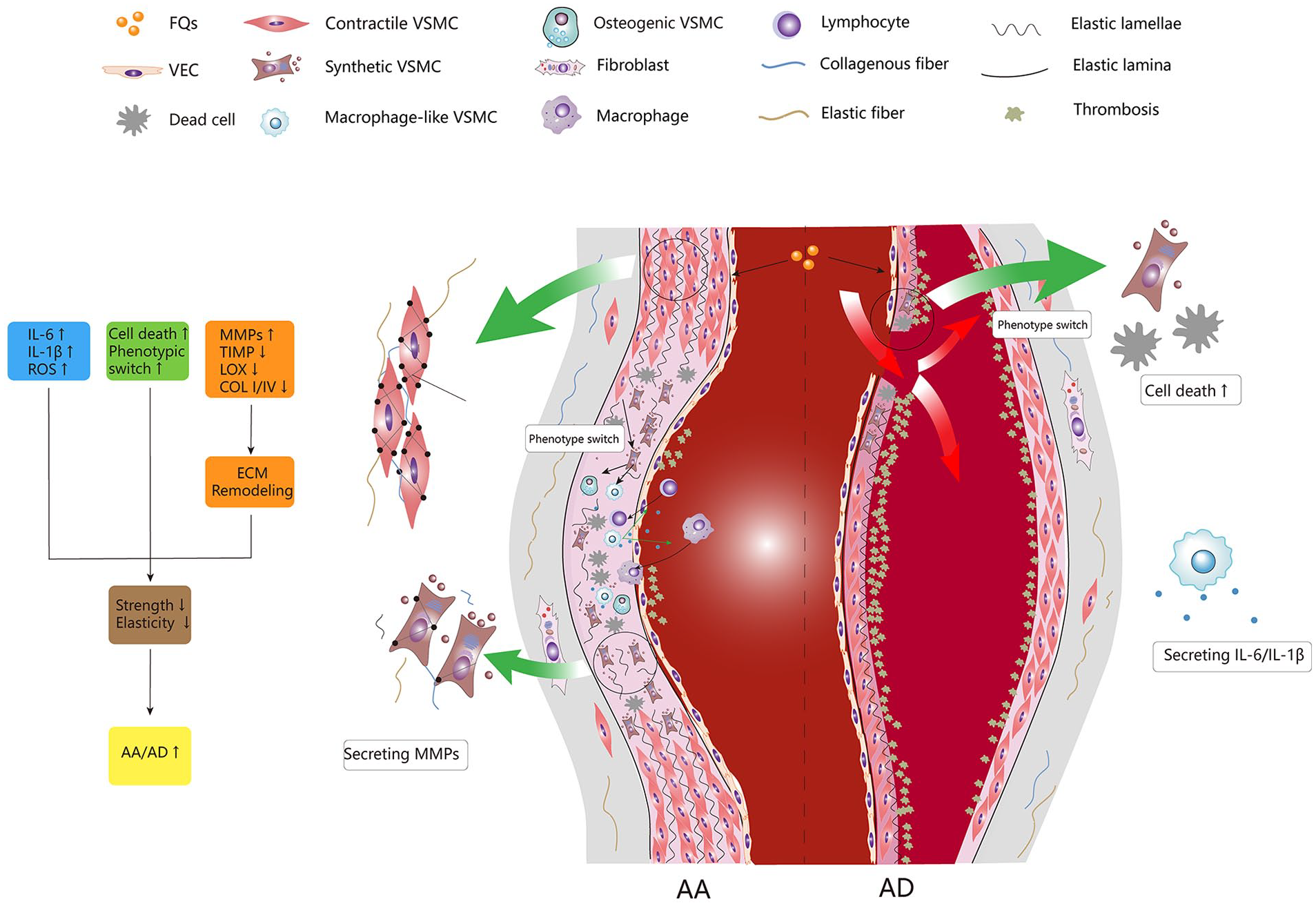
Fluoroquinolones (FQs) are associated with structural damage of the aorta. The aorta consists of three layers; from the inside out are the intima, the media, and the adventitia. There is an elastic lamina between each layer. The media consists of dozens of annular VSMCs and annular elastic lamellae arranged in a concentric circle structure. VSMCs are the highest proportion of cells in the media. Under the influence of FQs, VSMCs switch to other phenotypes from contractile phenotype. Among them, macrophage-like VSMCs secrete massive cytokines, recruit circulating macrophages and lymphocytes into the arterial wall, and aggravate the local inflammatory response. MMPs secreted by macrophages and macrophage-like VSMCs degrade collagen and elastin abundant in ECM, thus causing the degradation and disappearance of elastic lamellae. Moreover, under the influence of FQs, massive VSMCs died in the media, and the level of TIMP, LOX, and COL I/IV decreased, which further reduced the contractility and strength of the arterial wall. Together, these factors contribute to an increased susceptibility to AA/AD.
Ciprofloxacin (200 µg/mL) decreased the mRNA levels of COL I/IV in cultured human arterial smooth muscle cells (SMCs). 48 Ciprofloxacin (100 mg/kg/day) increases the activity of MMPs and the level of MMP-9 49 in the aortic wall of mice with AA and AD. Ciprofloxacin (100 mg/kg/day) significantly promotes the deterioration of aortic elastic fiber structure in rats with Marfan syndrome and considerably elevates the expression of Mmp2 and Mmp9. 50 Given that osteopontin (OPN) can induce the expression of MMP2/9, Inada et al. found that moxifloxacin (30 and 100 mg/kg/day) increased the expression of Opn in the aorta of angiotensin II-treated mice. Therefore, MMP2 overexpression and elastin breakdown are induced by FQs. 51 MMP activity is regulated by endogenous tissue inhibitors of MMPs (TIMPs). An imbalance in MMP/TIMP can cause excessive ECM degradation. 52 The expression of TIMP1/2 of VSMCs in a human aortic microphysiological model was significantly down-regulated after ciprofloxacin (200 µg/mL) treatment. 48 Furthermore, ciprofloxacin exposure significantly reduced TIMPs and collagen secretion in aortic myoblasts, resulting in a significant increase in the MMP-9/TIMP-2 ratio. 53
Ciprofloxacin not only directly or indirectly induces the enhancement of MMPs, but also destroys the structural stability of the ECM fibrin. Lysyl oxidase (LOX) is an amine oxidase involved in the assembly and stability of elastic fiber.54,55 Ciprofloxacin (100 mg/kg/day) reduced LOX levels in the aortic wall of aortic aneurysm and dissection (AA/AD) mice, promoted AA/AD formation, and increased susceptibility to aortic rupture. 49 Disruption of the ECM can lead to progressive enlargement of the aortic diameter. In clinical practice, the maximal AAA diameter is the most commonly used measure to assess the risk of AAA rupture. 56 In a recent study, Çulpan et al. observed that ciprofloxacin (100 mg/kg/day) significantly increased the abdominal aortic diameter in a CaCl2-induced AAA model in Sprague Dawley (SD) rats, which was linked to the duration of ciprofloxacin treatment. 57 However, short-term (4 weeks) use of FQs did not appear to cause significant expansion of the diameter of the normal aorta, but significantly aggravated the destruction of the aortic wall in the presence of risk factors (angiotensin II or elastase induction). Therefore, these results suggest that FQs disrupt the aortic ECM by inducing excessive ECM degradation and reducing the structural stability of fibrin, which significantly promote the progression of AA/AD and increase susceptibility to AA rupture.
VSMC damage and phenotypic transformation
Most VSMCs are located in the media, which is the primary cellular component of the aortic wall. Its status and quantity are closely related to the function of the aorta. 58 Ciprofloxacin can induce damage and increased apoptosis of aortic cells in mice, and the expression of apoptotic genes (such as Bcl2, Bax, Caspase-3) is significantly up-regulated. Ciprofloxacin decreases the activity of normal human aortic VSMCs in a dose-dependent manner and promotes cell death. It has been reported to cause subcellular nuclear and mitochondrial DNA damage in aortic VSMCs, followed by mitochondrial dysfunction and the production of reactive oxygen species. 49 Xiang et al. found that ciprofloxacin (200 µg/mL) induced mitochondrial dysfunction in human aortic VSMCs and promoted apoptosis via the P38 and ERK1/2 MAPK pathways. 48
Previously, VSMCs were classified as contractile and synthetic/proliferative phenotypes. As the primary type of VSMCs, contractile VSMCs not only maintain the integrity of the aortic wall, but also provide them with abundant tension and active elasticity to avoid excessive expansion of the aorta in response to blood pressure.47,59 Under the action of multiple injury factors inside and outside the blood vessels, the number of contractile VSMCs is excessively reduced owing to apoptosis, or the phenotypic transformation of VSMCs is caused by impaired mitochondrial function, which leads to decreased strength of the aortic wall, and VSMCs cannot complete the recovery of artery shape after cardiac pulse injection through contraction. This results in an overexpansion of the blood vessel wall and its eventual rupture. 60 Synthetic VSMCs have weak contractility but robust proliferative and secretory properties, including collagen, elastin, laminin, gelatin, and proteoglycan. 47 Synthetic VSMCs also play a pivotal role in preserving the vascular wall integrity. The dynamic balance between these two phenotypes is the basis of maintaining the physiological function of the aortic wall. In recent years, however, with the application of lineage tracing techniques and single-cell analysis techniques, researchers have newly defined macrophage-like, mesenchymal-like, fibroblast-like, and osteogenic VSMCs in addition to the above two phenotypes. 61 Macrophage-like VSMCs have a certain phagocytic capability and release MMPs and chemokines, which can exacerbate the local inflammatory response and cause excessive degradation of elastin, thereby accelerating the progression of AA/AD. Recent studies have demonstrated that ciprofloxacin exposure significantly increases apoptosis and decreases cell activity in the aortic structure of mice with Marfan syndrome. 53 Ciprofloxacin (200 µg/mL) decreases the expression level of contractile markers smooth muscle 22 alpha (SM22α) and calponin 1 (CNN1) in human arterial SMCs. Moxifloxacin inhibits Sm22 expression in the aorta of angiotensin II-treated C57 male rats. The decreased expression of contractile phenotypic markers suggests the transformation of VSMCs to synthesis, and the secretion of OPN and MMP-2 leads to remodeling of the aortic wall structure.48,51 Al-Neklawy et al. conducted pathological staining of sections from rats treated with ciprofloxacin, revealing multiple tissue losses in the tunica media, disordered arrangement of VSMCs, abnormal reduction in the number of VSMCs, and most notably, an increase in eosinophilic VSMCs, which may be the result of VSMCs injury. The diminished immunohistochemical staining of α-smooth muscle actin (αSMA) in the ciprofloxacin-treated group was indicative of phenotypic transition of VSMCs. 62 Moreover, as a DNA topoisomerase inhibitor, ciprofloxacin can cause nuclear and mitochondrial DNA damage and induce mitochondrial dysfunction and reactive oxygen production, thereby promoting the phenotypic transformation of VSMCs. 49 There are currently a small number of studies examining whether FQs influence the phenotypic transformation of VSMCs, and lineage tracing and single-cell analysis techniques are uncommon in this field. It is anticipated that inhibition of the transformation of VSMCs into macrophage-like phenotypes will become a crucial therapeutic target for AA/AD. More relevant and in-depth research in this field in the future is recommended.
Aortic inflammatory response
It has been established that inflammatory responses play a crucial role in the onset and progression of AA and AD. 63 A substantial number of chronic inflammatory cells, such as lymphocytes, macrophages, mast cells, and neutrophils infiltrate the tunica media and adventitia of AA and AD, which are essential for aortic injury and repair. Circulating neutrophils are involved in the early stages of AAA formation, but their numbers do not differ significantly in the late stages. 63 Mononuclear macrophages are the most abundant inflammatory cell type, and infiltrating macrophages can be polarized into either M1 or M2 subtypes. Stimulated by tissue inflammatory factors, M1-type macrophages produce proteolytic enzymes such as MMPs to participate in tissue remodeling, and secrete a variety of chemokines to recruit more monocytes/macrophages and lymphocytes into the aortic wall, forming a positive feedback loop. 64 In contrast, M2-type macrophages exert an antiinflammatory function, mainly by secreting cytokines such as interleukin (IL)-4, IL-10, and IL-13. 65 In AA and AD, the M1/M2 balance is disrupted, which triggers the onset or progression of local inflammation. It has been shown that the depletion of CD11c+ antigen-presenting cells (DCs) can significantly reduce the maximum AAA diameter. 66 Mast cell-derived proteases, such as trypsin, synergistically increase MMP expression and VSMCs apoptosis synergistically. 67 Notably, there is a positive correlation between the number of mast cells and the maximum AAA diameter. 68 As both CD4+ T cells and CD8+ T cells accumulate in AAA samples, studies have shown that IFN-γ released by CD8+ T cells can promote the recruitment of macrophages, thus increasing the expressions of proteolytic enzymes and chemokines. 69
IL-6 and IL-β1 are important proinflammatory cytokines that recruit monocytes into the arterial wall and are closely associated with AAA and preruptured AAA. 70 Ciprofloxacin induces the increased expression of proinflammatory factors (IL-6 and IL-1β) and reactive oxygen species in aortic SMCs. 49 In a recent study, Çulpan et al. discovered that ciprofloxacin administration (100 mg/kg/day) resulted in severe inflammatory changes and structural damage to the aortic wall in a AAA model of SD rats induced by CaCl2. 57 Inflammation influences the risk of aortic dilation and rupture and is involved in aortic remodeling. However, few studies have investigated fluoroquinolone-induced inflammatory responses in AA and AD. It is necessary to further study the number and type of inflammatory cells and the expression level of inflammatory factors in the aortic wall following FQ exposure, as this is believed to be an important mechanism by which FQs promote the advancement of AA and AD.
Mechanisms for future study
Changes in endothelial cell function also play important roles in the occurrence and development of AA/AD.71,72 Endothelial cells secrete vasoactive chemicals, participate in the interchange of fluids, form barriers, and contribute to coagulation and inflammation. Endothelial cells and the inner elastic membrane beneath them jointly form a vascular endothelial barrier that is compromised when endothelial cells die in significant numbers or when their function is impaired. Through the impaired intima, inflammatory cells penetrate the tunica media and release proinflammatory substances, resulting in additional degeneration of the tunica media. Hence, the occurrence and development of aneurysms and dissections is inevitable. Angiogenesis is prevalent in aneurysm tissues, and is most evident along the ruptured edge of an AA, indicating that it is associated with the evolution of AA. 73 In addition, angiogenic response is associated with inflammatory infiltration, which promotes aneurysm wall degeneration and rupture. 74 Possible mechanisms affecting the progression of AAA or dissection include the formation of neutrophil extracellular traps (NETs), loss of structural cells because of copper toxicity, and organelle destruction. However, to date, no relevant studies have been conducted. Exploration of the aforementioned directions is expected to further reveal the relevant mechanism of FQ-related AA/AD.
Summary
As the global population ages, researchers have predicted that the disease burden of AA/AD will continue to increase over the next decade.75,76 Recent basic studies have shown that FQs can increase susceptibility to AA/AD and aggravate its progression through multiple pathways (ECM degradation and remodeling, increased phenotypic transformation and loss of VSMCs, and vascular wall inflammation). However, the relevant mechanisms are still not well studied. Currently, except for reducing the use of FQs, there is no effective method for inhibiting the occurrence of FQ-related AA/AD. The phenotypic transformation of VSMCs, recruitment of inflammatory cells, changes in endothelial cell function, and neovascularization require further investigation. As FQs are one of the most widely used antibiotics, the use of alternative medicines or shorter courses to reduce unnecessary FQs treatments can help reduce the global burden of AA/AD. Based on the available evidence, we believe that health care professionals should be fully aware of the aortic risks associated with FQs. When FQs are prescribed, patients should stop taking them and seek medical attention if AA/AD-related symptoms occur. In patients with AA/AD, aortic dilatation, AA/AD high-risk patients (such as those with Marfan syndrome, Loys–Dietz syndrome, contractile-Goldman syndrome, familial thoracic aortic aneurysm, and dissection), and older adults, the benefit-risk should be carefully evaluated and FQs replaced with other antibiotics whenever possible. If FQ use is unavoidable in high-risk populations, aortic-related imaging should be considered during and after use of FQs. The use of FQs has saved the lives of numerous infected individuals. For low-risk patients with AA/AD, the use of appropriate FQs for infection control remains a first-line consideration, and optimal antiinfective therapy should not be delayed because of concerns regarding AA/AD.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the National Natural Science Foundation of China (Grant No. 81870354), Scientific Research Project of Shanxi Provincial Health Commission (Grant No. 202203021211031), and Graduate Education Innovation Program of Shanxi Provincial Education Institution (Grant No. 2022Y383).
