Abstract
Background:
We sought to assess the safety and efficacy of endovenous thermal ablation (EVTA) in treating large great saphenous veins (GSV) > 12 mm in diameter.
Methods:
We performed a systematic review according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) 2020 for comparative and noncomparative studies depicting EVTA in the treatment of GSV > 12 mm. Primary endpoints included GSV occlusion, technical success, deep vein thrombosis (DVT), and endovenous heat-induced thrombosis (EHIT). We conducted a comparative analysis between GSV > 12 mm and < 12 mm and a meta-regression analysis for two sets of studies, one including the whole dataset, containing treatment arms of comparative studies with GSV < 12 mm and one exclusively for GSV > 12 mm.
Results:
Seven studies, including 2564 GSV, depicting radiofrequency (RFA) and endovenous laser ablation (EVLA) were included. GSV > 12 mm occlusion, technical success, DVT, and EHIT estimates were 95.9% (95% CI: 93.6–97.8), 99.9% (95% CI: 98.9–100.0), 0.04% (95% CI: 0.0–3.4), and 1.6% (95% CI: 0.3–3.5). Meta-regression revealed a negative association between GSV diameter and occlusion for both the whole dataset (p < 0.01) and the > 12 mm groups (p = 0.04), GSV diameter and technical success for both groups (p < 0.01), (p = 0.016), and GSV diameter and EHIT only for the whole dataset (p = 0.02). The comparative analysis between GSV < 12 mm and GSV > 12 mm displayed an occlusion estimate of OR 1.79 (95% CI: 1.25–2.56) favoring small GSV.
Conclusion:
Whereas we have displayed excellent occlusion and technical success results for the EVTA of GSV > 12 mm, our analysis has illustrated the unfavorable impact of GSV diameter on occlusion, technical success, and EHIT outcomes regardless of the 12 mm threshold. Potential parameter or device adjustments in a diameter-oriented fashion could further enhance outcomes.
Keywords
Background
Endovenous thermal ablation (EVTA), including radiofrequency ablation (RFA) and endovenous laser ablation (EVLA), constitute the main pillars in the treatment of lower-limb venous insufficiency. Since the early years of their application, a debate has surfaced concerning their efficacy and safety in ablating large, great saphenous veins (GSV) of diameters larger than 12 mm.
Owing to their divergence in thermal energy conduction mediation, the ongoing controversy around RFA and EVLA stemmed from different qualities exhibited by the early generation of both apparatuses. On the one hand, the hemoglobin-specific affinity displayed by the original EVLA device which utilized a bare catheter tip (then, the only available configuration), in combination with the higher energy required when ablating larger GSV, resulted according to the literature in suboptimal occlusion outcomes and postinterventional complications such as pain and bruising.1 –3 On the other hand, since RFA ablation necessitates direct contact of the fiber tip with the venous endothelium and in an era where vein compression was achieved with the use of Esmarch rubber bandage, physicians were reluctant to employ RFA ablation for larger GSV.
Regardless of the limitations presented by the early generation of EVTA devices, modifications such as the shift from a < 1000 nm hemoglobin-specific, high energy-demanding EVLA device to the newer > 1000 nm water-specific lower energy-requiring apparatus in combination with the routine use of tumescent anesthesia and the development of contemporary techniques (multipass technique) have considerably expanded the capabilities of EVTA. 4
In line with these developments, the recently published European Society for Vascular Surgery (ESVS) guidelines have upgraded the role of EVTA in the treatment of very large GSV (> 12 mm) (class: IIa, level of evidence: C). We performed a systematic review, meta-analysis, and meta-regression in an effort to summarize evidence and investigate the safety and efficacy of EVTA in the treatment of large GSV.
Methods
Design
We performed a systematic review conforming to the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines 2020. The inclusion and exclusion criteria, along with the endpoints, were predetermined within a research protocol that was not registered. The corresponding author can provide the protocol upon request 5 (online Supplementary Figure 1).
Eligibility criteria
Inclusion criteria
We included randomized controlled trials, prospective and retrospective studies, depicting EVTA for GSV of diameters > 12 mm. We also included comparative studies with treatment arms displaying the effects of EVTA in the treatment of GSV < 12 mm. There were no time, sex, or language limitations.
Exclusion criteria
Studies providing GSV occlusion outcomes but failing to provide a definition on occlusion were excluded, as well as studies defining occlusion as a patent GSV for a length > 2 cm from the saphenofemoral junction (catheter tips are routinely positioned 1–2 cm distally to the GSV). Studies that included GSV ablation with EVLA devices < 1000 nm and studies depicting endovenous ablation without the use of tumescent anesthesia were also excluded. Studies displaying occlusion outcomes on a follow-up of under 6 months and studies with a total patient population of under 20 patients, or in the case of comparative analysis of under 10 patients per treatment arm, were also excluded. Lastly, we excluded studies describing ablation of the GSV, small saphenous vein, and/or accessory saphenous vein not providing separate data regarding GSV procedural outcomes.
Information sources, search strategy, selection, and data collection process
Investigators VB and AB performed a systematic search on Medline, Scopus, EMBASE, and Google Scholar. The research protocol was initiated synchronously by the two investigators in August 2022 and was concluded by October 2022. The search terms employed during the search process were identical for both researchers and included "great saphenous vein" AND "large", "GSV" AND "large", "great saphenous vein" AND "12 mm", "GSV" AND "12 mm", "saphenous vein" AND "12 mm", "saphenous vein" AND "large diameter". The researchers separately reviewed the abstracts of the recovered literature and identified publications that met the inclusion criteria. Following initial screening, the researchers retrieved the qualified articles in full and further assessed eligibility. When disagreement emerged, unanimity was achieved through consultation with a third researcher (AK). No automation tools were used during the screening process.
Data items
The primary endpoints included GSV occlusion by the end of follow-up, technical success, deep vein thrombosis (DVT), and endovenous heat-induced thrombosis (EHIT). Secondary endpoints included paresthesia, pigmentation-induration, superficial thrombophlebitis, and ecchymosis-hematoma.
Definitions
As successful GSV occlusion, we defined a noncompressible GSV and/or no evidence of flow upon duplex ultrasonography for the entire length of the ablated GSV segment or visible recanalization at a maximum length of 2 cm for the saphenofemoral junction. As technical success, we defined the successful passage of the catheter tip in proximity to the saphenofemoral junction followed by successful ablation of the GSV with no evidence of blood flow on duplex examination in the immediate postoperative period. As DVT, we defined any spontaneous thrombus formation into the deep venous system following ablation. EHIT was defined as the propagation of thrombus from the superficial venous system to the deep venous system categorized according to the endovenous heat-induced thrombosis classification including stages I to IV. 6
Study risk of bias assessment
Two investigators VB and AB individually assessed the risk of bias for the incorporated studies employing the methodological index for non-randomized studies (MINORS) tool. The MINORS checklist contains eight elements aiming to determine the risk of bias for noncomparative studies and 12 items (an extra four) for the evaluation of comparative studies. Checklist items are scored as 0 (not reported), 1 (inadequately reported), and 2 (adequately reported), with the maximum score for noncomparative studies being 16 and that of comparative studies 24. The risk of bias for noncomparative studies was labeled as 0–8 (high), 9–12 (moderate), and 13–16 (low). The risk of bias for comparative studies was designated as 0–12 (high), 13–18 (moderate), and 19–24 (low). We evaluated inter-observer agreement using the intra-class correlation coefficient (ICC) calculated with a two-way mixed effects model while we further measured skewness in order to identify possible trends.7,8
Effect measures and synthesis methods
We computed the relevant crude estimates for any given endpoint for each one of the included studies and reported outcomes as proportions. The consequent outcomes were thereafter meta-analyzed using the inverse variance method and the Freeman–Tukey double arcsine transformation of proportions. We calculated the pooled effect estimates as the back transformation of the weighted means of the transformed proportions, using the restricted maximum likelihood (REML) between study variance estimator and Hartung–Knapp confidence intervals. Given the variability in patient, anatomical, and interventional characteristics, the assumptions of fixed effect models were not met in our study. We therefore used random effects models for pooling effects estimates and presented them as proportions with their corresponding 95% CIs. Log odds ratios (OR) and log relative risks (RR) were generated by the Mantel–Haenszel method using the REML variance estimator and Hartung–Knapp CIs, back-transformed and presented as OR and RR, respectively, with their complementary 95% CI. The fraction of variance attributed to heterogeneity was estimated using the I2 test. We additionally performed meta-regression analyses for two groups of studies. The first group included the whole dataset, containing treatment arms of comparative studies with GSV < 12 mm, whereas the second group consisted only of studies and treatment arms depicting EVTA of GSV > 12 mm (aiming to evaluate the effect of covariates exclusively on GSV > 12 mm). Within the context of this review, study groups potentially encompassing GSV exhibiting a diameter precisely equivalent to 12 mm were assigned to the GSV > 12 mm subgroup. We used the mean GSV diameter in mm, the type of device used (EVLA or RFA), and the mean length of follow-up in months as covariates for the three primary endpoints. Given the absence of a null hypothesis and the inclusion of fewer than 10 studies, we did not proceed with the assessment of publication bias. For statistical analysis, we used R 4.1.0 (R Foundation for Statistical Computing, Vienna, Austria).9,10
Results
Baseline study characteristics
Seven studies, four comparative case series, and three noncomparative case series comprised of 2552 patients, and 2564 GSV were finally included. Three studies described interventions for GSV of diameters exceeding 12 mm, and four studies depicted interventions for GSV larger than 13 mm11–17 (Table 1).
Baseline study characteristics.
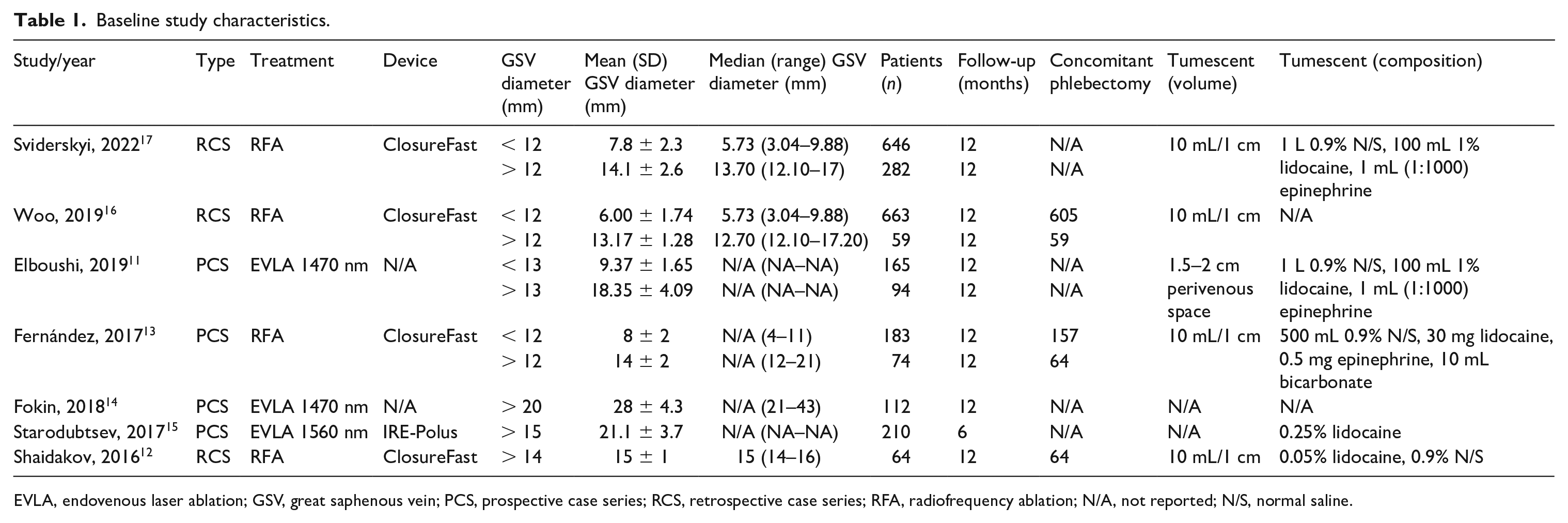
EVLA, endovenous laser ablation; GSV, great saphenous vein; PCS, prospective case series; RCS, retrospective case series; RFA, radiofrequency ablation; N/A, not reported; N/S, normal saline.
The mean MINORS score for noncomparative studies was 10.66 (SD = 0.62). The mean score for comparative studies was 16.75 (SD = 1.75). Interobserver agreement was excellent, ICC 0.93 (95% CI: 0.88–0.95), with a tendency toward higher scoring, skewness −0.83 (standard error of skewness, 0.20) (online Supplementary Table 1).
Overall endovenous thermal ablation efficacy and safety
Seven studies, four reporting on RFA and three on EVLA ablation, were included in the GSV occlusion endpoint. The crude and pooled GSV occlusion estimates by the end of follow-up were 95.4% (794/832) and 95.9% (95% CI: 93.6–97.8) (I2 = 22%, p = 0.26)11 –17 (Figure 1).
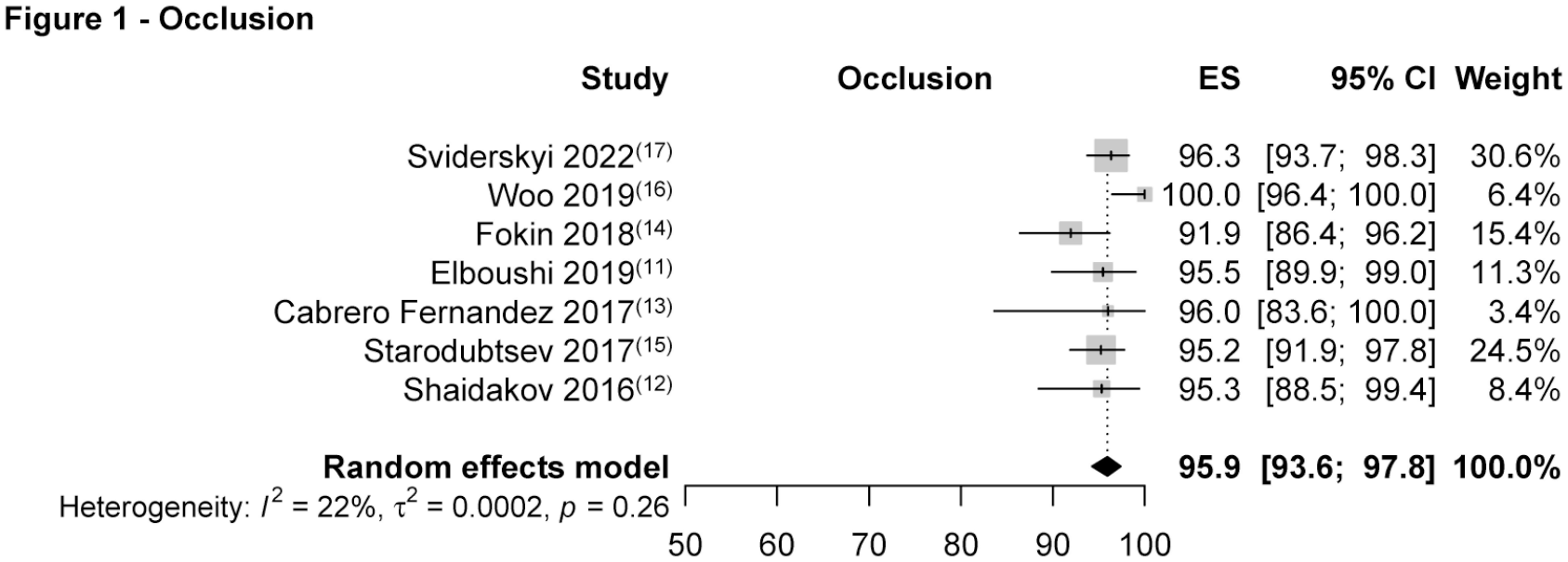
Great saphenous vein occlusion by the end of follow-up.
Technical success was reported by five studies, with three studies describing RFA and two EVLA ablation. The crude and pooled technical success estimates were 99.5% (630/633) and 99.9% (95% CI: 98.9–100.0) (I2 = 55%, p = 0.15).11,13,14,16,17 Three studies reported on DVT occurrence. The crude and pooled DVT estimates were 0.3% (1/368) and 0.04% (95% CI: 0.0–3.4) (I2 = 25%, p = 0.26).3,11,15 Three studies reported on EHIT occurrence with crude and pooled outcomes of 1.7% (8/465) and 1.6% (95% CI: 0.3–3.5) (I2 = 0%, p = 0.72).14,16,17 Out of the three included studies, two provided further information regarding EHIT classification with one recorded EHIT III and three EHIT II incidents.
Three studies reported on the paresthesia, pigmentation-induration, and ecchymosis-hematoma endpoints. The corresponding pooled estimates were 1.8% (95% CI: 0.0–8.5) (I2 = 39%, p = 0.2), 2.9% (95% CI: 0.0–18.2) (I2 = 77%, p = 0.01), and 2.1% (95% CI: 0.0–10.9) (I2 = 49%, p = 0.14).13,16,17 Two studies reported on superficial thrombophlebitis.13,16
Comparative analysis between ablation of GSV > 12 mm and GSV < 12 mm
Three studies portraying RFA ablation reported on GSV occlusion outcomes comparing GSV smaller and larger than 12 mm. One study investigating EVLA ablation reported on occlusion outcomes between GSV of diameters greater and smaller than 13 mm. In the analysis, we included all four studies describing 1844 GSV ablation events. We additionally controlled for the potential influence exhibited on our model by the study including GSV > 13 mm by performing a leave-one-out analysis. The OR between the smaller than 12 mm and larger than 12 mm subgroups was 1.79 (95% CI: 1.25–2.56) (I2 = 0%, p = 0.96), favoring GSV of diameters < 12 mm.11,13,16,17 Though the superiority of occlusion outcomes persisted throughout the leave-one-out analysis where the study by Elboushi et al. (< 13 mm) was omitted, outcomes failed to reach statistical significance, 1.77 (95% CI: 0.90–3.48) (I2 = 0%, p = 0.1) (Figure 2) (online Supplementary Figure 2).
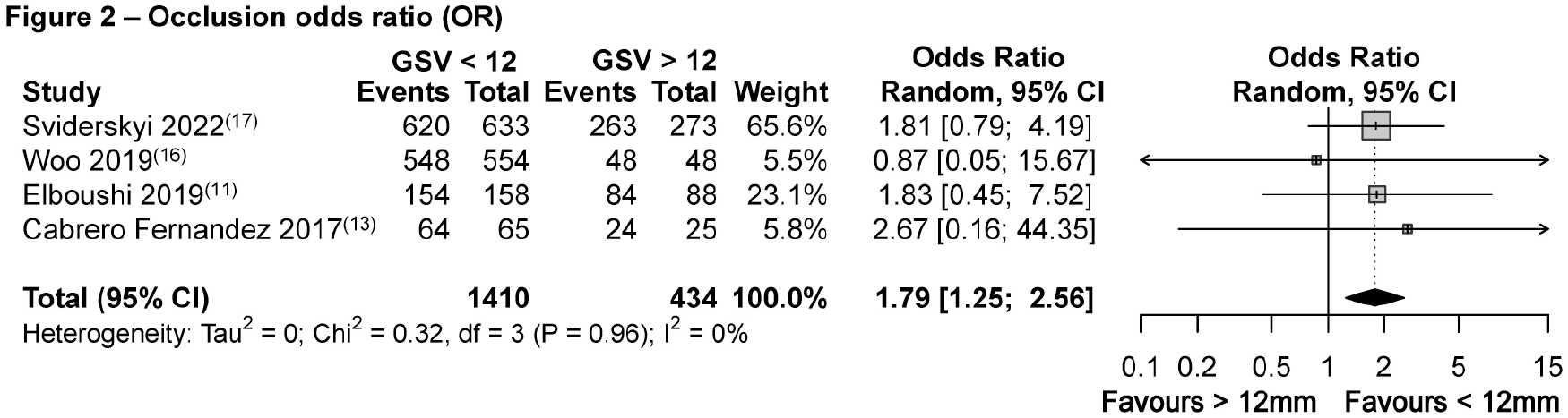
Occlusion odds ratio, comparative analysis between GSV > 12 mm and GSV < 12 mm.
Four studies reported on comparative outcomes regarding technical success with an OR of 2.45 (95% CI: 0.35–16.88) (I2 = 0%, p = 0.78), favoring small GSV.11,13,16,17 Two comparative studies reported on DVT outcomes between large and small GSV with a RR of 4.14 (95% CI: 0.35–48.98) (I2 = 0%, p = 0.58), favoring GSV of diameters < 12 mm.11,13 Two comparative studies depicting RFA interventions reported on EHIT occurrence with a RR of 3.62 (95% CI: 0.11–117.66) (I2 = 0%, p = 0.67), favoring small GSV.
Nonstatistically significant differences were identified between large and small GSV regarding paresthesia, ecchymosis hematoma, and pigmentation-induration with RRs of 0.7 (95% CI: 0.3–1.7) (I2 = 0%, p = 0.50), 1.0 (95% CI: 0.5–2) (I2 = 0%, p = 0.98), and 1.0 (95% CI: 0.6–1.6) (I2 = 28%, p = 0.25), respectively.13,16,17 Two studies reported on comparative outcomes regarding superficial thrombophlebitis.13,16
Meta-regression
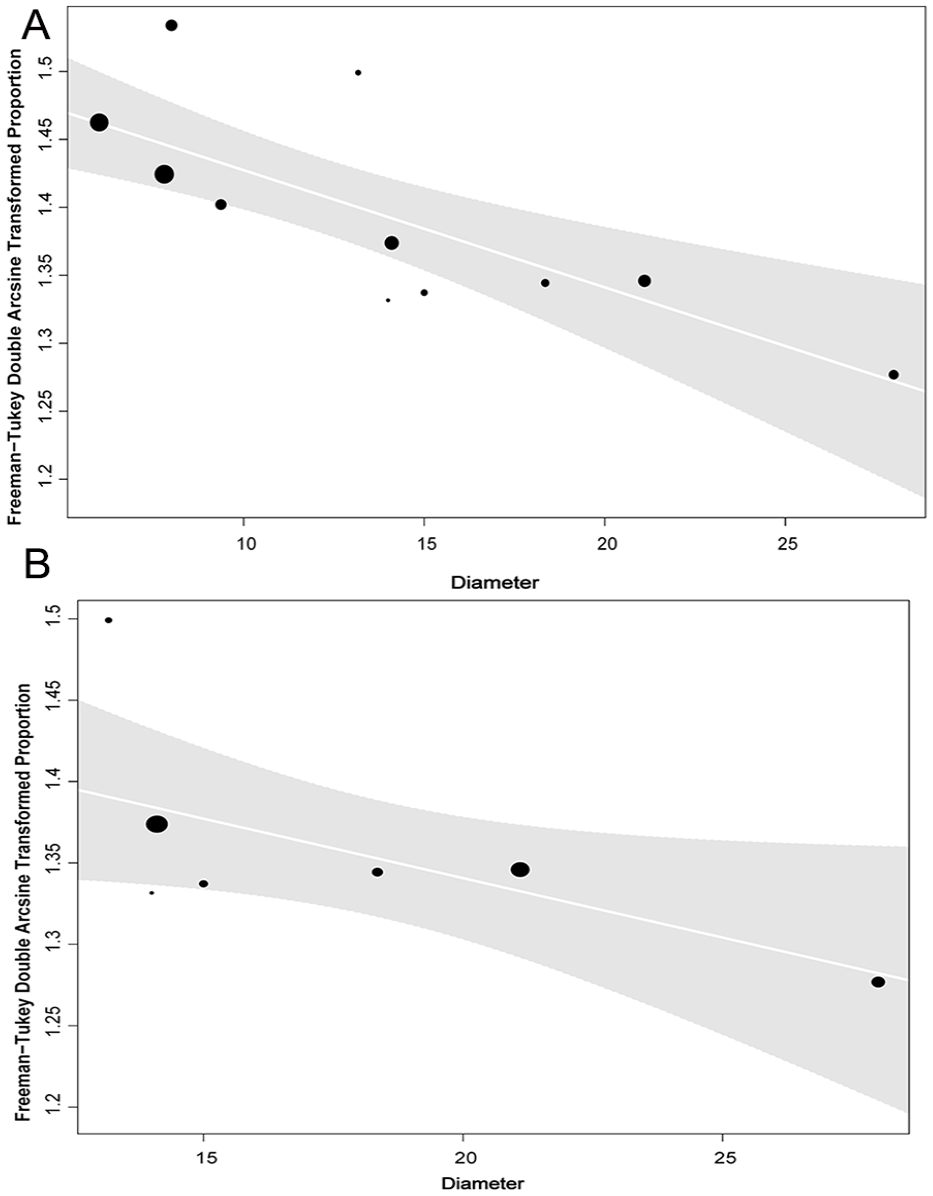
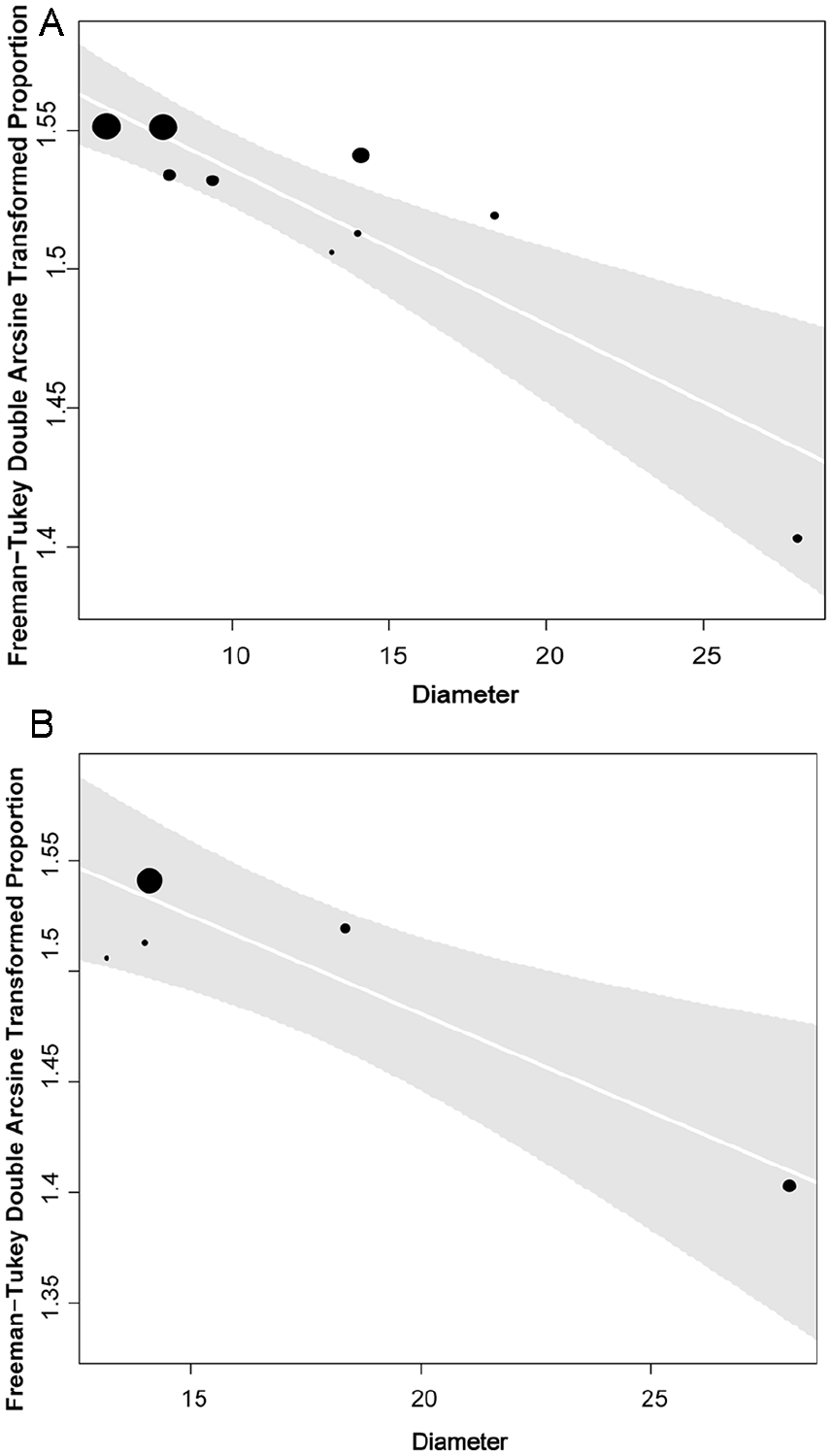
No association was identified between occlusion, the type of device used, and the length of follow-up for both the > 12 mm (β = 0.016, p = 0.75, R2 = 0.00%), (β = 0.0014, p = 0.89, R2 = 0.0%), and the whole datasets (β = 0.061, p = 0.22, R2 = 11.11%), (β = 0.01, p = 0.47, R2 = 0.0%). Meta-regression revealed a negative association between GSV diameter and occlusion (β = −0.0086, p < 0.01, R2 = 93.14%), an outcome which extended beyond the threshold of 12 mm (β = −0.0073, p = 0.04, R2 = 100%) (Figures 3A and 3B).
Meta-regression analysis displayed an association between technical success and GSV diameter for both the whole dataset (β = −0.0056, p < 0.01, R2 = 100%) and the > 12 mm groups (β = −0.0089, p = 0.016, R2 = 100%). No association was identified between technical success, the device used, and follow up duration for neither the whole dataset (β = 0.058, p = 0.06, R2 = 100%), (β = 0.0005, p = 0.94, R2 = 0%) nor the > 12 mm group (β = 0.01, p = 0.67, R2 = 100%), (β = −0.0045, p = 0.73, R2 = 0%) (Figures 4A and 4B).
We did not identify any correlation between DVT occurrence, the device used, GSV diameter, and follow-up duration for both the whole dataset (β = 0.32, p = 0.43, R2 = 100%), (β = −0.0002, p = 0.95, R2 = 71.27%), (β = 0.037, p = 0.61, R2 = 69.10%) and the > 12 mm groups (β = 0.11, p = 0.10, R2 = 100%), (β = −0.015, p = 0.18, R2 = 100%), (β = 0.01, p = 0.55, R2 = 0%).
During regression analysis of the whole dataset, we identified a positive association between EHIT occurrence and the diameter of the GSV (β = 0.0055, p = 0.02, R2 = 100%), a result which failed to reach statistical significance for the > 12 mm group (β = 0.0025, p = 0.38, R2 = 0%). Furthermore, our analysis displayed nonstatistically significant relationships between EHIT and the devices used for neither the whole dataset (β = −0.082, p = 0.23, R2 = 61.16%) nor the > 12 mm group (β = −0.036, p = 0.35, R2 = 0%). All included studies reported on a mean follow-up of 12 months (Figure 5).
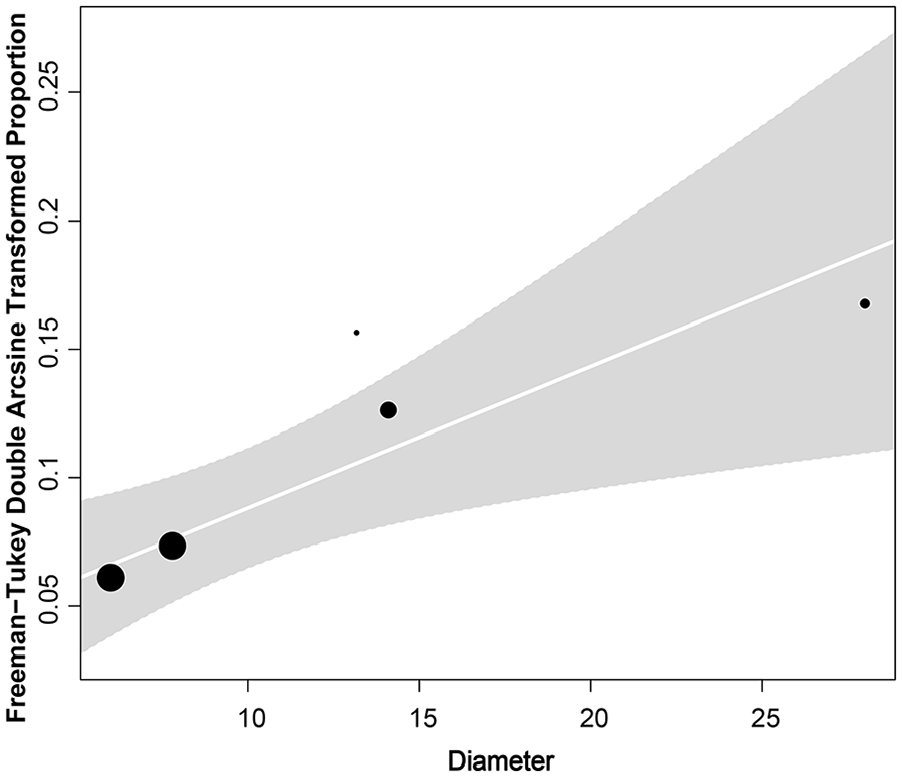
Meta-regression association of vein diameter and EHIT (whole dataset).
Discussion
In our analysis, we have presented excellent occlusion and technical success estimates for GSV > 12 mm in diameter. The EVTA of GSV < 12 mm increased the odds for occlusion by almost 80% compared to veins > 12 mm. Meta-regression analysis revealed the negative effect of GSV diameter on occlusion, technical success, and EHIT outcomes, results which extended beyond the cut-off of 12 mm for the former two endpoints.
Although many published studies have identified GSV diameters of various thresholds (ranging from 6 mm to 10 mm) as a significant predictor of recanalization, these reports carry limitations, such as the inclusion of < 1000 nm EVLA devices and the limited number of GSV over 12 mm in diameter. Namely, the study by van der Velden et al. including patients with a median GSV diameter of 6 mm identified diameter as a significant predictor for recanalization. Furthermore, and though the authors did not perform a subgroup analysis, 87.7% of the reported recanalization events concerning EVLA involved the use of < 1000 nm devices. 18 Equivalent findings were also displayed by Bissacco et al. and Zollmann et al. in their studies investigating EVTA in treatment of GSV with a mean diameter of 6 mm.19,20 Paradoxically, whereas Bissacco et al. identified GSV of diameters > 6 mm as a critical prognosticator for recanalization, they were unsuccessful in displaying similar outcomes for GSV exceeding 8 mm in diameter.
This analysis, in which we have summarized EVTA outcomes in the treatment of GSV with a minimum diameter of 12 mm, is in agreement with the above-mentioned studies. Moreover, meta-regression outcomes displaying the extension of the association between diameter and occlusion beyond the cut-off of 12 mm suggest the potentially arbitrary nature of the 12 mm threshold. Finally, despite failing to identify relevant research, the results of our analysis regarding the association of GSV and technical outcomes are only rational, given the relationship between GSV diameter and occlusion.
GSV aneurysms are defined as focal dilations of the GSV that exceed at least three times the average diameter of the vein or a GSV diameter > 20 mm. In our review, none of the included studies acknowledged the presence of GSV aneurysms or provided specific treatment precautions for such cases. 21 However, in two studies, the recorded mean GSV diameter exceeded 20 mm.14,15 Although these studies showcased favorable treatment outcomes, it is essential to exercise caution when drawing indirect inferences about the management of GSV aneurysms based solely on such minimal information. Given the need for a comprehensive understanding and guidance in the field of GSV aneurysmal disease, further research is paramount.
The standardized tumescent anesthesia protocol is comprised of 445 mL crystalloid solution along with 50 mL of 1% lidocaine, 1:100,000 epinephrine, and 5 mL of 8.4% sodium bicarbonate. 22 Tumescent anesthesia in the setting of EVTA has multiple purposes extending past its anesthetic and heat dissipation properties. Hydrodissection, which is accomplished through perivenous space infiltration, serves both in compression of the venous trunk and in protecting important surrounding structures such as the saphenous and sural nerves. The addition of epinephrine enhances vasoconstriction and delays the absorption of lidocaine, ultimately lengthening anesthetic duration, whereas sodium bicarbonate achieves improved pain outcomes throughout the alkalization of the solution (epinephrine and lidocaine exhibit highly acidic properties).23,24
Five out of the seven included studies provided data regarding the composition of tumescent anesthesia, with three studies employing the standardized format and two studies nonstandardized variations. Although the potential influence of the qualitative characteristics of tumescent anesthesia on the EVTA of large GSV has not yet been investigated, in reality only epinephrine could plausibly affect ablation outcomes owing to its innate vasoconstrictive properties. Resnick et al., in their study investigating the clinical success of EVLA performed under epinephrine-containing tumescent anesthesia versus an epinephrine-depleted solution, failed to display statistically significant differences between the two groups. 25 Although the macrovascular vasoconstrictive effect of epinephrine on the GSV should be further investigated, avoiding cardiovascular side effects warrants prudence not to exceed the maximum recommended dose of 0.07 mg/kg. 22
Despite the established standardized protocol regarding the composition of tumescent anesthesia, there has been no guidance addressing the issue of anesthetic volume. The two rules of thumb currently adopted by vascular surgeons, including the injection of perivenous anesthesia with the target of creating a 10 mm perivenous space, or the 10 mL/1 cm approach, do not account for the diameter of the GSV. 26 In our review, five studies reported on volumetric characteristics, with four authors using the 10 mL/1 cm approach and one aiming to create a perivenous space between 1.5 cm and 2 cm. These variations in the application of tumescent anesthesia observed throughout the literature could serve in interpreting the mosaic of reports ranging from studies displaying no association between GSV diameter and occlusion to studies identifying various diameters as critical thresholds. 27 Finally, whether the development of a diameter-oriented anesthetic protocol or the expansion of training programs into the efficacious application of such a protocol will sufficiently aid in improving GSV outcomes for larger veins is a subject of additional research.
DVT and EHIT differ significantly in terms of severity, natural history, and appearance on sonographic examination. 28 DVT is defined as thrombus formation into the deep venous system whereas EHIT is classified into four stages (I to IV) and refers to thrombus propagation from the GSV into the femoral vein as a result of coagulation-induced thrombosis. The course of EHIT regarding the foremost classes of the condition is considered benign, with guidelines reserving the initiation of anticoagulation therapy for high-risk patients of EHIT class II and all patients of EHIT classes III and IV. 6 According to the literature, the overall reported EHIT prevalence is 1.27% (regarding the clinically relevant stages of the condition) with GSV diameter, male sex, history of venous thromboembolism, and concomitant phlebectomy being identified among potential risk factors.2,29 Concerning GSV diameter, several studies have identified various cut-offs ranging from 7.5 mm to 10 mm as predictors for EHIT occurrence.30–32 In our review, though the regression analysis of the whole dataset revealed a statistically significant association between diameter and EHIT occurrence, both the comparative analysis (GSV > 12 mm versus GSV < 12 mm) and regression analysis including only GSV > 12 mm failed to reach statistical significance. These results both add to the growing body of evidence suggesting the negative effect of GSV diameter on EHIT and indicate a cut-off point well under the threshold of 12 mm. Ultimately, if one takes into account the minimal reported DVT estimates following EVTA of under 1%, the nonidentification of GSV diameter as a risk factor for the development of DVT is rather expected. 29
Paresthesia is one of the most commonly anticipated complications following EVTA. The reported paresthesia outcomes vary widely and range from 3% to 12%, though they heavily depend on multiple parameters, such as the treatment modality, or the specific device parameters.33,34 Given the aforementioned estimates, our pooled postoperative paresthesia outcome regarding large GSV of 2.13% is acceptable. Additionally, the comparative analysis failed to display statistically significant differences between large and small GSV. Similarly, ecchymosis-hematoma and pigmentation-induration composite endpoints depicted satisfactory estimates of 3.31% and 6%, respectively, with nonstatistically significant differences between large and small GSV.
Nonthermal nontumescent (NTNT) ablation modalities are a group of novel endovenous treatments currently including mechanochemical (MOCA) and cyanoacrylate (CAC) ablation. Despite the paucity of data regarding the correlation between GSV diameter and NTNT ablation results, there is increasing evidence suggesting the negative influence of GSV vein diameter on occlusion for both MOCA and CAC.35,36 Namely, findings from a recently published network meta-analysis comparing EVTA and NTNT ablation displayed a trend towards the superiority of EVTA (regarding occlusion success) when ablating larger GSV. 37
Considering the extensive use of endothermal ablation techniques and their well-known advantages over traditional surgery, the scarcity of high-quality data regarding the efficacy of EVTA in ablating large GSVs, came as a surprise to the authors of this review. Since the first publication on this subject dating back to the early 2000s, in a span of 20 years our research has failed to identify any randomized controlled trials investigating the impact of large GSVs (even GSVs over 10 mm) on EVTA outcomes. As newer thermal and NTNT ablation modalities become more and more accessible, providing physicians with a plethora of options in the treatment of lower-limb venous disease, the identification of characteristics such as the ability or not of a certain modality to ablate larger veins becomes progressively substantial.
Limitations
A major limitation of our analysis, is the inclusion of nonrandomized studies, most of which were retrospective in nature, leaving our review vulnerable to selection bias. Moreover, and despite avoiding the use of the DerSimonian–Laird heterogeneity variance estimator and implementation of the Hartung–Knapp method, we could not eliminate the limitations posed by the low number of included studies, which are known to threaten the statistical power and reliability of both meta-analytic and meta-regression outcomes.9,38 Additionally, we have failed to identify studies including microwave ablation in the treatment of large GSV and thus our review does not include the whole spectrum of available endothermal ablation modalities. Finally, the heterogeneity of the inclusion criteria could be a potential source of bias.
Conclusions
We report excellent occlusion and technical success results for the EVTA of GSV > 12 mm. Nonetheless, meta-regression analysis illustrated the unfavorable impact of GSV diameter on occlusion, technical success, and EHIT outcomes. Although the negative effect of diameter on occlusion and technical success extended beyond the threshold of 12 mm, this was not true for the EHIT occurrence, suggesting the lack of a definitive GSV diameter cut-off. Potential device and/or tumescent anesthesia parametrization in a diameter-oriented manner, irrespective of cut-off points, could further enhance outcomes.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231183997 – Supplemental material for Endovenous thermal ablation in the treatment of large great saphenous veins of diameters > 12 mm: A systematic review meta-analysis and meta-regression
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231183997 for Endovenous thermal ablation in the treatment of large great saphenous veins of diameters > 12 mm: A systematic review meta-analysis and meta-regression by Vangelis Bontinis, Alkis Bontinis, Andreas Koutsoumpelis, Anastasios Potouridis, Argirios Giannopoulos, Vasileios Rafailidis, Angeliki Chorti and Kiriakos Ktenidis in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X231183997 – Supplemental material for Endovenous thermal ablation in the treatment of large great saphenous veins of diameters > 12 mm: A systematic review meta-analysis and meta-regression
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X231183997 for Endovenous thermal ablation in the treatment of large great saphenous veins of diameters > 12 mm: A systematic review meta-analysis and meta-regression by Vangelis Bontinis, Alkis Bontinis, Andreas Koutsoumpelis, Anastasios Potouridis, Argirios Giannopoulos, Vasileios Rafailidis, Angeliki Chorti and Kiriakos Ktenidis in Vascular Medicine
Supplemental Material
sj-pdf-3-vmj-10.1177_1358863X231183997 – Supplemental material for Endovenous thermal ablation in the treatment of large great saphenous veins of diameters > 12 mm: A systematic review meta-analysis and meta-regression
Supplemental material, sj-pdf-3-vmj-10.1177_1358863X231183997 for Endovenous thermal ablation in the treatment of large great saphenous veins of diameters > 12 mm: A systematic review meta-analysis and meta-regression by Vangelis Bontinis, Alkis Bontinis, Andreas Koutsoumpelis, Anastasios Potouridis, Argirios Giannopoulos, Vasileios Rafailidis, Angeliki Chorti and Kiriakos Ktenidis in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
