Abstract
Introduction:
Although favorable results of fluoropolymer-based drug-eluting stent (FP-DES) treatment for femoropopliteal lesions have been reported, it is unclear whether minimal lumen area (MLA) after FP-DES implantation affects clinical outcomes. This study aimed to reveal the association between intravascular ultrasound (IVUS)-evaluated MLA and the 1-year risk of restenosis and aneurysmal degeneration after FP-DES implantation for femoropopliteal lesions.
Methods:
A subanalysis of the CAPSICUM (Contemporary outcomes After Paclitaxel-eluting peripheral Stent implantation for symptomatic lower limb IsChemia with sUperficial feMoral or proximal popliteal lesion) study analyzed 718 limbs in 686 patients with available IVUS-evaluated MLA data. The association of MLA with the 1-year risk of restenosis and aneurysmal degeneration was analyzed using the generalized propensity score method.
Results:
The 1-year incidence rate of restenosis was estimated to be 8.8% (95% CI, 6.1% to 12.5%) for the upper quartile of MLA (21.1 mm2) versus 14.3% (95% CI, 10.7% to 18.7%) for the lower quartile of MLA (15.2 mm2), with an odds ratio of 0.58 (95% CI, 0.36 to 0.93; p = 0.024), whereas the 1-year incidence rate of aneurysmal degeneration was 23.8% (95% CI, 19.5% to 28.8%) for the upper quartile versus 16.8% (95% CI, 12.6% to 22.0%) for the lower quartile, with an odds ratio of 1.55 (95% CI, 1.04 to 2.32; p = 0.031).
Conclusion:
A large MLA after FP-DES implantation for femoropopliteal lesions was associated with decreased restenosis risk but increased aneurysmal degeneration risk. These findings suggest that MLA is a valuable predictor of clinical outcomes.
Keywords
See commentary: Monteleone P, Farr A, Bunte MC. Looking into the future: Endovascular intervention with intravascular ultrasound. Vasc Med 2023; 28: 422–424
Introduction
Symptomatic lower-extremity artery disease often comes from femoropopliteal arterial lesions. Newer femoropopliteal endovascular treatments including drug-eluting therapy dramatically reduce the rate of restenosis and target lesion revascularization. Accordingly, the latest guidelines recommend endovascular treatment (EVT) as the first-line treatment for short and intermediate femoropopliteal lesions, though EVT could be considered for long, complex lesions in patients with severe comorbidities.1–3 Clinical studies have reported that a fluoropolymer-based drug-eluting stent (FP-DES) (Eluvia; Boston Scientific, Marlborough, MA, USA) has durable and consistent patency,4–8 and is now widely used in everyday femoropopliteal practice.
The literature suggested that polymer-free drug-coated stent (PF-DCS) (Zilver PTX; Cook Corporation, Bloomington, IN, USA) performance was suboptimal in cases with a small minimal lumen area (MLA). 9 However, whether a small MLA would similarly affect the clinical outcomes of FP-DES remain unknown. Although FP-DES was reported to have higher patency rates than PF-DCS,4–7 the incidence of aneurysmal degeneration after FP-DES implantation has recently raised clinical concern.5,8 The current study aimed to reveal the association between intravascular ultrasound (IVUS)-evaluated MLA and clinical outcomes, including 1-year risk of restenosis and aneurysmal degeneration, after EVT with FP-DES implantation for femoropopliteal lesions in patients with symptomatic lower-extremity artery disease.
Methods
We retrospectively analyzed the database of the Contemporary outcomes After Paclitaxel-eluting peripheral Stent implantation for symptomatic lower limb IsChemia with sUperficial feMoral or proximal popliteal lesion (CAPSICUM) study. 8 The CAPSICUM study was a prospective, multicenter, observational study that registered 1204 limbs of 1097 adult patients (age ⩾ 20 years) who underwent FP-DES implantation for symptomatic atherosclerotic femoropopliteal artery disease between February 2019 and June 2020 at 60 cardiovascular centers across Japan. The list of participating centers is provided in online Supplemental Appendix A. All participants were asked to visit their centers 12 ± 2 months after FP-DES implantation for evaluation of restenosis occurrence and presence of aneurysmal degeneration, regardless of ischemic symptoms. Of the 1204 registered limbs in 1097 patients, data on IVUS-evaluated MLA were available for 746 limbs. Owing to the risk of over-expansion of the stent, 28 limbs with an MLA value larger than the expected luminal area of 6-mm FP-DES were excluded. The current study evaluated the association between IVUS-evaluated MLA and clinical outcomes in 718 limbs.
The study was conducted in accordance with the Declaration of Helsinki and was approved by the ethics committees of the participating centers. Informed consent was obtained from the participants or, if not possible, from their families.
Measures and definitions
Outcome measures were 1-year restenosis and aneurysmal degeneration. Restenosis was defined as a peak systolic velocity ratio > 2.4 on duplex ultrasonography 10 or > 50% diameter stenosis or occlusion of follow-up angiography. 11 Aneurysmal degeneration was defined as a dark halo around the stenting site on Doppler ultrasonography. 12 Calcium circumferential grade was determined by IVUS according to the presence of the most extensively distributed calcified area in the four quadrants of 0−90°, 91−180°, 181−270°, and 271°−360°. Reference vessel diameter was assessed using fluoroscopy at a healthy distal site without atherosclerotic plaque. An IVUS-evaluated external elastic membrane (EEM) cross-sectional area was measured at the distal reference segment. The FP-DES size was selected to match the distal vessel diameter as closely as possible to achieve a 1:1 stent-to-EEM ratio. After FP-DES treatment, IVUS evaluated the in-stent area of the cross-section in the entire implanted stent using the automatic pull-back system. An IVUS image slice with MLA in the cross-section with the smallest lumen area was selected for analysis. The MLA was measured by tracing each strut’s center, not the inner or outer edge.
A team of certified specialists from each participating institution, recognized by the relevant academic society in Japan, conducted the analysis of all parameters. These evaluations were performed in accordance with the current clinical expert consensus document for EVT imaging, 13 as shown in Figure 1.
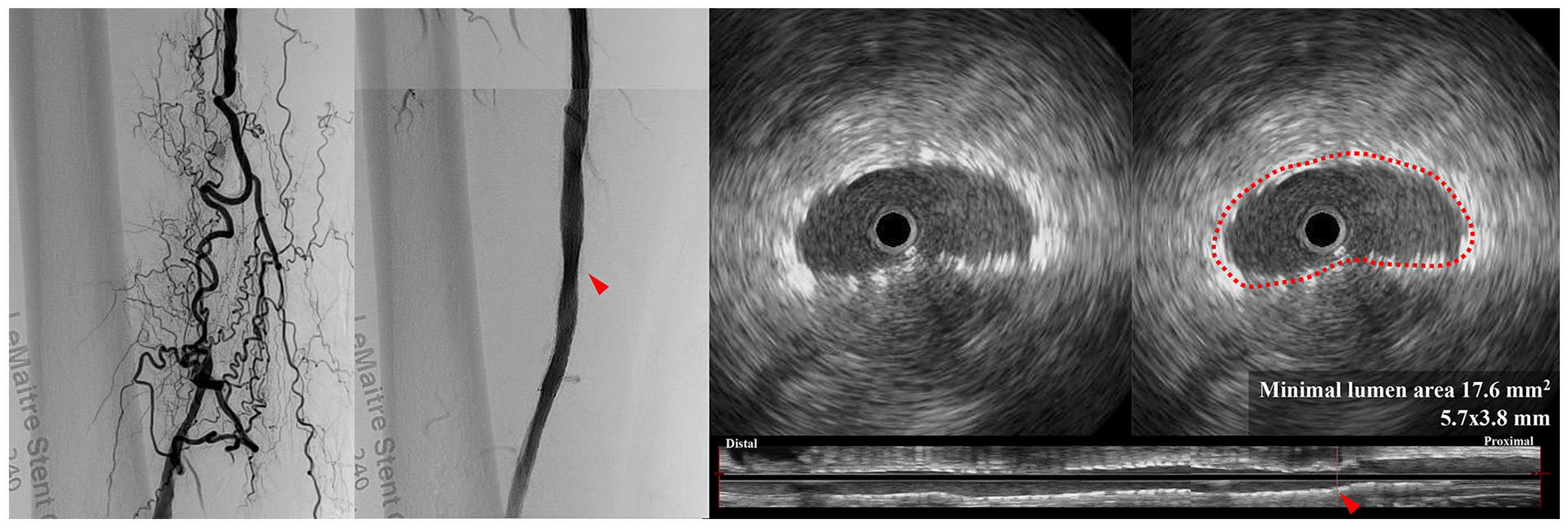
IVUS measurement process for MLA (arrowhead). (1) After FP-DES treatment, IVUS assesses the entire implanted stent’s in-stent cross-sectional area using an automatic pull-back system. (2) The IVUS image slice with the smallest lumen area, representing the MLA, is selected for analysis. (3) To measure the MLA, the center of each strut is traced (dotted outline), rather than the inner or outer edges.
Statistical analysis
Data on baseline characteristics are presented as mean ± SD values for continuous variables and frequency (percentage) values for categorical variables, if not otherwise mentioned. Statistical significance was considered for p < 0.05, and 95% CIs were reported where appropriate. The association of MLA with the 1-year risk of restenosis and aneurysmal degeneration was analyzed using the generalized propensity score (GPS) method, an extension of propensity score methods to analyze continuous exposures. 14 To calculate GPS, we first developed the ordinary least square (OLS) regression model of continuous MLA exposure based on baseline covariates. Baseline covariates entered as explanatory variables in the OLS regression model were sex, age, height, body weight, body mass index, smoking, diabetes mellitus, renal function, cerebrovascular disease, coronary artery disease, statin use, chronic limb-threatening ischemia, history of revascularization, presence of a popliteal lesion, reference vessel diameter, lesion length, chronic total occlusion (CTO), calcification, spot stenting, IVUS-evaluated subintimal wire passage, IVUS-evaluated reference EEM area, and IVUS-evaluated calcification. Using the OLS regression model, we subsequently calculated GPS as the conditional probability density function of continuous MLA exposure for the observed baseline covariates. The balancing property of GPS was checked using the blocking method. 14 In brief, we divided the study population into four subgroups according to the quartiles of continuous MLA exposure. We thereafter compared baseline covariates between one arbitrary subgroup and the others using t-statistics with GPS stratification evaluated at the average of the exposure stratum. We finally estimated the dose–response function of continuous MLA exposure in relation to risk of restenosis and aneurysmal degeneration. For the estimation, we regressed restenosis risk on continuous MLA exposure and the estimated GPS (evaluated based on the observed MLA value) using the logistic regression model in which MLA, GPS, and their interaction term were entered as explanatory variables. Based on the developed regression model, we estimated the risk of restenosis and aneurysmal degeneration corresponding to an arbitrary value of continuous MLA exposure and GPS evaluated based on the MLA value of each individual. The dose–response function was then obtained by averaging the estimates in the overall study sample. 14 The dose–response function demonstrates the average response that would occur in the population if ‘everyone’ had an arbitrary MLA. Since the dose–response function was expected to be nonlinear, we graphically demonstrated the function along with the derivative of the function. We also demonstrated the difference in restenosis risk between the quartiles of the observed MLA value.
We supplementarily performed two sensitivity analyses. First, we extracted the subset of patients who had bilateral limbs treated and performed an intra-individual comparison in clinical outcomes between one limb with the larger MLA and the other limb with the smaller MLA. Second, we performed multiple logistic regression analyses and calculated the odds ratios of MLA for outcomes with adjustment for covariates that demonstrated a significant association with MLA in the OLS regression model. However, the sample size and the number of observed events in these sensitivity analyses were too small that p-values derived from the statistical tests would make little clinical sense. Therefore, a numerical assessment was performed.
Missing data were addressed using multiple imputations by the chained equations method. In this procedure, we generated 10 imputed datasets and combined the analytic results according to Rubin’s rule. All statistical analyses were performed using R Statistical Software version 4.1.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Baseline characteristics are summarized in Table 1. Mean age was 75 ± 9 years, and 68.7% were men. Prevalence rates of diabetes mellitus, dialysis, and chronic limb-threatening ischemia were 57.3%, 26.8%, and 33.6%, respectively. Mean reference vessel diameter and lesion length were 5.2 ± 0.9 mm and 19.3 ± 9.8 cm, respectively. Proportions of CTO and bilateral wall calcification were 56.8% and 40.9%, respectively. IVUS evaluation revealed a subintimal wire passage in 10.7% of cases and a reference EEM area of 27.9 ± 12.1 mm2. Regarding the severity of calcification revealed on IVUS assessment, no calcification, ⩽ 180° calcification, and > 180° calcification were found in 20.7%, 39.2%, and 40.0% of cases, respectively. Mean MLA after FP-DES implantation was 18.4 ± 4.3 mm2. Data on 1-year restenosis and aneurysmal degeneration were available for 586 (81.6%) and 456 (63.5%) limbs, respectively. The crude 1-year proportions of restenosis and aneurysmal degeneration by the quartiles of MLA are illustrated in Figure 2.
Baseline characteristics of the overall population and by quartiles of minimal lumen area (MLA).
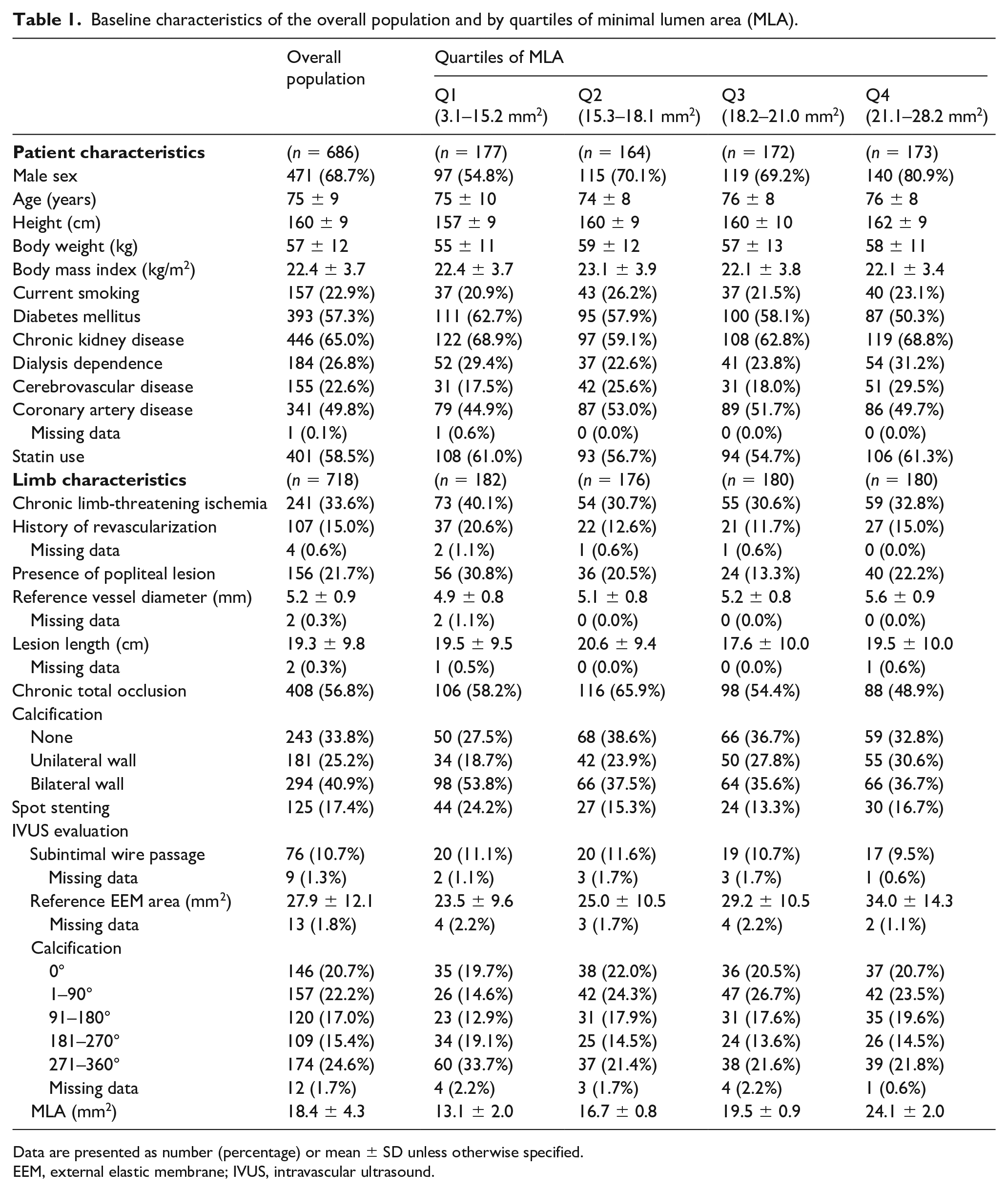
Data are presented as number (percentage) or mean ± SD unless otherwise specified.
EEM, external elastic membrane; IVUS, intravascular ultrasound.
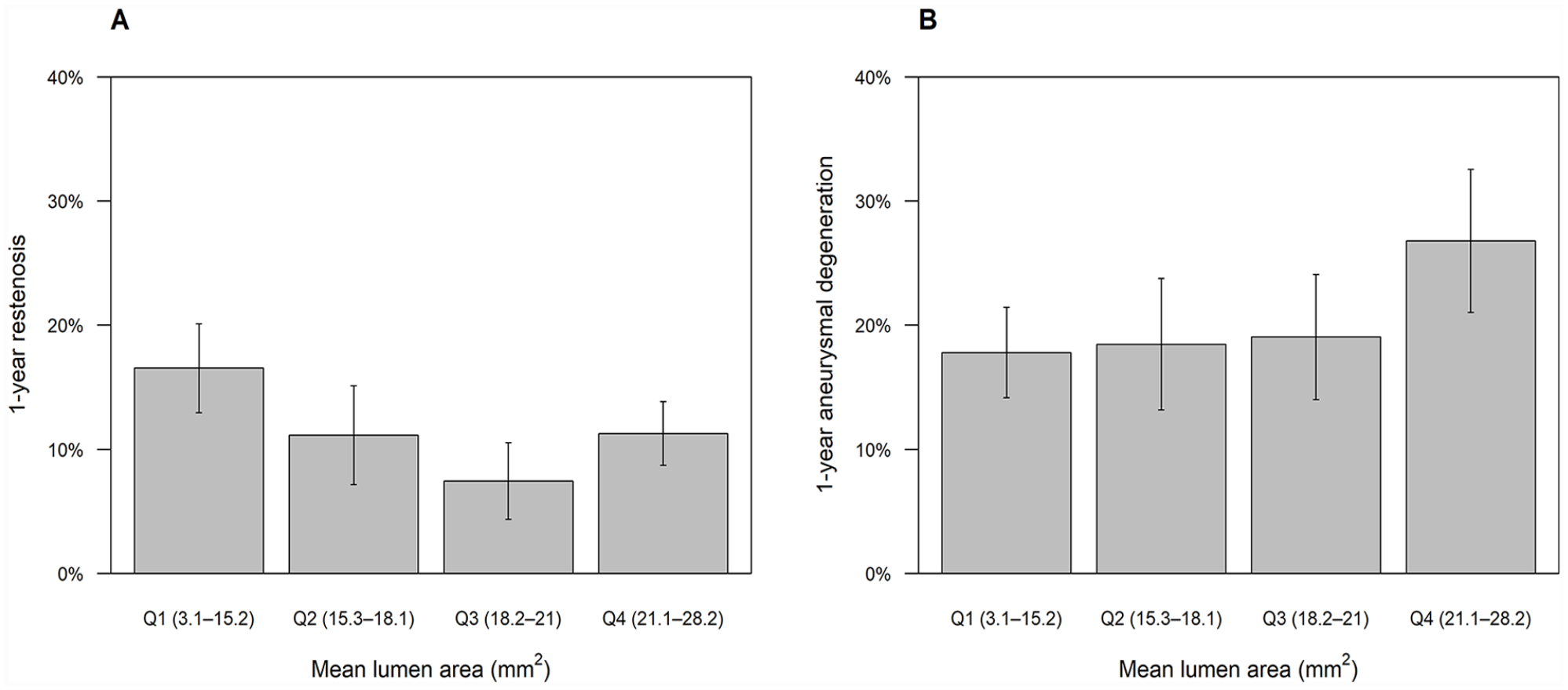
Unadjusted 1-year proportion of restenosis (
The OLS regression analysis demonstrated that baseline characteristics independently associated with MLA were diabetes mellitus (β = −0.84, p = 0.007), reference vessel diameter (β = 0.99 per 1-mm increase; p < 0.001), CTO (β = −0.76, p = 0.032), IVUS-evaluated reference EEM area (β = 0.09 per 1-mm2 increase, p < 0.001), and IVUS-evaluated calcification (β = −0.33 per category, p = 0.048) (Table 2). The adjusted R2 of the multivariate OLS regression analysis was 0.19 (Figure 3). Based on the multivariate OLS regression results, we developed the GPS. After GPS adjustment, absolute values of t-statistics were smaller than 1.96 (i.e., not significant) for all covariates (Figure 4), suggesting that the GPS adjustment balanced them well. The GPS-adjusted dose–response function for restenosis and aneurysmal degeneration is illustrated in Figures 5 and 6, respectively. In general, MLA was inversely associated with restenosis risk and positively associated with aneurysmal degeneration. The 1-year incidence rate of restenosis was estimated to be 8.8% (95% CI, 6.1% to 12.5%) for the upper quartile of MLA (21.1 mm2) versus 14.3% (95% CI, 10.7% to 18.7%) for the lower quartile of MLA (15.2 mm2), with an odds ratio of 0.58 (95% CI, 0.36 to 0.93; p = 0.024), whereas the 1-year incidence rate of aneurysmal degeneration was 23.8% (95% CI, 19.5% to 28.8%) for the upper quartile versus 16.8% (95% CI, 12.6% to 22.0%) for the lower quartile, with an odds ratio of 1.55 (95% CI, 1.04 to 2.32; p = 0.031) (Table 3).
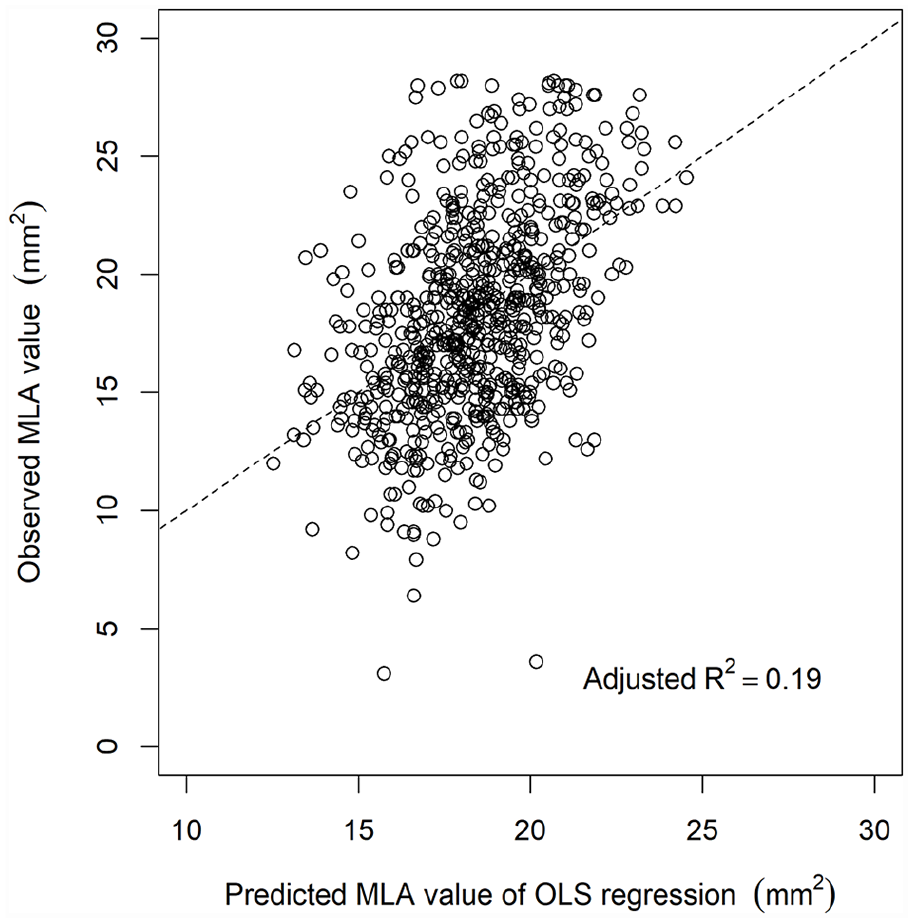
Scatter plot of the association between predicted MLA values obtained on OLS regression analysis and observed MLA values. Predicted MLA values were derived from the multivariate OLS regression analysis results shown in Table 2.
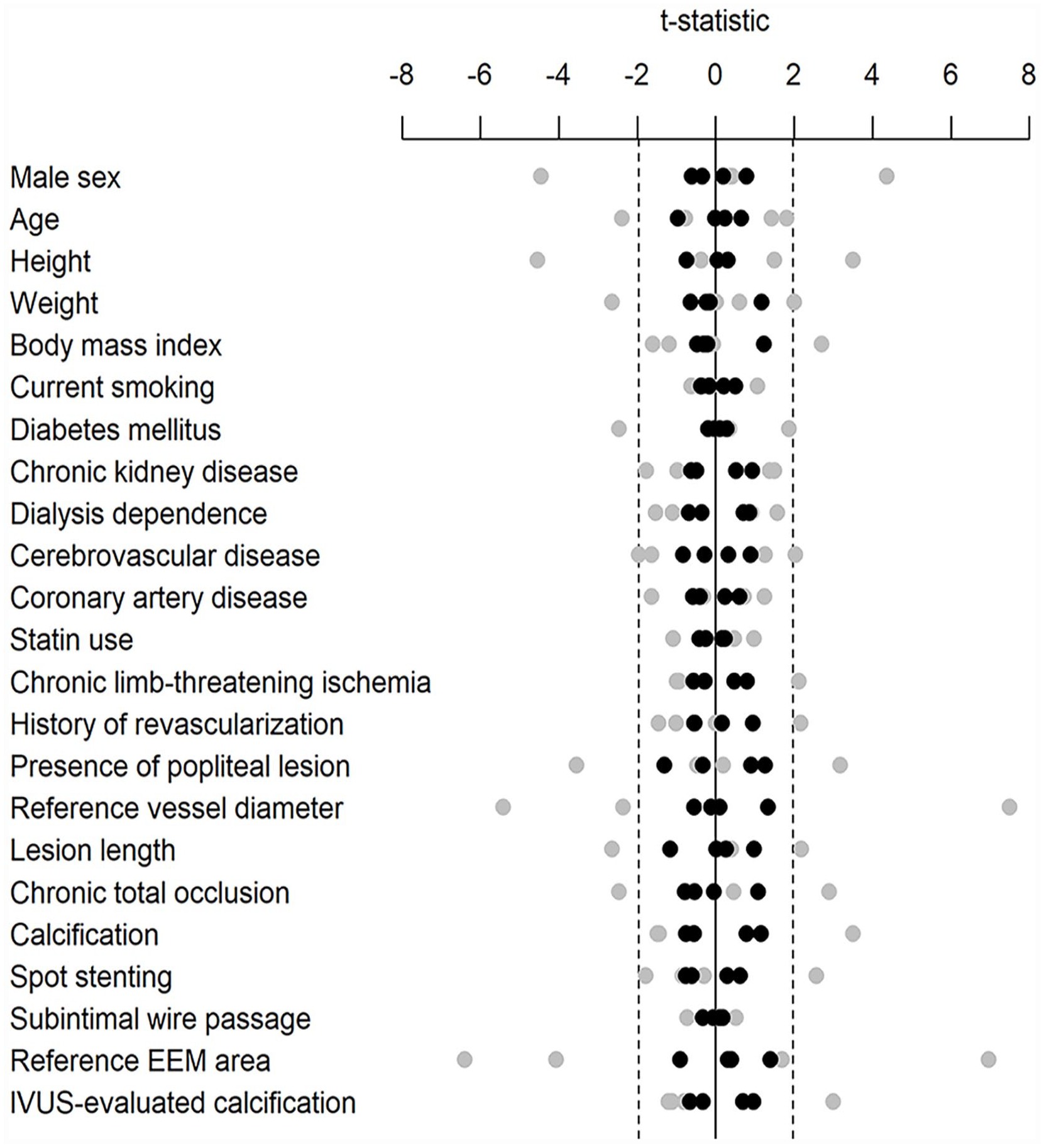
Balance of baseline covariates before (gray circles) and after (black circles) GPS adjustment.
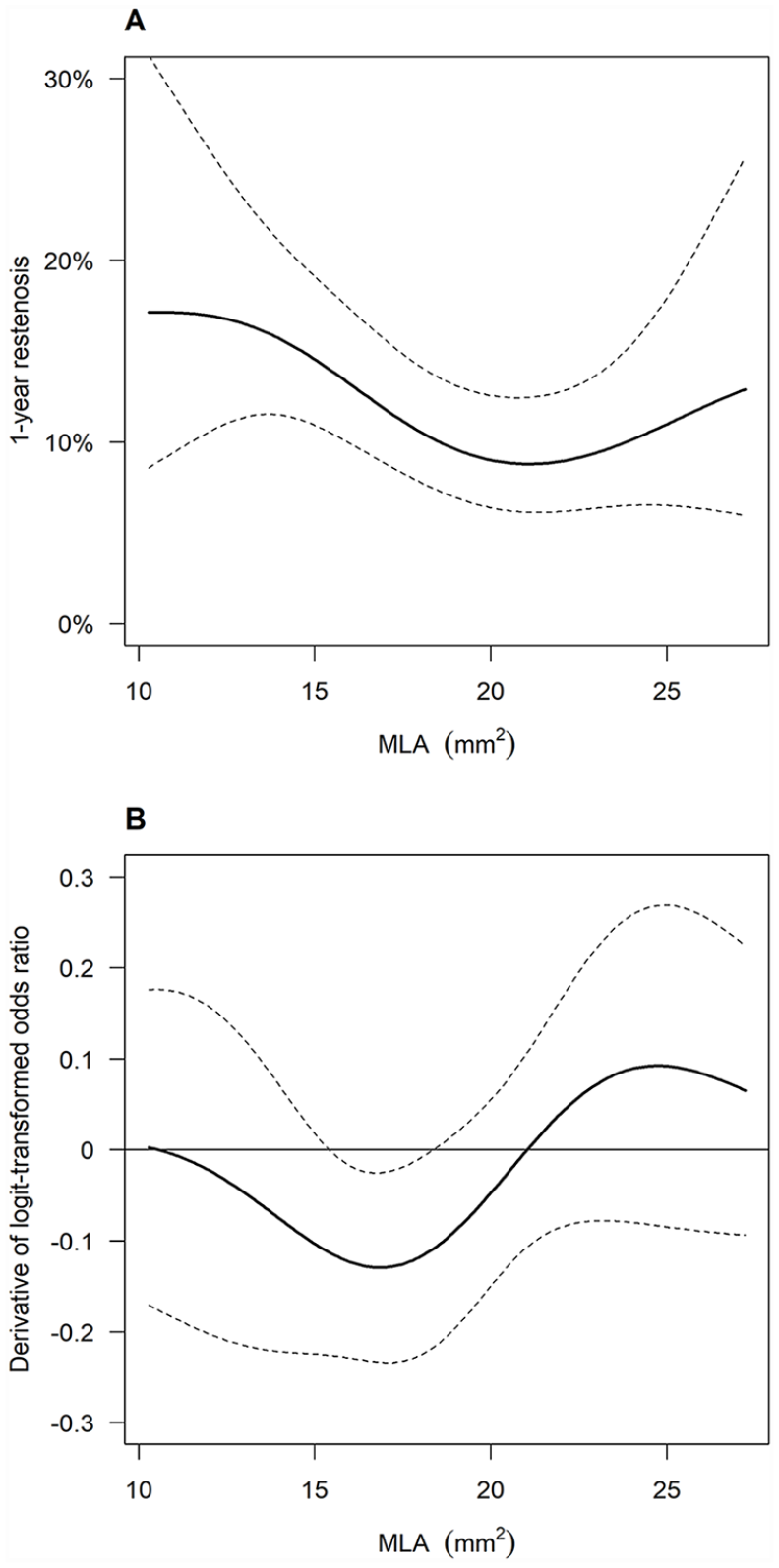
Dose–response function of MLA for restenosis risk. (
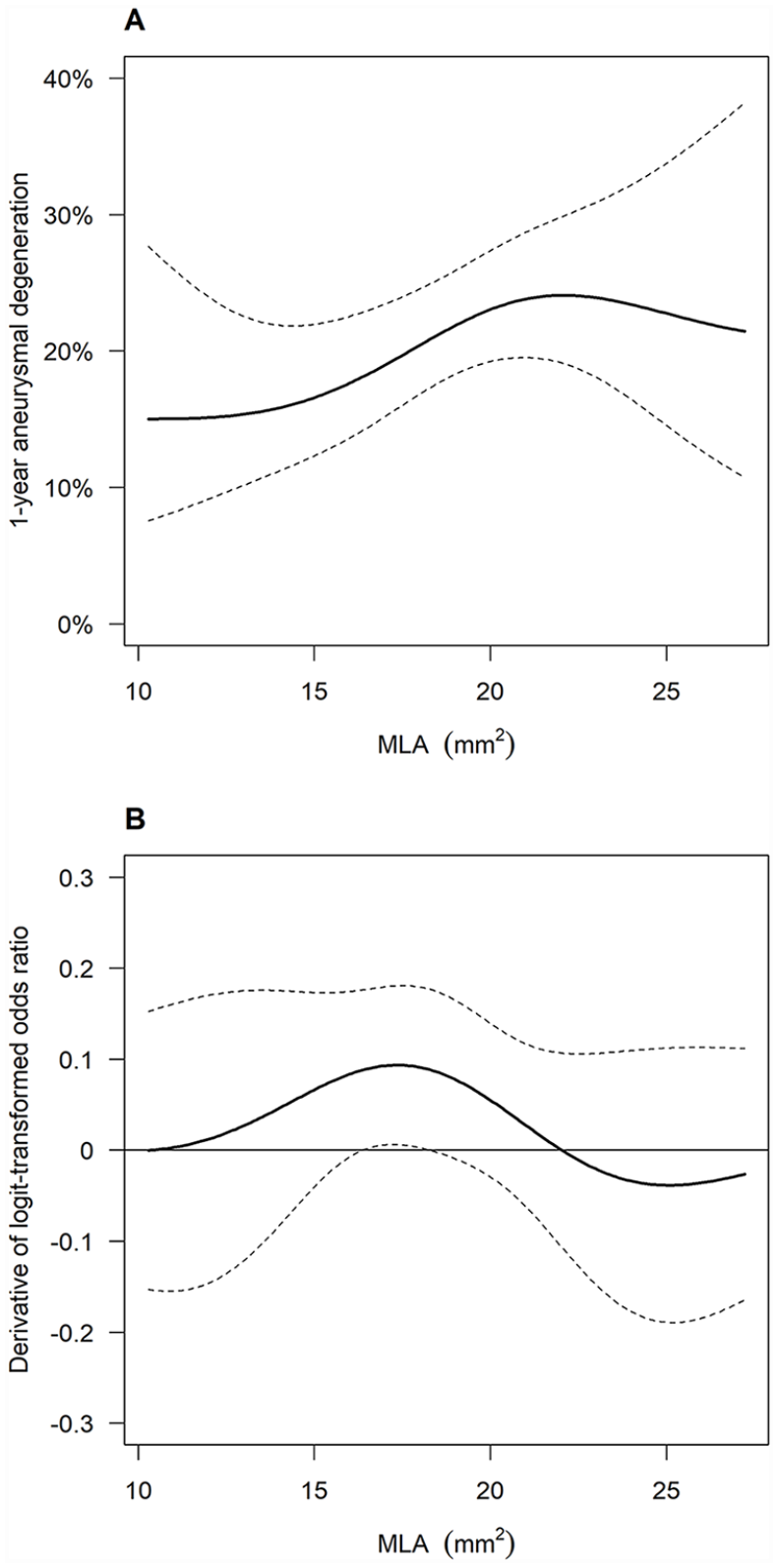
Dose–response function of MLA for aneurysmal degeneration risk. (
Ordinary least square regression analysis of minimal lumen area based on baseline covariates.
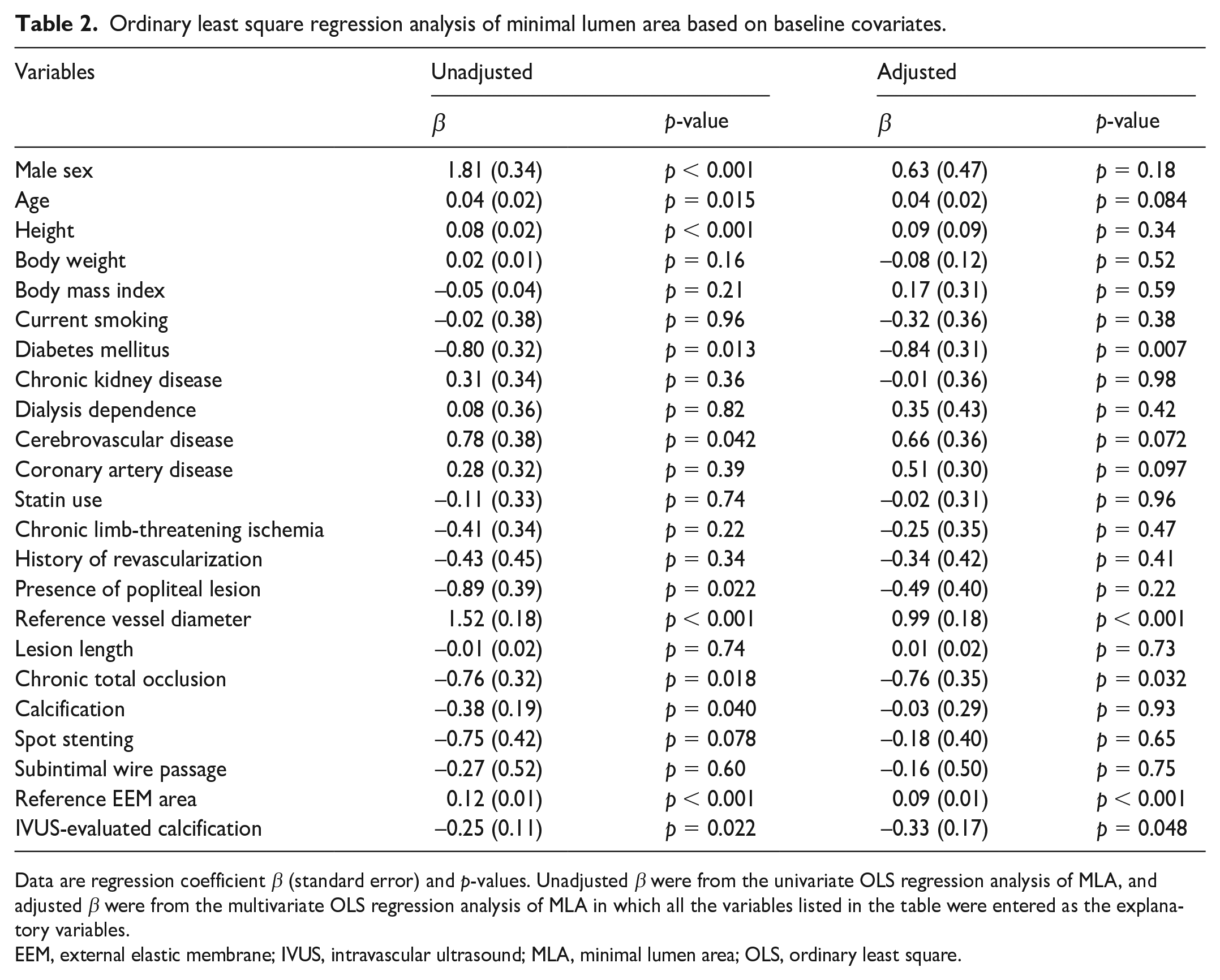
Data are regression coefficient β (standard error) and p-values. Unadjusted β were from the univariate OLS regression analysis of MLA, and adjusted β were from the multivariate OLS regression analysis of MLA in which all the variables listed in the table were entered as the explanatory variables.
EEM, external elastic membrane; IVUS, intravascular ultrasound; MLA, minimal lumen area; OLS, ordinary least square.
Restenosis and aneurysmal degeneration risks corresponding to tertiles of minimal lumen area.

Data are GPS-adjusted estimates [95% CIs].
GPS, generalized propensity score.
Sensitivity analysis
First, we extracted the subset of patients who had bilateral limbs treated (n = 32) and performed an intra-individual comparison. The mean MLA was 20.1 ± 3.4 mm2 in the limbs with the larger MLA and 16.2 ± 3.2 mm2 in those with the smaller MLA. Those with the larger MLA had a numerically lower 1-year proportion of restenosis (9.7% [0.0% to 21.0%] vs 11.6% [0.1% to 23.1%]) and a numerically higher 1-year proportion of aneurysmal degeneration (14.7% [0.0% to 30.5%] vs 13.8% [0.6% to 26.9%]), although the difference did not reach statistical significance (both p > 0.99). Second, we calculated adjusted odds ratios of MLA for outcomes using multiple logistic regression models. Consequently, MLA had a numerically inverse association with restenosis and a numerically positive association with aneurysmal degeneration, although the associations did not reach statistical significance; adjusted odds ratios of MLA per 1-SD increase were 0.87 (95% CI, 0.66 to 1.44; p = 0.30) for restenosis and 1.16 (0.92 to 1.47; p = 0.20) for aneurysmal degeneration, respectively. The numerical results in these sensitivity analyses were in line with the GPS-based findings.
Discussion
The current study evaluated the association between MLA and clinical outcomes in patients treated with FP-DES implantation. After adjusting baseline characteristics using the GPS method, a large MLA after FP-DES implantation was associated with decreased risk of restenosis and increased risk of aneurysmal degeneration.
Previous reports showed that MLA after use of bare nitinol stents, PF-DCS, or drug-coated balloons (DCB) was significantly associated with the patency rate.9,15,16 The findings of the present study consisted of those previous reports. MLA would still be a key factor modulating device performance, even when lesions are treated with FP-DES, providing superior patency rates among existing devices. A large MLA, indicating the relatively large stent lumen, would reduce restenosis risk, even if a substantial neointimal proliferation occurs as a response to stent implantation. Therefore, achieving a large MLA would maximize FP-DES performance and contribute to a lower restenosis rate. Additionally, MLA is expected to be largely dependent on the vessel size. Furthermore, the presence of calcification is a major obstacle to vessel dilation, 17 and CTO has been reported as an independent factor associated with MLA. 18 It would be no surprise that reference vessel diameter, CTO, and IVUS-evaluated reference EEM area, as well as IVUS-evaluated calcification, were positively associated with MLA during the OLS regression analysis. The OLS regression analysis also revealed that diabetes mellitus was associated with MLA, which might be more frequent arterial calcification in patients with diabetes mellitus.19,20
The present study also revealed that a large MLA was significantly associated with increased risk of aneurysmal degeneration. A larger MLA could be gained by sufficient vessel preparation using a large balloon. However, it would potentially increase the risk of overextending and thus damaging the target vessel. After FP-DES treatment, the vessel was continuously exposed to paclitaxel eluted with fluoropolymer over time. Continuous exposure of the damaged vessel to paclitaxel might induce aneurysmal degeneration formation. We previously reported in the CAPSICUM study that IVUS use was significantly relevant to the occurrence of aneurysmal degeneration after FP-DES implantation.8,21 During IVUS assessment, the vessel diameter is usually determined by EEM size, whereas it is based on lumen diameter in angiography. The vessel diameter measured using IVUS is generally larger than that measured in angiography. 22 Consequently, IVUS use would be associated with the use of larger balloons, resulting in aneurysmal degeneration formation. Although it is still unclear whether aneurysmal degeneration after FP-DES implantation affects clinical events, a case of aneurysmal degeneration progression has been reported, 23 and long-term follow-up is warranted.
Study limitations
First, the present study included only Japanese patients, and clinical results may be different for other ethnicities; therefore, results should also be investigated in non-Japanese populations. Second, this study was a single-arm model and did not compare other scaffolds, especially PF-DCS. Third, a core laboratory analysis was not used for the angiography and IVUS evaluation of initial treatment and Doppler ultrasound evaluation in the follow-up period. Although this may compromise the reliability of restenosis assessment, each participating center had pivotal clinical trial experience for femoropopliteal EVT; accumulation of experiences minimized variation. Fourth, the data on stent and balloon sizes, angiographically measured luminal diameter, and the location of lesions in the superficial femoral artery were not available. Therefore, we could not assess whether the association of restenosis or aneurysmal degeneration risks with MLA would be explained by the stent/balloon size. Neither could we translate the present findings (the association between MLA and outcomes) into the association of angiographically measured luminal diameter with outcomes. Furthermore, our GPS adjustment did not include the information on the location of lesions in the superficial femoral artery. Further clinical studies are necessary to elucidate these issues. Fifth, the IVUS data collected in the present study were limited. We could not show whether MLA was superior to other indices in the risk assessment. Sixth, the present report was just a study that demonstrated the association of MLA with outcomes and was not a study that determined the cutoff point of MLA. Future studies are needed to find what threshold to aim for during EVT. Seventh, the data on aneurysmal degeneration were available only in 63.5%, which potentially led to an inherent bias. However, aneurysmal degeneration per se is seldom accompanied by any clinical symptoms and cannot be detected without a medical examination. In this sense, there would not be a relationship between the propensity of a value to be missing and its values. The missing data can be regarded as so-called ‘missing at random (MAR)’ (which statistically means that there is a systematic relationship between the propensity of missing values and the observed data, but not the missing data). It is known that the multiple imputations can, at least partially, solve the problem based on MAR. Therefore, we believe that the adaptation of the multiple imputation method in the present study could minimize the influence of the potential bias, although the potential bias could not be completely eliminated. Finally, our follow-up period was 12 months, and further studies with longer follow-up periods are necessary to confirm our results and evaluate the efficacy and safety between the IVUS-evaluated MLA and clinical outcomes.
Conclusion
The present study demonstrated that a large MLA after EVT with FP-DES implantation for femoropopliteal lesions was not only associated with a decreased risk of restenosis but also with an increased risk of aneurysmal degeneration. These findings suggest that MLA is a valuable predictor of clinical outcomes.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231182742 – Supplemental material for Impact of postprocedural minimum lumen area on clinical outcome after femoropopliteal drug-eluting stent implantation
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231182742 for Impact of postprocedural minimum lumen area on clinical outcome after femoropopliteal drug-eluting stent implantation by Takuya Haraguchi, Mitsuyoshi Takahara, Osamu Iida, Yoshimitsu Soga, Terutoshi Yamaoka, Masahiko Fujihara, Daizo Kawasaki, Shigeo Ichihashi, Amane Kozuki, Katsuhiko Sato and Tsutomu Fujita in Vascular Medicine
Footnotes
Acknowledgements
The authors thank the cardiac catheterization laboratory medical staff and clinical research coordinators at the participating centers.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Osamu Iida and Yoshimitsu Soga are consultants who received honoraria from Boston Scientific. Terutoshi Yamaoka, Masahiko Fujihara, Daizo Kawasaki, Shigeo Ichihashi, and Amane Kozuki received remuneration from Boston Scientific Japan. The remaining authors have no conflicts of interest to report.
Funding
The CAPSICUM study was supported by the Research Association for Lower Limb Artery Revascularization (LIBERAL) sponsored by the following companies (in alphabetical order): Boston Scientific Japan K.K.; OrbusNeich Foundation; Terumo Corp.; and Kaneka Medix Corporation. The funding companies played no role in study design, selection of enrolled patients, treatment strategy, revascularization procedures or equipment, or collection, analysis, or interpretation of data.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
