Abstract

What is an intracranial aneurysm?
The walls of blood vessels (both arteries and veins) are made up of three layers: the innermost layer (closest to the blood flow) is called the intima, the middle layer is known as the media, and the outermost layer is the adventitia. Arteries, the vessels that deliver oxygen and nutrients to all the tissues in the body, tend to have thicker and more muscular walls than veins. When a layer (or layers) of the artery wall is weakened, it creates a “ballooning” or bulge, called an aneurysm.1–3 When these bulges in the artery wall occur in the brain, they are referred to as intracranial aneurysms (meaning inside the skull; also known as brain aneurysms or cerebral aneurysms) (Figure 1). Small, unruptured intracranial aneurysms rarely cause symptoms or problems. However, over time, more blood can accumulate in these “pockets” of the weakened vessel wall, leading to expansion of the aneurysm and further destruction of the vessel layers. 1 If this goes unchecked, the aneurysm can rupture (i.e., burst), leading to bleeding within the brain. This bleeding can lead to serious health complications such as stroke, brain damage, hydrocephalus (i.e., fluid build-up in the brain), coma, and death. In less common cases, large unruptured intracranial aneurysms can cause health problems by pressing on the surrounding nerves and structures, leading to symptoms such as weakness, speech issues, facial drooping, and vision changes.2–4
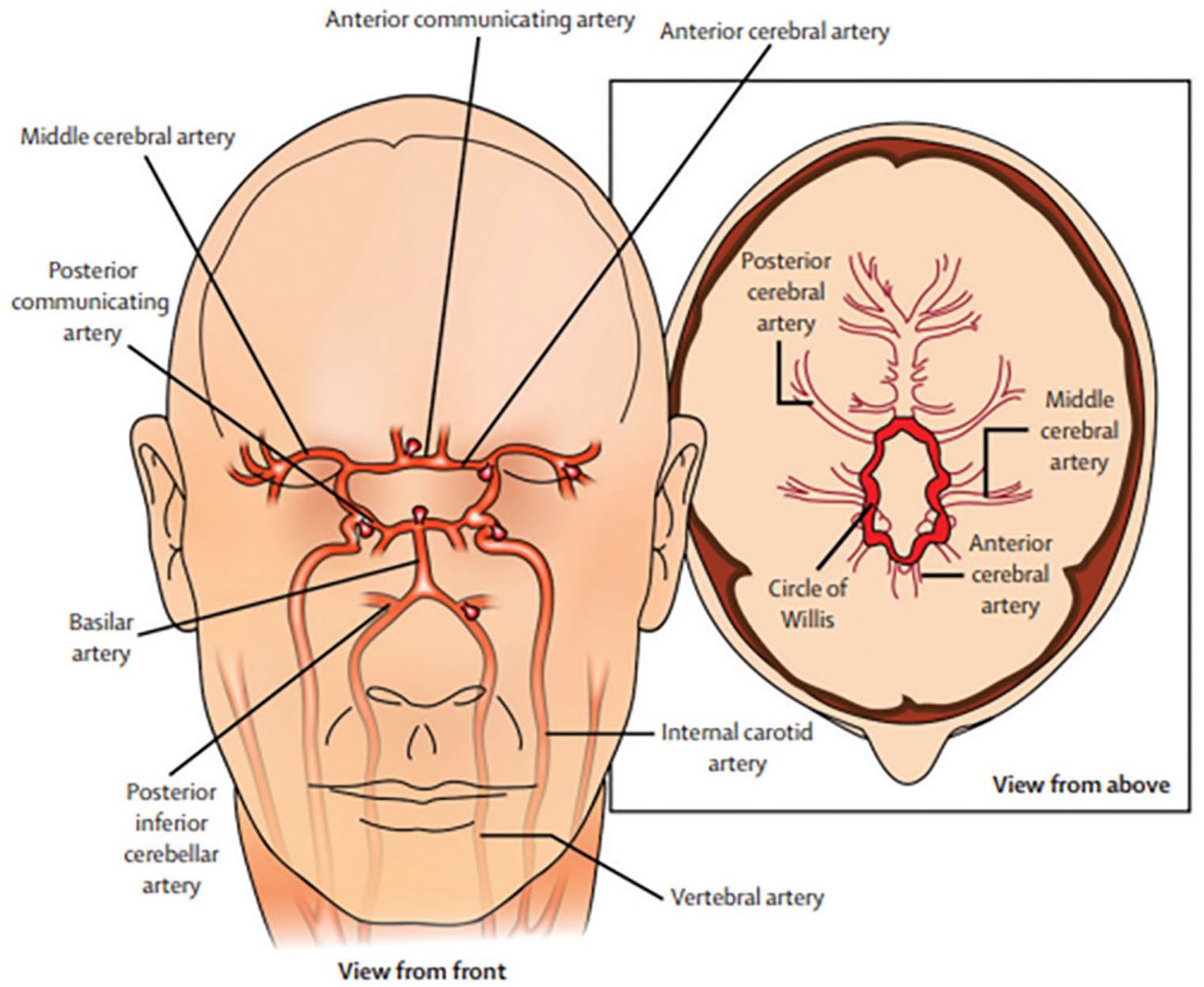
Common sites of formation of intracranial saccular aneurysms.
Intracranial aneurysms are described by their size, location, shape, and cause.3–5 For example, giant aneurysms have a diameter greater than 25 mm, and small ones are less than 7 mm. 2 Saccular aneurysms are the most common; they have a shape resembling a berry and therefore are also known as berry aneurysms. Fusiform aneurysms are spindle-shaped, meaning they are wider in the middle and tapered at the ends. Traumatic aneurysms are caused by blunt or penetrating forces. Dissecting aneurysms occur when there is a tear in the vessel wall. Intracranial aneurysms can occur anywhere in the brain but are most common in the major arteries at the base of the brain (known as the Circle of Willis 3 ), and they are frequently located near vessel branching points, called bifurcations (Figure 1).1–3 Given their potential to be life-threatening, intracranial aneurysms are a major health concern and should be taken seriously.
How common are intracranial aneurysms?
Though intracranial aneurysms can occur in anyone and at any age, they are mostly seen in adults between the ages of 30 and 60, with women affected more often than men.2,4 The chance of having an aneurysm increases with age, with the peak between ages 60 and 70. 1 Because many aneurysms do not cause symptoms and are found incidentally, the exact number of people living with intracranial aneurysms is unknown; however, studies suggest that 1–6% of people will have an intracranial aneurysm in their lifetime. 5 In the United States, roughly 6 million people are affected, with 30,000 Americans per year suffering an intracranial aneurysm rupture. Globally, 500,000 people die each year from ruptured intracranial aneurysms, and 3–5% of all new strokes are from brain bleeds related to an intracranial aneurysm rupture. 5
What are the signs and symptoms of intracranial aneurysms?
Unruptured intracranial aneurysms
Most unruptured aneurysms do not cause symptoms; however, large unruptured aneurysms can cause symptoms, including:
Headache
Double vision/changes in vision
Pain above the eye
Pupil dilation
Paralysis/drooping of one side of the face
Numbness and tingling on one side of the face
Dizziness
Loss of balance
Difficulty speaking.
Headache is the most common symptom, with stroke being the second most common presentation. 5
Ruptured intracranial aneurysms
The most feared complication is aneurysm rupture and bleeding within the brain (known as subarachnoid hemorrhage [SAH], a type of intracranial hemorrhage [ICH]). SAHs often begin with a sudden, excruciating headache. It is often described as a thunderclap headache, or the worst headache of one’s life. Other symptoms may include neck pain or stiffness, sudden confusion, loss of consciousness (blacking out), seizures, and weakness in the arms or legs.2,5 If any of these symptoms occur, patients should seek medical attention immediately.
Who is at risk for intracranial aneurysm?
Risk factors for aneurysm development
How brain aneurysms form is not completely understood, but three main factors may play a role:
1) Genetics (family history of aneurysms, connective tissue disorders, autosomal dominant polycystic kidney disease [ADPKD]);
2) Environmental factors that increase the risk of vessel disease/atherosclerosis (e.g., smoking, high blood pressure, high cholesterol, diabetes); and
3) Hemodynamic stress (increased tension or pressure on the artery walls).1–6
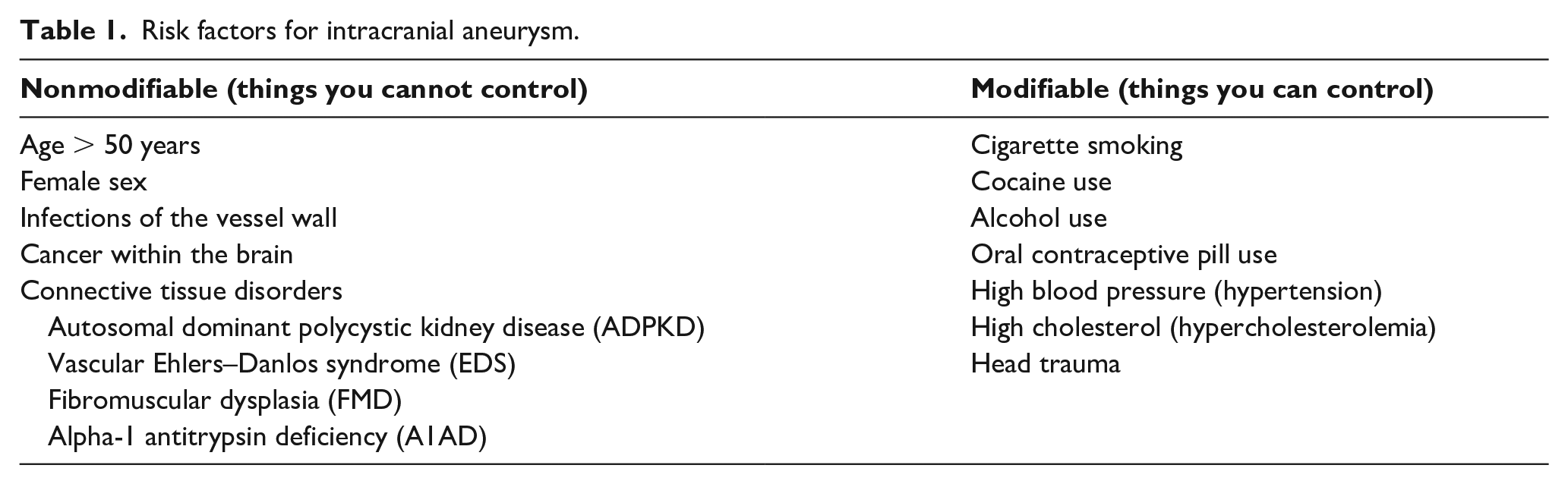
Other risk factors include age, female sex, drug use (i.e., cocaine, amphetamines), heavy alcohol use, and severe head injury. Approximately one out of five patients with aneurysm or SAH report having someone in their family with those diagnoses. 5 Cigarette smoking and high blood pressure (hypertension) independently increase the risk of aneurysm formation, and the presence of both creates an even greater risk of aneurysm and severe complications.2,3,5,7–10 The vascular disease fibromuscular dysplasia (FMD) has been associated with intracranial aneurysm. 11 For a more complete list of risk factors for aneurysm development, see Table 1.
Risk factors for intracranial aneurysm.
Risk factors for aneurysm growth and rupture
Many factors that increase the risk for aneurysm development also increase the risk for growth and rupture. Aside from smoking, female sex, and hypertension, the characteristics of the aneurysm itself are also important. Factors like larger initial aneurysm size, the presence of multiple aneurysms, the shape of the aneurysm (i.e., oblong-shaped, multilobed aneurysms), and the location of the aneurysm (i.e., at branchpoints, posterior circulation aneurysms in the back of the brain) can increase the risk of more rapid growth and potential rupture.5,10 The longer an aneurysm is present, the more likely it is to grow, which is the rationale for routine imaging to monitor the aneurysm long-term 1 .
Aneurysm rupture is a potentially fatal complication. Some studies report a 17-fold increased risk in rupture in those with two or more first-degree relatives with SAH. 2 Generally, the larger the aneurysm, the greater the risk of rupture: less than 5 mm (or 0.5 cm) have a minor risk of rupture, 10 mm or greater (1.0 cm) have an increased risk, and greater than 20 mm have the highest risk of rupture. 2
How are intracranial aneurysms diagnosed?
Most intracranial aneurysms are found incidentally on routine head imaging (i.e., when looking for other brain-related illnesses or trying to find the cause of brain or nerve-related symptoms). 1 If there is suspicion of a bleeding brain aneurysm, the best initial study is a computed tomography (CT) scan of the head without contrast. This study can quickly detect bleeding in the brain (though not perfectly accurate), allowing for quick treatment if needed. If there is no bleed seen on the CT scan, but suspicion remains high, a brain magnetic resonance imaging (MRI) examination is the next best option (MRI is more accurate than CT).1,2,5 If there are symptoms of an intracranial aneurysm but there is no concern for bleeding, CT angiography (CTA) of the head is the most appropriate test. This study looks at the major arteries of the brain by visualizing the vessels with intravenous administration of contrast. MRI of the arteries (or MRA) can also be helpful; however, this study is less likely to identify small aneurysms (< 3 mm).2,11 The gold standard for identifying aneurysms is an imaging study known as digital subtraction angiography (DSA).2,5,9,10 This is a slightly invasive procedure that involves placing a catheter (a flexible, small plastic tube) within an artery to visualize the blood vessels (using contrast). This imaging technique uses 3-D rotation that can identify the small aneurysms that may be missed by MRA. Despite the accuracy of DSA, it is not always performed given its potential risk of complications including stroke, bleeding, and artery damage. Patients with a history of at least two family members with intracranial aneurysms or SAH should be offered screening for intracranial aneurysm with CTA or MRA.2,5,9 Patients with ADPKD or FMD should also be offered screening.5,9,12 Figure 2 shows examples of aneurysms found using different imaging techniques.
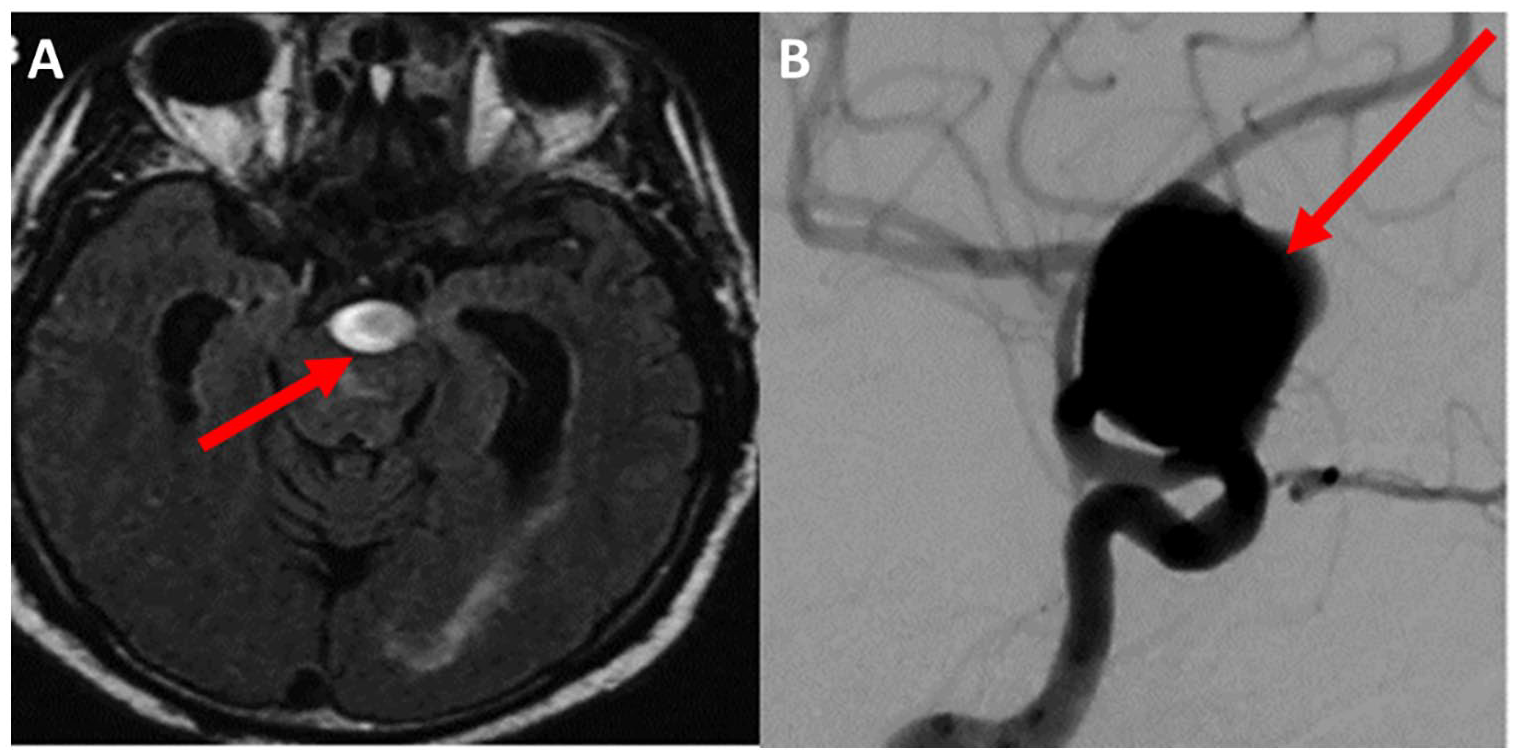
Intracranial aneurysms.
Once an intracranial aneurysm is discovered, how is it monitored?
In patients with a low-risk, unruptured aneurysm, it is reasonable to monitor with MRA or CTA at 6 to 12 months after the aneurysm is first discovered. 9 After the first year, the MRA or CTA may be repeated every year for 3 years, followed by every 2 years if the aneurysm is stable in size. 2
What are the treatment options?
Ruptured intracranial aneurysms warrant immediate intervention; however, intervening on unruptured aneurysms is not as straightforward. Deciding to intervene on an unruptured aneurysm is a complex medical decision and several factors are considered by the medical team, including the patient’s medical history and other medical conditions, age, history of prior aneurysm rupture, and family history of aneurysm rupture. Aneurysm characteristics such as the location, size, shape, and type are also considered.1,5,9 As with any procedure or surgery, there should be a review of the risks and benefits of the different approaches, followed by shared decision-making between the patient and medical team. Most experts agree that intervention on unruptured aneurysms should be reserved for large aneurysms or aneurysms with other high-risk features and symptomatic patients who have a high risk of rupture. The intervention should also take place in a dedicated neurovascular center to minimize the risk of procedural complications. 5 The most common surgical and endovascular approaches for the treatment of cerebral aneurysms are discussed below.
Surgical and endovascular options
Craniotomy and clipping
First reported in 1938, 5 surgical clipping is a major open surgical procedure in which the neurosurgeon opens the skull (called a craniotomy) and places a small, metal, clothespin-like clip across the neck (or base) of the aneurysm (Figure 3). Placement of the clip closes off blood flow to the aneurysm, causing it to shrink in size until it scars. The benefit of clipping is that it is usually a permanent solution. 9 The downside is that it requires a craniotomy and a lengthy recovery.
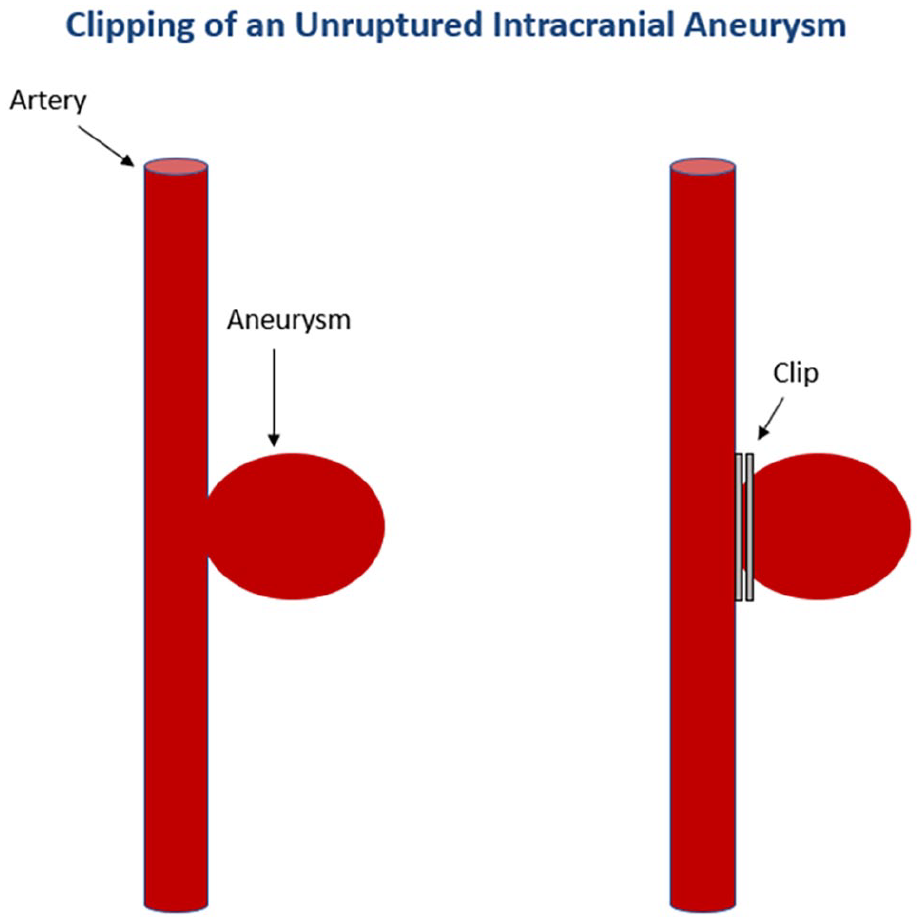
Craniotomy with clipping of an unruptured intracranial aneurysm. After craniotomy, a small metallic clip can be placed across the neck of a saccular aneurysm.
Endovascular therapy
First reported in 1990, 5 endovascular coiling is a minimally invasive procedure in which a catheter is placed into a thigh or arm artery to gain access to the site of the artery in the brain where the aneurysm is located. Once the aneurysm is identified, a variety of techniques can be employed to remove the aneurysm from the circulation. The first endovascular treatments involved placing platinum coils from the catheter into the aneurysm, sealing it off from the normal circulation. The coil prevents blood flow from entering it by inducing clot formation (embolization) of the aneurysm 10 (Figure 4). In patients who are at increased risk of complications with coil alone, sometimes a stent can be placed within the artery first, followed by coil placement into the aneurysm. The stent is thought to keep the coil within the aneurysm, preventing it from being dislodged into the healthy artery (Figure 5). Other endovascular procedures can be used, such as flow diversion, where a stent is placed in an upstream and larger (or parent) artery, reducing blood flow into the aneurysm (usually downstream) itself (Figure 6). Endovascular procedures to treat intracranial aneurysms do not require opening of the skull and are therefore gaining popularity due to quicker recovery.
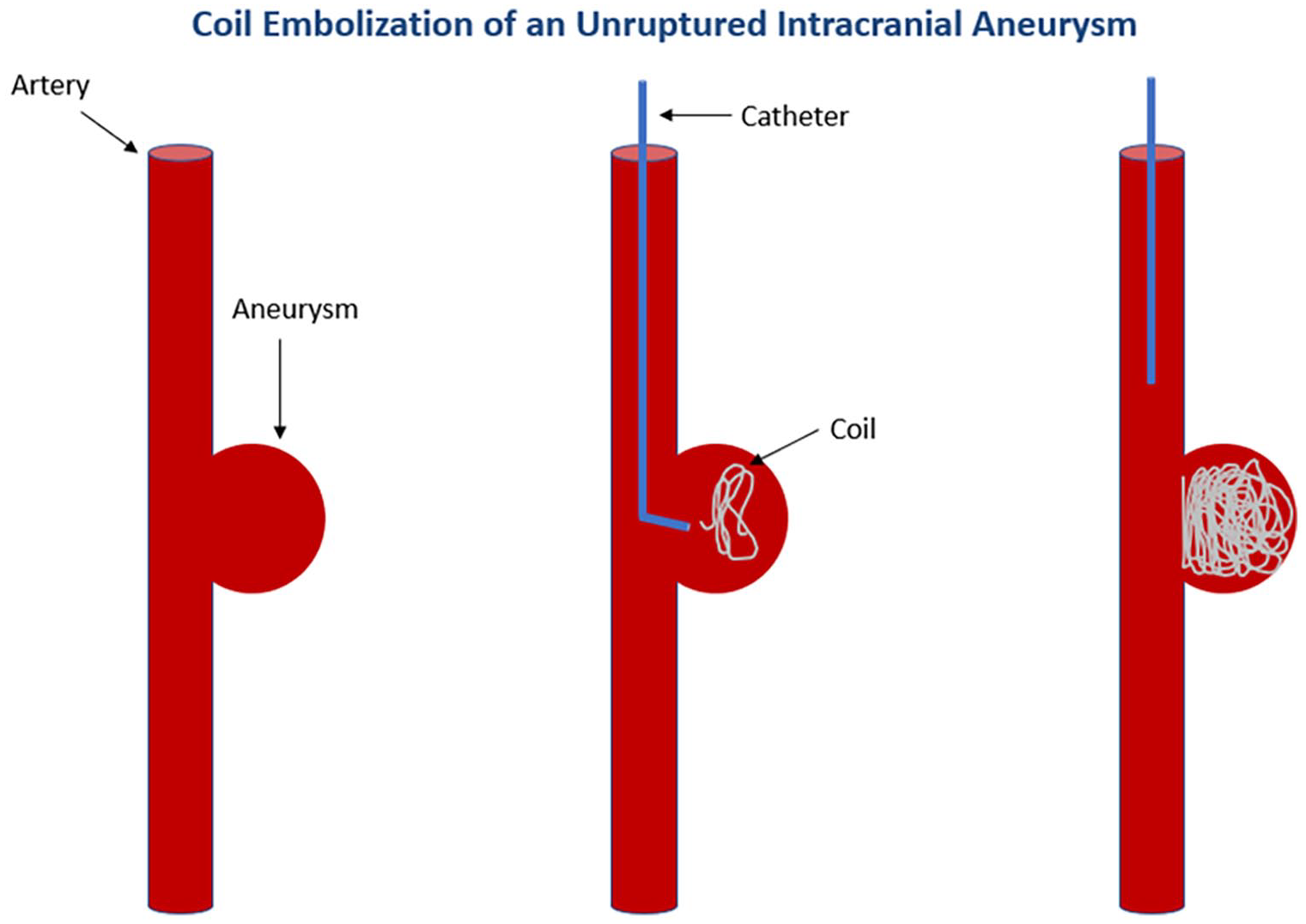
Coil embolization of an unruptured intracranial aneurysm using the Guglielmi detachable coil system. Tiny platinum coils are placed directly into the aneurysm.
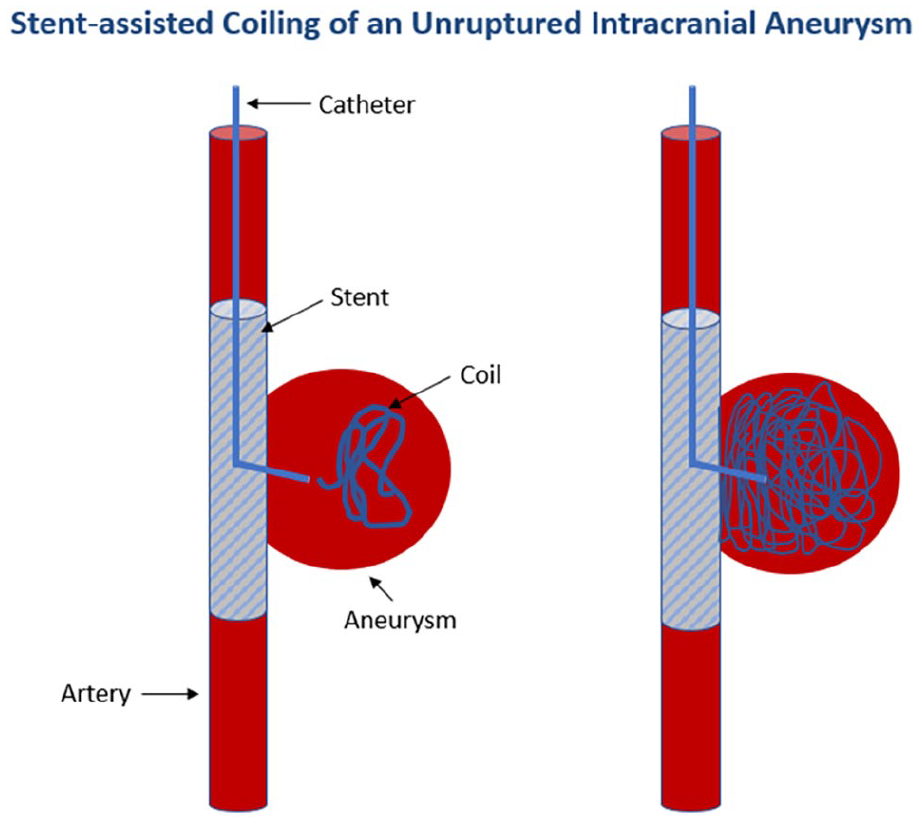
Stent-assisted coiling of an unruptured intracranial aneurysm. For some aneurysms that cannot be treated with coiling alone due to the risk of complications, a stent can be placed first and coils then placed into the aneurysm via a catheter that is advanced through the stent into the aneurysm. The stent prevents the coils from becoming displaced from the aneurysm and entering the parent artery.
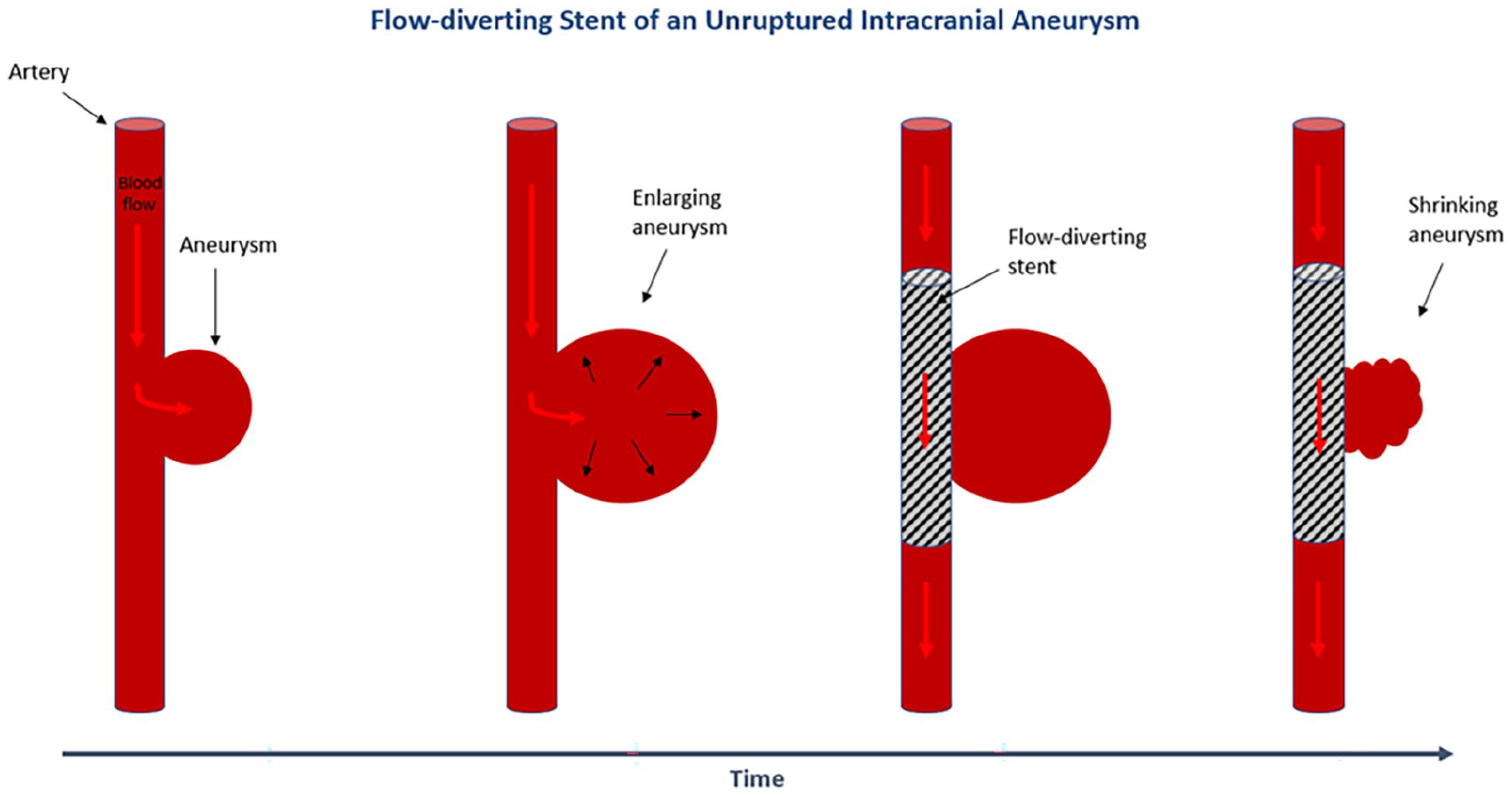
Flow-diverting stent of an unruptured intracranial aneurysm. This image depicts how blood flow into an aneurysm enlarges it over time. With placement of the diverting stent, blood flow to the aneurysm is reduced, causing it to eventually shrink in size.
Risk factor modification
Unruptured aneurysms that do not have high-risk features can be managed conservatively via risk factor modification. 1 Smoking cessation and maintaining a normal blood pressure are the mainstays of noninvasive treatment. Lowering the consumption of alcohol is also important, as heavy alcohol use increases the risk of SAH. 5
Summary
Intracranial aneurysms occur when the wall of an artery within the brain weakens and creates a bulge. Over time, this bulge can increase in size and rupture, causing a brain bleed. The most common symptom of rupture is an excruciating headache but can also include vision changes and face or limb paralysis. Risk factors for developing an aneurysm include smoking, hypertension, and genetics. Aneurysms are detected by CTA or MRA of the brain and its major vessels. Screening for intracranial aneurysms should be offered to all patients with ADPKD, FMD, and those with two or more close relatives with a history of SAH. A ruptured aneurysm is a medical emergency and requires surgical or endovascular intervention, most commonly clipping or coil embolization, respectively. Surveillance with repeat head imaging should occur within at least 1 year after the aneurysm is discovered, followed by regular imaging follow-up based on the individual’s risk factors. Management should always include quitting smoking, reducing alcohol consumption, and maintaining normal blood pressure.
The ‘Vascular Disease Patient Information Page’ is a regular feature of Vascular Medicine. All articles in the collection are available for free online at http://journals.sagepub.com/vmjpatientpage. The Vascular Disease Patient Information Page is provided for educational purposes only and is not a substitute for medical advice.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
