Abstract
Introduction:
The Society for Vascular Surgery Threatened Limb Classification System (‘WIfI’) is used to predict risk of limb loss and identify peripheral artery disease in patients with foot ulcers or gangrene. We estimated the diagnostic sensitivity of multiple clinical and noninvasive arterial parameters to identify chronic limb-threatening ischemia (CLTI).
Methods:
We performed a single-center review of 100 consecutive patients who underwent angiography for foot gangrene or ulcers. WIfI stages and grades were determined for each patient. Toe, ankle, and brachial pressure measurements were performed by registered vascular technologists. CLTI severity was characterized using Global Limb Anatomic Staging System (GLASS stages) and angiosomes. Medial artery calcification in the foot was quantified on foot radiographs.
Results:
GLASS NA (not applicable), I, II, and III angiographic findings were seen in 21, 21, 23, and 35 patients, respectively. A toe–brachial index < 0.7 and minimum ipsilateral ankle–brachial index < 0.9 performed well in identifying GLASS II and III angiographic findings, with sensitivity rates 97.8% and 91.5%, respectively. The diagnostic accuracy rates of noninvasive measures peaked at 74.7% and 89.3% for identifying GLASS II/III and GLASS I+ angiographic findings, respectively. The presence of medial artery calcification significantly diminished the sensitivity of most noninvasive parameters.
Conclusions:
The use of alternative noninvasive arterial testing parameters improves sensitivity for detecting PAD. Abnormal noninvasive results should suggest the need for diagnostic angiography to further characterize arterial anatomy of the affected limb. Testing strategies with better accuracy are needed.
Keywords
Introduction
The global vascular guidelines on the management of chronic limb-threatening ischemia (CLTI) – defined as ‘the presence of peripheral artery disease (PAD) in combination with rest pain, gangrene, or a lower-limb ulceration > 2 weeks duration’ – recommends considering revascularization for patients with foot ulcers or gangrene who have significant PAD (Global Anatomic Staging System [GLASS] stages I, II or II) 1 and who are at low, moderate, or high risk for limb loss (i.e., Society for Vascular Surgery’s Wound-Ischemia-foot Infection [WIfI] clinical stages 2–4). 2 With revascularization, one-year limb salvage rates of 89–92% have been reported in clinical trials 3 and in observational series of patients in WIfI clinical stages 2 and 3. 4 In contrast, the natural history of limbs with foot ulcers and CLTI is poor, with 1-year amputation rates of approximately 40–50%.5,6 As such, failing to identify CLTI in patients with foot ulcers or foot gangrene – in essence, de facto triaging to natural history rather than to revascularization – could contribute to poor limb outcomes and potentially avoidable amputations.
Considering this, we studied the sensitivity and diagnostic accuracy of clinical and noninvasive lower-extremity arterial testing parameters in a cohort of patients who underwent angiography for foot ulcers or foot gangrene with the intent of optimizing the identification of CLTI.
Methods
Study objective, patient population, and study sample
The population of interest was all patients with gangrene or nonhealing ulcers (> 4 weeks in duration) of the foot (limb distal to the malleoli) who underwent digital subtraction angiography because of suspicion for CLTI. Patients with prior transfemoral or transtibial amputations or any prior infrainguinal revascularization of the index limb were excluded. Patients with bilateral foot wounds who underwent dedicated angiography for each foot were recorded as separate episodes. Only the index angiogram for patients who underwent multiple angiograms of the same leg during the selective date range were included.
The study sample consisted of 100 consecutive patients at the Michael E. DeBakey Veterans Affairs Medical Center (Houston, TX, USA) who met the inclusion and exclusion criteria starting from January 1, 2020. The study sample was identified with the computerized scheduling software (OR ControlTM; Healthcare Control Systems, Inc., Austin, TX, USA) used to register and schedule all procedures done at this hospital. As it was a retrospective study, individual consent was not obtained. Approval of this research by the Baylor College of Medicine Institutional Review Board and the Michael E. DeBakey Veterans Affairs Hospital was obtained (protocols H-34858 and IRBNet ID 1684733-2, respectively).
Demographic and clinical characteristics
Clinical and demographic characteristics, including age, sex, hemoglobin A1c, serum creatinine, estimated glomerular filtration rate, the presence or absence of diabetes, heart failure, and end-stage renal disease were obtained from the electronic medical record, using the most recent values prior to angiography. The electronic medical record was also used to identify access complications within 30 days following angiography.
The extent or severity of the index foot wound and any accompanying infection were quantified using the wound and ischemia grades of the WIfI scoring system based on wound descriptions, vital signs, laboratory results, and radiographic findings recorded in the electronic medical record. Ischemia grades of the WIfI score were assessed based on noninvasive arterial testing of the index lower extremity obtained within 1 year prior to angiography using the previously described categorization. 2 Patients without noninvasive testing were categorized based on the minimum WIfI clinical stage determined by the wound and infection components when estimating the diagnostic performance of various noninvasive and clinical parameter thresholds (e.g., WIfI clinical stage 2 or higher).
Noninvasive arterial parameters
Noninvasive arterial studies done at our facility were performed by one of three registered vascular technologists in a vascular laboratory accredited by the Intersocietal Accreditation Commission using Parks System 2100-SW Flo-Lab machines (Parks Medical Electronics, Inc., Aloha, OR, USA). Our technologists routinely obtain two ankle pressure measurements and plethysmography readings at the ankle and toe level. Additionally, they obtain toe pressure measurements for all patients that have either a first or second toe without a wound too tender to allow cuff placement. Our lab uses conventional reporting of the ankle–brachial index (ABI) as the higher of the two ankle pressures on the index limb divided by the higher of the two brachial (arm) pressures. The toe–brachial index (TBI) of the index limb was calculated as the digit pressure divided by the higher of the two brachial pressures. For this analysis, we additionally calculated a ‘minimum’ ABI as the lower of the two ankle pressures of the index divided by the higher of the two brachial pressures, as this has previously been reported as a more sensitive parameter in identifying PAD. 7 The diagnostic accuracy of plethysmography is part of a separate study by our group and not reported here.
Angiographic assessment using GLASS
All angiography was performed by one of four board-certified vascular surgeons on Philips Allura (Amsterdam, The Netherlands) floor-mounted imaging systems in hybrid operating rooms. Digital subtraction angiography was used for all diagnostic imaging. Iodixanol was the preferred contrast agent, with carbon-dioxide angiography reserved for patients with advanced chronic kidney disease. Vascular surgeons at our center have been recommending angiography to all patients with foot ulcers or foot gangrene who also have toe pressures below 60 mmHg, weak or nonpalpable pedal pulses, heel or dorsal foot ulcers, those with large ulcers, and those with advanced infection. Our group has generally employed BEST-CLI trial eligibility criteria to evaluate aortoiliac inflow, with patients without normal femoral pulses getting computed tomographic angiography of the abdomen, pelvis, and lower extremities instead of conventional angiography. 8
The Society for Vascular Surgery Global Limb Anatomic Staging System (GLASS) was used to quantify the complexity of femoropopliteal and infrapopliteal (tibial) disease in affected limbs as seen on angiography, with not applicable (NA) reflecting intact in-line flow to the ankle and stages I–III reflecting an increased burden of disease. Severity of atherosclerotic disease in pedal vessels was classified with the GLASS inframalleolar modifier. 1 An intact target-artery path (TAP) to the most relevant angiosome was defined as in-line blood flow from the groin to the area of the index foot ulcer through named infrainguinal and at least one pedal artery that had outflow vessels visualized in the area of the foot ulcer. In general, the most relevant angiosome arteries were the anterior tibial-dorsalis pedis or the posterior tibial-lateral plantar arteries for the toes, the anterior tibial-dorsalis pedis artery for dorsal foot wounds, the posterior tibial-plantar arteries for the plantar forefoot proximal to the toes and the plantar midfoot, and the posterior tibial or peroneal artery for the heel.9–12 Patients with an intact TAP to the ankle but without in-line arterial circulation to the foot and no named vasculature were classified as ‘desert foot’ (i.e., GLASS inframalleolar disease descriptor P2, 1 Kawarada type 3 13 ). Finally, whether an in-line TAP could plausibly be obtained, either through endovascular or surgical revascularization, was determined for all patients with GLASS I+ findings by two board-certified vascular surgeons.
Medial artery calcification (MAC) scoring
The extent of medial artery calcification (MAC) in the foot was quantified in the manner described by Liu and colleagues. 14 Specifically, foot radiographs within 6 months prior to the index angiogram were reviewed by two board-certified vascular surgeons. One point was given for the presence of ‘tram track’ (circumferential) calcifications in the following locations if over a certain length: 2 cm or longer in the dorsalis pedis, medial or lateral plantar artery, or first plantar metatarsal artery or its lateral branch proximal to the interphalangeal space; or 1 cm or longer in the lateral plantar hallucial artery or any of the plantar digital arteries of the lesser toes.
Diagnostic accuracy of various parameters
We estimated the sensitivity of multiple clinical parameters, including ischemia grade, modified WIfI stage, and various noninvasive arterial measures for identifying various GLASS stages. Sensitivity was calculated as the proportion with a particular diagnostic finding among all those at or above the GLASS stage of interest. Diagnostic accuracy was calculated as the proportion of true negatives and true positives among total cases. Categorical data were compared using Pearson’s chi-squared test and continuous variables using Student’s t-test. WIfI staging and angiography findings are reported as counts and proportions.
Results
Baseline patient characteristics
We identified 100 limbs in 90 consecutive patients that met inclusion and exclusion criteria. The median age was 70 years, and 98.9% were men (89 men, 1 woman). Seventy-six patients (84.4%) had diabetes, with a median HbA1c of 7.3%. The mean preoperative creatine was 2.3 mg/dL with an average eGFR of 58.3 mL/min/1.73 m2. Fifteen (16.7%) had end-stage renal disease, and 25 (27.8%) had chronic heart failure. Patient characteristics and demographics are listed in Table 1. In 61 limbs, the index wound was located on a toe or toes; five on the dorsal foot; 12 on the plantar forefoot or midfoot; 18 on the heel; two just below the malleoli; and two along incisions of prior transmetatarsal (partial foot) amputations. No 30-day access-related complications occurred.
Baseline demographics and other characteristics of the study sample (n=90).
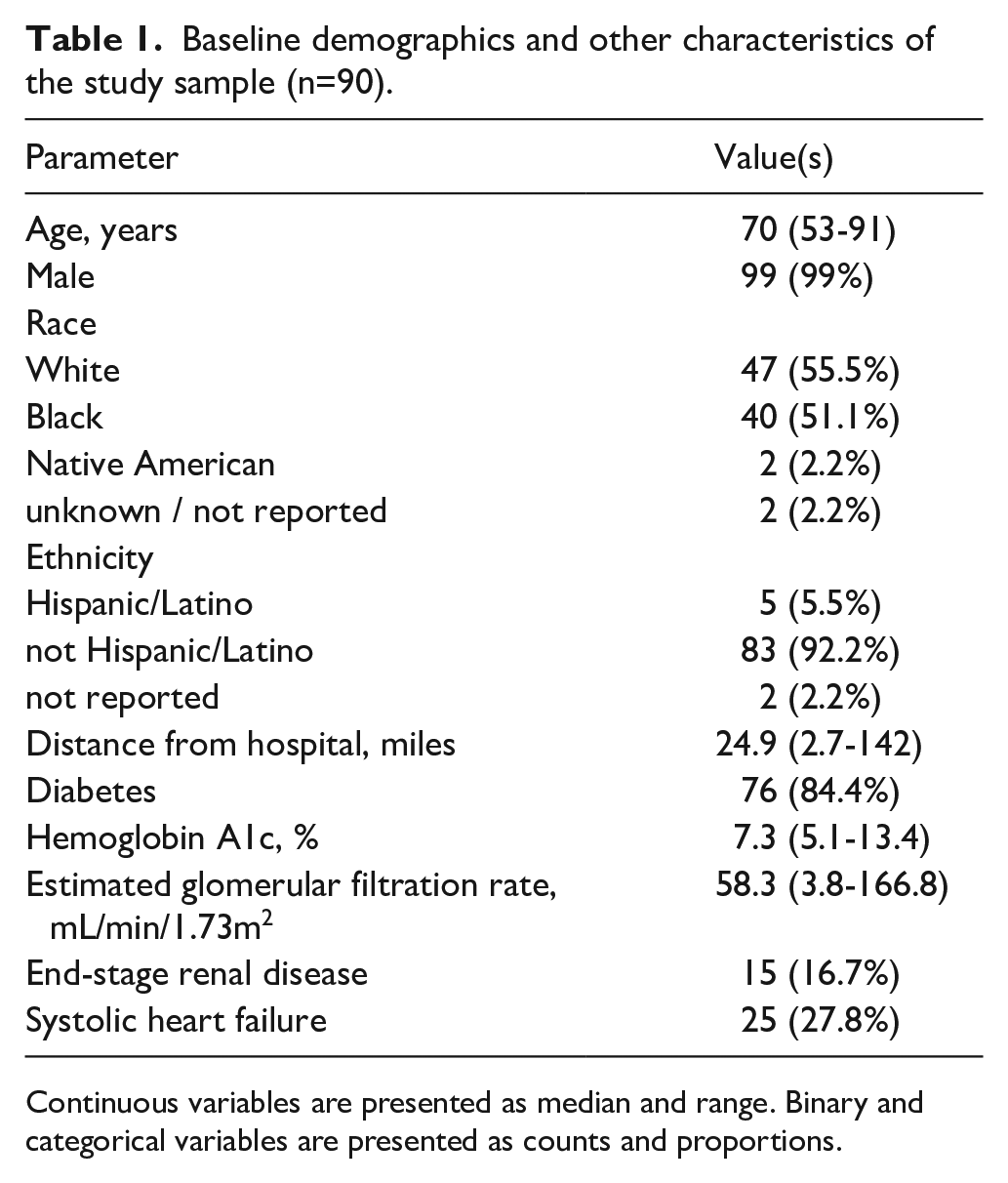
Continuous variables are presented as median and range. Binary and categorical variables are presented as counts and proportions.
Of the 100 limbs, 93 had adequate information available to allow for all three components of the WIfI score to be calculated. Of these, 16 were WIfI stage 1, 19 stage 2, 29 stage 3, and 29 stage 4. The WIfI ischemia grade was determined by ABI in 51 patients (54.8%) and by toe pressure in 42 patients (45.2%) (Table 1). Of the 100 limbs, 21 (21%) were categorized as GLASS NA (intact in-line flow to the ankle), 21 (21%) as GLASS stage I, 23 (23%) as GLASS stage II, and 35 (35%) as GLASS stage III. Eighteen patients (19.4%) had in-line flow to the angiosome and nine (9.7%) had intact flow to the foot via both the anterior and posterior tibial arteries (Table 2). Wound blush prior to any intervention was seen in 63 limbs (63%), of which 34 limbs (34%) had GLASS II or III disease.
Relationship between WIfI ischemia grade and angiographic findings among patients with foot ulcers or foot gangrene (n = 93).
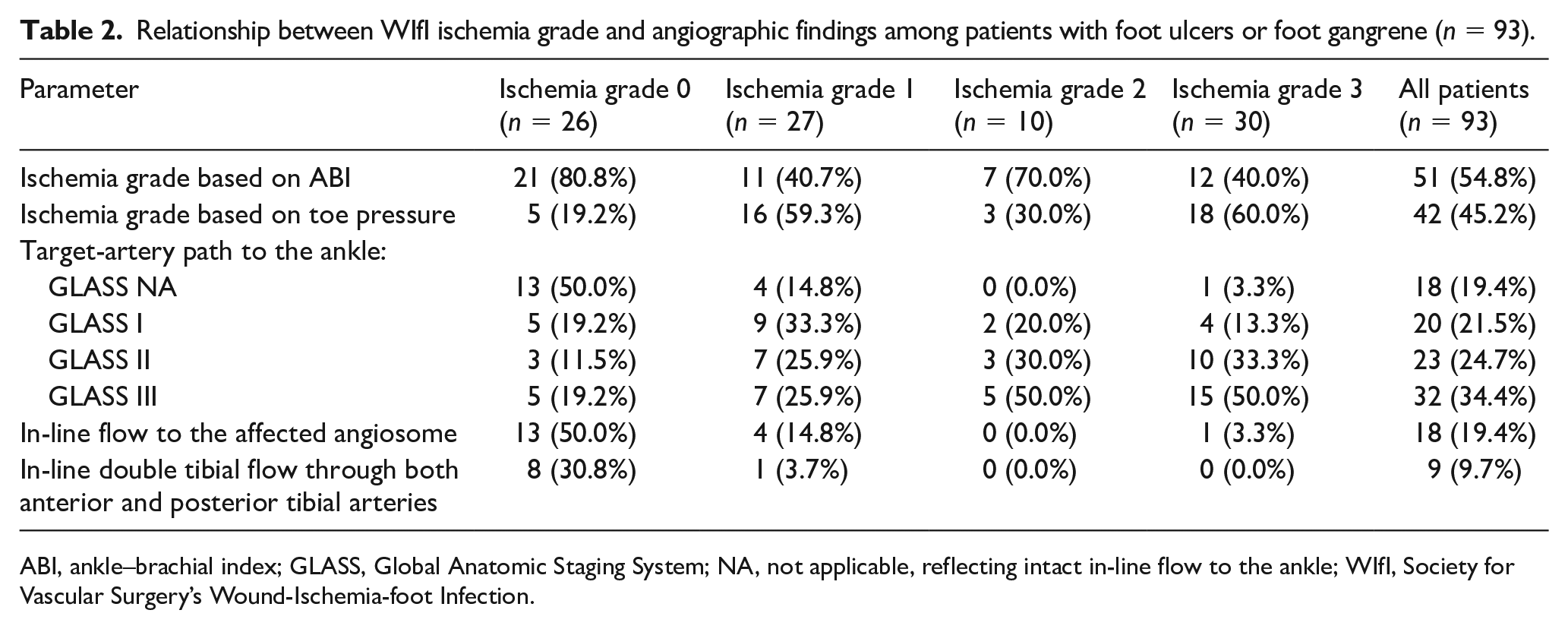
ABI, ankle–brachial index; GLASS, Global Anatomic Staging System; NA, not applicable, reflecting intact in-line flow to the ankle; WIfI, Society for Vascular Surgery’s Wound-Ischemia-foot Infection.
Sensitivity of clinical and noninvasive parameters to identify varying GLASS stages
Overall, WIfI ischemia grade did not reliably correlate with angiographic findings (Figure 1). Thirteen of the 26 limbs (50.0%) with a WIfI ischemia grade of 0 lacked in-line flow to the ankle. This included five limbs (19.2%) categorized as GLASS I, three limbs (11.5%) categorized as GLASS II, and five (19.2%) categorized as GLASS III (Table 2). Twenty-three of the 27 limbs (85.8%) with a WIfI ischemia grade of 1 lacked in-line flow to the ankle. This included nine limbs (33.3%) categorized as GLASS I, seven limbs (25.9%) categorized as GLASS II, and seven limbs (25.9%) categorized as GLASS III (Table 2). All 13 ischemia grade 0 limbs and all four ischemia grade 1 limbs that had GLASS NA in-line flow also had in-line flow to the affected angiosomes.
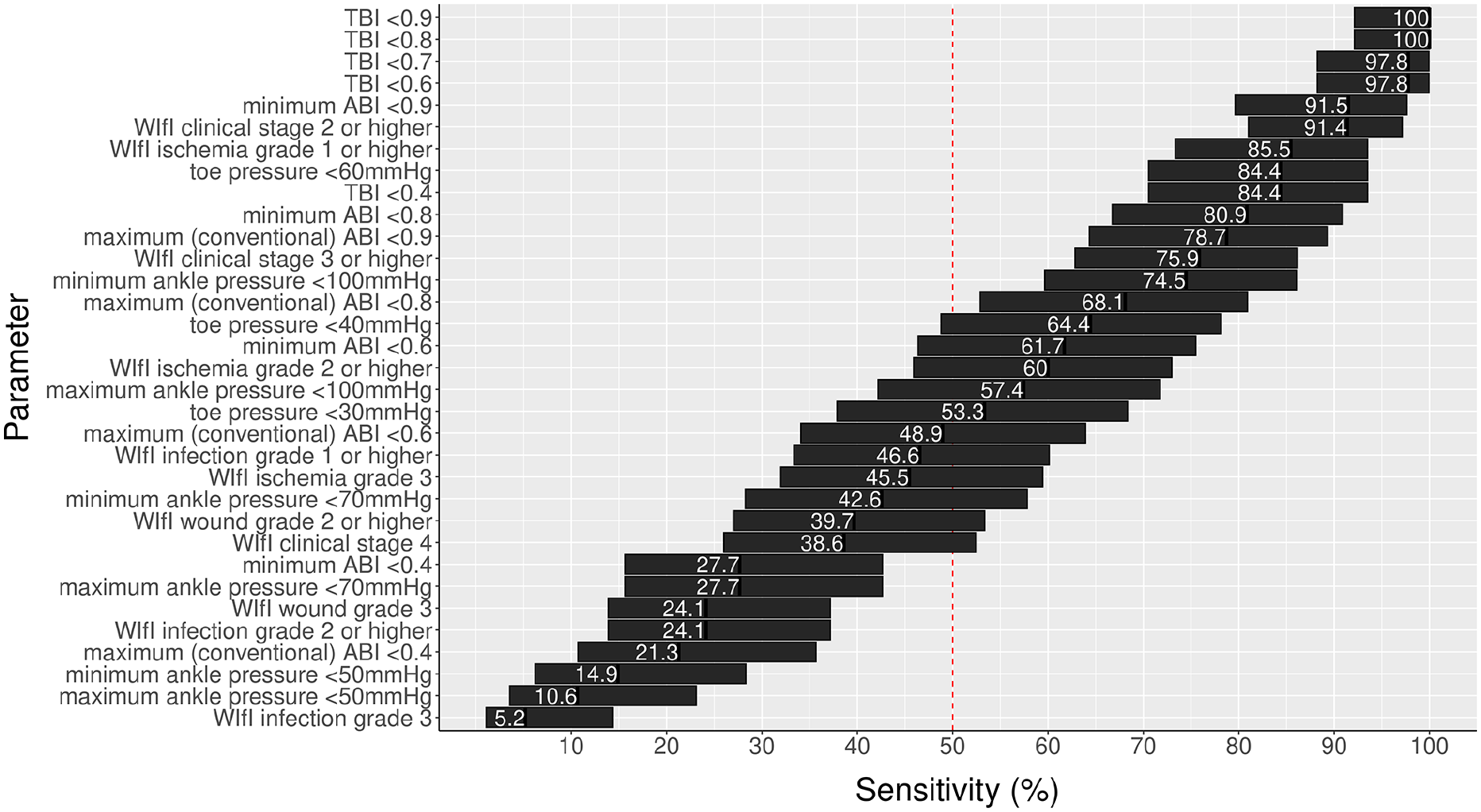
Sensitivity rates of various clinical and noninvasive parameters to identify GLASS II or III angiographic stage. All parameters refer to those obtained from the index limb. The bars span the upper and lower 95% CI around point estimates. Corresponding specificity rates are presented in Supplemental Figure E (available online).
The sensitivity rates of 33 different clinical and noninvasive parameters for identifying GLASS II or III angiographic findings are shown in Figure 1 and Table 3. These sensitivity rates ranged from 5.2% for grade 3 infection to 100% for a TBI < 0.8. Some general trends were apparent: toe–brachial indices had higher sensitivity rates than absolute toe pressures; ankle–brachial indices had higher sensitivity rates than the corresponding absolute ankle pressures; ankle–brachial indices using the lower of the two ankle pressures had higher sensitivity rates than ankle–brachial indices using the higher of the two ankle pressures; and noninvasive arterial parameters were generally better than parameters that incorporated other clinical data (e.g., WIfI clinical stage, wound grade).
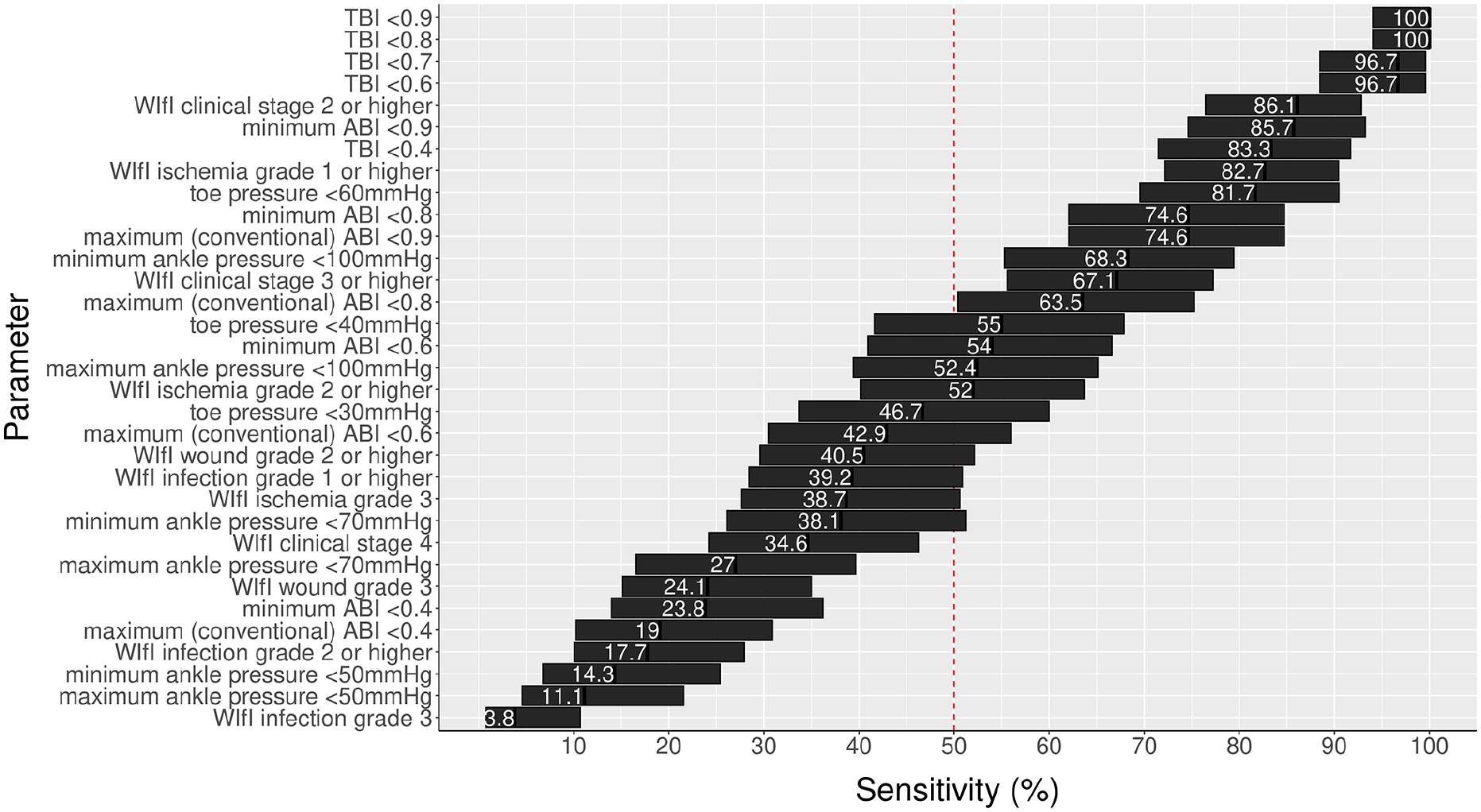
Sensitivity rates of various clinical and noninvasive parameters to identify GLASS I higher angiographic stage. All parameters refer to those obtained from the index limb. The bars span the upper and lower 95% CI around point estimates. Corresponding specificity rates are presented in Supplemental Figure F (available online).
Diagnostic performance of various non-invasive parameters in detecting GLASS II or III angiographic findings.
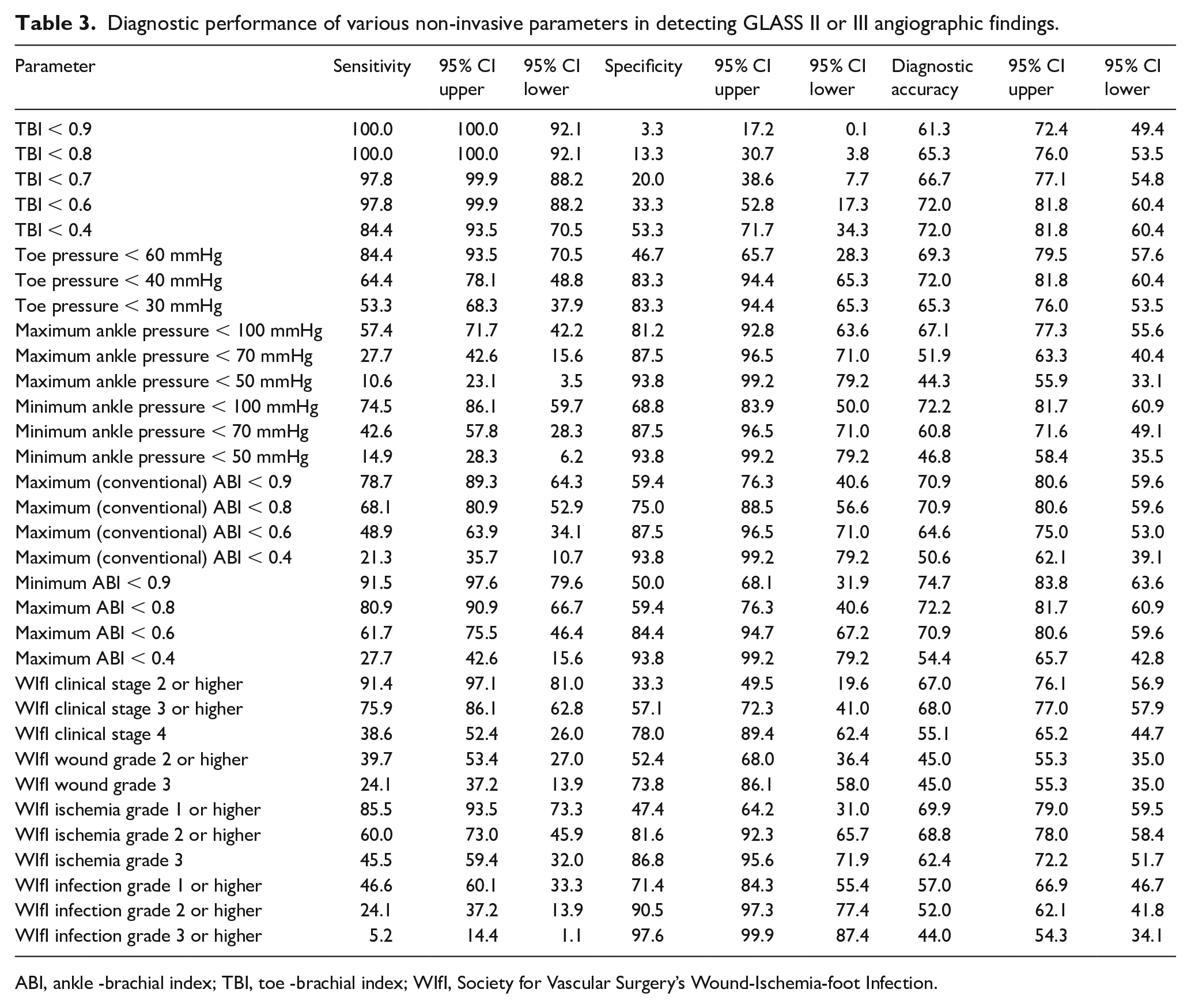
ABI, ankle -brachial index; TBI, toe -brachial index; WIfI, Society for Vascular Surgery’s Wound-Ischemia-foot Infection.
The sensitivity rates of these various parameters in identifying GLASS I+ disease (a threshold for revascularization suggested for all patients with WIfI clinical stage 2 or higher) are shown in Figure 2. Aside from sensitivity rates being slightly lower for some parameters, the ordering of tests was largely the same with few exceptions: minimal ABI < 0.9 lower in ranking, WIfI ischemia grade 2 or 3, WIfI wound grade 2 or 3, and WIfI clinical stage 3 or higher somewhat higher in ranking. Online Supplemental Table A lists the sensitivity rates of these parameters for GLASS I+ angiographic stages. Graphs showing the relationship between several noninvasive parameters and GLASS angiographic stages for individual patients is shown in online Supplemental Figures A–D.
Diagnostic accuracy and specificity of clinical and noninvasive parameters to identify varying GLASS stages
The diagnostic rates of the various clinical and noninvasive parameters for identifying GLASS II or III angiographic findings and GLASS I+ angiographic findings are shown in Figures 1 and 2, respectively. At 74.7%, minimal ABI of < 0.9 had the best diagnostic accuracy rate for identification of GLASS II or III angiographic findings. The diagnostic accuracy of a TBI < 0.7 was 66.7%, but the sensitivity was only 20.0%. In general, an inverse relationship was present between sensitivity and specificity in these parameters identifying GLASS II and III angiographic findings (Table 3 and Supplemental Figure E).
At 89.3%, TBI < 0.6 had the highest diagnostic accuracy in the identification of GLASS I or higher stage angiographic findings. It also had a specificity of 60.0% and a sensitivity of 96.7% (Table 3 and Supplemental Figure F). Minimum ABI < 0.9 also had good performance, with diagnostic accuracy, specificity, and sensitivity rates of 82.3%, 68.8%, and 85.7%, respectively.
The impact of foot MAC on identification of varying GLASS stages
A total of 95 limbs had available foot X-rays within 1 year of angiography, including 37 (38.9%) with a MAC score 0, 10 (10.5%) with a MAC score of 1, 10 (10.5%) with a MAC score of 2, eight (8.4%) with a MAC score of 3, 16 (16.8%) with a MAC score of 4, and 14 (14.7%) with a MAC score of 5. In total, 58 patients (61%) had a foot MAC score greater than zero.
A multivariate logistic regression suggested that the presence of diabetes mellitus and end-stage renal disease were significantly and positively associated with the presence of any foot MAC (odds ratios of 0.36 and 0.39, respectively; p-values 0.006 and 0.002, respectively). Bivariate chi-squared analysis also suggested an inverse relationship between GLASS femoropopliteal segment grades and the presence of any foot MAC (p = 0.048).
The sensitivity rates of various noninvasive testing and clinical parameters of patients without foot MAC (i.e., foot MAC score of 0) were compared to those with foot MAC (i.e., foot MAC of 1–5). This demonstrated that the sensitivity of TBI and minimum ABI thresholds seemed to have minimal change in presence of MAC, whereas absolute toe pressures, absolute ankle pressures, and maximum (conventional) ABIs were notably lower in the presence of pedal MAC (Table 4).
Diagnostic performance of various noninvasive parameters for detecting GLASS II or III angiographic findings in patients with or without pedal MAC.
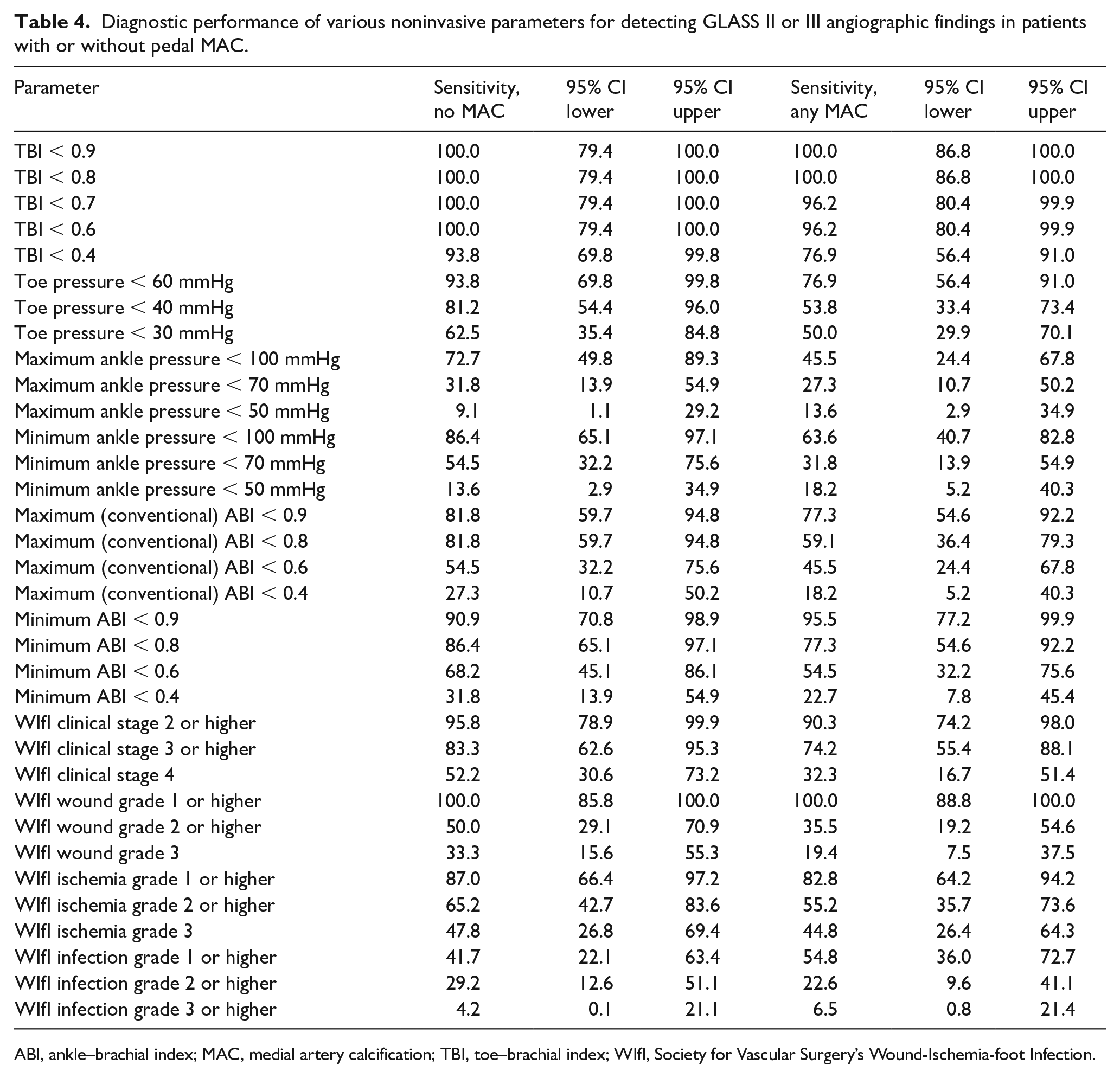
ABI, ankle–brachial index; MAC, medial artery calcification; TBI, toe–brachial index; WIfI, Society for Vascular Surgery’s Wound-Ischemia-foot Infection.
Discussion
The intersocietal global vascular guidelines 1 and the Society for Vascular Surgery’s Threatened Limb Classification System 2 have both established objectives systems to characterize clinical presentation and angiographic findings. Both have provided sound guidance to vascular specialists deciding when and how to perform revascularization for patients with CLTI. Both of these documents should continue to inform the management of CLTI.
The focus of this analysis is not the mode or outcomes of revascularization for this patient population. Instead, the focus of this analysis was to further assess the sensitivity of clinical features and noninvasive arterial testing parameters in identifying CLTI. Our aim is to eliminate missed diagnoses of CLTI among patients with foot ulcers – in other words, reducing the number of patients with ulcers erroneously categorized as being ‘neuropathic’ or the number of patients who have noninvasive testing that erroneously suggests that arterial insufficiency is not present or not severe. Our hope is that this would then minimize the number of patients erroneously triaged to wound care without revascularization, a situation that could clearly contribute to otherwise-avoidable amputations.
As endovascular intervention increasingly becomes first-line treatment for patients with CLTI, there is a risk of conflating the value of angiography to identify CLTI with the value of concomitant endovascular interventions to treat CLTI. In this study, we purposely focused only on the identification of CLTI in patients with foot ulcers or foot gangrene. Literature in our field suggests that differences in the outcomes of endovascular versus open revascularization for CLTI are small compared to differences between revascularization of any form compared to the natural history of untreated or unrecognized CLTI. 15
Whereas previous studies have most often used the presence of an atherosclerotic lesion, often > 50% or > 75% luminal stenosis,16–23 as the gold standard when calculating the diagnostic performance of noninvasive parameters, this study used two thresholds in the novel GLASS categorization. As such, this study represents the most detailed analysis of the diagnostic performance of noninvasive arterial testing to date. We focused on GLASS II and III angiographic stages because we anticipated that most vascular specialists would offer some form of revascularization to patients with foot ulcers or foot gangrene with these findings. We estimated the sensitivity rates of various parameters in identifying GLASS I or higher angiographic findings also, as the global guidelines suggest this as a threshold for revascularization in patients with a WIfI clinical stage 2 or higher. We recognize that the decision to treat is more nuanced than the GLASS categorization of angiographic findings alone; nonetheless, binary categorization is helpful both in clinical practice and in the estimation of diagnostic performance.
The results of this study further refine the previously published decision analysis.24–28 Specifically, TBIs have a lower false negative rate (higher sensitivity) than absolute toe pressures. We would suggest preferentially using an ipsilateral limb TBI of > 0.7 as the single best test to exclude CLTI in patients with foot ulcers or foot gangrene. In patients without a first or second toe or who have a tender wound that precludes cuff placement (approximately 50% of patients in our study), the minimum ABI (lower of the two ankle pressures on the index limb divided by the higher of the two brachial pressures) should be obtained. Results below either of these thresholds should suggest angiography as the next step. In contrast to absolute toe and ankle pressure thresholds, TBI and minimum ankle ABI seem relatively unaffected by the presence of foot MAC.
The weak correlation between noninvasive parameters and GLASS angiographic stages in this study also suggests that the noninvasive testing parameters examined here can be useful in identifying PAD and should not be used to estimate the severity of angiographic findings. Although the physiological intent of noninvasive testing is appealing, the large number of patients with GLASS stage III angiographic findings and toe pressures, ankle pressures, and ABIs that are in the normal range should caution against the idea of noninvasive testing supplanting digital subtraction angiography as the gold standard for identifying and quantifying PAD. Findings of this study suggest that abnormal results of noninvasive testing should be viewed as ruling in CLTI, though normal results do not conclusively rule it out.
The parameters with the highest sensitivity rates had disappointingly low specificity rates. Clearly, more accurate noninvasive means to identify CLTI in patients with foot ulcers are needed. This may be through newer testing modalities or further optimization of the performance of existing testing modalities. 29 As illustrated by the impact of foot MAC on sensitivity rates in this study, test results alone may be insufficient; considering clinical characteristics – especially age, diabetes, end-stage renal disease, and smoking history 30 – may also be important to the optimal use of noninvasive testing. 31 Nonetheless, with no access-related complications seen in this series of 100 consecutive angiograms done with ultrasound guidance, the clinical benefit seems to strongly favor a low threshold for diagnostic angiography. Patient characteristics (especially baseline ambulatory ability), hospital resource availability, and monetary costs (especially that incurred directly by the patient) should be considered in the decision for diagnostic angiography as well.
Our team has been liberal in offering angiography to most patients with foot ulcers and either an abnormal pedal pulse exam or abnormal noninvasive test. This study represents a subset of patients presenting to our hospital with foot ulcers, and it is possible that some patients with CLTI seen during this study period did not undergo angiography and were therefore not included in the study. We see fewer young patients than many centers, so the PAD prevalence in the study cohort may be higher at our center than in others. Finally, although our hospital’s population is racially and ethnically diverse, the study sample is almost exclusively comprised of men. We are unaware of prior findings that would suggest a different diagnostic accuracy of noninvasive testing in women; nonetheless, these findings should be corroborated in women with foot ulcers.
Conclusion
In summary, findings from this study suggest that testing to exclude significant PAD most reliably should be ordered as follows: (1) TBI above 0.7 and (2) minimum ABI above 0.9. Abnormal results should be viewed as ruling in PAD. Normal or mildly abnormal maximum ABI numbers viewed as equivocal, not conclusively ruling out the presence of PAD. Angiography should remain an important diagnostic test to identify advanced PAD in patients with foot ulcers, foot infections, and gangrene.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X221147945 – Supplemental material for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X221147945 for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia by Justin Chin-Bong Choi, Jorge Miranda, Erin Greenleaf, Michael S Conte, Marie D Gerhard-Herman, Joseph L Mills and Neal R Barshes in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X221147945 – Supplemental material for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X221147945 for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia by Justin Chin-Bong Choi, Jorge Miranda, Erin Greenleaf, Michael S Conte, Marie D Gerhard-Herman, Joseph L Mills and Neal R Barshes in Vascular Medicine
Supplemental Material
sj-tiff-3-vmj-10.1177_1358863X221147945 – Supplemental material for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia
Supplemental material, sj-tiff-3-vmj-10.1177_1358863X221147945 for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia by Justin Chin-Bong Choi, Jorge Miranda, Erin Greenleaf, Michael S Conte, Marie D Gerhard-Herman, Joseph L Mills and Neal R Barshes in Vascular Medicine
Supplemental Material
sj-tiff-4-vmj-10.1177_1358863X221147945 – Supplemental material for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia
Supplemental material, sj-tiff-4-vmj-10.1177_1358863X221147945 for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia by Justin Chin-Bong Choi, Jorge Miranda, Erin Greenleaf, Michael S Conte, Marie D Gerhard-Herman, Joseph L Mills and Neal R Barshes in Vascular Medicine
Supplemental Material
sj-tiff-5-vmj-10.1177_1358863X221147945 – Supplemental material for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia
Supplemental material, sj-tiff-5-vmj-10.1177_1358863X221147945 for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia by Justin Chin-Bong Choi, Jorge Miranda, Erin Greenleaf, Michael S Conte, Marie D Gerhard-Herman, Joseph L Mills and Neal R Barshes in Vascular Medicine
Supplemental Material
sj-tiff-6-vmj-10.1177_1358863X221147945 – Supplemental material for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia
Supplemental material, sj-tiff-6-vmj-10.1177_1358863X221147945 for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia by Justin Chin-Bong Choi, Jorge Miranda, Erin Greenleaf, Michael S Conte, Marie D Gerhard-Herman, Joseph L Mills and Neal R Barshes in Vascular Medicine
Supplemental Material
sj-tiff-7-vmj-10.1177_1358863X221147945 – Supplemental material for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia
Supplemental material, sj-tiff-7-vmj-10.1177_1358863X221147945 for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia by Justin Chin-Bong Choi, Jorge Miranda, Erin Greenleaf, Michael S Conte, Marie D Gerhard-Herman, Joseph L Mills and Neal R Barshes in Vascular Medicine
Supplemental Material
sj-tiff-8-vmj-10.1177_1358863X221147945 – Supplemental material for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia
Supplemental material, sj-tiff-8-vmj-10.1177_1358863X221147945 for Lower-extremity pressure, staging, and grading thresholds to identify chronic limb-threatening ischemia by Justin Chin-Bong Choi, Jorge Miranda, Erin Greenleaf, Michael S Conte, Marie D Gerhard-Herman, Joseph L Mills and Neal R Barshes in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
