Abstract
The American University of Beirut (AUB)-HAS2 risk index is a recently published tool for preoperative cardiovascular evaluation. It is based on six data elements: history of
Introduction
Vascular surgeries are usually considered intermediate–high risk, with some of them entailing long procedures with significant hemodynamic stress. Furthermore, patients with vascular disease tend to have a high prevalence of comorbidities including diabetes, smoking, renal insufficiency, pulmonary disease, and a very high prevalence of concomitant cardiovascular diseases.1,2 This is why they are usually at a high risk for developing postoperative cardiovascular events.3,4 Thus, preoperative cardiovascular risk assessment is a very essential step in the initial evaluation of vascular patients to estimate their risk and optimize their peri-operative management.5,6 Several risk models have been derived and utilized for such an evaluation. Table 1 lists the most commonly used of these indices.1,2,7–14 We have recently reported a new risk index that is simple to acquire and has a powerful discriminatory ability to predict cardiovascular events post-noncardiac surgery.10–12 The AUB-HAS2 cardiovascular risk index was derived from a prospectively enrolled cohort of 3284 patients undergoing noncardiac surgery at the American University of Beirut (AUB) and it was validated in the large American College of Surgeons National Surgical Quality Improvement Program (ACS NSQIP) database.
15
It is based on six easily acquired data elements: history of
Risk indices commonly utilized in vascular surgery.

BMI, body mass index; COPD, chronic obstructive pulmonary disease; MI, myocardial infarction ; NSQIP, National Surgical Quality Improvement Program.
Methods
The study population consisted of 90,476 vascular surgeries registered between 2008 and 2012 in the ACS NSQIP database. 15 Datasets after 2012 were not included in our study because they did not capture cardiac history on patients, which is one of the essential elements in the AUB-HAS2 index. The performance of the AUB-HAS2 index was studied in seven vascular surgery groups: carotid endarterectomy (CEA), open abdominal aortic aneurysm surgical repair (OAAA), endovascular aortic aneurysm repair (EVAR), supra-inguinal bypass (SUPRA), infra-inguinal bypass (INFRA), lower extremity thrombo-endarterectomy (LE-TE), and lower extremity angioplasty (LE-angioplasty). Each patient was assigned a modified AUB-HAS2 score of 0, 1, 2, or > 2 based on the number of data elements present. The score elements are: history of heart disease, symptoms of heart disease (angina or dyspnea), age ⩾ 75 years, anemia (hemoglobin < 12 mg/dL), and emergency surgery (the vascular surgery element present in the original AUB-HAS2 index was not included since all of the patients had vascular surgery to begin with). Patients were designated as having history of heart disease if they had history of prior myocardial infarction (MI), coronary angioplasty, cardiac surgery, heart failure, atrial fibrillation, or moderate/severe valvular disease confirmed by echocardiography. The primary outcome measure was death, MI, or stroke at 30 days after surgery. Death was defined as mortality secondary to any cause occurring during the intraoperative period or at any point within 30 days after the principal operative procedure. MI was defined by ECG changes indicative of an acute MI (one or more of the following three: ST elevation > 1 mm in two or more contiguous leads, new left bundle branch, or new q-waves in two or more contiguous leads), or new elevation in troponin greater than three times the upper level of the reference range in the setting of suspected myocardial ischemia. Stroke was defined as the new occurrence of a motor, sensory, or cognitive dysfunction which persists for more than 24 hours. In accordance with the AUB’s guidelines (which follow the US Code of Federal Regulations for the Protection of Human Subjects), consent for our analysis by institutional review board approval was waived because the data were collected as part of a quality assurance activity.
The ACS NSQIP database
The ACS NSQIP is a nationally validated, risk-adjusted, outcomes-based program to measure and improve the quality of surgical care. It consists of a large multicenter database that collects data on patients undergoing major surgical procedures from more than 300 participating sites on more than 150 variables, including preoperative risk factors, intraoperative variables, and 30-day postoperative mortality and morbidity outcomes. The sites are mainly from the USA, but other international hospitals are also part of the program, including the AUB Medical Center. The database is updated on a yearly basis, and access to the updated data is granted for all participating hospitals. All NSQIP participating hospitals use an American College of Surgeons (ACS)-validated, systematic sampling protocol. Each hospital assigns a trained Surgical Clinical Reviewer (SCR) to collect preoperative through 30-day postoperative data on randomly assigned patients. The data are subject to regular inter-rater reliability audits to assess the quality. Data are entered online in a HIPAA-compliant, secure, web-based platform that can be accessed 24 hours a day. Surgeries are entered in the database using the International Classification of Diseases (ICD) codes. The ACS provides SCR training, ongoing education opportunities, and auditing to ensure data reliability. A surgeon champion assigned by each hospital leads and oversees program implementation and quality initiatives. The SCR and surgeon champion at each site adjudicate clinical events, which are audited on a regular basis along with all variables by the central ACS NSQIP administration.
Statistical analysis
Descriptive analysis was performed and presented in the respective tables. Categorical variables are presented as number and percentage and continuous variables as mean ± SD. The comparisons among the different vascular groups in Table 2 were performed using the chi-squared test for categorical variables and the analysis of variance (ANOVA) test for continuous ones. The performance of the AUB-HAS2 index was studied by measuring and comparing the event rates among the different score groups (0, 1, 2, and > 2) in the various surgical groups. The comparisons among the different AUB-HAS2 groups were performed using the chi-squared test for categorical variables. The Cochran–Armitage test for trend was used to evaluate the trend in the proportions of the outcome across the levels of the AUB-HAS2 index. The discriminatory power of the AUB-HAS2 index was further studied by constructing receiver operating characteristics (ROC) curves and measuring the respective area under the curve (AUC). SAS software, version 9.4 (SAS Institute Inc., Cary, NC, USA) was used for data management and analyses. Statistical significance was set at the 0.05 alpha level.
Clinical characteristics of the vascular surgery groups.

Values are mean ± SD or %.
ASA, American Society of Anesthesiology; BMI, body mass index; CEA, carotid endarterectomy; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; Cr, creatinine; CVD, cardiovascular disease; EVAR, Endovascular aneurysm repair – endovascular repair; h/o, history of; INFRA, infra-inguinal bypass; LE-Angio, lower extremity angioplasty; LE-TE, lower extremity thrombo-endarterectomy; MI, myocardial infarction; OAAA, open abdominal aortic aneurysm repair; PCI, percutaneous coronary intervention; SUPRA, supra-inguinal bypass.
Results
Table 2 shows the baseline clinical characteristics of the patients enrolled in the different vascular surgery groups. Overall, 83% of patients had hypertension and 36% had history of heart disease. 87% of the surgeries were done under general anesthesia and more than 90% had an American Society of Anesthesiology (ASA) class of ⩾ 3. AAA patients had the highest prevalence of male sex (79%) and COPD. Diabetes was most prevalent in INFRA and LE-angioplasty patients. CEA patients had the highest prevalence of prior stroke (24%).
Table 3 and Figure 1A show the incidence of the primary outcome in each of the groups. The lowest 30-day event rate was seen in the CEA group (3.3%) and the highest in the OAAA group (13.1%). The CEA group had the highest stroke rate (1.7%). Overall, patients with an AUB-HAS2 score of 0 had an event rate of less than 3%, except for the SUPRA group, which had an event rate of 3.8%. On the other hand, most of patients with a score > 2 had a high event rate around 10% or more, except for the CEA patients who had an event rate of 6.8%. Figure 1B compares the event rates among the different AUB-HAS2 groups within each type of surgery. In all vascular surgery groups, there was a gradual and significant increase in risk as the AUB-HAS2 index increased. Within each surgical subgroup, comparisons of the primary outcome measure (death, MI, or stroke) between all the AUB-HAS2 score groups were all statistically significant with a p < 0.0001.
Comparison of outcomes among AUB-HAS2 score groups.

Values are n (%).
CEA, carotid endarterectomy; EVAR, endovascular aneurysm repair; INFRA, infra-inguinal bypass; LE-Angio, lower extremity angioplasty; LE-TE, lower extremity thrombo-endarterectomy; MI, myocardial infarction; OAAA, open abdominal aortic aneurysm repair; SUPRA, supra-inguinal bypass.
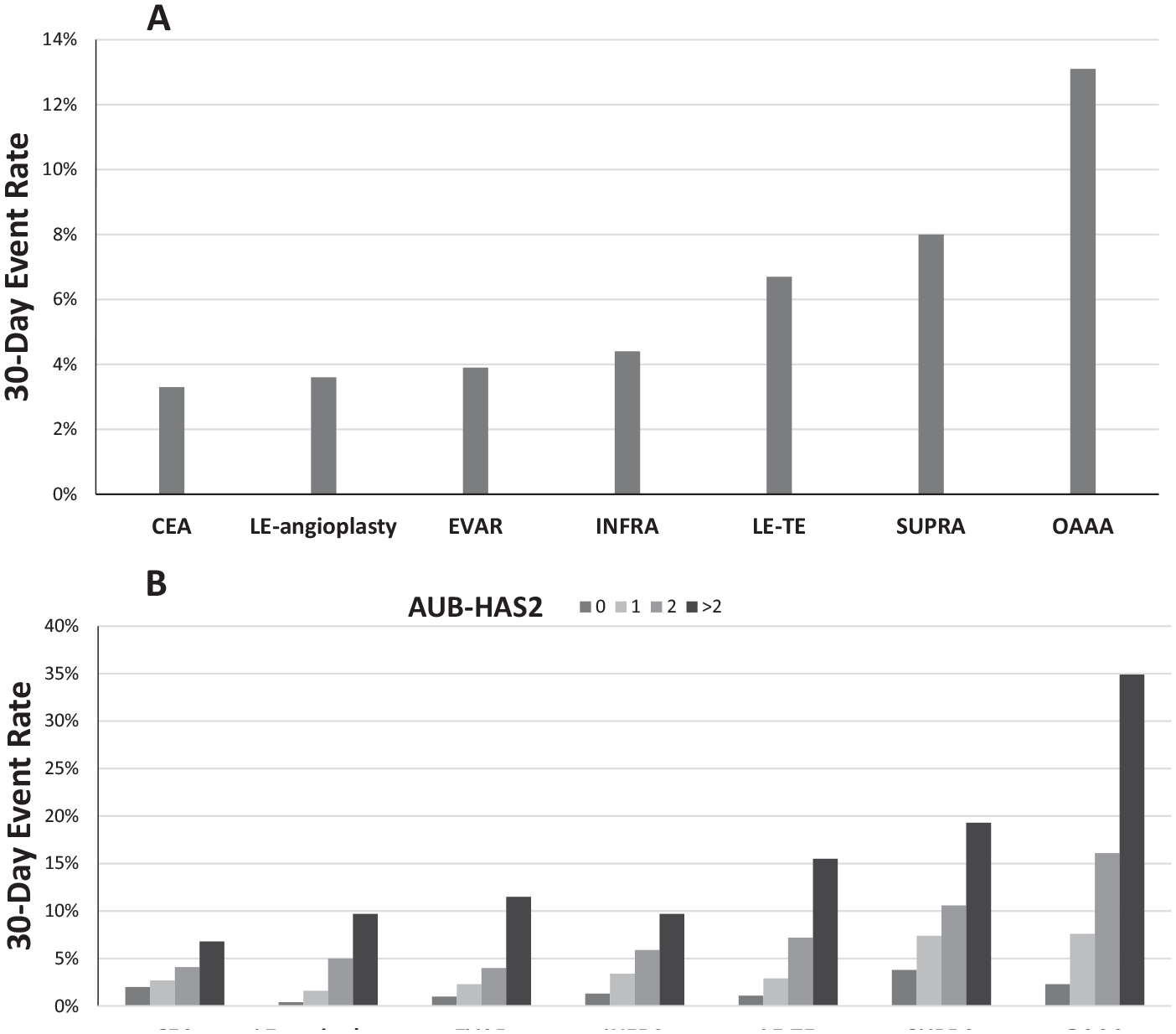
The 30-day event rates (death, MI, or stroke) for the different types of vascular surgeries (A), and comparison of these event rates among the different AUB-HAS2 score groups within each type of vascular surgery (B).
The performance of the AUB-HAS2 index was further studied by constructing ROC curves in each of the groups and measuring the AUC (Table 4). The AUC in the whole group was 0.71 and it ranged from 0.60 in CEA patients to 0.75 in OAAA and LE-angioplasty patients.
AUCs of the AUB-HAS2 score in the various vascular surgery groups.

AUC, area under the curve.
Discussion
In spite of the major advances in surgical and anesthesiology techniques, vascular surgery continues to pose a high risk for cardiovascular complications in the short and long term.1–4 Preoperative cardiovascular evaluation in those patients is routinely performed as recommended by the guidelines to assess their risk and guide their management.5,6 Over the past two decades, multiple indices have been derived for this purpose either for vascular patients in general or for specific operations such as carotid endarterectomy or aortic aneurysm repair (Table 1).1,2,7–13 The AUB-HAS2 risk index was recently derived and validated for the risk assessment of noncardiac surgeries.10–12 It had an excellent discriminatory power and was able to stratify patients into low, intermediate, and high-risk groups. The current study extends the validation of the AUB-HAS2 index to a wide spectrum of vascular surgeries where it was also able to stratify the risk in the majority of patients into low (< 3%, score of 0), intermediate (3–10%, score of 1–2), and high (> 10%, score > 2). Although each type of vascular surgery has its own peculiarities in terms of risk factors and potential cardiovascular events, and therefore might be better assessed by its own specific risk model, having one simple comprehensive index with good discriminatory power would be very helpful and practical for utilization by physicians and house staff in their busy daily practice. The objective of this report was to study the performance of the AUB-HAS2 index in a comprehensive and wide variety of vascular surgeries that are representative of current practice. Although lower extremity angioplasty and EVAR are percutaneous procedures, they still portend significant risk. As a matter of fact, the 30-day event rates of these procedures in our study were 3.6% and 3.9%, respectively. The AUB-HAS2 score was very effective in stratifying risk in these percutaneous procedures into low, intermediate, and high similar to open surgeries. It is important to note that the AUB-HAS2 index does not have creatinine in its data elements, which is present in many other risk indices for vascular or noncardiac surgery in general. Our index, however, has anemia as one of its elements, which is not present in the other indices. Anemia has been noted to be an important marker of events across the spectrum of patients undergoing cardiovascular interventions or surgery.5,6 It seems that there might be an interaction between anemia and renal function in which one of them might replace the other in risk prediction.
The Vascular Study Group Cardiac Risk Index (VSG-CRI) is a commonly used index for the preoperative cardiac assessment in vascular patients. 1 It was derived from a large group of patients undergoing multiple surgeries (CEA, INFRA, EVAR, and OAAA). The VSG-CRI outcome measures consisted primarily of cardiac complications excluding mortality. This index is more complex than the AUB-HAS2 index, consisting of nine elements that are listed in Table 1. Three of those elements (age, coronary artery disease, congestive heart failure) are also present in the AUB-HAS2 index. However, although it is derived from a vascular surgery cohort (compared to a noncardiac surgery cohort of the AUB-HAS2 index) and consists of more data elements (nine compared to five), it had the same exact discriminatory power with an AUC of 0.71, albeit in two different databases but both consisting of large multicenter registries. Furthermore, the AUC of the AUB-HAS2 index, specifically in OAAA patients (0.75), was higher than that of the VSG-CRI (0.68). The advantage of the AUB-HAS2 index, other than its simplicity, is that it is more comprehensive in its outcome measures, which include, in addition to MI, both mortality and stroke, which are particularly relevant to patients undergoing vascular surgery.
Multiple other models were derived either for one specific outcome measure, such as myocardial infarction, or for one specific surgery, such as CEA or AAA. The Vascular Quality Initiative Cardiac Risk Index (VQI-CRI) was derived to predict postoperative MI. 2 It has eight elements (listed in Table 1) with age, coronary artery disease, and congestive heart failure being common to the AUB-HAS2 index. The discriminatory ability of the index, as measured by the AUC, was similar to the AUB-HAS2 index with a range of 0.62 to 0.72 across the various vascular surgeries. Ramanan et al. derived a specific index predicting mortality after OAAA. 13 It consisted of six weight-adjusted data elements (Table 1). Age and dyspnea are common elements with the AUB-HAS2 index. Its discriminatory power (AUC of 0.72) was also similar to our index. Paradoxically, the index did not have any elements related to cardiac disease. Also, a ‘low’ 30-day mortality rate was set at a < 7% cut-off, which is higher than the normally accepted low mortality risk. The AUB-HAS2 index had the lowest performance in the CEA group, with an AUC of 0.60. In general, most derived indices for vascular surgery have a low performance in CEA patients. Gupta et al. derived a CEA-specific index from a group of 17,692 asymptomatic patients from the NSQIP database. 14 The index consisted of six weight-adjusted data elements consisting of age, dyspnea, angina, COPD, functional status, and previous peripheral revascularization. Although it was derived from a CEA population with weight-adjusted elements, it had a modest discriminatory power like the AUB-HAS2 index, with an AUC of 0.64. Volkers et al. evaluated more than 20 CEA prediction models in an independent external database and found that they all had weak–modest discriminatory performance, with an AUC ranging from 0.49 to 0.64. 16 One of the possible explanations for this low performance is the lack of high-risk patients in the derivation cohorts either because clinicians are not operating on them or because they are sending them to carotid stenting instead, and thus it would be difficult to identify them in any risk prediction model for surgical CEA.
In general and in routine clinical practice, there are two main objectives from preoperative cardiovascular risk assessment: first, is to provide patients as well as treating physicians (surgeon, anesthesiologist, cardiologist, housestaff, etc.) an estimation of the postoperative cardiovascular risk. The second objective is to try to use these data to improve patients’ outcomes. The first objective is essential for patients to be informed about their risk and thus provide a real well-informed consent. In this regard, having three categories of risk (low, middle, and high) would be more helpful and represents a middle ground between a very broad risk assessment (low, high) that is used by the ACC/AHA guidelines 5 and a very quantitative and specific one where mere numbers are provided to patients, as done in other risk indices. 9 The second important objective of preoperative risk assessment is to utilize the data to improve outcomes. This is an area where there is a paucity of data. This is reflected in the ACC/AHA guidelines themselves, 5 where most of the recommendations regarding patient management and further testing based on risk assessment carries a class II level, being primarily based on consensus and clinical judgement rather than large randomized clinical trials. Nevertheless, this is a very relevant question and would be important to address in large prospective studies.
Study limitations
Our study has several strengths, the most important of which is that it validates a prospectively derived risk index in a large external dataset with a wide spectrum of vascular surgery groups. Several of the other commonly used indices were found to have significantly lower discriminatory power when applied in external datasets other than the ones in which they were derived.16,17 On the other hand, one of our study limitations is that the NSQIP dataset is a comprehensive registry of all types of surgery and thus it does not record some data elements that might be significant for a specific type of surgery such as vascular surgery. Hence, we could not perform direct comparison with other indices because some of their data elements are not coded in the NSQIP database.
Conclusion
Our study extends the validation of the AUB-HAS2 index to a broad spectrum of vascular surgeries. The index is simple and can quickly and effectively stratify risk in the majority of those patients into low (< 3%, score of 0), intermediate (3–10%, score of 1–2), and high (> 10%, score > 2). Further prospective validation is warranted to define its role in the preoperative cardiovascular evaluation of patients undergoing these surgeries.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
