Abstract
To compare the disappearance of the telangiectasias after sclerotherapy with 75% glucose (HG) versus 0.2% sodium tetradecyl sulfate (STS). This prospective, randomized clinical trial compared the results of sclerotherapy of the telangiectasias with HG and STS. The primary efficacy end point was telangiectasia disappearance within 14, 28, 42, and 56 days following treatment. The clearing of the vessels was assessed using a six-point scale (from 0 to 5). A total of 159 women were treated (81 in the STS group; 78 in the HG group). The median score of the vessels clearing (IQR) was significantly lower in the STS group than in the HG group: 3 (2–4) versus 4 (3–5) after 56 days, p < 0.001. Pigmentation was frequently observed in the STS group (38.3% vs 2.6%; p < 0.001). In conclusion, throughout the entire follow-up period, sclerotherapy of telangiectasias with glucose was significantly superior to that with STS. Moreover, pigmentation and intravascular clots frequently occurred with STS treatment.
Introduction
Telangiectasias are primarily treated to improve one’s aesthetics. Sclerotherapy is the gold standard for treating telangiectasias using detergents such as polidocanol (POL) or sodium tetradecyl sulfate (STS). However, many sclerotherapy-related minor adverse events have been reported, including pigmentation and telangiectatic matting, which significantly worsen the aesthetic treatment results achieved with detergents.1–6 According to some studies, a very low frequency of pigmentation and telangiectatic matting occurs when sclerotherapy with hypertonic glucose (HG) is employed.7,8 We hypothesized that the less aggressive sclerosing agent, 75% glucose, may be more effective and safer in the treatment of telangiectasias.
Methods
The study protocol was approved by the regional ethics committee (No. 3, 11.27.2017, Ethics Committee of the Medical Faculty of Derzhavin Tambov State University). The trial was registered at the Russian Registry of Treatment of Chronic Venous Diseases (RRT CVD); ID: VRCVD 1.005.
This study was designed as a prospective, randomized, comparative clinical trial in an outpatient department specializing in venous disease (~500 sclerotherapy procedures per year) at three multidisciplinary clinics.
Patients
The inclusion criterion was women aged between 18 and 70 years with telangiectasias unrelated to the reticular veins in C1 patients observed between September 2018 and June 2019.
Patients with C2–C6 venous insufficiency and those who had undergone sclerotherapy within 12 weeks on the ipsilateral leg or within 4 weeks on the contralateral leg, laser treatment, or surgery were excluded. Patients with C1 disease who had telangiectasias communicating with saphenous or reticular veins were excluded. Other exclusions were patients with a history of acute superficial or deep vein thrombosis, leg oedema, arterial occlusive disease, clinically relevant electrocardiogram abnormalities, acute systemic disease, poor general health, severe generalized infection, allergic reaction to STS or corn, history of high ferritin, haemochromatosis, iron storage disorders or those who took minocycline, hydroxychloroquine or iron supplements, acute febrile states, and symptoms of microangiopathy, neuropathy or inflammatory skin disease in the treatment area. Each participant provided written informed consent prior to procedure initiation and participation in the study.
Procedures
A standardized detailed ultrasound protocol was performed to exclude underlying reticular and saphenous vein reflux.
The treatment areas were captured with a high-definition digital camera (Samsung Galaxy S8). The photos were taken by the investigator. To ensure unmistakable follow-up of the exact location of the treatment area and maintain the same image quality throughout the study, images were captured at a distance of 0.5 metres from the camera according to anatomical features (birthmarks or other marks on the skin) in a room under artificial light with the blinds shut to exclude the impact of natural light. The maximal diameter of the telangiectasias was measured by dermatoscopy and they were divided into two groups: small (< 0.5 mm) or large (⩾ 0.5 mm). Thereafter, eligible patients were allocated by a computerized randomization procedure using Mersenne Twister to receive either 0.2% STS (STS group) or 75% HG (HG group) (1:1).
Since the glucose was viscous, the sclerosing agent was administered with a 1-mL syringe through a 30 G needle. To provide equal conditions, a 1-mL syringe and a 30 G needle were used in both groups. Injections were performed until complete whitening of the vessel was achieved. All sclerotherapy procedures were carried out by one physician from each clinic. After the procedure, elastic compression stockings with a pressure of 23–32 mmHg (RAL-GZ 387; stockings were made by different hosiery companies, i.e., Sigvaris, Medi, and Venosan) were applied for 24 h. Patients wore the stockings during the day for 5 days.
An 8-week follow-up period was employed in the present study. Each patient came back to the clinic every 2 weeks. During control visits, the treatment areas were captured at a distance of 0.5 metres from the camera according to anatomical features in a room under artificial light with the blinds shut. Then the images were made and sent to the blinded experts, who were phlebologists working at other clinics and had great experience in sclerotherapy. The primary efficacy end point was the disappearance of the telangiectasias within 14, 28, 42, and 56 days after treatment. The clearing of the vessels was assessed on a six-point scale (from 0 to 5 points): 5, total disappearance of telangiectasias (100% efficacy) – extremely good improvement; 4, disappearance of ~80% of telangiectasias – very good improvement; 3, disappearance of ~60% of telangiectasias – good improvement; 2, disappearance of ~40% of telangiectasias – fair improvement; 1, disappearance of ~20% of telangiectasias – minimal improvement; and 0, no changes – no improvement. The results were estimated by two independent and blinded medical experts using images. Safety outcomes were immediately analysed post-treatment and after 14, 28, 42, and 56 days. These outcomes included side effects such as visual disturbances, headache and migraine, anaphylaxis, skin necrosis, pigmentation, intravascular clots, telangiectatic matting, and bruising.
Continuous variables were expressed as median and IQR, while categorical variables were expressed as absolute and relative frequencies. The estimate of the significance of the correlation between the variables was achieved with the Spearman rank correlation coefficient (rs). The qualitative characteristics of samples were tested with the χ2 test. For group comparisons of the quantitative values, the Mann–Whitney U test was used. Analysis was carried out using the website ‘Social Science Statistics’ (https://www.socscistatistics.com/tests/). A p-value less than 0.05 was considered to indicate statistical significance.
Sample size was calculated with the online calculator ‘Sample Size Calculator’ on the website www.calculator.net. The selected parameters were CI 5% and CI 80%. The sample size must include at least 158 participants. To account for the possibility of patient dropout, 5% of participants was added to the sample size. The total calculated sample size was 166 subjects. To achieve adequate separation among the three clinics, the number of subjects was increased to 168.
Results
A total of 168 women were randomized to receive either 0.2% STS (STS group: 84 patients) or 75% HG (HG group: 84 patients). Nine patients prematurely terminated their participation in the study and were excluded from the analysis. The most frequent causes of termination were noncompliance with the study protocol, personal reasons, or loss to follow-up. As a result, 81 patients in the STS group and 78 patients in the HG group completed the study and were analysed. Both groups had similar characteristics, as shown in Table 1. The maximal diameter of the telangiectasias was 1 mm in each group. The outer thigh was identified as the most frequent treatment area (STS group: 30%, HG group: 41%).
Baseline characteristics of the study participants.
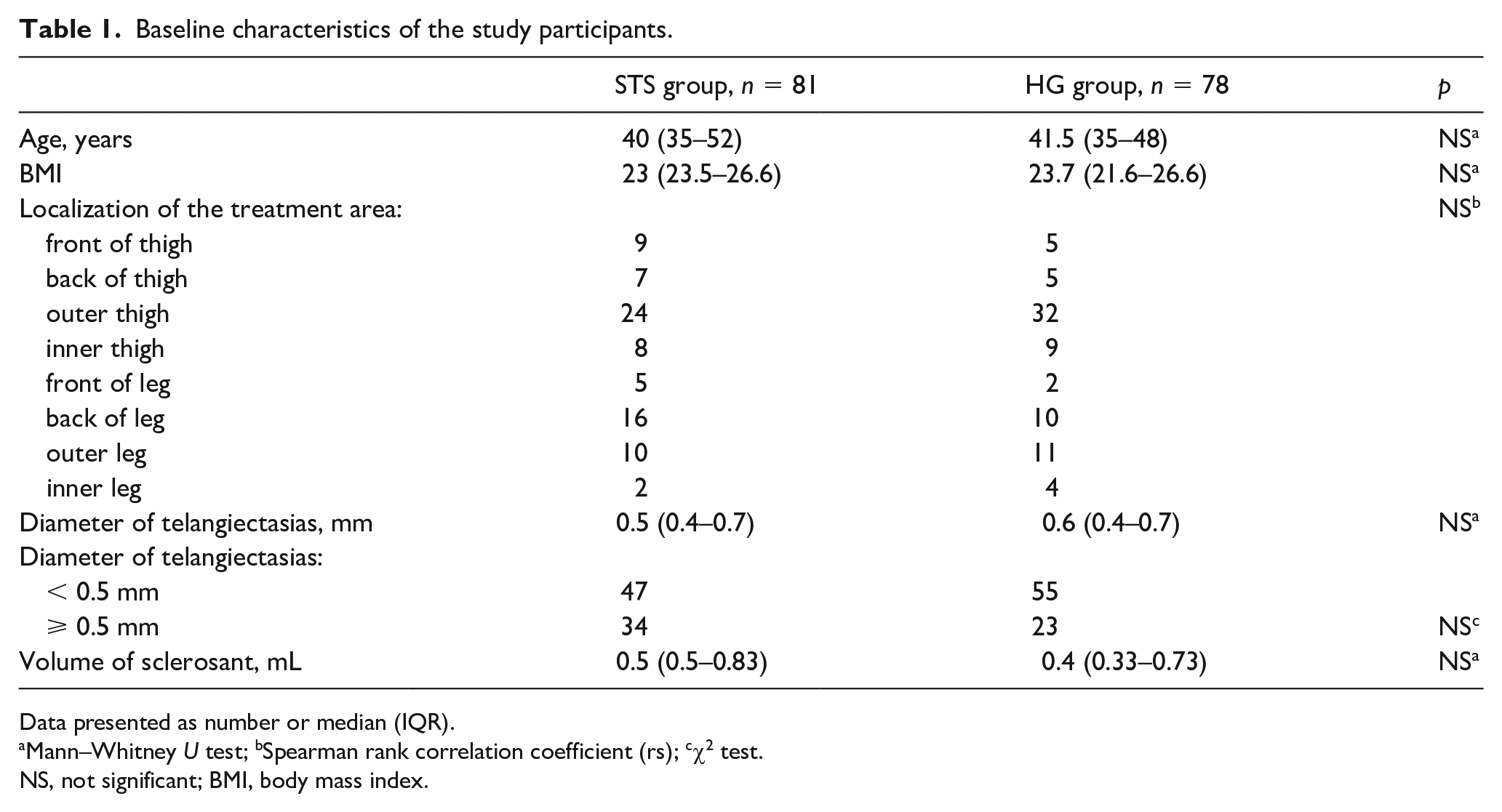
Data presented as number or median (IQR).
Mann–Whitney U test; bSpearman rank correlation coefficient (rs); cχ2 test.
NS, not significant; BMI, body mass index.
The median score of the vessels clearing (IQR) was significantly lower in the STS group than in the HG group: 2 (0–3) versus 4 (2–4) after 14 days, p = 0.00006; 2 (1–4) versus 4 (3–4) after 28 days, p = 0.00044; 3 (2–4) versus 4 (3–5) after 42 days, p = 0.00199; and 3 (2–4) versus 4 (3–5) after 56 days, p = 0.00017. Independent experts were reporting the same values. A strong correlation was identified between the estimates of the experts (r = 0.76). At the first visit, 15 patients (19%) in the HG group had excellent results (5 points); however, only one patient (1.2%) in the STS group had such a result, p < 0.001 (Figure 1). At the end of the follow-up period, the results were assessed as good and excellent (4 and 5 points) in 31 patients in the STS group (38.3%) and 50 patients in the HG group (64%), p = 0.002. At the end of the follow-up period, the results were deemed poor (0 and 1 points) in 30 patients (37%) in the STS group and 15 patients (19%) in the HG group, p = 0.012 (Figure 2).
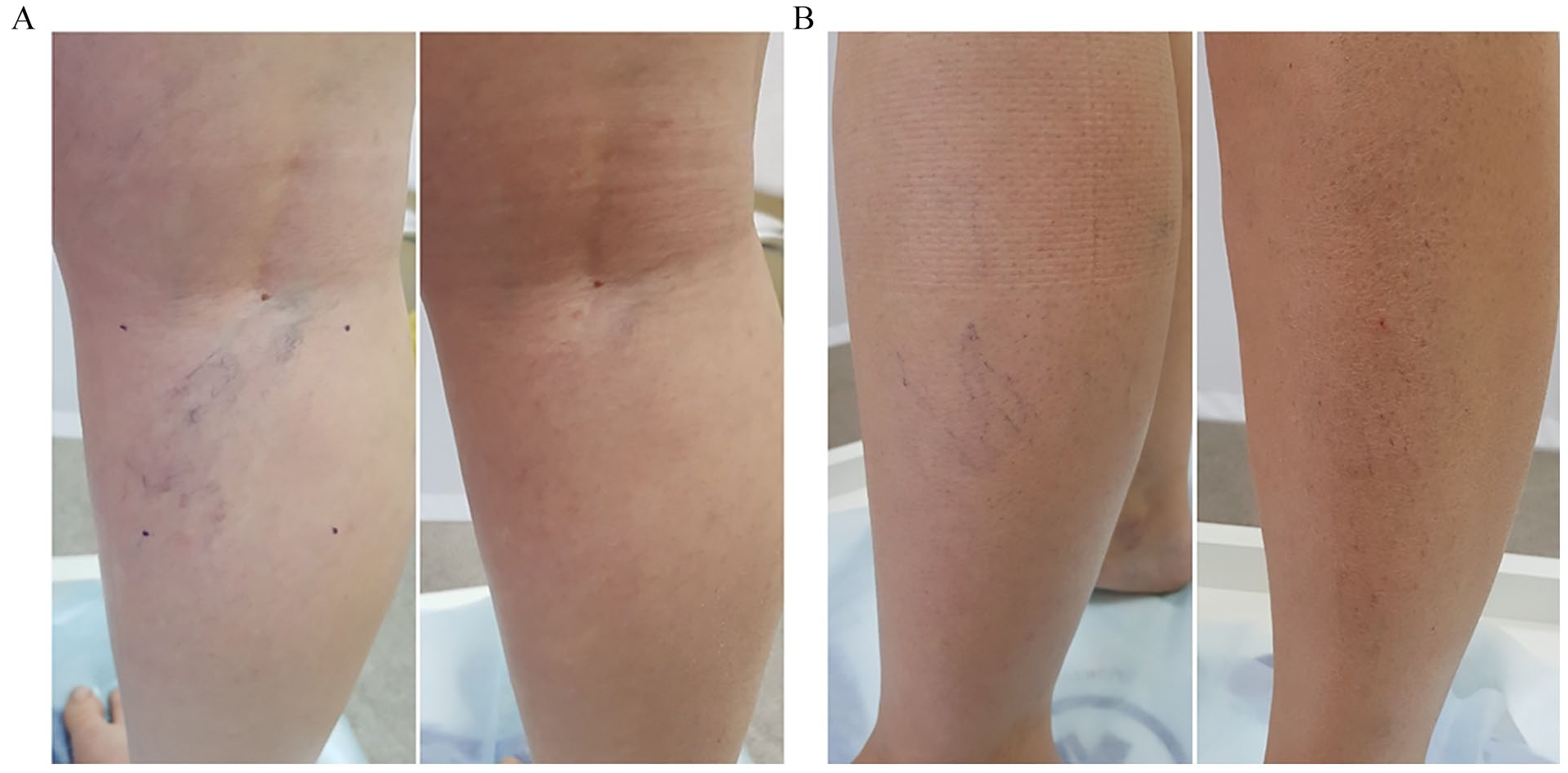
Example image of score 5 within 14 days after treatment with (A) 75% HG and (B) 0.2% STS.
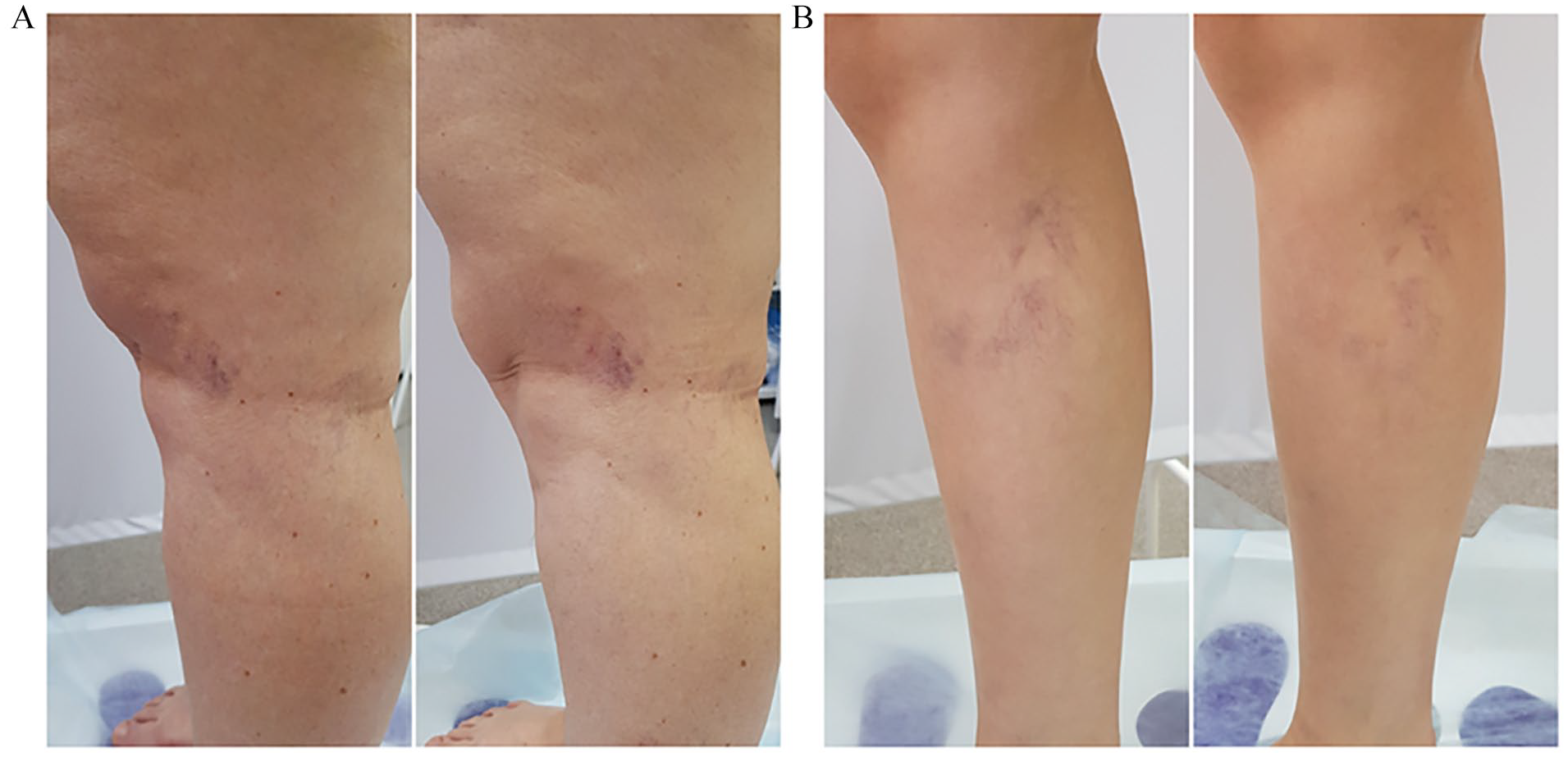
The score 0 within 56 days after treatment with (A) 75% HG and (B) 0.2% STS.
During the follow-up period, there was no significant association between the score and the diameter of the telangiectasias for patients in the STS group. For those in the HG group, no significant association was found after 14 and 56 days. A better result was achieved after sclerotherapy of small telangiectasias (i.e. < 0.5 mm) with glucose after 28 and 42 days (Table 2). There was no correlation between the score and localization of treatment area.
The efficacy end points achieved during the follow-up period.
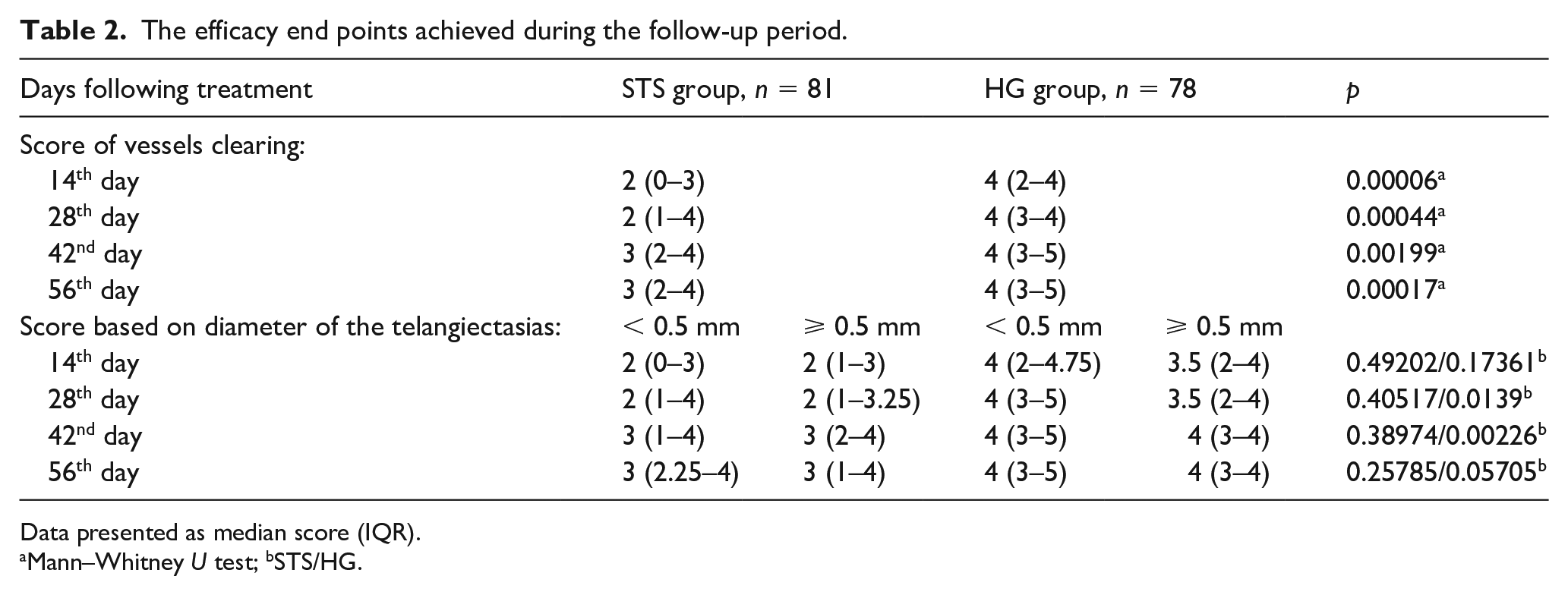
Data presented as median score (IQR).
Mann–Whitney U test; bSTS/HG.
Following sclerotherapy, more pigmentation was frequently observed in patients in the STS group than the HG group (38.3% vs 2.6%, p < 0.001) at the end of the follow-up period. Additionally, pigmentation was identified in 18 of the 34 patients (52.9%) with large telangiectasias and 13 of the 47 patients (27.7%) with small telangiectasias in the STS group (p = 0.021). We recognized many cases of intravascular clots (23.5%) following injection with STS. Following treatment with STS, one patient (1.2%) experienced a headache (Table 3). However, no serious adverse events were observed following sclerotherapy in both groups.
Sclerotherapy-related minor adverse events.
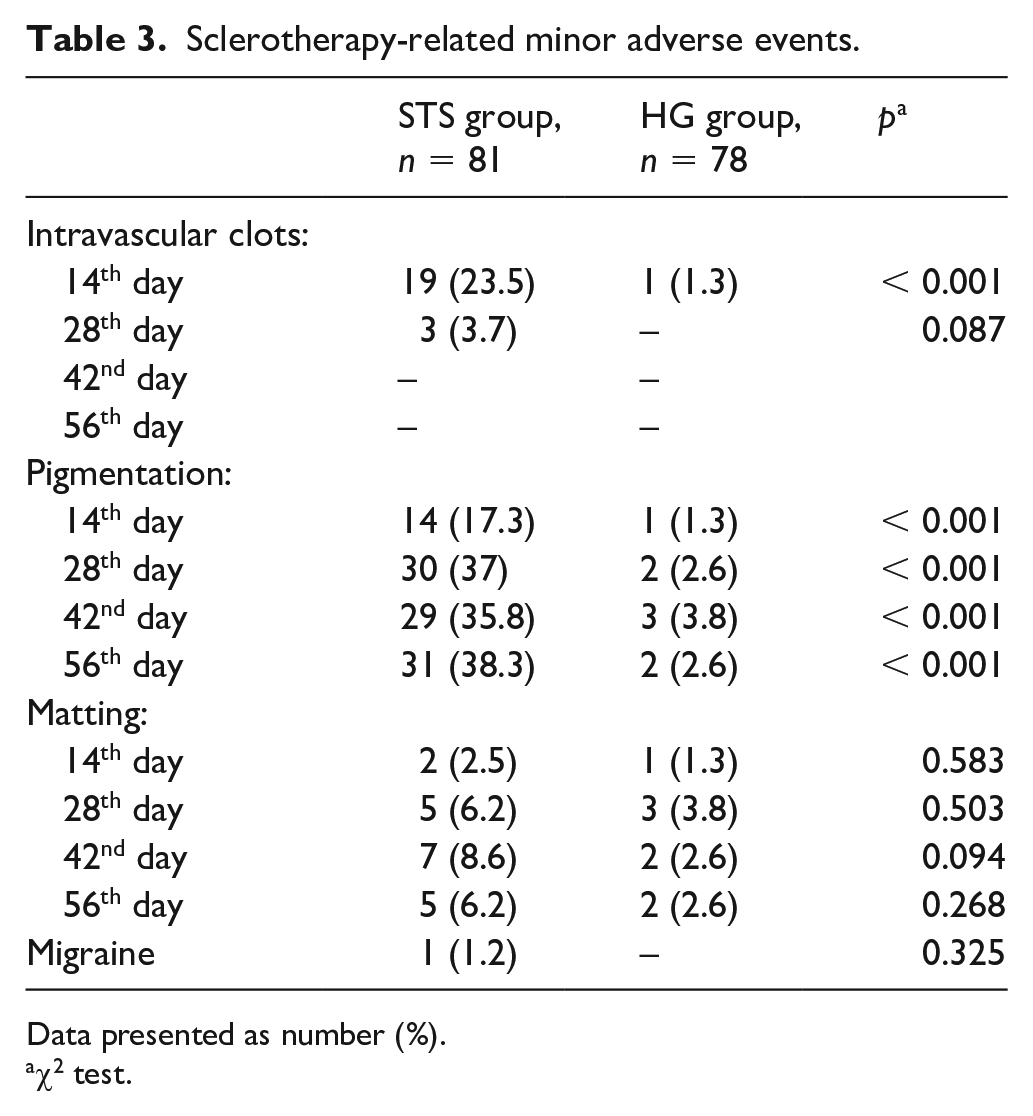
Data presented as number (%).
χ2 test.
Discussion
During telangiectasia treatment, clinicians primarily focus on improving one’s aesthetics. As a result, a treatment that can cause excellent results in the earliest attainable timeframe is warranted. However, many sclerotherapy-related minor adverse events, including pigmentation and telangiectatic matting, cause a significantly worse aesthetic treatment outcome when a detergent is employed.1–6 In recent studies, a more aggressive or concentrated agent was found to more frequently induce matting and pigmentation. Rabe et al. 2 conducted a double-blind, randomized, comparative clinical trial with 316 subjects administered either 0.5% POL, 1% STS, or placebo as a treatment agent for telangiectatic veins. These researchers detected matting and pigmentation in 21% and 78% of patients treated with STS and 14% and 65% of those administered POL, respectively. However, we believe that the 1% STS used to treat sclerotherapy telangiectasias is a very high concentration. By treating patients with telangiectatic veins with 0.5% POL or long-pulsed 1064 nm Nd:YAG laser, Parlar et al. 3 observed a high frequency of matting with both therapies (prevalence, 20%). Moreover, pigmentation was observed in 67.9% of patients treated with 0.5% POL.
In a histological study, Bush and Bush 6 demonstrated that 0.15% STS and 0.31% POL caused total endothelial lysis and substantial changes when sclerotherapy of relatively large telangiectasias (0.8–1 mm) was carried out. Additionally, smooth muscle wall alterations were observed; however, these were mild. When the STS concentration was > 0.2%, muscle wall alterations were marked and, in some cases, considerable wall thinning was noted. A POL concentration > 0.31%, or an STS concentration > 0.2%, consistently caused intraluminal clumping of cellular debris and visible red cells in a 1-mm telangiectasia. Based on our findings, too, 0.2% STS is an extremely aggressive agent on telangiectasias. Although pigmentation was observed in both treatment groups, its frequency was greater in patients treated with 0.2% STS, with a significant difference between the treatment groups (38.3% vs 2.6%; p < 0.001). Pigmentation was dependent on the diameter of the telangiectasias and was frequently observed in patients with large telangiectasias (52.9% vs 27.7%; p = 0.021).
Early studies performed in 1969 demonstrated that hypertonic solutions cause dehydration of endothelial cells through osmosis and did not strongly disrupt the vessel wall.9,10 Some studies have estimated the effect of HG as a sclerosing agent.7,8,11 These studies revealed that 75% HG does not cause pigmentation and matting when it is injected into telangiectasias. However, this finding was not confirmed in the present study. In fact, we identified three matting cases and three pigmentation cases following glucose injection. However, a dark pigmentation was not recognized. There was no skin necrosis, bruising, visual disturbance, headache, or anaphylaxis in patients treated with 75% HG.
The findings presented herein clearly demonstrate that over the entire follow-up period, HG was more effective at eliminating telangiectasias than the detergent, as evidenced by consistently higher vessel clearing scores and significantly fewer intravascular clots and less pigmentation. Within 14 days, we recognized significant disappearance of the telangiectasias following treatment with HG and many cases of intravascular clots (23.5%) following injection with STS; the latter finding caused the worse result and served as the main reason for the increased frequency of pigmentation. Nonetheless, the result (disappearance of telangiectasias) improved on day 56 in both groups and a statistical difference remained between the groups.
Because 75% HG is a very mild sclerosing agent, the elimination of all telangiectasias could not be achieved. However, this issue could be resolved by reintroducing this agent into previously treated vessels. A further study with a different design is warranted to confirm this hypothesis.
Study limitations
Some limitations of the present study should be emphasized. The treatment was not blinded because of the different consistencies of the sclerosing agents. This study did not include a sham-procedure placebo group. The 3-week compression therapy was not used in this study. 12 We did not use any devices for visualization of the potential residual patent veins. The conclusions relied completely on subjective assessment.
Conclusions
Throughout the entire follow-up period, we found that sclerotherapy of telangiectasias with 75% HG caused a significantly superior result compared with 0.2% STS in the selected study population. However, both treatments were recognized to be safe and no serious adverse events were observed in either group. Nonetheless, in the STS group, pigmentation, intravascular clots, and matting were identified as the most common minor adverse events.
Footnotes
Acknowledgements
We would like to thank Igor Zolotukhin, a cardiovascular surgeon, Professor at the Russian Academy of Sciences, and Professor in the Department of Faculty Surgery (Moscow, Russia), and Matvey Parikov, a cardiovascular surgeon at the Innovative Vascular Centre (Saint Petersburg, Russia) for their help in assessing the clearing of vessels.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
