Abstract

What is VITT?
The pandemic caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has led to the rapid development of highly effective vaccines, which have proven to be the most important tool in combating the coronavirus disease 2019 (COVID-19) pandemic. Initial clinical trials reported no major adverse events beyond rare cases of anaphylaxis. As of May 19, 2021, over 1.4 billion vaccine doses have been administered. 1
As a result of the remarkable increase in COVID-19 vaccine administration, new reports of adverse events were reported. In particular, thrombosis in atypical vascular beds and thrombocytopenia were reported following administration of ChAdOx1 nCov-19 (AstraZeneca), a recombinant chimpanzee adenoviral vector encoding the spike glycoprotein of SARS-CoV-2, and Ad26.COV2.S (Johnson & Johnson/Janssen [JJ]), a recombinant adenovirus type 26 vector encoding the SARS-CoV-2 spike glycoprotein. This syndrome has been termed ‘vaccine-induced immune thrombotic thrombocytopenia’ (VITT).2,3 The exact incidence of VITT appears to be extremely low. Published estimates have been extrapolated from small case series and were initially estimated at one per 100,000 individuals. 4
How do you recognize VITT?
VITT is characterized by thrombosis (arterial or venous), thrombocytopenia, and a positive immunoassay for antiplatelet factor 4 (PF4). Symptoms can occur between 4 and 28 days (peak time for initial symptoms is 6–14 days) following AstraZeneca or JJ COVID-19 vaccination. 5 Thrombotic events have typically been reported in unusual locations such as cerebral venous thrombosis or splanchnic vein thrombosis (e.g., portal, mesenteric veins). It has also been reported that patients have presented with extremity deep vein thrombosis, pulmonary embolism, and arterial thrombosis. Afflicted patients were typically younger than the age of 50, and more than two-thirds were female. 5 Clinical symptoms that should trigger consideration of VITT include severe and persistent headache, abdominal pain, back pain, leg swelling or pain, and chest pain with or without shortness of breath in the setting of recent vaccination. Initial workup should include:
Symptom-guided imaging tests for presumed thrombosis (e.g., computed tomography or magnetic resonance imaging venogram of the head for severe headache);
Standard coagulation studies (e.g., complete blood count with platelet count, D-dimer, fibrinogen);
Anti-PF4 immunoassay (PF4 heparin-induced thrombocytopenia enzyme-linked immunosorbent assay (ELISA) is considered the most reliable).
Platelet counts ranged from 10 to 113 × 109/L in the reported case series. 2 Nearly 100% of reported cases of VITT had a positive anti-PF4 ELISA with an optical density greater than 2.0. 3 An unusually high optical density is one distinguishing feature of VITT that is not typical for heparin-induced thrombocytopenia (HIT). HIT shares certain features of VITT including haptenized platelet antigens and the development of antiplatelet immunoglobulins (IgG) that can degranulate platelets upon platelet Fc receptor (FcR) ligation. This promotes platelet aggregation, thrombosis, and consumptive thrombocytopenia. 6 Unlike HIT, functional assays such as standard serotonin release assays can be negative and therefore are not required to confirm the diagnosis of VITT. The constellation of thrombosis, thrombocytopenia, and a positive PF4 ELISA 4–28 days following AstraZeneca or JJ vaccination without heparin exposure is suggestive of VITT.
What is VITT mechanistically?
The exact pathogenesis of VITT remains unclear. However, just as antiplatelet IgG develops in HIT in the presence of alpha granule PF4/heparin complexes, a hallmark of VITT appears to be fragments of vaccine/PF4 complexes evoking an immune response, and subsequent development of antiplatelet IgG in a manner resembling HIT but without prior exposure to heparin products.6,7 A commonality shared by HIT and VITT is platelet FCγRIIA ligation by the fixed domain of antigen-crosslinked circulating autoantibodies in patients. This causes exocytosis of pro-thrombotic and inflammatory alpha granules that contain PF4, fibronectin, Factor V, and p-selectin; dense granules that contain adenosine diphosphate (ADP) and serotonin; and Weibel–Palade bodies that contain p-selectin and von Willebrand factor (vWF). 8 There is also a recent opinion that VITT may be intertwined with dysregulated activation of the complement system, though primary data in support of this hypothesis are lacking at this time. 9
Platelets are highly dynamic entities with morphology and intricate granule content truly only appreciated by high-resolution electron microscopy (EM). A summarized hybrid schematic of platelet activation in VITT is noted below in the platelet of a healthy subject from whom washed platelets were prepared utilizing our Institutional Review Board protocol (Figure 1).
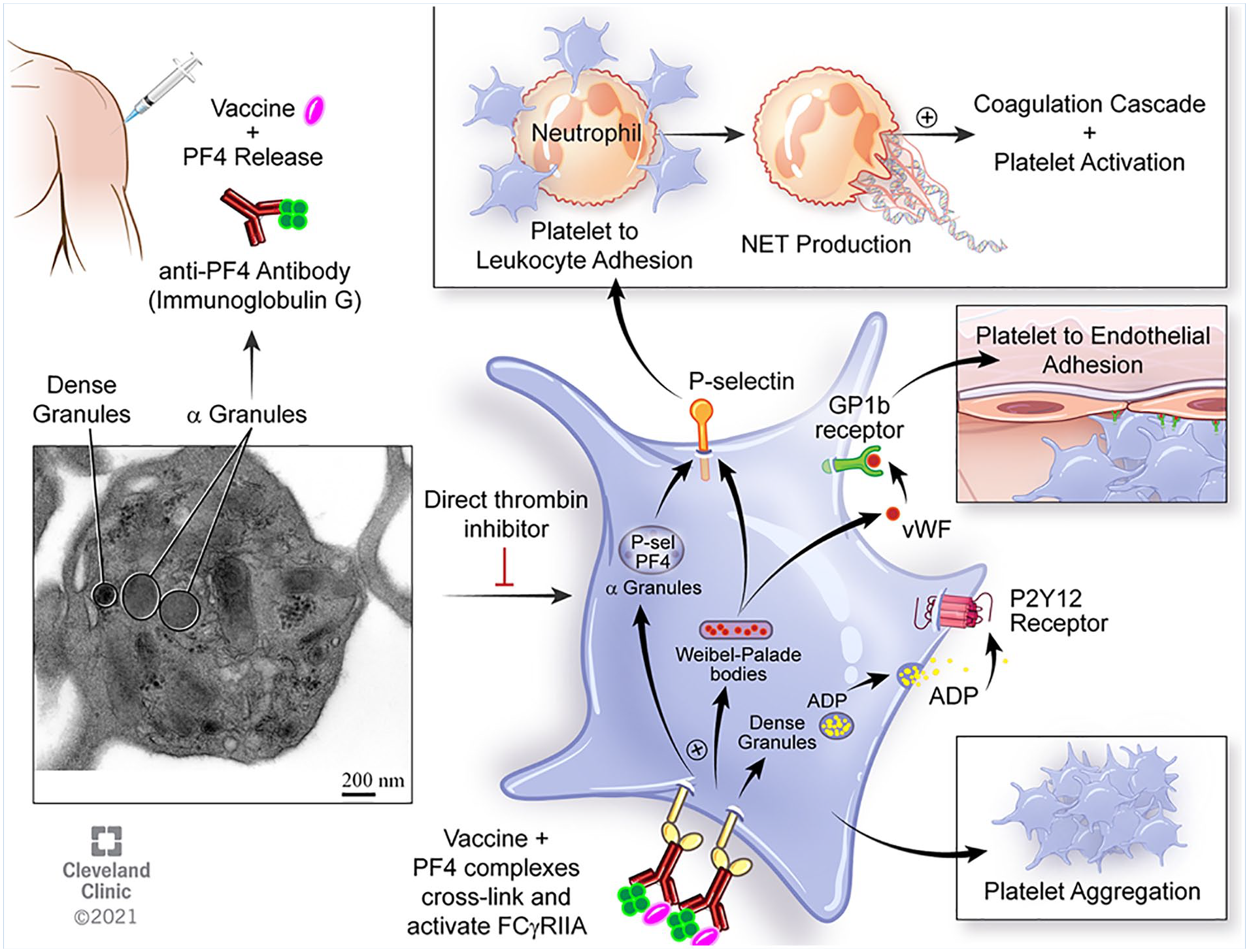
Electron microscopic depiction of platelet activation in VITT. Vaccination using non-mRNA-based technology in rare cases causes the formation of PF4/vaccine complexes that invoke an immune response. Immunoglobulin development and platelet receptor ligation by the fixed domain of the antibody activates and degranulates platelets to promote thrombosis, and the formation of neutrophil extracellular traps.
How do we treat VITT?
Given the low incidence of VITT and the limited availability of high-quality data on this complication at the present time, current treatment recommendations resemble the treatment for HIT and are derived mainly from expert consensus recommendations. Treatment should begin immediately if VITT is strongly suspected. In patients presenting with thrombosis, thrombocytopenia, and a positive anti-PF4 ELISA 4–28 days following AstraZeneca or JJ vaccination, treatment with intravenous immune globulin (IVIG) and nonheparin anticoagulation is recommended. 5 High-dose glucocorticoids can be considered. IVIG should be dosed at 0.5–1 g/kg daily for 2 days. Considerations for nonheparin anticoagulation include direct thrombin inhibitors (e.g., argatroban, bivalirudin), direct oral anticoagulants (e.g., apixaban, rivaroxaban), or fondaparinux. Anticoagulation should be reserved for platelet counts over 20 × 109/L and in patients without serious bleeding. Platelet transfusions and utilization of heparin products should be avoided. Consultation with an expert in thrombosis, such as vascular medicine or hematology, is recommended. It should be noted that thrombosis in unusual locations can also be seen in patients infected with SARS-CoV-2. Although rare, in published case series, splanchnic vein thrombosis and cerebral venous thrombosis were noted as complications of COVID-19. In contrast to VITT, cases of thrombosis in the setting of COVID-19 can be treated with intravenous unfractionated heparin.10,11
Why do we think this is happening with the adenoviral vector-based vaccines but not the mRNA-based vaccines?
Whereas the Moderna and Pfizer mRNA-based vaccines utilize ribosomes resident in any cell to initiate translation of an immunogenic peptide fragment-based region of the SARS-CoV-2 spike protein, both ChAdOx1 nCov-19 (AstraZeneca) and Ad26.COV2.S (JJ) use engineered adenovirus technology. Devoid of the ability to replicate, adenovirus-based vaccines still require nuclear translocation for transcription before subsequent cytoplasmic translation (the starting point for mRNA-based vaccines). In the cytoplasm, spliceosome-mediated fragments of transcribed RNA is thought to ultimately result in solubilization of multiple components of translated but fragmented SARS-CoV-2 spike protein, which can—for unclear reasons—act as a thrombogenic immunological substrate. Nonpeer-reviewed preliminary data regarding this hypothesis are intriguing, 12 but additional mechanistic experiments are required to fully understand VITT.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The work of Scott J. Cameron was supported by the National Heart, Lung, and Blood Institute (HL158801-01).
