Abstract
Previous studies on everolimus-eluting bioresorbable vascular scaffolds (BVS) have shown promising 1-year primary patency rates in infrapopliteal arteries. Literature from large cohorts on long-term outcomes with the infrapopliteal Absorb BVS (Abbott Vascular) is lacking. The aim of this study is to pool published and unpublished data to provide a more precise estimate of the 24-month outcomes of Absorb BVS for the treatment of infrapopliteal disease. For the pooled analysis, updated original and newly collected data from three cohorts on treatment with the Absorb BVS for de novo infrapopliteal lesions were combined. The primary endpoint was freedom from restenosis. Secondary endpoints were freedom from clinically driven target lesion revascularization (CD-TLR), major amputation and survival. The pooled analysis included a total of 121 patients with 161 lesions, treated with 189 Absorb BVS in 126 limbs. The mean age of the patients was 73 years, 57% had diabetes mellitus, and 75% were classified as Rutherford–Becker class 5 or 6. Of the 161 lesions, 101 (63%) were calcified and 36 (22%) were occlusions. Successful deployment was achieved with all scaffolds. Freedom from restenosis was 91.7% and 86.6% at 12 and 24 months, respectively, and freedom from CD-TLR was 97.2% and 96.6%. Major amputation occurred in 1.6% of the limbs. Overall survival was 85% at 24 months. In conclusion, this pooled analysis represents the largest reported analysis of mid-term results of the Absorb BVS for the management of chronic limb-threatening ischemia. At 24 months, the Absorb BVS was safe with promising clinical outcomes for the treatment of infrapopliteal disease.
Keywords
Introduction
Patients with chronic limb-threatening ischemia (CLTI) represent a complex and high-risk subset of peripheral artery disease (PAD) with poor prognosis. 1 According to the European Society of Vascular Medicine guidelines on PAD management, endovascular revascularization is recommended as first-line therapy for the treatment of infrapopliteal disease in patients with CLTI. 2 The preferred endovascular technique for revascularization in infrapopliteal arteries is still a debated topic. Guidelines agree that balloon angioplasty remains a reasonable primary endovascular approach, as evidence to support other (more expensive) techniques is lacking.2,3
Yet, two meta-analyses showed a tendency toward improved results with drug-eluting stents (DES) when compared to balloon angioplasty and bare metal stents (BMS).4,5 DES improved rates of patency, freedom from target lesion revascularization (TLR) and major amputation for the treatment of infrapopliteal disease at 1 year of follow-up.4,5 The favorable results of DES over BMS may be due to the antirestenotic drug that has been added into the polymer of DES. The antirestenotic drug lowers the risk of neointimal hyperplasia and ultimate reocclusion by reducing induration of chronic inflammation 6 caused by a reaction of the metal stent platform on the vessel wall. 7 However, this is only effective as long as the antirestenotic drug is present on the polymer of the stent. As a result, studies reported improved patency rates after 1 year,8,9 but failed to show improved patency rates after 3 years. 10
Stents without a metal platform, bioresorbable vascular scaffolds (BVS), could help overcome some of the limitations of infrapopliteal DES. The Absorb everolimus-eluting BVS (Abbott Vascular, Santa Clara, CA, USA) consists of a poly(
Previous studies on the use of the Absorb BVS in infrapopliteal artery disease reported promising primary patency rates of 86–96% at 1 year.12–14 However, the cohorts are relatively small and 2-year data have not been published from all centers. Accordingly, the aim of this study is to pool the published and unpublished data of the three centers that evaluated the use of Absorb BVS for the treatment of infrapopliteal artery disease to provide a more precise estimate of 24 months’ outcomes.
Methods
Study design
This was a pooled analysis of three cohorts undertaken at Changi General Hospital (CGH), Singapore; University of Chicago Medical Center (UCMC), Chicago; and Prince of Wales Hospital (PWH), Sydney. The cohorts were identified by a systematic literature search. Details on search, eligibility criteria, and study selection are available in the online supplementary material. The cohorts had an identical design but were conducted independently. The Institutional Review Board of all participating centers approved the study protocols, which were conducted according to the principles of the Declaration of Helsinki and in compliance with local regulatory requirements.
Patient selection
Eligibility criteria for the three cohorts have been published elsewhere.14–16 In brief, in all cohorts, patients were eligible if they were suffering from chronic lower limb ischemia (Rutherford–Becker class 3–6) and had de novo lesions of > 50% or > 60% in severity in below-the-knee (BTK) arteries (distal popliteal or tibial arteries) with a diameter between 2.5 mm and 4 mm. In one cohort (PWH), 16 the length of the stenotic lesions had to be ⩽ 5 cm in length to be eligible for the procedure. In two cohorts (UCMC, PWH), patients were excluded if they could not provide informed consent, had a life expectancy of < 12 months, could not tolerate dual antiplatelet therapy or where angiography was impossible due to renal insufficiency.15,16 In one cohort (CGH), patients were excluded if the stenotic lesions were located in the distal 8 cm above the ankle joint or if the patient had arterial thrombosis or restenotic lesions. 14
Endovascular procedure
The details of the procedure have been described previously and were performed in a similar fashion.12–14 Briefly, all patients received antiplatelet therapy prior to the procedure. Angiography was performed via antegrade or crossover access. After angiography, systemic heparinization was done as per institutional protocol. Inflow lesions were treated in the same procedure, if present. All infrapopliteal target lesions were pre-dilated with angioplasty. The diameter of the balloon used for angioplasty was matched on a 1:1 basis on the reference vessel diameter (RVD), which was determined on angiography. The size of the Absorb BVS was also chosen on a 1:1 basis to treat the target lesions. If more than one scaffold was required, consecutive scaffolds were abutted or overlapped with a maximum overlap of 1 mm when necessary for total lesion coverage. Each BVS was deployed gently by increasing the inflation pressure with 2 atm every 5 seconds until the desired diameter and pressure were reached. Maximum inflation was continued for 30–60 seconds to establish complete scaffold coverage. Post-dilatation was left at the operator’s discretion. Dual antiplatelet therapy was continued for at least 6 months, followed by aspirin 81–100 mg daily for life.
Patients had a duplex ultrasound follow-up at 12 and 24 months, and more frequently when needed. Clinical follow-up differed among the participating centers and included history and physical examination, as well as wound management.
Data collection
Once the authors of the cohorts agreed to participate, databases were updated and collected. All centers were provided with a uniform database for data collection. For two cohorts (UCMC, PWH), data from their previous studies were used and updated.15,16 For one cohort (CGH), 14 additional data of the outcomes at 24 months were collected, which have not been published previously. Data were derived from electronic medical records, clinical records, and imaging/laboratory reports. Angiographic data were derived by analyzing the procedural angiographic images by experienced interventionalists. The database of the three cohorts were merged into one database for analysis. The merged database included retrospectively collected data from two centers (CGH, UCMC) and prospectively collected at one center (PWH).
Endpoints and definitions
The primary endpoint of the pooled analysis was freedom from restenosis at 24 months. Restenosis was defined in the three cohorts as a peak systolic velocity (PSV) ratio > 2.0 or PSV > 2 m/s (equivalent to > 50% stenosis) or a PSV of 0 m/s (equivalent to thrombosis or occlusion) as seen on color-flow Doppler examination. 17 Secondary endpoints were freedom from clinically driven target lesion revascularization (CD-TLR), procedural and 30-day outcomes, and rates of amputation and survival at 24 months. CD-TLR was defined as avoidance of any clinically driven repeat percutaneous intervention of the target lesion or bypass surgery of the target vessel performed for restenosis or other complication of the target lesion. Technical success was defined as successful deployment of the Absorb BVS in the target lesion without residual stenosis of > 30%.
Statistical analysis
Baseline and procedural characteristics were described using median (IQR or range), when non-normally distributed, mean ± SD, when normally distributed, and count (percentage). Quantile-quantile (Q-Q) plots were analyzed to determine whether continuous variables followed a normal distribution. If the points in the Q-Q plot were present on a straight diagonal line, the data were defined as normally distributed. Freedom from restenosis and freedom from CD-TLR were estimated by Kaplan–Meier (KM) analysis and were performed on a per scaffold basis. Statistical significance was defined as p < 0.05. Statistical analysis was performed using SPSS, version 23 software (IBM Corp., Armonk, NY, USA).
Results
Patient characteristics
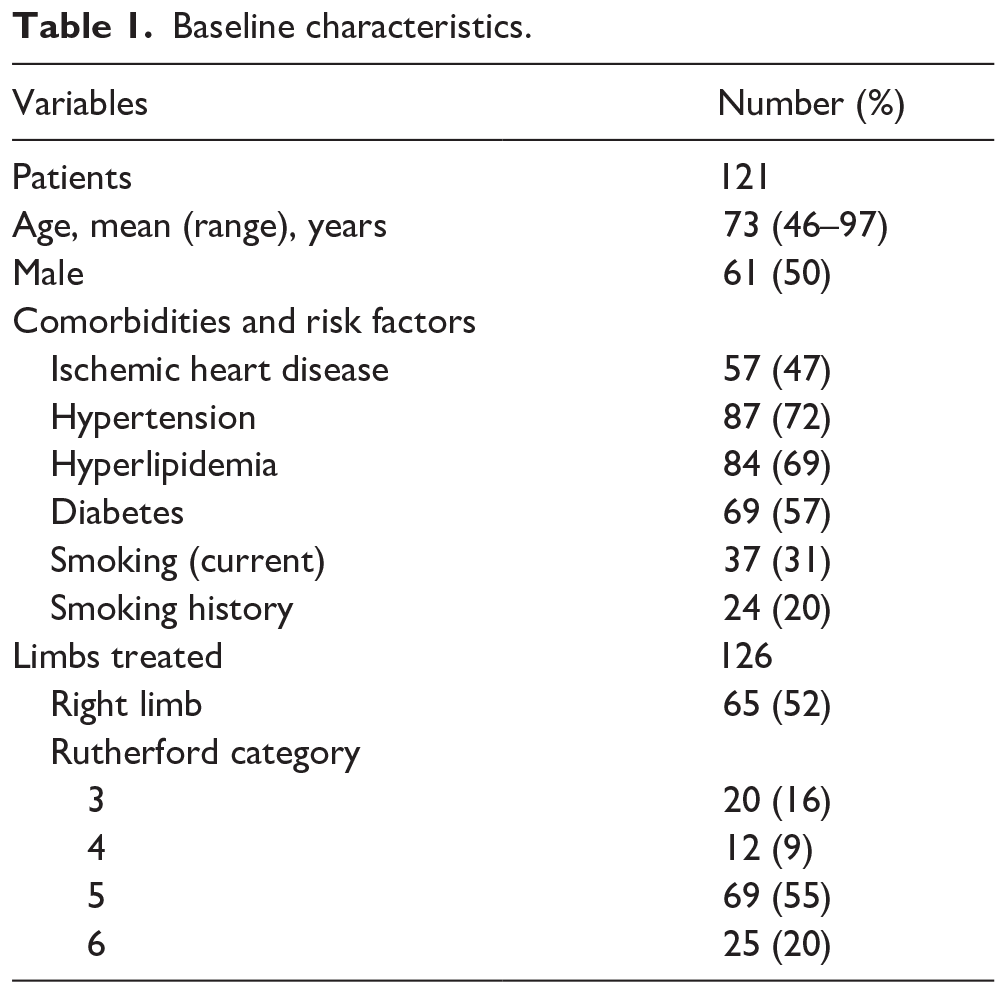
The pooled analysis included a total of 121 patients with 161 lesions treated with 189 scaffolds in 126 limbs; 41 patients were treated in CGH, 31 in UCMC and 49 in PWH. Sixty-one patients (50%) were male and the mean age was 73 years (46–97 years). Hypertension was present in 87 (72%), hyperlipidemia in 84 (69%), and diabetes in 69 (57%) patients. Seventy-five per cent of patients presented with Rutherford–Becker class 5 or 6 ischemia (Table 1).
Baseline characteristics.
Lesion and scaffold details
The details of lesion and scaffold characteristics are provided in Table 2. Overall, the median severity of stenosis was 80% (IQR: 80–95%). Calcification was present in 63% and total occlusion in 22% of the lesions. The median lesion length was 21 mm (IQR: 15–30 mm) and the median vessel diameter was 3.1 mm (IQR: 3.0–3.5 mm). The median scaffold length was 28 mm (IQR: 18–28 mm) and the median scaffold diameter was 3.0 mm (IQR: 3.0–3.5 mm).
Lesion and scaffold characteristics.
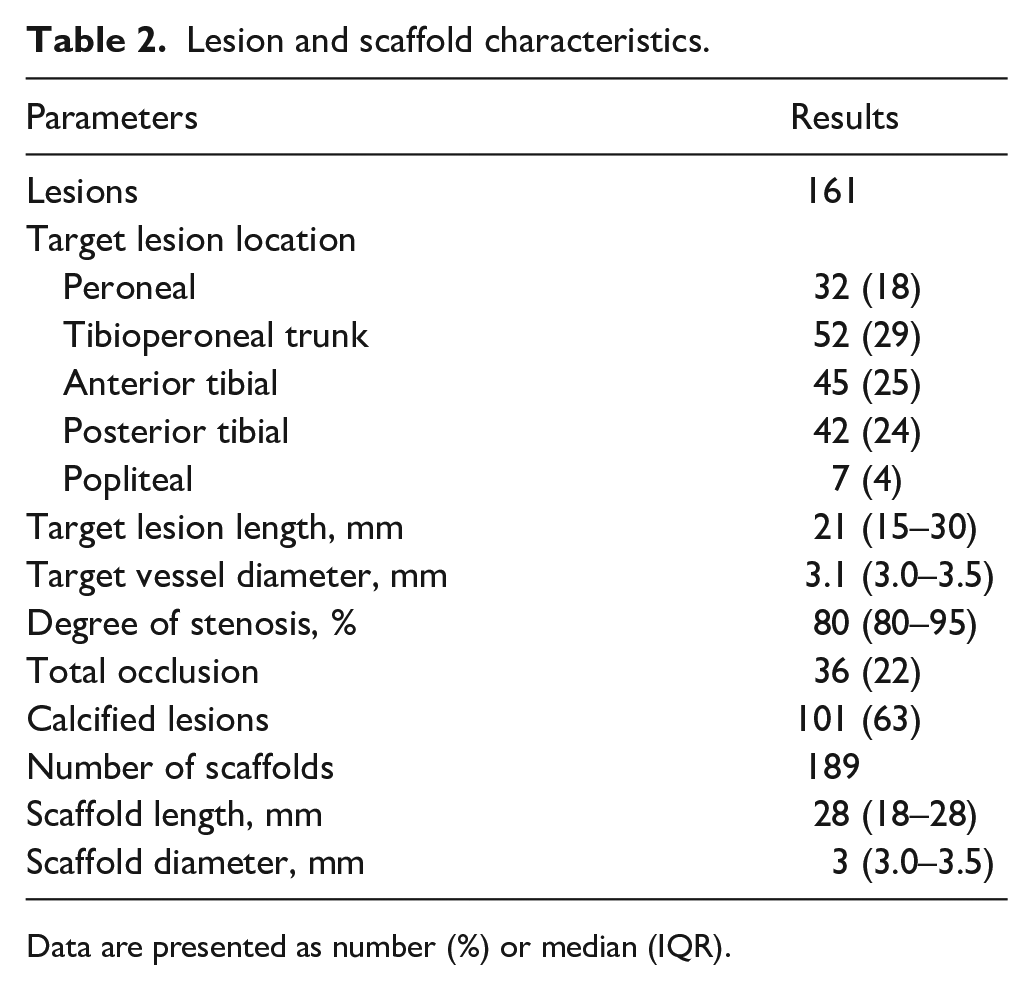
Data are presented as number (%) or median (IQR).
Procedural and 30-day outcomes
All 189 scaffolds were successfully deployed. One patient underwent CD-TLR in two scaffolds on the second postoperative day due to a thrombosis. There was one major amputation within 30 days of the index procedure, due to infected gangrene of the heel. This was not deemed to be related to the procedure. No deaths occurred within the 30 days after the index intervention.
Clinical outcomes at follow-up
The median clinical follow-up time was 29 months (IQR: 25–49) and the median time of radiological follow-up was 25 months (IQR: 12–41). Within 24 months, restenosis occurred in 21 scaffolds, resulting in an overall patency rate of 91.7% and 86.6% at 12 and 24 months, respectively. The median time to restenosis was 11 months (IQR: 6–13 months). The KM curve for the primary endpoint of freedom from restenosis is shown in Figure 1. Target lesion revascularization was performed in six scaffolds, resulting in freedom from CD-TLR of 97.2% and 96.6% at 12 and 24 months, respectively (Figure 2). The median time to CD-TLR was 5 months (IQR: 0–12 months). Beyond 30 days, one patient underwent a major amputation due to deteriorating tissue loss. A total of 18 deaths occurred within 24 months. The overall survival rate was 85% at 24 months.
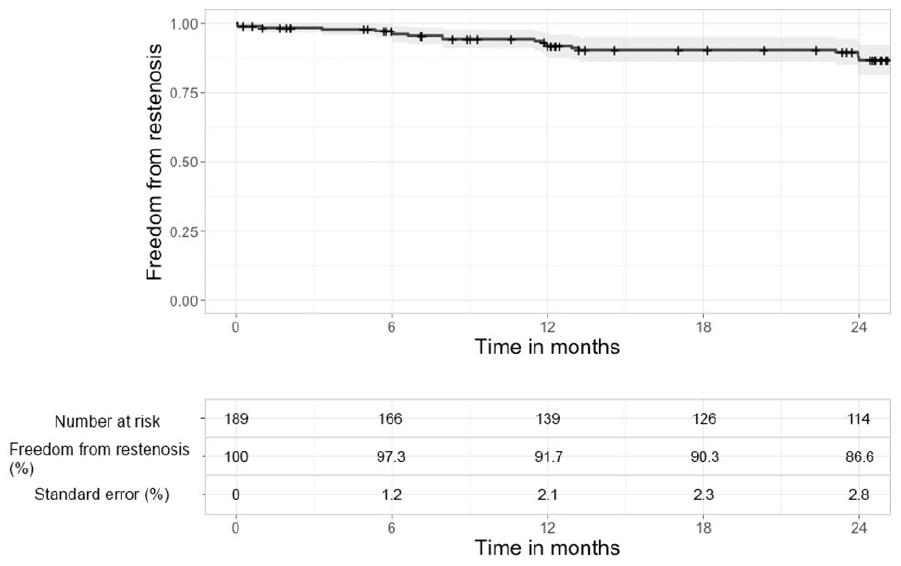
Kaplan–Meier analysis for freedom from binary restenosis with 95% CI.
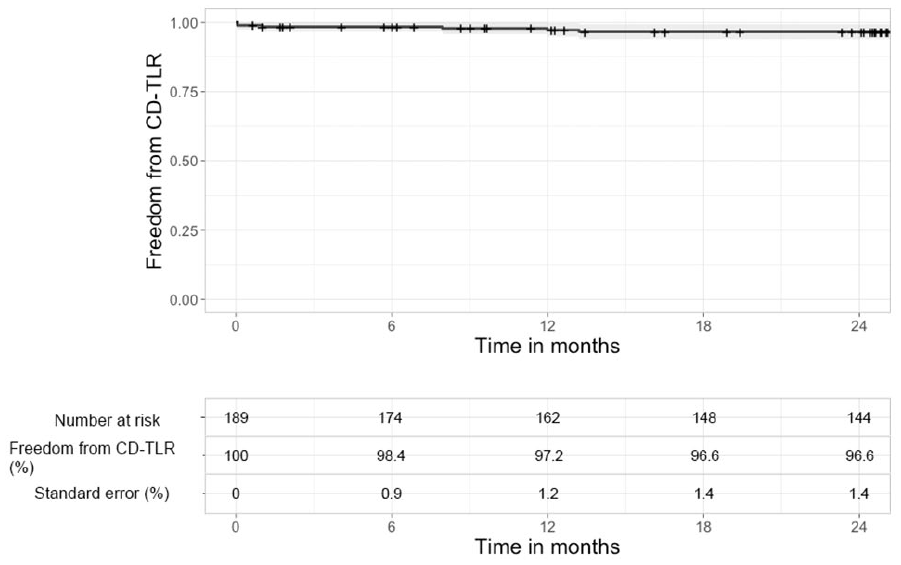
Kaplan–Meier analysis for freedom from CD-TLR with 95% CI.
Discussion
This pooled analysis shows that, with long-term follow-up, the Absorb BVS has favorable primary patency rates and freedom from reintervention rates. In addition, it was found to be feasible and safe.
In the overall cohort, patency rates at 12 and 24 months were 91.7% and 86.6%, respectively. This is equivalent to historical results from studies that evaluated non-resorbable DES. The primary patency rates with the Xience-Prime Everolimus-Eluting Stent (Abbott Laboratories, Abbot Park, IL, USA) and a combination of Xience Everolimus Eluting Stent (Abbott) and Promus Everolimus Eluting Stent (Boston Scientific, Marlborough, MA, USA) in BTK disease was approximately 80% 18 and 74%, 19 respectively, at 24 months.
Although the patency rates of historical studies on DES are comparable with the patency rates of BVS, the BVS has the advantage of being temporary. Nowadays, stenting is used less frequently than percutaneous transluminal angioplasty (PTA) because of the permanent implant, which may act as a future impediment. A BVS has the benefits of DES over PTA (improved patency rates, elimination of acute and subacute recoil, residual stenosis, and flow-limiting dissection), 20 but not the burden of DES, a permanent implant. So far, only one study has reported the patency rates at 3 years’ follow-up, which were promising (81.1%). 16 Future studies are necessary to fully investigate the benefits of BVS compared to DES or PTA in infrapopliteal arteries.
One patient underwent CD-TLR in two scaffolds, due to occlusion on the second day of follow-up in our study. The patient was on warfarin but was stopped prior to the index procedure and post-procedural antiplatelet therapy was not initiated. This is the most likely explanation for that early occlusion, which was successfully treated by endovascular revascularization. The 24-month freedom from CD-TLR of 96.6% in our study was similar to that found in the long-term in previous studies using non-resorbable DES (81–91%).18,19,21,22
Because studies on the use of BVS in infrapopliteal arteries in CLTI patients are limited, this article contributes to the knowledge of its use for the treatment of infrapopliteal disease. Although the results are promising, the Absorb BVS is currently not commercially available. However, the bioresorbable MOTIV scaffold (REVA Medical, San Diego, CA, USA) is now available in Europe and may allow some operators to have access to the BVS technology.
There still is a high unmet need for better treatment strategies for infrapopliteal disease. BVS may become a reasonable treatment strategy in the future, but more research is needed to assess this further. One known, recently started trial is the LIFE-BTK trial, which will study the new-generation ESPRIT BVS (Abbott Vascular, Santa Clara, CA, USA) in infrapopliteal arteries in patients with CLTI. The LIFE-BTK is a multicenter trial that has planned to enroll 235 patients, who will be randomized to ESPRIT BVS or PTA, with a follow-up of 5 years. 23 The results of this trial will be eagerly awaited. For current clinical practice, the present study could be a helpful contribution.
Limitations
The present study is not without limitations. First, most of the data were collected retrospectively and, therefore, some inaccuracy and bias is likely. Second, because no control group was used in this study, direct comparisons between BVS and DES in infrapopliteal disease are not possible. Third, no cost-effectiveness analyses have been conducted and the effect on quality of life was not evaluated. In addition, the studied lesions were short, yet long lesions are common in BTK arteries.
Conclusion
This pooled analysis showed that the Absorb BVS can be safely used for the treatment of patients with CLTI due to infrapopliteal artery disease with favorable rates of patency, reinterventions, and amputations at long-term follow-up. Future large-scale, randomized controlled trials may help further assess the significance of our findings.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X20977907 – Supplemental material for Mid-term outcomes of an everolimus-eluting bioresorbable vascular scaffold in patients with below-the-knee arterial disease: A pooled analysis of individual patient data
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X20977907 for Mid-term outcomes of an everolimus-eluting bioresorbable vascular scaffold in patients with below-the-knee arterial disease: A pooled analysis of individual patient data by Eline Huizing, Steven Kum, Jetty Ipema, Ramon L Varcoe, Atman P Shah, Jean-Paul PM de Vries and Çağdaş Ünlü in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Steven Kum: consultant for Abbott Vascular; Ramon L Varcoe: consultant for Abbott Vascular and Medtronic; Atman P Shah: consultant and proctor for Abbott; the remaining authors have nothing to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
