Abstract

What is COVID-19, and how does it affect the body?
SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2) is the virus responsible for coronavirus disease 2019 (COVID-19). As of October 1, 2020, SARS-CoV-2 has infected over 34 million people and has resulted in over 1 million deaths worldwide. In the United States alone, there are over 7 million confirmed cases and greater than 200,000 deaths. 1 Elderly individuals and those with chronic medical conditions are at the highest risk, but the disease can strike people in good health and those in any age group, and it may be carried and transmitted to others by people with no symptoms. SARS-CoV-2 is now well known to cause lung disease, including pneumonia or more severe respiratory complications. COVID-19 is spread mostly through exhaled air and respiratory droplets. Wearing masks protects those around us from contracting SARS-CoV-2 and may protect the wearer from getting another person’s infection. Pneumonia-related COVID-19 has gained the most attention, as patients with severe illness frequently have low blood oxygen levels and may require mechanical ventilation (i.e. life support with a ‘breathing machine’), often for prolonged periods of time. But COVID-19 can affect multiple organs in the body beyond the lungs, such as the heart, gastrointestinal tract, kidneys, bone marrow, liver, and skin. 2 The reason so many body systems are affected by COVID-19 may be due to the fact that SARS-CoV-2 infects a wide variety of human cells, including the cells that make up the surface of our blood vessels that supply circulation to these organs, called ‘the endothelium’.
How does SARS-CoV-2 cause disease?
SARS-CoV-2 binds and infects cells that express a protein called angiotensin converting enzyme 2 (ACE2) on their surface, including cells that line blood vessels. The cells take up the virus, which then uses the cell as a place to grow and multiply. The uptake of virus causes an ACE2 deficiency that alone may start the disease process. Additionally, though not completely understood, growth of the virus may cause disease through direct damage to the cells in organs, damage to the blood vessels that supply those organs, and a dysregulated immune response known as a ‘cytokine storm’. 3
Does COVID-19 cause blood clots?
Early on during the COVID-19 pandemic, it was observed that patients suffering from COVID-19, especially those requiring intensive care unit (ICU)-level care, had high rates of blood clotting, or thrombosis. These blood clots may come in the form of deep vein thrombosis (DVT), clots in the veins of the legs, or pulmonary embolus (PE), which occurs when a DVT dislodges and travels to the lungs and blocks blood flow to the lung. 4 PE worsens any existing lung disease and heart problems and can cause severe lung and heart disease itself. If large enough, a PE can cause sudden death. In addition to vein blood clots, patients with COVID-19 have also been found to have thrombosis in the arteries of the brain, causing strokes, 5 and in the arteries of the arms and legs. 6 Patients with severe COVID-19 are also at increased risk for heart damage, some of which may be due in part to clots in the coronary arteries, the blood vessels that feed the heart muscle. Clotting is not limited only to patients with severe SARS-CoV-2 infection. For example, ‘COVID toes’, a term that describes purple discoloration of the feet and toes, are a more common manifestation of the disease in younger adults or children. It is believed to be caused by inflammation due to the virus and clotting of the small blood vessels of the skin, 7 leading to a lack of blood flow in the small arteries of the feet (Figure 1).
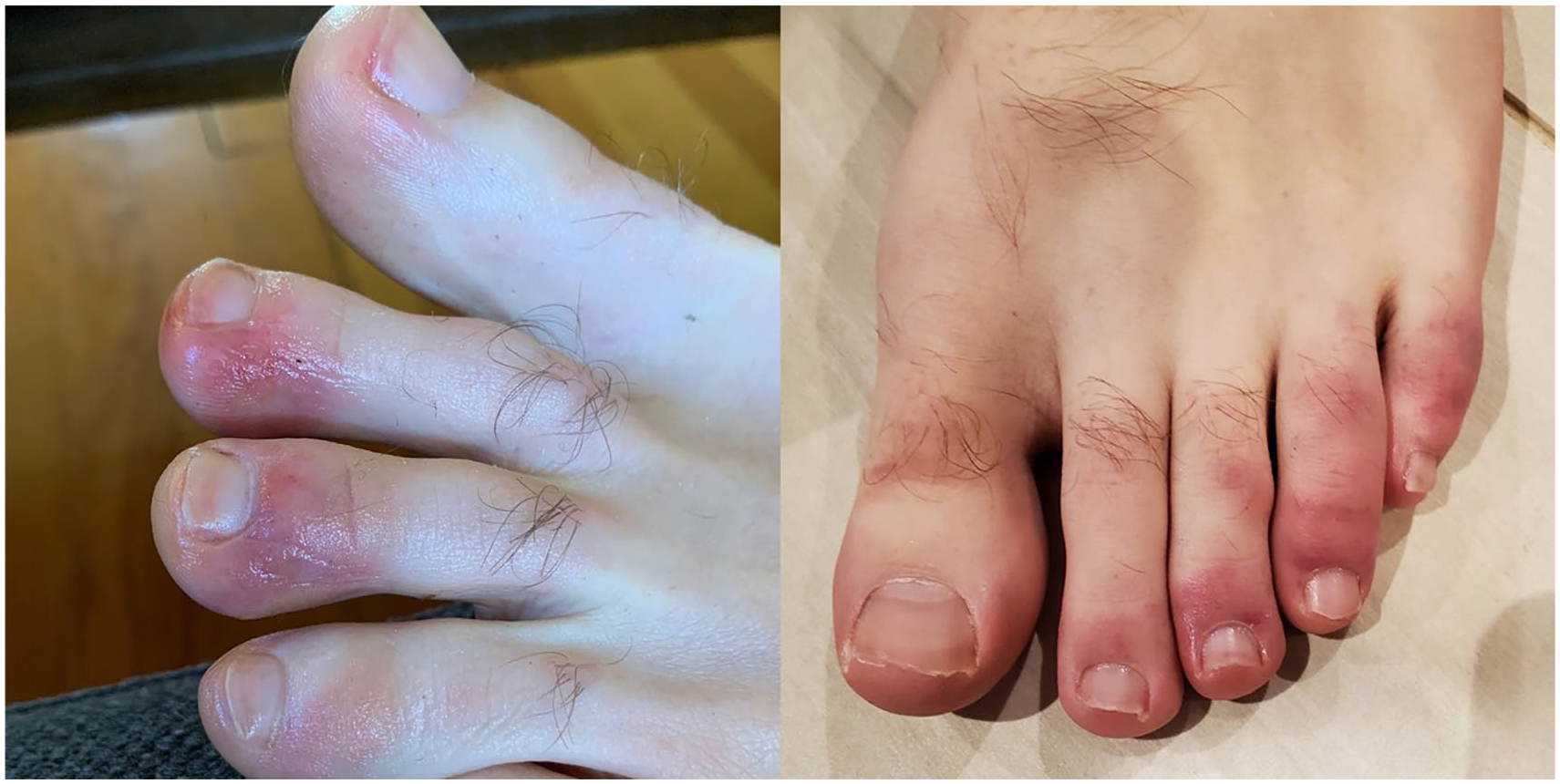
‘COVID toes’ are a form of thrombosis in COVID-19. COVID toes are reddish-purple discolorations on the toes or feet that may be mildly itchy or painful. These lesions are due to small blood clots that plug small blood vessels in the toes. COVID toes alone do not require any treatment with anticoagulation.
Why does COVID-19 cause blood clots?
This question is a topic of intense investigation. There are likely multiple causes, and researchers have only a partial understanding currently. All patients who are critically ill are at increased risk for thrombosis due to the severity of the illness itself. They are at increased risk due to immobility, which leads to slow blood flow in the legs. Clots can also form in and around large intravenous (IV) lines. Patients with COVID-19 receiving care in an ICU may have a higher rate of clotting compared with patients in an ICU who do not have COVID-19. 8 It is a challenge to establish the true rates of COVID–19-related thrombosis because of the inability to test every patient suspected of a blood clot with the appropriate test—a CT scan of the lungs or an ultrasound of the legs—due to limited resources and infection-control concerns. The question is complex because many of the risk factors for severe illness in COVID-19 overlap with risk factors for venous thrombosis, such as advanced age, obesity, smoking, diabetes, heart disease, lung disease, cancer, and sickle cell disease.
Several mechanisms may explain why COVID-19 is associated with a higher risk of blood clotting, but the exact reasons are not yet known. COVID-19 appears to cause severe inflammation, and many inflammatory factors themselves create a predisposition to form clots. In fact, some researchers have argued that blood clotting in small vessels (known as the microvasculature) may actually be the cause of COVID-19 lung disease itself. In the lung and other organs, severe COVID-19 may largely be a blood vessel disease and, therefore, abnormal blood clotting is a fundamental aspect of COVID-19 in general (Figure 2).
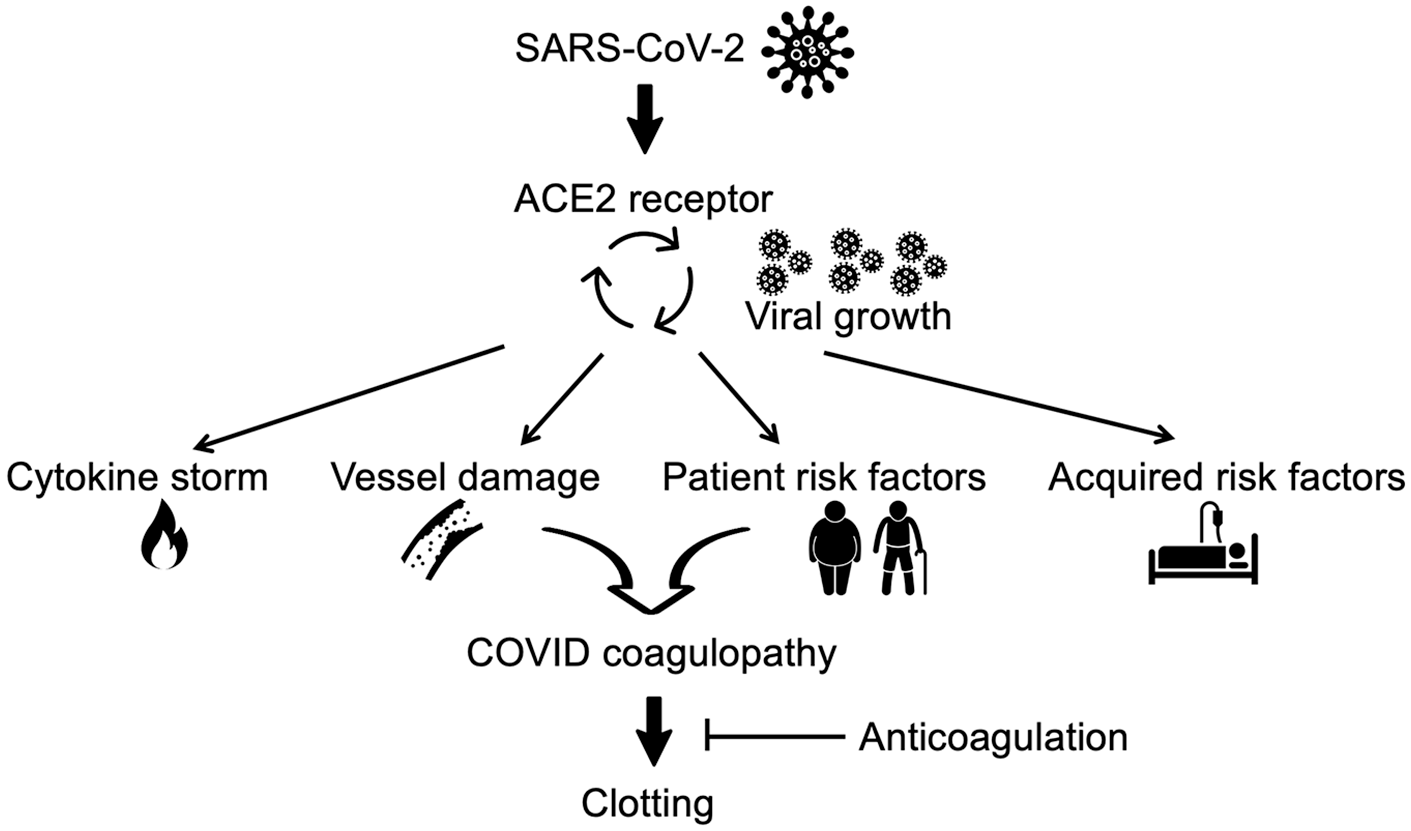
COVID-19 causes blood clotting by various mechanisms. SARS-CoV-2 enters cells, including cells that make up blood vessels (‘the endothelium’), via the ACE2 protein expressed on the cell surface. Inside the cell, growth of SARS-CoV-2 can lead to direct blood vessel cell damage and severe inflammation (‘cytokine storm’) that creates a tendency toward clotting. Patient risk factors for severe COVID-19 are also risk factors for clotting in general, such as advanced age, obesity, smoking, diabetes, heart disease, lung disease, cancer, and sickle cell disease. Acquired risk factors in hospitalized patients include surgery, prolonged immobility, and large IV lines. To prevent blood clotting, the dose of anticoagulation is adjusted based on the severity of COVID-19 coagulopathy.
What is COVID-19 coagulopathy?
Patients with moderate to severe SARS-CoV-2 infection usually have significant abnormalities in blood tests used to measure blood coagulation (clotting). Abnormal coagulation is known as ‘coagulopathy’.
8
The tests used include the (1) prothrombin time, (2) fibrinogen, and (3) D-dimer: The
How can the risk of blood clots be reduced in patients with COVID-19?
In most patients, the development of blood clots is due to multiple causes. For example, if an 80-year-old man breaks his hip, has hip surgery, and is on prolonged bed rest after surgery, he is at high risk for venous thrombosis because male sex, older age, major orthopedic surgery, being in the hospital, and immobility are all risk factors for blood clots. Despite different thrombosis risks among different patients, most patients admitted to the hospital receive what is called ‘thrombosis prophylaxis’—preventive anticoagulation (low-dose blood thinners) to help reduce the risk of blood clots. Most of the time, a standard thrombosis prophylaxis regimen fits all patients, even patients with a history of blood clots, although sometimes the length of treatment is prolonged. However, patients with COVID-19 often present with abnormal coagulation times, and because thrombosis is common among patients with COVID-19, a standard approach to thrombosis prophylaxis may not be appropriate (Figure 2). Instead, a more aggressive anticoagulation regimen is often utilized.
In patients with COVID-19, the best approach to prevention of thrombosis is to treat the coagulopathy (blood-clotting tendency) aggressively before it gives rise to a large clot such as a DVT or PE. Many hospitals have used a strategy of adjusting the dose of thrombosis prophylaxis based on the severity of illness (e.g. if the patient is admitted to a standard medical ward, or requires care in an ICU) or the degree of activated coagulation, as determined by the D-dimer level. For example, a normal D-dimer is 500 ng/dL, increasing by 100 ng/dL per decade after 60 years of age. Patients with COVID-19 and a lower D-dimer (less than 1000 ng/mL) who do not require ICU-level care will often receive standard doses of thrombosis prophylaxis that are similar to what hospitalized patients without COVID-19 receive. Patients with a D-dimer between 1000 and 2000 ng/dL, or who require care in an ICU, may receive intermediate-dose anticoagulation, either via an injection called a low-molecular-weight heparin given twice daily or an IV medication called unfractionated heparin. Higher-risk patients require higher-intensity thrombosis prophylaxis. These include patients with COVID-19 who have a history of blood clots. Other patients with COVID-19 who may receive higher-dose anticoagulation are those who are suspected of having thrombosis but cannot undergo confirmatory testing, have a hemodialysis line that frequently clots, or have another reason for anticoagulation such as the abnormal heart rhythm atrial fibrillation or an artificial heart valve. These patients receive treatment-dose anticoagulation with a low-molecular-weight heparin or unfractionated heparin infusion. Some institutions provide full-dose anticoagulation for all patients with COVID-19 in the ICU or with very high D-dimer levels (greater than 3000 ng/dL, for example). The dosing schedule listed above is just an example. Different hospitals are using different thrombosis prophylaxis regimens for COVID-19. As the degree of COVID-19 coagulopathy increases or decreases based on laboratory testing, doctors may adjust the patient’s anticoagulation medications. The medical team also makes decisions about the dose of anticoagulation based on the risk of bleeding, which is unique for each patient.
As researchers try to learn more about COVID-19 and thrombosis, several studies are underway to evaluate different thrombosis prophylaxis regimens to learn what is most effective and safe in different groups of patients. Refer to clinicaltrials.gov and search under ‘COVID-19’ and ‘anticoagulation’ to see the list of ongoing clinical trials.
How is COVID-19 coagulopathy treated after hospital discharge?
The clotting disorder associated with COVID-19 illness does not end when the patient goes home. Any patient with documented thrombosis needs treatment-dose anticoagulation, which is different from prophylactic doses. Most patients require at least 3 months of anticoagulation, often with an oral medication known as a direct oral anticoagulant (DOAC). 10 Because patients recovering from COVID-19 are also recovering from related coagulopathy, they may be discharged on anticoagulation for thrombosis prophylaxis even though they have not had a blood clot. Patients may go home with a low-molecular-weight heparin injection or an oral anticoagulant for as long as 30–40 days after discharge. D-dimer levels can be followed as an outpatient to look for resolution of coagulopathy and to monitor for new blood clots.
Summary
COVID-19 can vary from causing no symptoms in some people to a life-threatening illness requiring intensive care and mechanical ventilation in others. COVID-19 affects the body in many different ways, including causing blood clots such as DVTs and PEs. Thrombosis in COVID-19 may be due to direct damage to blood vessels, ‘out-of-control’ inflammation, or other mechanisms that are not completely understood. Many patients with moderate-to-severe COVID-19 have ‘coagulopathy’ (a predisposition to form blood clots) that can be measured by laboratory testing. Patients hospitalized with COVID-19 are given thrombosis prophylaxis (blood thinners) to prevent blood clotting. Instead of a one-size-fits-all approach, in patients with COVID-19, thrombosis prophylaxis regimens have been altered to provide higher doses of preventive-dose blood thinners. These doses can be adjusted based on the degree of coagulopathy, as determined by laboratory testing. Full-intensity, treatment-dose anticoagulation is prescribed if there is a documented thrombosis or in certain patients with COVID-19 at very high risk for thrombosis. There is still much that doctors do not understand about COVID-19, and ongoing studies will provide understanding of the best ways to prevent and treat thrombosis in COVID-19.
The ‘Vascular Disease Patient Information Page’ is a regular feature of Vascular Medicine. All articles in the collection are available for free online at http://journals.sagepub.com/vmjpatientpage.
The Vascular Disease Patient Information Page is provided for educational purposes only and is not a substitute for medical advice.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
