Abstract
Calf muscle pump (CMP) promotes venous return from the lower extremity and contributes to preload and cardiac output. Impaired CMP function may reflect a measure of frailty or cumulative disease burden or may impede cardiac function. The study objective was to test the hypothesis that impaired CMP negatively impacts survival. Consecutive adult patients who underwent venous strain gauge plethysmography at the Mayo Clinic Gonda Vascular Laboratory (January 1, 1998 – December 31, 2011) were assessed for overall survival. Patients with venous incompetence, venous obstruction or unilateral calf pump dysfunction were excluded. Risk of mortality was assessed with Cox proportional hazard ratios and after adjusting for Charlson Comorbidity Index variables. Over the study period, 2728 patients were included in the analysis. Compared to patients with normal CMP, those with impaired CMP were older (p < 0.001), predominantly female (p = 0.01) and had higher mean Charlson scores (p < 0.001). Patients with impaired CMP had a higher mortality rate at 5 (8.9% vs 2.4%), 10 (17.5% vs 5.9%), and 15 years (22.8% vs 8.3%) compared to those with normal CMP (p < 0.001 for each comparison). Of patients with heart failure, those with impaired CMP had worse survival at each 5-year increment compared to those with normal CMP (p < 0.05 at each increment). In conclusion, impaired CMP appears to be an independent predictor of poor outcomes after adjusting for variables within the Charlson Comorbidity Index. The association between impaired CMP, heart failure, and mortality may represent a negative impact on circulatory function or a surrogate measure of frailty.
Introduction
The calf muscle pump (CMP) is a contributing component of the cardiovascular system providing venous return from the lower extremity to the right atrium. Components of a normally functioning CMP include effective muscular contractile elements of the foot and calf in addition to highly compliant thin-walled veins, competent venous valves, and unobstructed venous outflow.1–4 With ambulation, venous blood is sequentially pumped against gravity from compartments of the foot, calf, and thigh to the inferior vena cava. The CMP is an important component due to its large venous capacitance and ability to generate high venous pressures. Under normal conditions, the CMP can generate venous pressures in excess of 250 mmHg.
Impaired CMP has been implicated in the genesis of chronic venous insufficiency and consequently indolent non-healing venous ulcers.1,5,6 Impaired CMP may result from neuromuscular disease, joint pathology, deconditioning, and general frailty. CMP dysfunction correlates with chronic venous disease severity as determined by the CEAP classification. Calf muscle strength has been objectively measured in patients with chronic venous insufficiency and showed a linear relationship with worsening venous insufficiency as calf muscle strength decreased. 6 Impaired CMP results in decreased venous return and edema with frequent temporal progression to dermal inflammation and ulceration. The physiology of venous return has important systemic implications with a direct relationship observed between venous return, stroke volume, and cardiac output.
Intermittent calf compression devices to enhance venous return and increase stroke volume, by mimicking calf pump function, have been used in patients with orthostatic hypotension to prevent syncope and postural tachycardia. 7 A lower calf muscle index was associated with orthostatic hypotension in a community study involving elderly patients. 8 Having a chronically low venous return may be detrimental to cardiac output over time and this may have important implications in patients with heart failure.
We sought to assess whether impaired CMP adversely impacts overall survival in adult patients who underwent venous strain gauge plethysmography testing at the Mayo Clinic Gonda Vascular Laboratory (January 1, 1998 – December 31, 2011).
Methods
Study design and patients
Consecutive patients over 18 years of age undergoing venous hemodynamic evaluation by strain gauge plethysmography at the Mayo Clinic Gonda Vascular Laboratory between January 1, 1998 – December 31, 2011 were analyzed. Patients were excluded if there was evidence of venous obstruction or valvular incompetence, unilateral calf pump dysfunction or lack of research authorization. The study protocol was reviewed and approved by the Mayo Clinic Institutional Review Board.
The Mayo Clinic Gonda Vascular Laboratory database contains all vascular studies performed since inception. The Vascular Laboratory study date was considered the index date for each patient. Clinical data were collected from a centralized system that contains complete records of all patients treated and followed at Mayo Medical Center. The Mayo Clinic medical record for each patient contains the details for every inpatient hospitalization, every outpatient visit regardless of the provider, every radiology examination, and all laboratory and pathology results (including autopsy reports), death certificates, and relevant correspondence. Heart failure was identified using in-house SAS-based tools including International Classification of Diseases, Ninth Revision (ICD-9) codes to search the electronic Mayo Clinic medical record.
Study date intervals for this project were determined for three reasons. First, 1996 began the era of the electronic medical record at Mayo Clinic. Second, during this time interval, strain gauge plethysmography was the method used for venous hemodynamic testing in the laboratory until 2011, when it was replaced by air plethysmography. This time interval provided a unified methodology for venous hemodynamic testing. Third, this time interval provides sufficient duration of time for follow-up mortality assessment.
Strain gauge plethysmography
Venous physiology was assessed for each leg by trained technicians dedicated to the Gonda Mayo Clinic Noninvasive Vascular Laboratory using strain gauge plethysmography (Phlebotest™; Eureka Company, Sweden; Figure 1A). This testing included three sequenced steps providing measured outcomes at each step: strain gauge outflow plethysmography (SGOP) to assess venous obstruction; passive drainage and refilling (PDR) to assess venous valvular incompetence; and exercise venous plethysmography (EVP) to assess CMP function.9–11
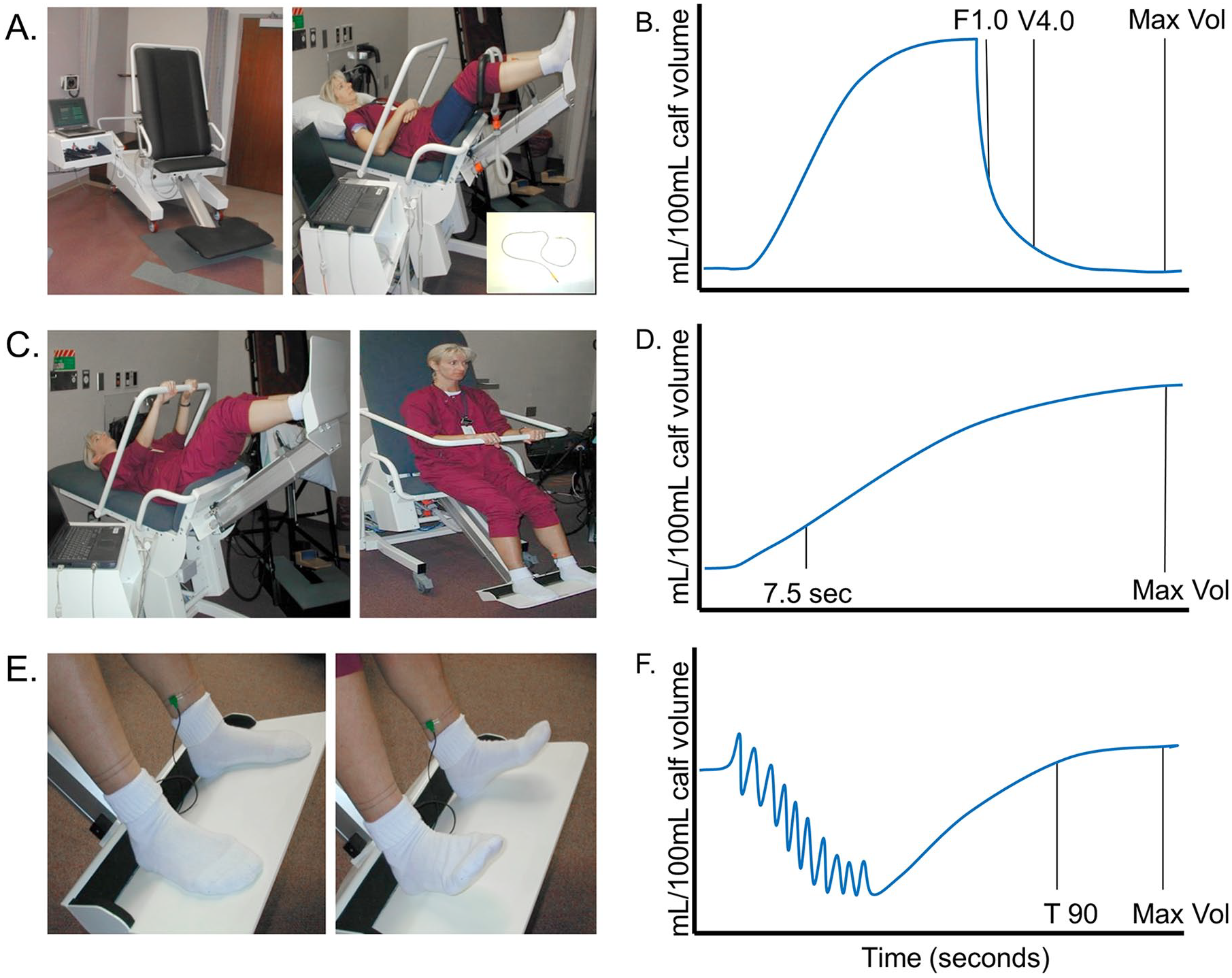
Venous plethysmography.
The patient was positioned on the Phlebotest chair with feet placed flat on the platform and adjusted so the inner angle between posterior thigh and calf is 90° (Figure 1B). After lowering the secure bar, the patient was instructed to hold gently onto the rail prior to chair tilt. The chair was then tilted until the patient achieved the supine position. Appropriate-sized blood pressure cuffs were placed around both thighs, and strain gauges were applied at the mid-calf. Once the patient was properly positioned and equipped, a series of three tests were performed as follows.
Venous outflow obstruction
Thigh cuffs were then inflated to 60 mmHg, allowing arterial inflow but occluding venous outflow (Figures 1A, 1B). Subsequent changes in calf volumes were then determined by strain gauge measures. Once a steady-state calf volume (Vsec) was achieved, the thigh cuffs were rapidly deflated allowing venous blood to return rapidly from the legs. Measurements included: (1) flow rate within the first second (F1.0) and (2) changes in calf volume (expelled volume) at 4 seconds (V4.0). Values for F1.0 and V4.0/Vsec were plotted and interpreted for venous outflow obstruction.9,10 If there was any evidence of venous obstruction in either limb by this methodology, patients were excluded from the analysis.
Venous valvular incompetence
PDR was used to provide a measure of valvular incompetence by assessing the rate of venous refilling with gravity (Figures 1C, 1D). With the patient still in the supine position, the strain gauges were moved to the mid-ankle. The chair tilt was then rapidly reversed, returning the patient to the sitting position. Venous valvular incompetence leads to an increased rate of venous refilling as measured by changes in calf volume per unit of time. Venous valvular incompetence was defined by a passive refill rate of > 5 mL/100 mL of calf volume per minute. If there was any evidence of venous valvular incompetence in either limb by this methodology, patients were excluded from the analysis. 9
Calf muscle pump (CMP)
CMP function was analyzed by exercise venous plethysmography (Figures 1E, 1F). While still in the seated position with legs dependent, the patient was instructed to perform 15 ankle flexes and then remain still for the final measures. The volume required to refill the lower limb was recorded. This refill volume is used as an estimate of calf pump function with normal values defined as a volume of ⩾ 1.0 mL/100 mL and impaired CMP as a refill volume < 1.0 mL/100 mL. 11
Assessment of covariates
The Charlson Comorbidity Index (CCI) was used to assess the impact of comorbid conditions on overall survival.12–17 All diagnoses codes comprising the CCI were extracted starting 3 years prior to the index date up to 1 year post index date. Only those diagnoses that appeared twice and were at least 30 days apart were considered. Previously established and validated coding algorithms for ICD-9 diagnoses were used to extract data on comorbidities comprising the CCI (online Supplementary Table 2). 17 A total of three CCI scores were calculated as: Sum of Diseases, Severity Weighted Sum of Diseases, and Severity and Age Weighted Sum of Diseases. Heart failure was ascertained as a part of the CCI comorbidities using ICD-9 codes. Further subclassification into heart failure with preserved or reduced ejection fraction was confirmed using echocardiographic data.
Outcome measure
Study outcome was all-cause mortality. Mortality data were obtained from the National Death Index. 18 Death rates were analyzed at 5, 10, and 15 years from the index vascular laboratory study date.
Statistical analysis
Clinical measurements and descriptive characteristics were summarized using frequencies for categorical variables and mean, SD, minimum, and maximum for continuous variables. Differences between groups were analyzed using chi-squared test for categorical variables and t-test for continuous variables. Kaplan–Meier plots were created to analyze the difference in survival time between impaired bilateral and normal bilateral muscle pump. The probabilities of survival between subcategories of bilateral pump function in the setting of heart failure were also plotted. A Cox proportional hazards model was used to adjust for all the relevant covariates in the study, with time to death as the outcome of interest. Bilateral impaired CMP, sex, age, Charlson score, creatinine level, and whether a patient had experienced myopathy, embolism, neuropathy, arthritis or heart failure were all included as covariates. The results of the model are reported using the hazard ratios and corresponding 95% CIs. In all cases, a two-tailed p-value of less than 0.05 was considered statistically significant. Statistical analysis was done using SAS statistical software (SAS version 9.4; SAS Institute Inc., Cary, NC, USA).
Results
During the study period (January 1, 1998 – December 31, 2011), 2726 unique patients underwent venous plethysmography at the Mayo Clinic Gonda Vascular Laboratory, met inclusion and exclusion criteria, and were included in the analysis. A total of 1788 patients had bilateral impaired CMP and 938 had normal CMP.
A number of clinical variables distinguished these two groups (Table 1). Patients with impaired CMP were on average nearly 6 years older compared to those with normal CMP (60.8 ± 17.2 vs 54.9 ± 14.8; p < 0.001) and had a higher percentage of females (69.5% vs 64.6%; p = 0.01). They had a higher prevalence of neuropathy (13.5% vs 8.0%; p < 0.001), arthritis (9.4% vs 5.1%; p < 0.001), myopathy (5.1% vs 2.0%; p < 0.001), and heart failure (15.4% vs 8.3%; p < 0.001) compared to patients with normal CMP. Atrial fibrillation (5.0% vs 2.5%; p = 0.001), hypertension (25.1% vs 16.3%; p < 0.001), dyslipidemia (21.1% vs 17.6%; p = 0.028), diabetes mellitus (9.2% vs 5.2%; p < 0.001), and coronary disease (11.9% vs 5.4%; p < 0.001) were also more common for those patients with impaired CMP. The mean Charlson score was higher amongst the impaired CMP group (0.7 ± 1.4 vs 0.4 ± 1.2; p < 0.001). There was no significant difference in the prevalance of smoking, prior VTE or cancer between these two groups. There were no differences in body mass index (BMI) comparing patients with poor pump function and normal pump function.
Baseline demographics.
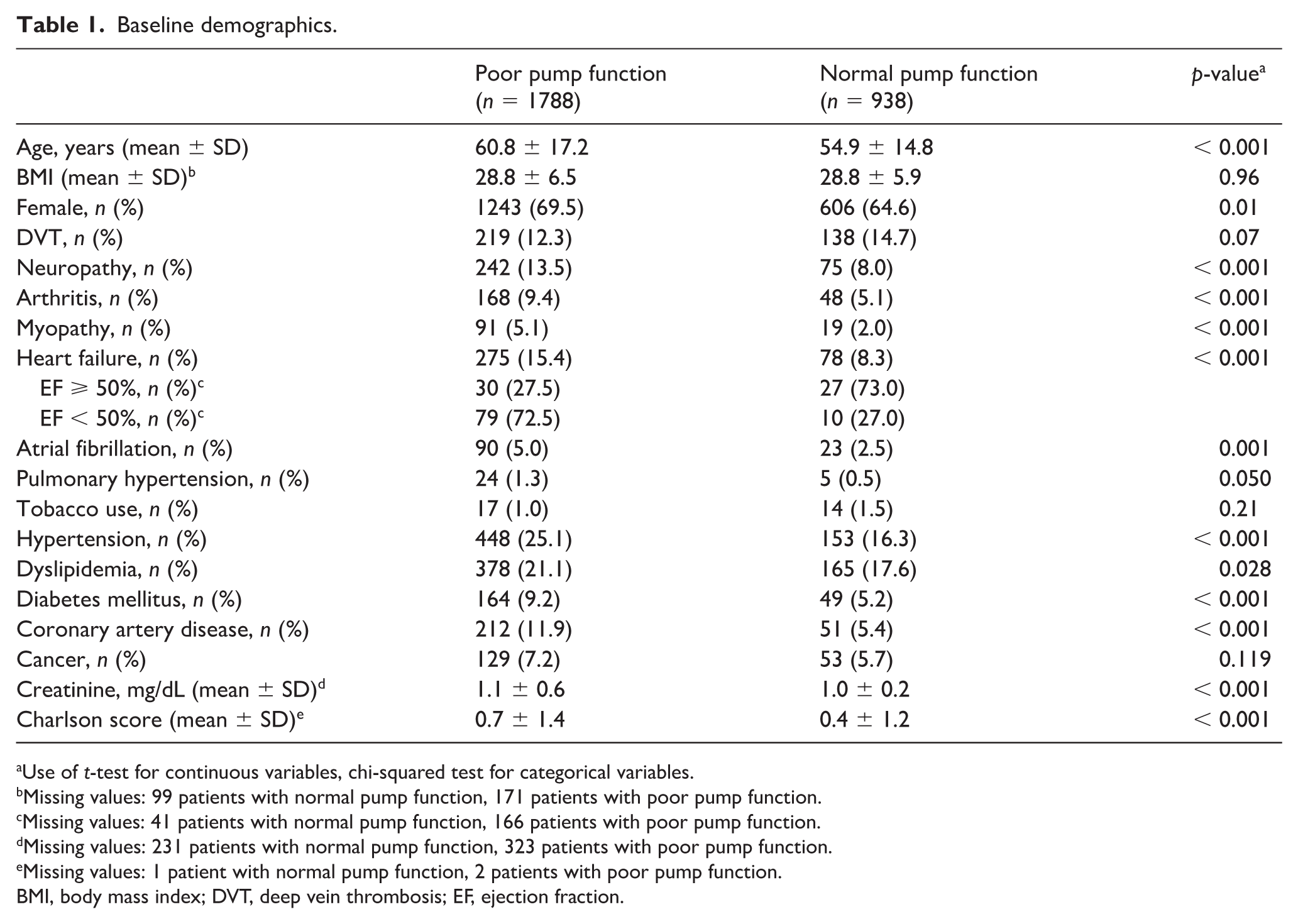
Use of t-test for continuous variables, chi-squared test for categorical variables.
Missing values: 99 patients with normal pump function, 171 patients with poor pump function.
Missing values: 41 patients with normal pump function, 166 patients with poor pump function.
Missing values: 231 patients with normal pump function, 323 patients with poor pump function.
Missing values: 1 patient with normal pump function, 2 patients with poor pump function.
BMI, body mass index; DVT, deep vein thrombosis; EF, ejection fraction.
Mortality assessment
The overall mortality rates for patients with impaired CMP were significantly higher at years 5, 10, and 15 compared to those with normal CMP (Table 2, Figure 2). There were 353 patients with heart failure enrolled in this study. As anticipated, patients with heart failure had greater all-cause mortality and worse survival compared to those without heart failure at each of the 5-year increments (Table 2, Figure 3). Amongst patients with heart failure, impaired CMP was associated with greater all-cause mortality with worse survival compared to those heart failure patients with normal CMP at each time interval (log-rank p = 0.03). Among the 2373 patients without a diagnosis of heart failure, those with impaired CMP had significantly greater all-cause mortality rates compared to those with normal CMP (log-rank p < 0.001).
All cause mortality rates at 5-year time intervals (5–15 years).
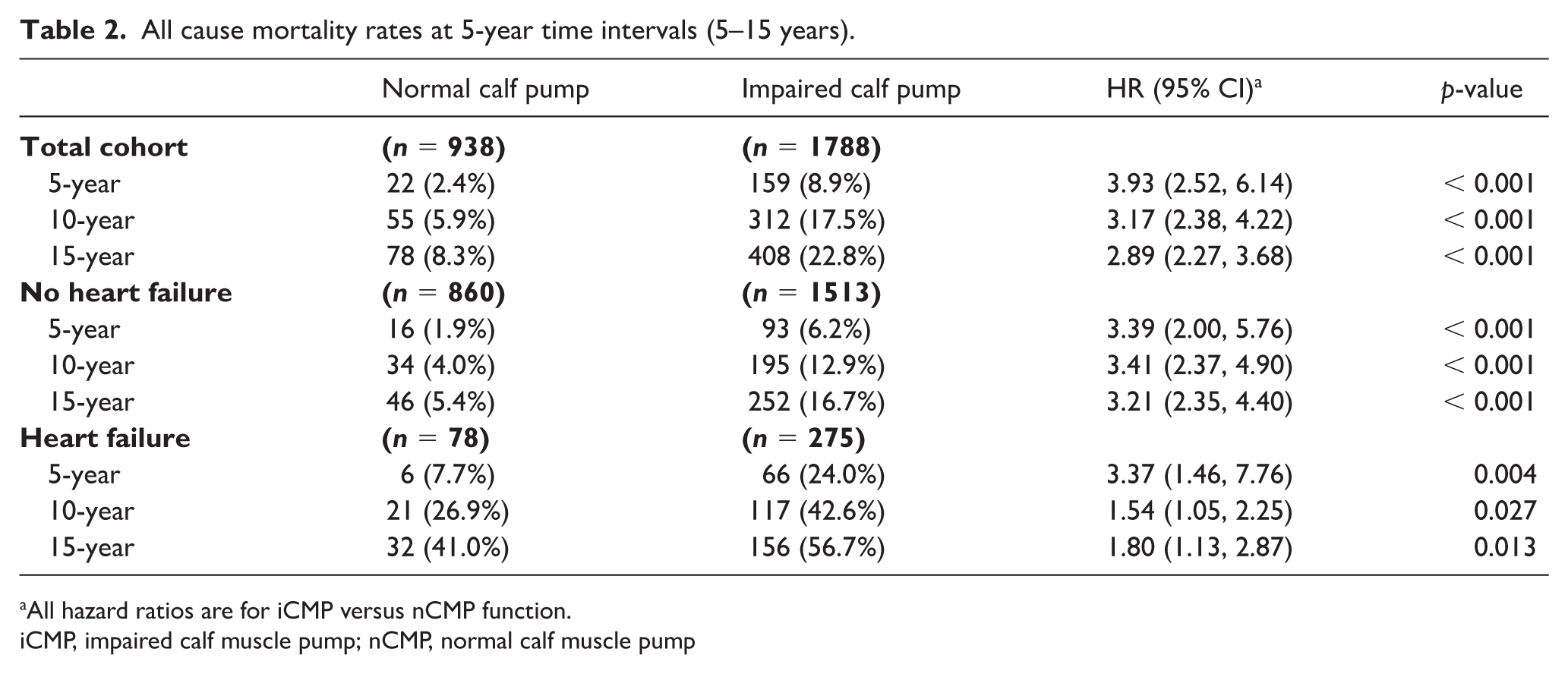
All hazard ratios are for iCMP versus nCMP function.
iCMP, impaired calf muscle pump; nCMP, normal calf muscle pump
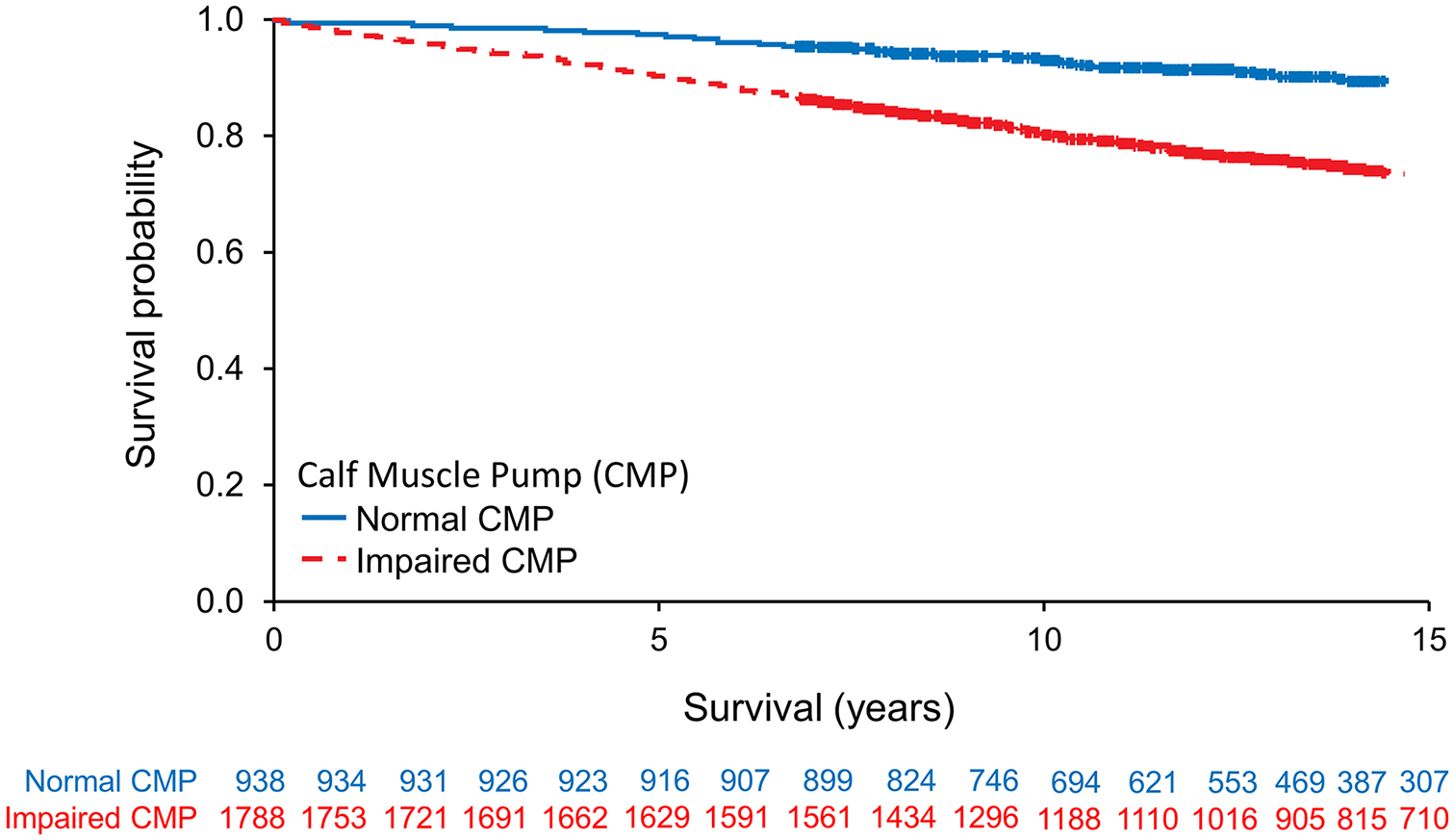
Kaplan–Meier survival stratified by CMP function.
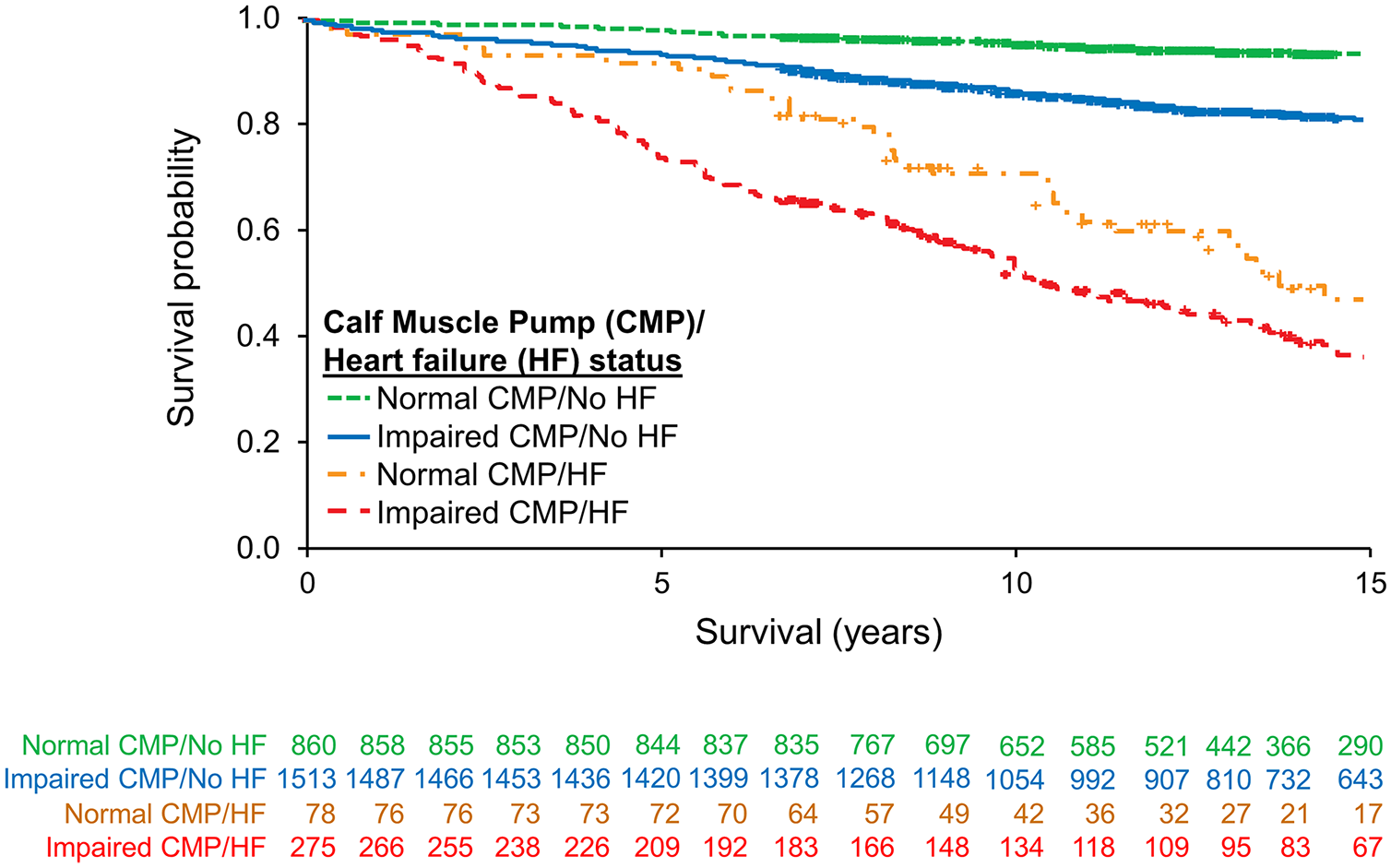
Kaplan–Meier survival stratified by CMP function and heart failure status.
Cox proportional hazards model for survival at 15 years.
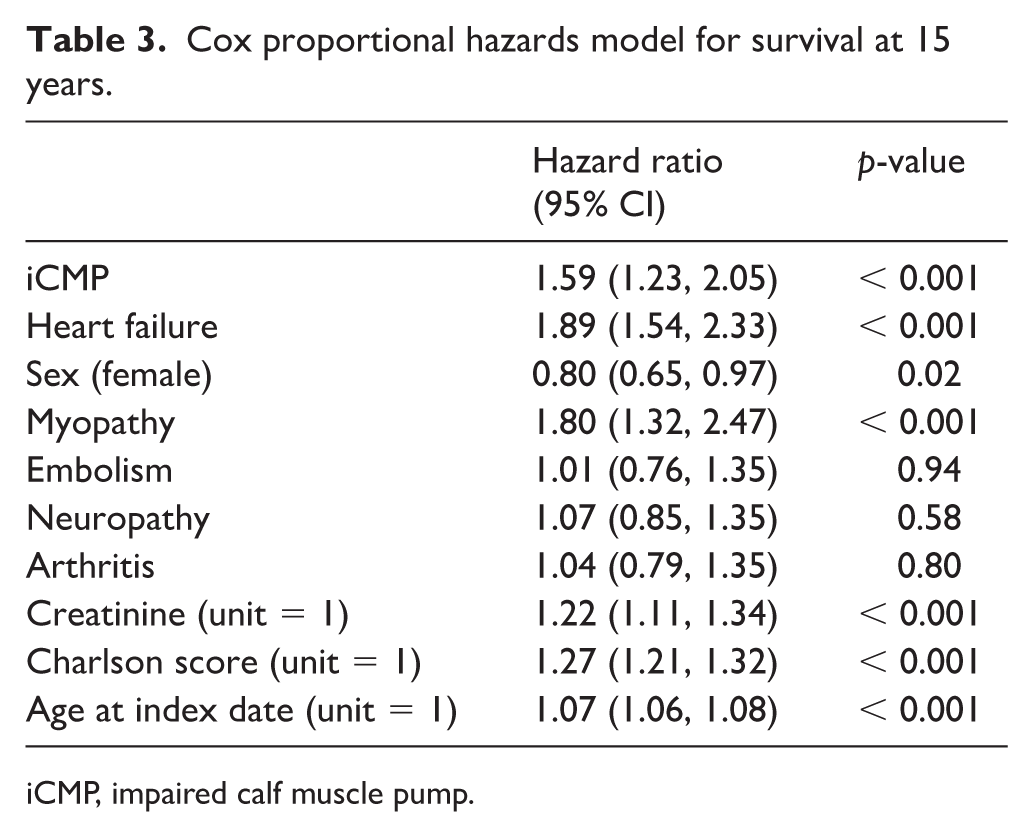
iCMP, impaired calf muscle pump.
The two most common causes of death amongst patients with CMP dysfunction were cardiovascular disease and cancer (Table 4). This was also true for patients with normal calf pump function. Of the combined cohort, there were 49 deaths due to unknown causes.
Cause of death throughout entire study.
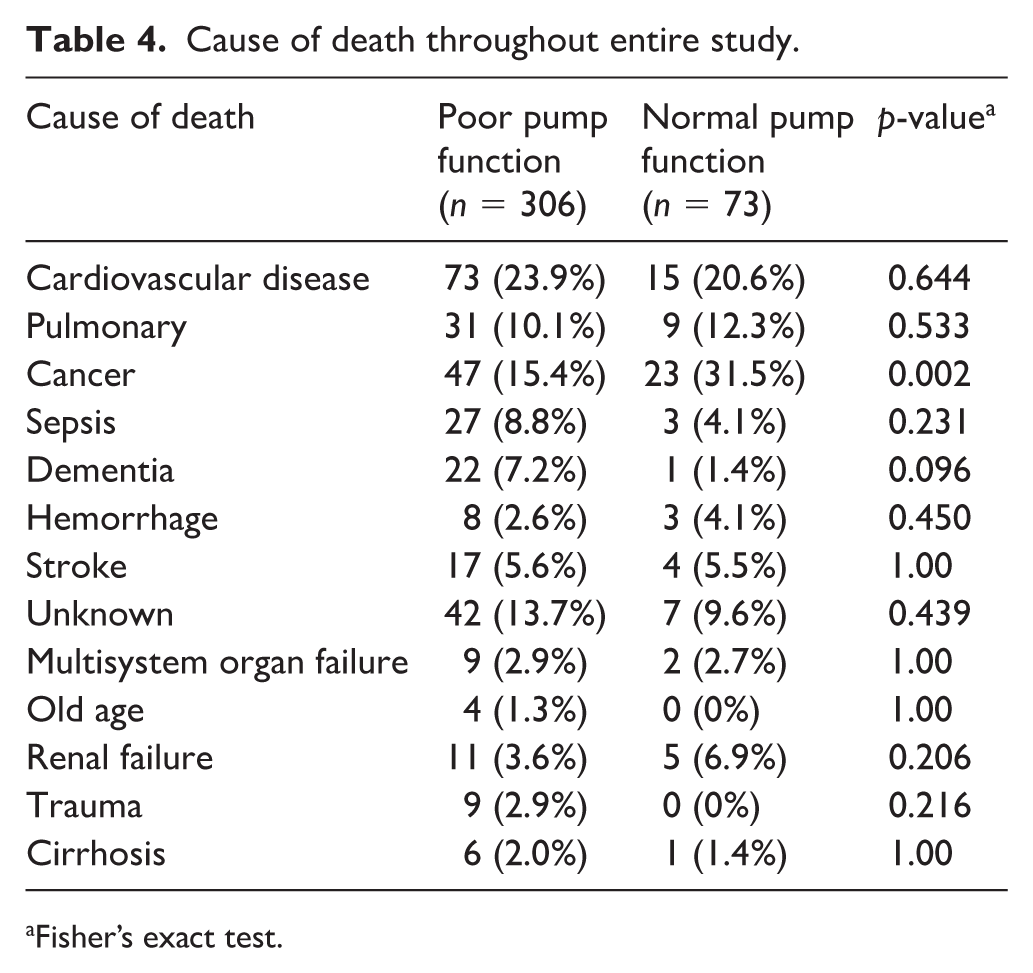
Fisher’s exact test.
Discussion
The central findings of this study are: (1) patients with impaired CMP have significantly greater all-cause mortality rates at 5, 10, and 15-year time intervals compared to patients with normal CMP; and (2) amongst patients with heart failure, impaired CMP appears to be associated with increased all-cause mortality rates compared to patients with heart failure and normal CMP. While there are a number of relevant comorbid conditions which differ amongst the two populations in this study, impaired CMP function remained an independent predictor of all-cause mortality after adjusting for these comorbid variables. While an association does not prove causality, a number of hypotheses for this finding could be proposed. An impaired CMP may simply reflect a measure of frailty or cumulative disease burden. Alternatively, by limiting cardiac preload, poor CMP function could impair cardiac output, reduce organ perfusion, and activate the renin-angiotensin aldosterone system with all the associated direct and indirect adverse consequences. Regardless of the physiologic consequences, this relatively simple, noninvasive, rapid, and inexpensive measure might help further refine disease severity and prognosis for patients with or without heart failure.
Lower calf muscle density measured by computerized tomography and decreased muscle strength have been associated with increased mortality in patients with peripheral artery disease. 19 Sarcopenia, loss of muscle strength, and increased fat infiltration of skeletal muscle have been associated with higher all-cause and cardiovascular mortality in the geriatric population.20–23 In contrast, low fat mass and high muscle mass is associated with lower cardiovascular and all-cause mortality in patients with cardiovascular disease. 24 Low muscle strength is associated with inflammation and mortality in dialysis patients. 25 Calf circumference was the strongest predictor of mortality in a population of emphysematous patients. 26 While decreased muscle mass, density, and strength in the context of other comorbid conditions could predict adverse outcomes and survival, the impact of such parameters on outcomes in healthy individuals has also been reviewed. Those with higher upper and/or lower body muscle strength had a lower mortality regardless of age in a recent meta-analysis of approximately 2 million adults. 27
Heart failure is known to negatively impact survival.28–30 Non-cardiovascular causes of mortality have been noted to contribute to 54.3% of heart failure deaths. 30 Calf circumference, used to measure sarcopenia, has been shown to be an independent predictor of future heart failure events as well as being associated with poor survival in patients with heart failure. 31 Skeletal muscle wasting has been associated with poor exercise capacity in patients with heart failure. 32 Small studies assessing calf muscle volume, calf muscle size and gait characteristics in patients with heart failure have found structural and functional impairment of the soleus muscle.32–34 There are several unanswered questions to be addressed with future studies: (1) can strength training improve impaired CMP function; (2) can knowledge of CMP function help direct therapy for patients with heart failure; and (3) do medications which impact pre-load differentially affect HF patients with and without impaired CMP. Our long-term goal is to determine whether strategies for improving calf muscle strength and endurance will improve calf muscle function. The ultimate goal is to determine whether exercise prescription for calf muscle strengthening resulting in improvements of CMP will improve longevity.
Study limitations
There are several study limitations that should be acknowledged. First, there may be a referral bias where only patients with symptoms sufficient to warrant evaluation in the Vascular Laboratory would have been studied. Cases were identified from an institutional vascular laboratory database which may not be representative of other community-based populations. Only those patients referred to the Mayo Clinic for venous hemodynamic assessment were included, thus risking a referral bias. These patients may be sicker than other community-based patients, which could contribute to sampling bias and higher mortality that may not be generalizable to the rest of the population with chronic venous diseases undergoing calf pump function assessments. Second, strain gauge plethysmography has largely been replaced by air plethysmography for calf pump function assessment. Third, lifestyle-related factors such as physical activity status, diet, obesity, and other relevant health history not included in the Charlson Comorbidity Index could impact outcomes. Furthermore, of the three components of the venous physiology, exercise venous plethysmography requires patient effort. Despite aggressive coaching, complete patient participation may not have been maximized. Tobacco exposure, which could negatively impact calf muscle circumference, function, and outcomes, may be underrepresented in our study sample due to underreporting, missing data, or inaccurate electronic record entry. Fourth, the retrospective study design limits any causality assumptions. Fifth, providers referring patients to the Gonda Vascular Center for venous evaluation have two testing strategy options. These include physiologic testing in the Noninvasive Vascular Laboratory and anatomic testing in Vascular Ultrasound. Both laboratories are located on Gonda 4, are physically adjacent to each other, and cross refer for various testing strategies. The work-flow in the Noninvasive Vascular Laboratory includes prompt referral for ultrasound imaging if there is any evidence of venous obstruction by physiologic testing. Ultrasound imaging is not obtained on all patients without a specific referral. It is possible that this strategy may miss subtle disease, including non-occlusive venous thrombi.
Conclusions
After adjusting for age, sex, and other comorbid conditions that comprise the Charlson Comorbidity Index, impaired CMP function was associated with an increased all-cause mortality amongst patients with and without heart failure. Impaired CMP due to structural and functional mechanisms may represent a surrogate measure of frailty. Alternatively, impaired CMP may negatively impact survival due to the inherent interaction between preload and overall cardiac functionality.
Supplemental Material
Supplementary_Table_1 – Supplemental material for Calf muscle pump function as a predictor of all-cause mortality
Supplemental material, Supplementary_Table_1 for Calf muscle pump function as a predictor of all-cause mortality by Meghana Halkar, Jose Medina Inojosa, David Liedl, Waldemar Wysokinski, Damon E Houghton, Paul W Wennberg, Grace Lin, Garvan Kane, Karen Fischer, Thom W Rooke, Rayya Saadiq, Amanda Bonikowske and Robert D McBane II in Vascular Medicine
Supplemental Material
Supplementary_Table_2_ICD_9_Codes – Supplemental material for Calf muscle pump function as a predictor of all-cause mortality
Supplemental material, Supplementary_Table_2_ICD_9_Codes for Calf muscle pump function as a predictor of all-cause mortality by Meghana Halkar, Jose Medina Inojosa, David Liedl, Waldemar Wysokinski, Damon E Houghton, Paul W Wennberg, Grace Lin, Garvan Kane, Karen Fischer, Thom W Rooke, Rayya Saadiq, Amanda Bonikowske and Robert D McBane II in Vascular Medicine
Supplemental Material
Supplementary_Table_3_Cox_Proportional_Hazard_Models – Supplemental material for Calf muscle pump function as a predictor of all-cause mortality
Supplemental material, Supplementary_Table_3_Cox_Proportional_Hazard_Models for Calf muscle pump function as a predictor of all-cause mortality by Meghana Halkar, Jose Medina Inojosa, David Liedl, Waldemar Wysokinski, Damon E Houghton, Paul W Wennberg, Grace Lin, Garvan Kane, Karen Fischer, Thom W Rooke, Rayya Saadiq, Amanda Bonikowske and Robert D McBane II in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
