Abstract

Patients with or at risk of arterial or venous thromboembolism (VTE) often require long-term oral anticoagulant therapy (OAT), but anticoagulation is a risk factor for intracranial hemorrhage, a devastating complication, which occurs in about five in 1000 patients per year. 1 This incidence is higher in patients with risk factors, such as older age, prior stroke, uncontrolled hypertension, prior intracranial hemorrhage, and in those who are treated with vitamin K antagonists (VKAs) rather than with direct oral anticoagulants (DOACs). 1 All types of intracranial hemorrhage (intracerebral, subdural, and subarachnoid) have the potential to cause serious morbidity as well as mortality, but intracerebral bleeding (ICH) has the worst prognosis: about half of such patients will suffer progressive neurological deterioration during the first 24–48 hours after admission, about half will eventually die at 30 days after an index event, and about two-thirds of those surviving will experience significant disability. 2 In those who survive, about one in 10 will suffer a recurrent ICH and this risk is highest for patients with lobar ICH, often caused by amyloid angiopathy, and in whom the risk of recurrent ICH can be at least 20%. 3
Whether and when to resume OAT after an initial ICH remain vexing questions. Many clinicians choose not to restart OAT because of the fear of recurrent ICH. Indeed, about one-half to three-quarters of patients with atrial fibrillation (AF) do not resume OAT after ICH in clinical practice despite observational data suggesting that the reintroduction of OAT in such patients is associated with lower rates of ischemic stroke and mortality when compared with no OAT resumption. 4 Thus, a meta-analysis of eight observational studies that included 5306 AF patients with ICH showed that OAT resumption halved the risk of thromboembolism without a significant increase in ICH recurrence, but these findings may be prone to selection bias. 4 Recognizing the limitations in the evidence, the 2016 European Society of Cardiology AF guideline recommended that the decision to resume anticoagulation after ICH ‘should be taken by a multidisciplinary team’, without a clear guidance on when, in whom, and how to resume. 5
To address the issue of when to resume anticoagulation, Pappas and Burke modelled the time-dependent risk of recurrent ICH in a large nationwide database study that included a total of 5339 patients with AF hospitalized for anticoagulant-related ICH to determine the earliest time when resuming OAT would result in a net clinical benefit. 6 Net clinical benefit was calculated by weighing the harm (in terms of excess in ICH) with the benefit (in terms of reduction in ischemic stroke) if anticoagulation was resumed, and in doing so, they were able to identify the earliest time when the net clinical benefit exceeded zero.
Their analyses revealed several key findings pertinent to the optimal timing of resuming anticoagulation after ICH. First, unlike ischemic stroke, the risk of recurrent ICH declined rapidly over time: about 50% of recurrent ICH occurred within 38 days, and about 80% by 4 months. Second, given the declining risk of ICH after an index event, resuming anticoagulation eventually led to a net clinical benefit over time, but the timing of resumption was critical to assure such benefit, and was highly influenced by the risk of recurrent ICH and, to a lesser extent, by the risk of ischemic stroke. Thus, in a study population with an annualized rate of ICH of about 2.5% and a median CHA2DS2-VASc score of 4, the benefit in preventing ischemic stroke exceeded the harm of anticoagulant therapy if it was resumed 11 days after the index discharge, but the confidence interval was wide, with a 95% upper margin of 42 days. In sensitivity analyses, the best estimate was slightly reduced to 9 days if a higher rate of ischemic stroke (10% per year) was assumed, and conversely, it increased to 19 days (with a 95% upper margin of about 9 weeks) if the risk of recurrent ICH (7.2%) was assumed to be about threefold higher.
Although other studies reported different estimates for the optimal time to resume anticoagulation, the upper margin of the estimates in the study by Pappas and Burke overlapped with those. In a study by Majeed and colleagues, which included 234 patients with warfarin-associated ICH with an indication for long-term anticoagulation (e.g. AF, VTE, or mechanical valve), and in which the risk of recurrent ICH was much higher than in the current study (10% vs 2.5%), the optimal time to restart warfarin was later and estimated to be between 10 and 30 weeks after an index ICH. 7 In another study from the Swedish Stroke Registry that included 2619 patients with AF and ICH, 96 (3.7%) patients had recurrent ICHs, and the optimal time to resume anticoagulant therapy occurred between 7 and 8 weeks after the initial ICH. 8 The variability in these estimates is not surprising and is likely accounted for by differences in the baseline risks of recurrent ICH and thrombotic complications, as well as by differences in the types of analytical models and underlying assumptions.
The key strengths of this study by Pappas and Burke are the highly relevant findings and the insights it provides when trying to integrate the time-dependent decline in ICH recurrence in the risk–benefit analysis to determine the optimal time to restart anticoagulation. 6 Several limitations, of which some were acknowledged by the investigators, include: (a) the relatively imprecise estimates of net clinical benefit that also led to considerable uncertainty in the time to consider OAT resumption; (b) the lack of stratification by ICH subtype to verify that the time-dependent decline in ICH applied to all risk subtypes and whether and when a net clinical benefit is achieved in patients with the highest risk of recurrent ICH (e.g. cerebral amyloid angiopathy); (c) the potential for case ascertainment bias and missed ICH recurrences given the use of the International Classification of Diseases (ICD) and administrative codes to identify this outcome; (d) uncertainty about the robustness of assuming a constant risk of ischemic stroke when other studies have shown a higher risk of ischemic complications in those with a recent major bleed (especially after intracranial bleeding) 9 ; and (e) the limited definition for net clinical benefit that did not take into account other potential benefits (e.g. reductions in VTE and myocardial infarction) and harms of anticoagulation (e.g. extracranial bleeding and cost) that are also important to patients. Nonetheless, Pappas and Burke reported on one of the largest cohorts of patients with AF and ICH, and the insights provided are of considerable interest to clinicians.
How does this study inform clinical decision-making? The current study shows that resuming anticoagulation is likely to be of net harm in the first 2 weeks after an initial ICH in a population with a 2.5% risk of ICH recurrence, but this period could possibly extend to at least the first 10 weeks if the risk of ICH recurrence is higher or the tolerability for a new ICH is low. However, uncertainty about net clinical benefit remains in some patients with the highest risk of ICH recurrence (e.g. patients with lobar ICH secondary to cerebral amyloid angiopathy) given the lack of reporting on this specific ICH subtype. Consequently, there is scope for clinical judgement and the engagement of patients to individualize clinical decisions on resuming anticoagulation.
Several issues pertinent to net clinical benefit after ICH merit discussion. First, the issue of which anticoagulant (and which dose) to resume was not specifically addressed. Although this study modelled the net clinical benefit associated with VKAs, the DOACs are as effective as VKAs for stroke prevention in AF and have lower risk of ICH, 10 and their use (with or without dose reduction) would likely enhance the net clinical benefit of anticoagulation in this population. Second, anticoagulation is not the only available strategy. Novel mechanical approaches, such as left atrial appendage closure, have the added advantage of mitigating thromboembolism without increasing bleeding and are therefore promising as bridging measures or useful adjuncts in the setting of lower intensity antithrombotic therapy. 11 Finally, although the study does not directly address the issue of resuming anticoagulation after ICH in other thrombotic disorders such as VTE, it would be reasonable to apply similar considerations using the appropriately adjusted weight for the thrombotic outcome to inform on the optimal timing for OAT resumption. Data informing on resumption of anticoagulation are even more scarce for patients with VTE, although a few small observational studies found that resuming anticoagulation was associated with a trend to fewer thromboembolic complications and reduced mortality without a significant increase in recurrent ICH. 12 Based on low-quality evidence, the 2018 American Society of Hematology VTE management guideline suggests resumption of OAT within 90 days after ICH rather than discontinuation in patients with a moderate to high risk of VTE recurrence. 13
In summary, resuming OAT in patients with ICH who have a long-term indication for anticoagulation remains a clinical dilemma. In the absence of randomized trials, observational studies like the current one provide useful insights on the time-dependent change in ICH recurrence and the net clinical benefit to assist in the decision to resume anticoagulation in patients with AF and ICH, but the findings require further validation in randomized controlled trials (RCTs). With several RCTs recently initiated (Table 1), we can expect more definite answers to the questions of in whom, when, and how to resume anticoagulation in these patients in the coming years. However, much more remains to be done to resolve these questions in other thrombotic conditions.
Ongoing randomized trials evaluating the benefit of resuming anticoagulation after ICH in patients with atrial fibrillation.
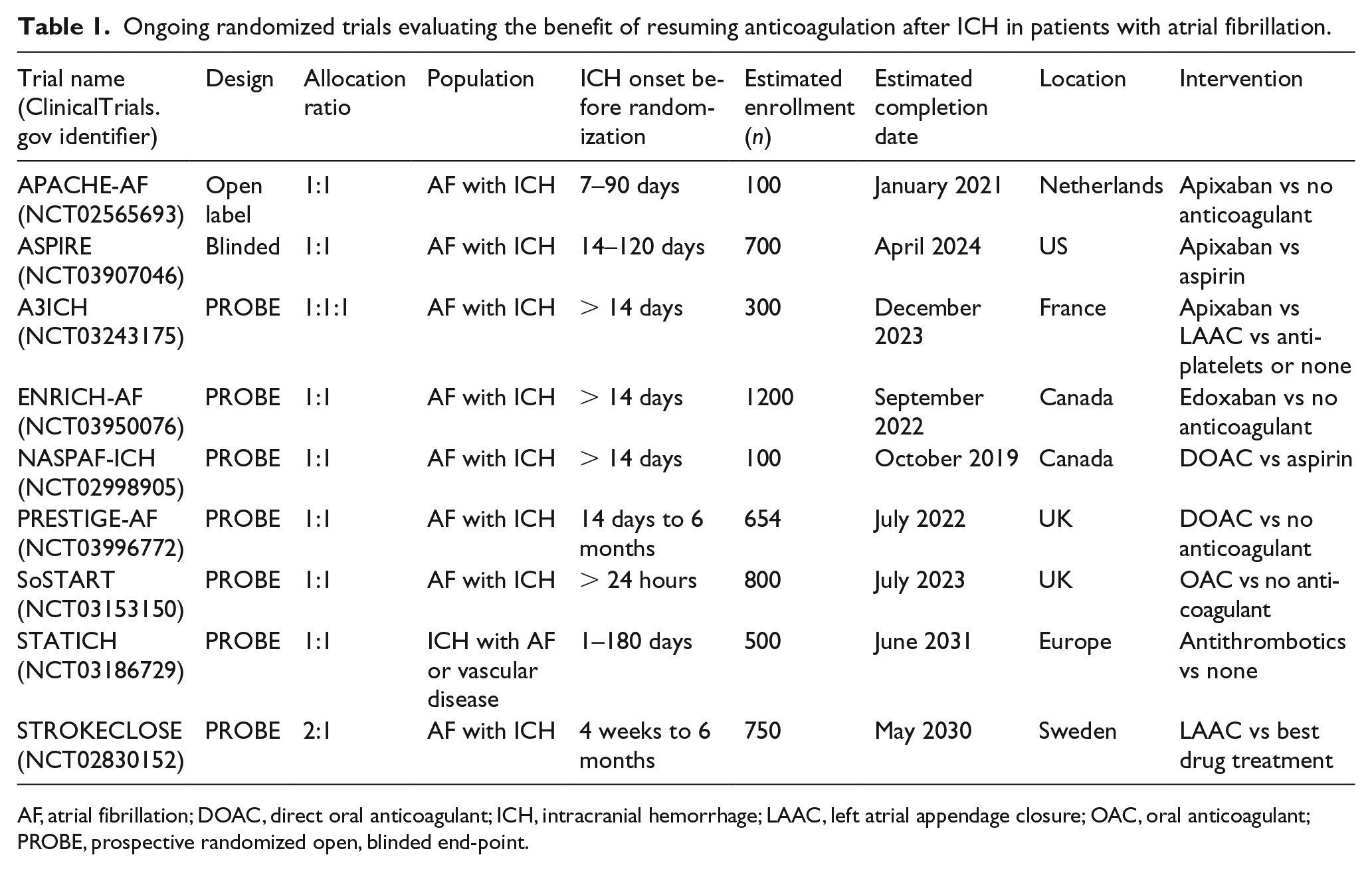
AF, atrial fibrillation; DOAC, direct oral anticoagulant; ICH, intracranial hemorrhage; LAAC, left atrial appendage closure; OAC, oral anticoagulant; PROBE, prospective randomized open, blinded end-point.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
