Abstract
Fibrosing mediastinitis (FM) is a rare disorder of inflammation and fibrosis involving the mediastinum. The formation of fibroinflammatory mass in the mediastinum can lead to obstruction of mediastinal structures and cause severe debilitating and life-threatening symptoms. Superior vena cava syndrome (SVCS) is a dreaded complication of FM with no medical therapy proven to be efficacious. Spiral vein grafting has long been utilized as first-line therapy for SVC syndrome due to FM. Endovascular repair with stents and angioplasty for malignant causes of SVC syndrome is well established. However, there are limited data on their utility in SVC syndrome due to FM. We present two cases of SVC syndrome due to FM treated with endovascular stenting and a detailed review of current literature on its utility in SVCS due to benign causes.
Keywords
Introduction
Fibrosing mediastinitis (FM) is a rare disorder of inflammation and fibrosis involving the mediastinum. The pathogenesis is hypothesized to be a severe fibroinflammatory host response to various antigens, such as Histoplasma, Mycobacterium, or Treponema.1,2 Autoimmune conditions, such as granulomatosis with polyangiitis, Behcet’s disease, and IgG4 disease, have also been associated with FM. 3 Histoplasmosis is the most common etiology for FM in the United States. However, in a large outbreak, only three out of 100,000 patients diagnosed with histoplasmosis developed FM. 4 The immune response begins in the lymph nodes, leading to mediastinal granulomatous lymphadenitis. The antigen may escape into the mediastinum, causing a hypersensitivity reaction and subsequent fibrosis. This process may result in compression of mediastinal structures and obstructive symptoms. FM is a diagnosis of exclusion, once other causes of mediastinal lymphadenitis have been ruled out. Superior vena cava syndrome (SVCS) is known to occur in more than 42% of patients with FM. 5 The most common symptoms include headache, upper extremity and facial swelling, vision changes, and cerebral edema. Medical treatments of anti-fungal therapy, steroids, or tamoxifen have been utilized with varying degrees of success, but no prospective trials have demonstrated efficacy.5,6
Spiral vein bypass grafting has long been utilized as first-line surgical treatment for SVCS secondary to FM. 7 Spiral vein grafting is a surgical technique developed by Chiu et al. for the repair of large-caliber vessels such as the superior vena cava (SVC). An autologous saphenous vein is harvested and longitudinally resected. It is then wrapped spirally around a large bore cylindrical tube, similar to an elastic bandage around a limb, and then secured by running sutures. The tube is then removed, and the resultant tubular spiral graft is used to repair or bypass the SVC. 8 Spiral vein grafts offer long-term patency and may be superior to synthetic grafts (polytetrafluoroethylene grafts). 9
Minimally invasive procedures, such as percutaneous transluminal angioplasty (PTA) or a self-expanding metallic stent placement, have shown significant short-term symptomatic improvement in SVCS secondary to malignant conditions. 10 Studies have also evaluated the effectiveness of endovascular procedures in SVCS resulting from benign conditions. 11 However, information on the effectiveness of endovascular stent placement in SVCS from fibrosing mediastinitis is limited. We report two cases of SVCS from fibrosing mediastinitis, with significant clinical improvement following endovascular self-expanding stent placement, and present a focused review of the available literature.
Methods
We highlight two case studies of successful treatment of SVCS due to FM, using endovascular stent placement. We conducted a nonsystematic, focused review of the literature on endovascular stenting in SVCS due to FM. PubMed Central, Medline, and individual scholarly journals were searched for keywords including endovascular stenting, covered stents, superior vena cava syndrome in fibrosing mediastinitis, fibrosing mediastinitis, anticoagulation after venous stenting, spiral vein graft bypass, and percutaneous transluminal angioplasty. All the relevant literature that focused on SVCS, endovascular stenting for venous disease or fibrosing mediastinitis was thoroughly reviewed. Case series, retrospective studies, and prospective studies in the English language literature were included in the review due to the rarity of the disease and limited literature on the topic. Studies with SVCS due to benign causes including FM were included in the review and are summarized in (Table 1).
Studies assessing the role of endovascular stenting and open surgical repair in SVCS due to benign causes.
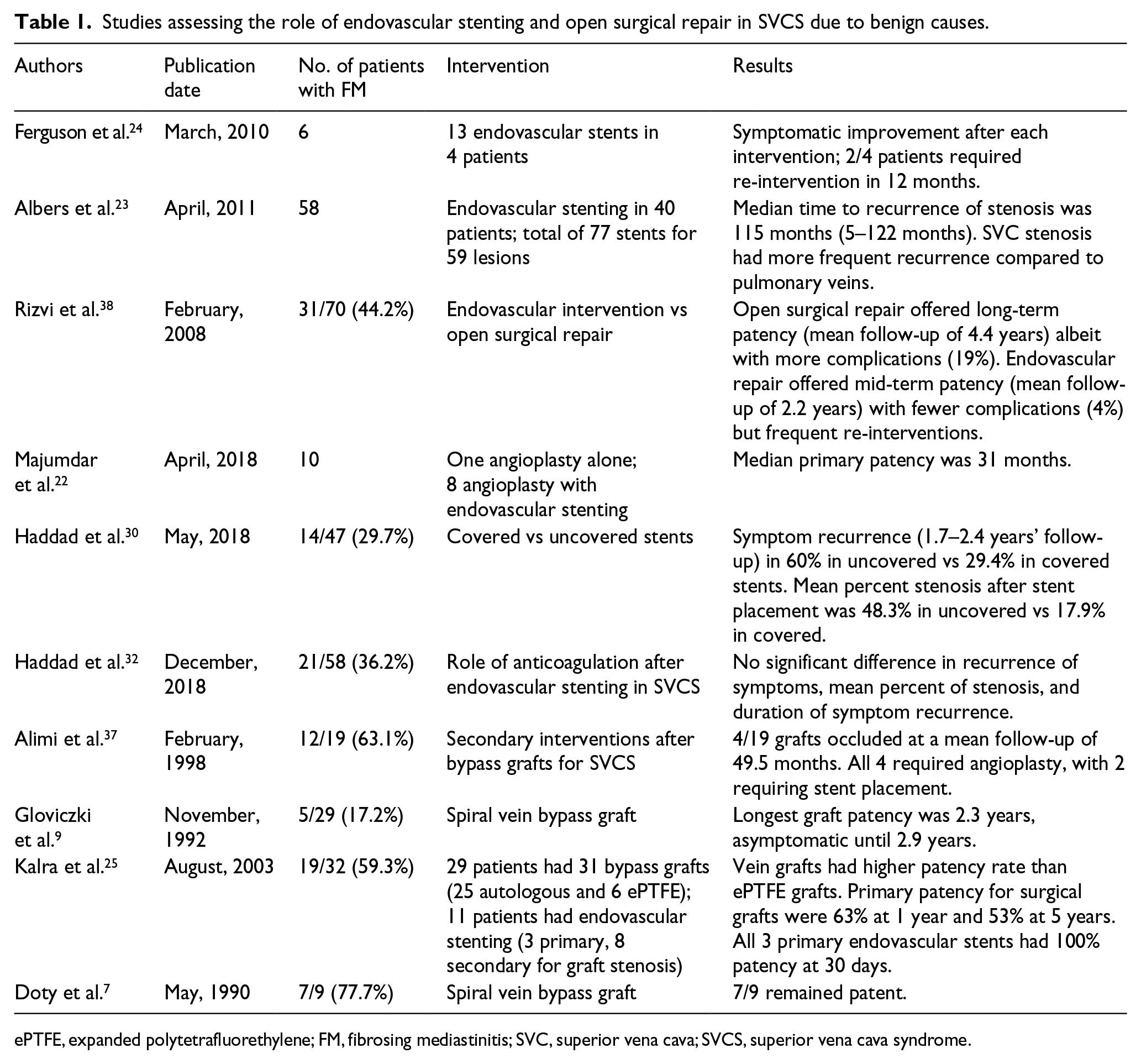
ePTFE, expanded polytetrafluorethylene; FM, fibrosing mediastinitis; SVC, superior vena cava; SVCS, superior vena cava syndrome.
Case 1
A 26-year-old Caucasian female resident of Southwest Ohio with a 10 pack-year smoking history, presented with a dry cough, head pressure, and distended neck veins of 6 weeks’ duration. She reported a sudden onset of sharp, stabbing chest pain that woke her up at night and prompted her to seek medical care. Her past medical history was significant for migraines that were controlled with ibuprofen. Her initial examination was significant for mild puffiness of the face and distended superficial veins of the neck and chest. A fungal panel was negative for Aspergillus species (< 1:8) and Coccidioides. However, she had elevated titers of Coccidioidomycosis complement fixation (1:2) and the Histoplasma yeast complement fixation titer was (> 1:8).
A computed tomography (CT) of the chest with contrast revealed a large right paratracheal mass encasing the SVC and multiple nodules with calcifications in both lungs (Figure 1). She underwent bronchoscopy with transtracheal fine needle aspiration (FNA) of the paratracheal mass. Cytology revealed benign lymphocytes, bronchial epithelial cells, macrophages, and fragments of dense fibrous connective tissue. 12 No granuloma or neoplasm was identified. A diagnosis of fibrosing mediastinitis secondary to histoplasmosis was made. She was treated with prednisone 20 mg daily, and with itraconazole 200 mg daily for 2 months along with rivaroxaban 20 mg daily indefinitely.
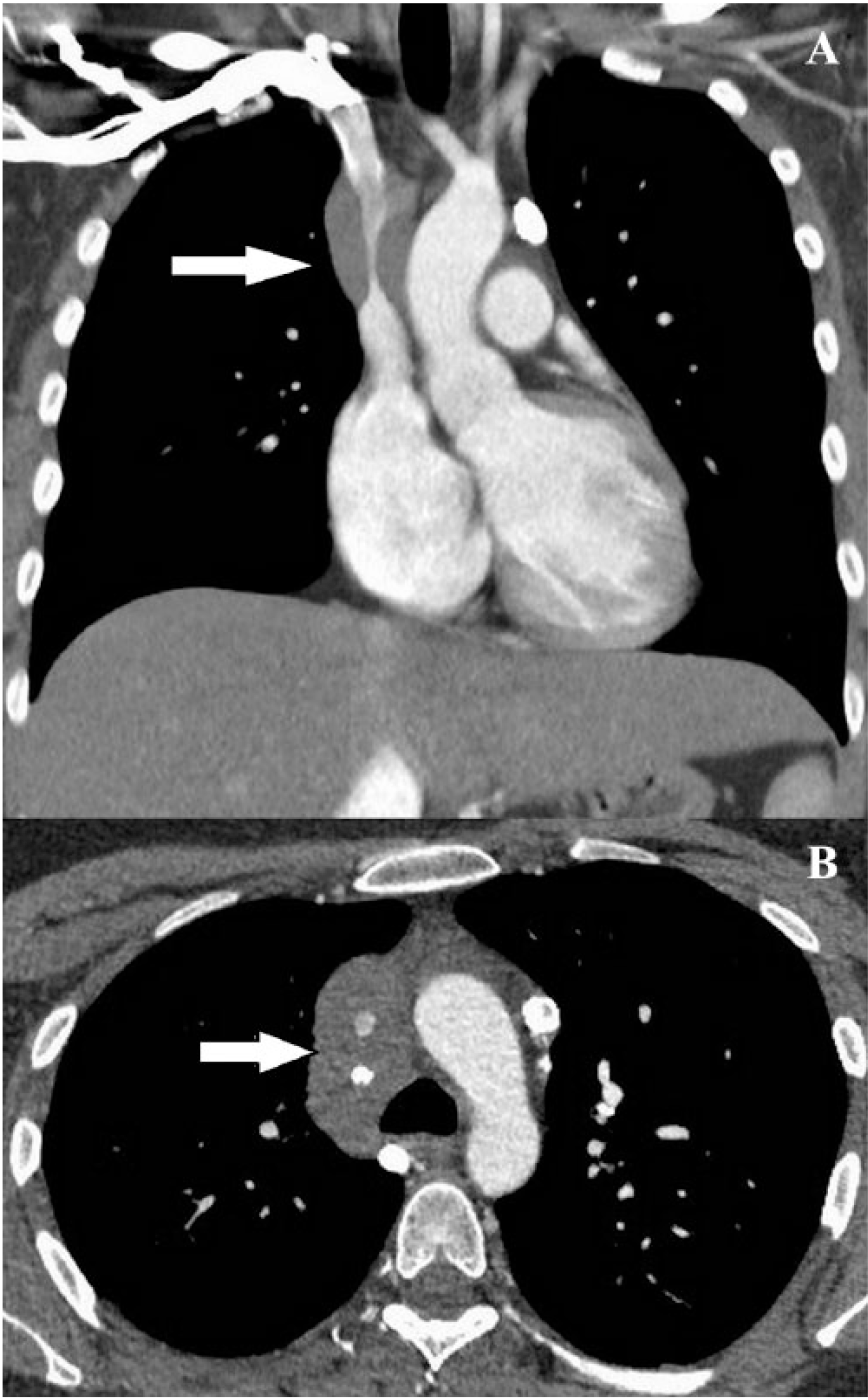
Coronal reformatted maximum intensity projection image (A) and axial image (B) from a contrast-enhanced CT venogram of the chest showing a large paratracheal mass encasing the SVC (arrow).
Her symptoms continued to progress with medical therapy, prompting the need to pursue SVC angioplasty. A venogram revealed narrowing of the SVC diameter to 1.5 mm. She initially underwent serial balloon angioplasty of the stenosis with 8 mm, 10 mm, and 12 mm balloons, leading to improvement in stenosis and an increase in SVC diameter to 6.4 mm (Figure 2). However, her symptoms did not improve for the next 4 weeks, and a repeat chest CT with contrast showed minor progression in her SVC compression. A repeat venogram showed a severe SVC stenosis extending 3 cm in length with reflux of contrast into the left innominate vein. Pre-dilation with a 10 × 40 mm balloon was conducted, followed by a Viabahn® VBX 11 × 39 mm balloon expanding stent (Gore Medical, Flagstaff, Arizona, USA) placement across the SVC, from the base of the innominate vein to the confluence of the distal SVC (Figure 3). After stent placement, the mean right internal jugular (RIJ) pressure decreased from 20 mmHg to 10 mmHg, and the mean right atrial pressure increased from 5 mmHg to 10 mmHg, suggesting an improved venous return to the heart. The patient reported remarkable improvement in her symptoms. Itraconazole and prednisone were discontinued, and she was maintained on rivaroxaban 20 mg daily. In the past 3 months of follow-up, the patient has had no recurrence of her symptoms.
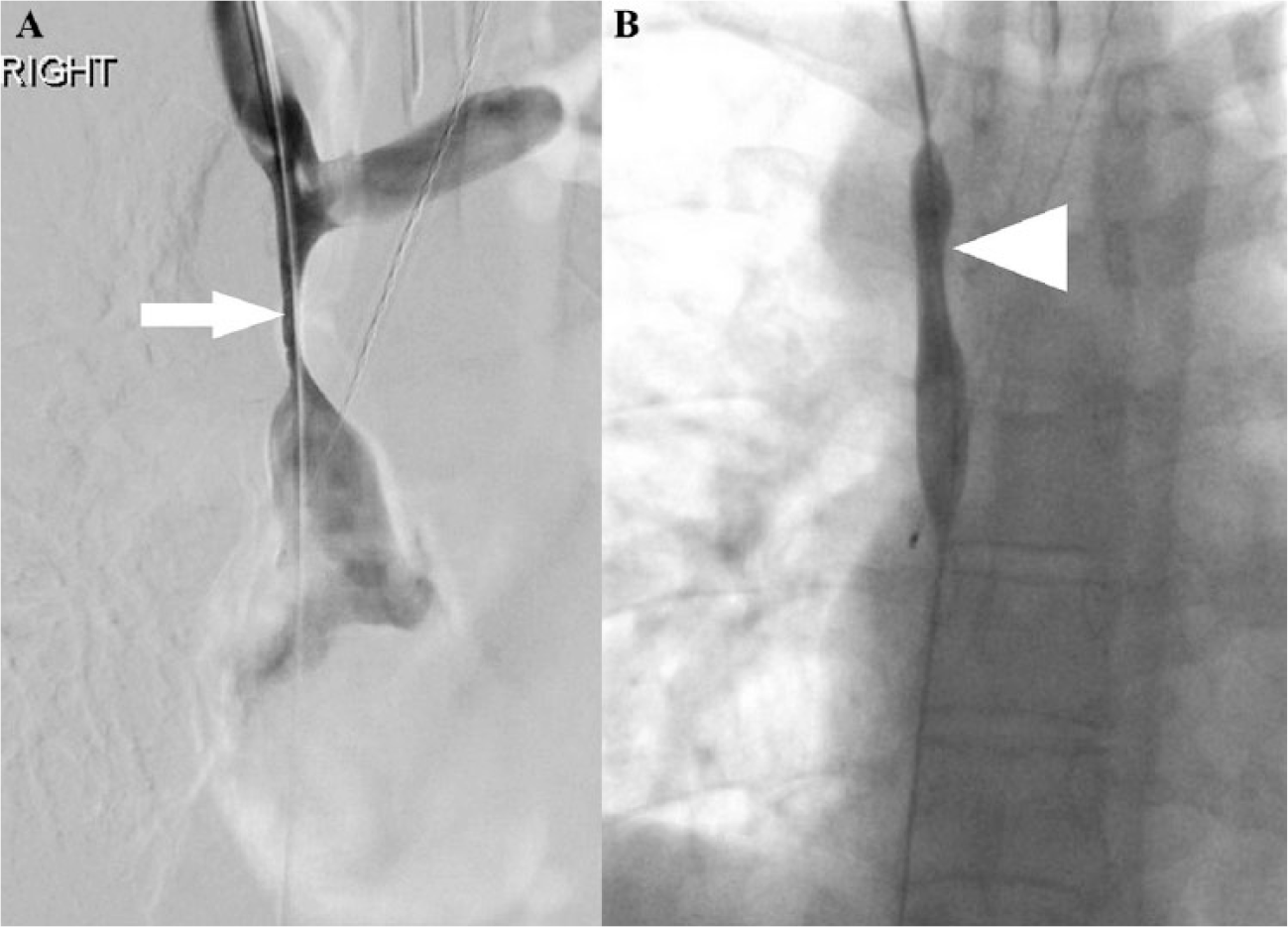
(A) Venogram revealing narrowing of the SVC down to 1.5 mm in diameter (arrow). (B) Serial balloon angioplasty of the stenosis with 8 mm, 10 mm, and 12 mm balloons, with improvement of the stenosis to a 6.4 mm diameter (arrowhead).
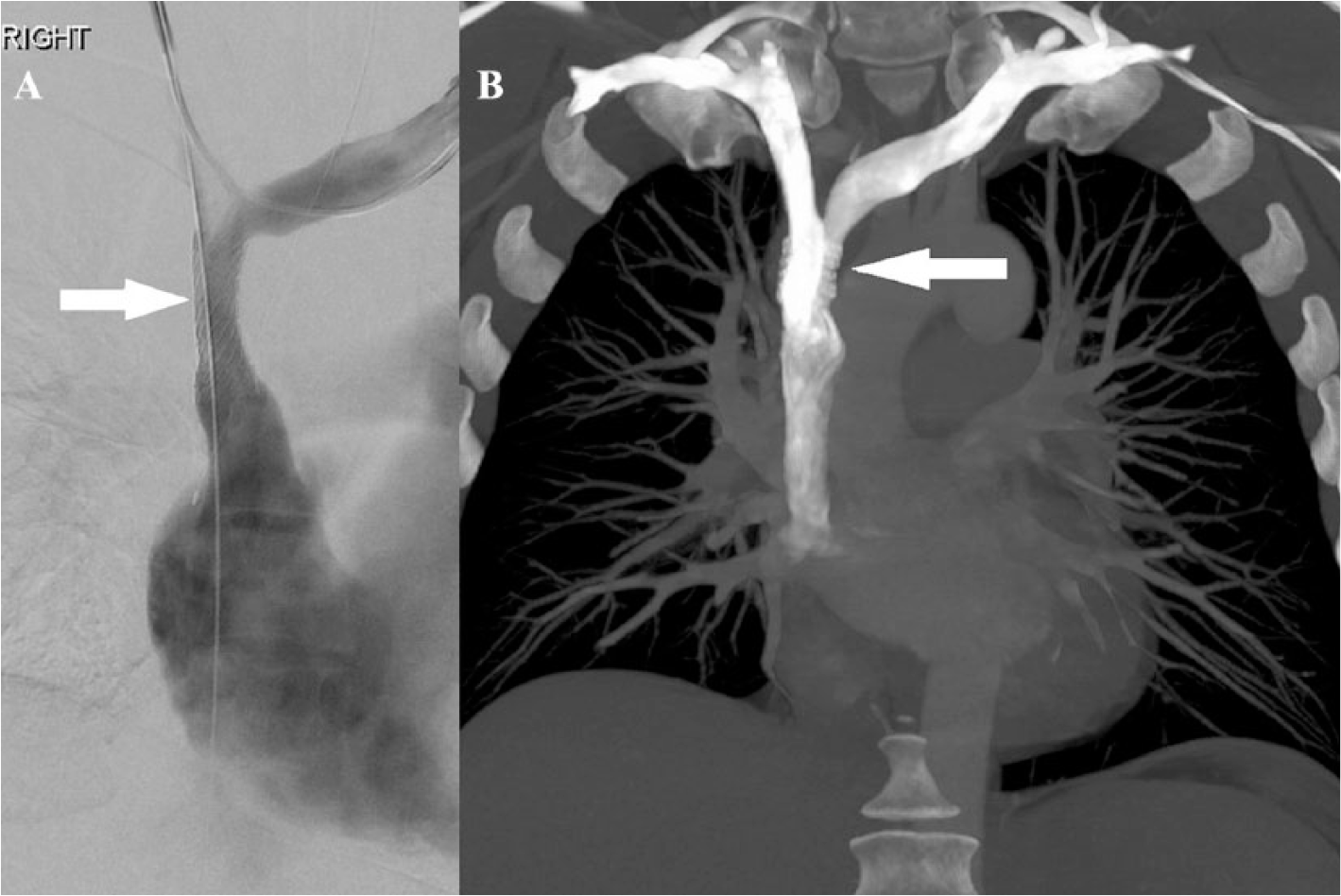
(A) Digital venogram after stent placement (arrow). (B) Coronal reformatted maximum intensity projection image showing successful stent placement (arrow).
Case 2
A 48-year-old African American male Ohio resident presented with recurrent syncopal episodes, exertional dyspnea, fatigue, facial swelling, and paresthesia of his hands and fingers of 6 months’ duration. He had a family history of amyloidosis, acute myeloid leukemia, lung cancer, and coronary artery disease. His medical history was otherwise unremarkable. Physical examination was remarkable for facial edema and distended neck veins. His laboratory workup, which included a tuberculosis screen, a fungal panel, and autoimmune serologies, were negative. A CT scan of the chest revealed a left lower lobe (LLL) nodule and mediastinal lymphadenopathy. Video-assisted thoracoscopic surgery (VATS) with biopsy of the LLL nodule revealed bland hyalinizing collagen with interspersed lymphocytes and plasma cells consistent with FM. Owing to the endemicity of histoplasmosis in Northeast Ohio, it was considered to be the most likely etiology. The patient was empirically treated with itraconazole 200 mg daily and prednisone 20 mg daily for 3 months without improvement in his symptoms. Rituximab therapy was also offered for 6 months but did not resolve his symptoms. A follow-up chest CT (Figure 4) with contrast revealed confluent mediastinal adenopathy involving the right paratracheal, subcarinal and bilateral hilar regions. Soft tissue infiltration circumferentially involved the bilateral mainstem bronchi and proximal lobar airways. Mass effect was noted on the SVC, though it appeared patent.
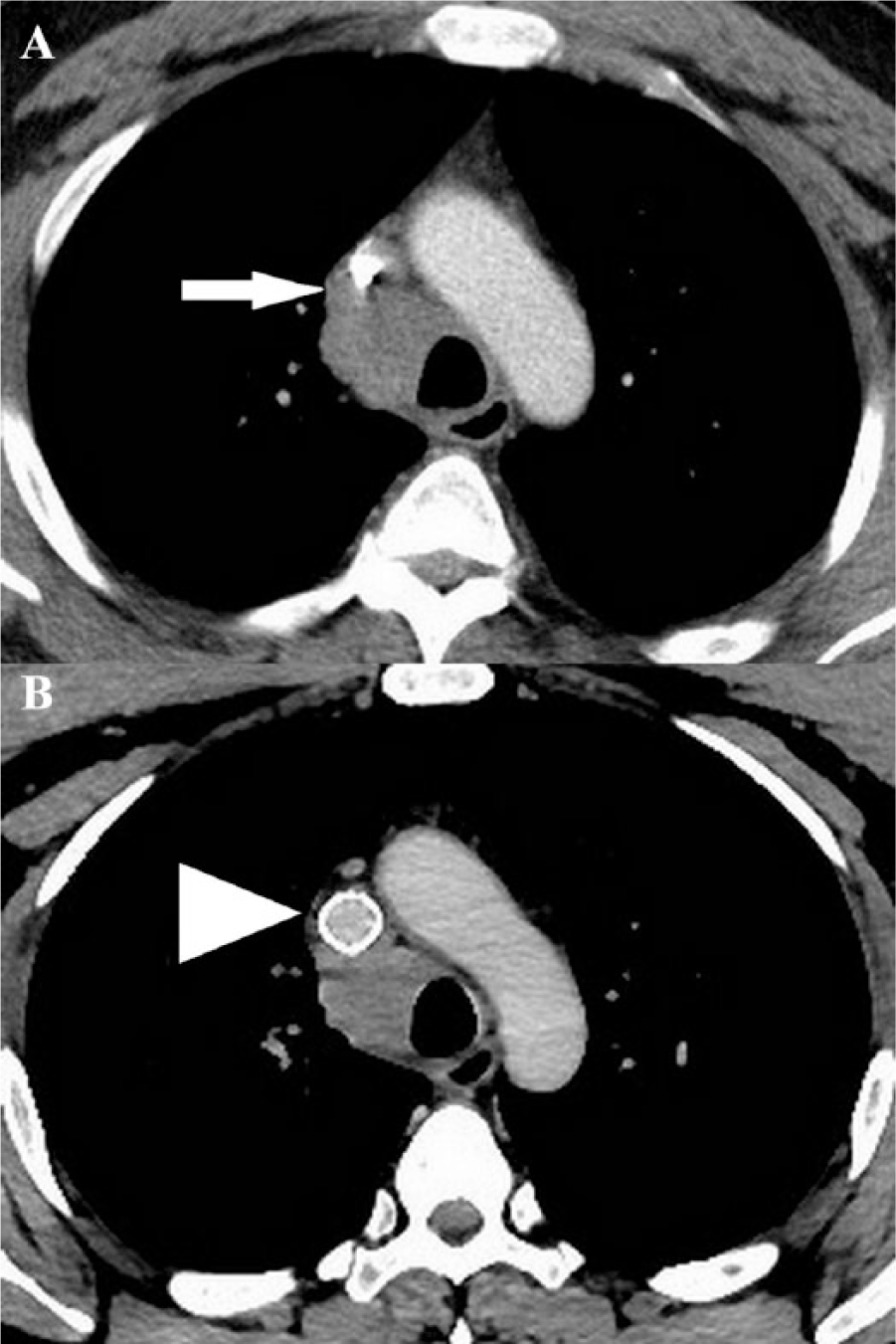
(A) Axial contrast-enhanced CT images which show the right paratracheal mass encasing the SVC (arrow). (B) Improved SVC lumen size with in situ SVC stent (arrowhead).
Owing to significant arms and facial edema, the patient underwent an SVC venogram that showed moderate short segment stenosis of the SVC from the confluence of the innominate veins to the divergence of the azygous vein. He underwent balloon dilation with improvement of stenosis from 8 mm to 12 mm. However, after several minutes, a repeat venogram showed a reduction in SVC diameter to 10 mm. Hence, a Viabahn® VBX 11 × 29 mm (Gore Medical, Flagstaff, Arizona, USA) balloon-expandable covered stent was placed successfully, with dilation of the SVC to 16 mm (Figure 5). He was discharged home on rivaroxaban 20 mg daily and reported improvement in his energy and facial swelling. At the 3-month follow-up, he reported no recurrence of facial edema or fatigue, but his arm paresthesia persisted, for which he was started on gabapentin 100 mg three times a day with subsequent improvement.
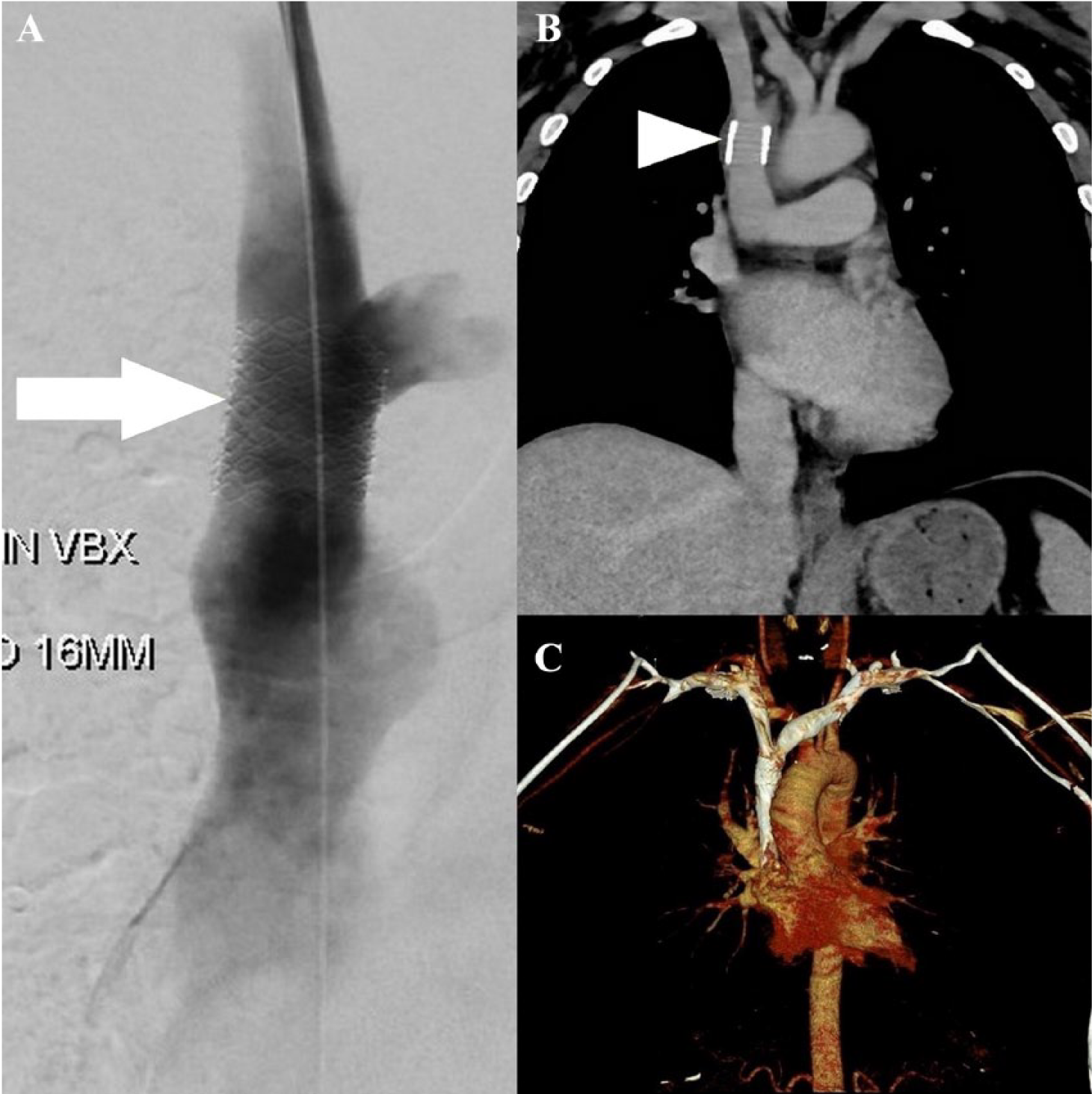
(A) Venogram after successful stent placement showing relief of the SVC stenosis (arrow). (B) Coronal maximum intensity projection reformatted images from a contrast-enhanced CT venogram of the chest showing a patent stent (arrowhead). (C) 3-D volume-rendered image reconstructed from the same CT showing no obstruction to flow or venous collateralization.
Discussion
FM is a rare fibroinflammatory disorder that results in the compression of mediastinal structures, such as pulmonary arteries and veins, central veins, the esophagus and airways. The pathogenesis is not clearly understood but is thought to be either an autoimmune or a host immune response to a fungal (Histoplasma), parasitic, mycobacterial or bacterial antigen13,14 (Table 2). Patients may remain asymptomatic for several months to years before significant obstruction of mediastinal structures occurs. Shortness of breath, hemoptysis, dysphagia, headaches, facial swelling, syncope, and chest pain are common symptoms. 5
Various etiologies of fibrosing mediastinitis.
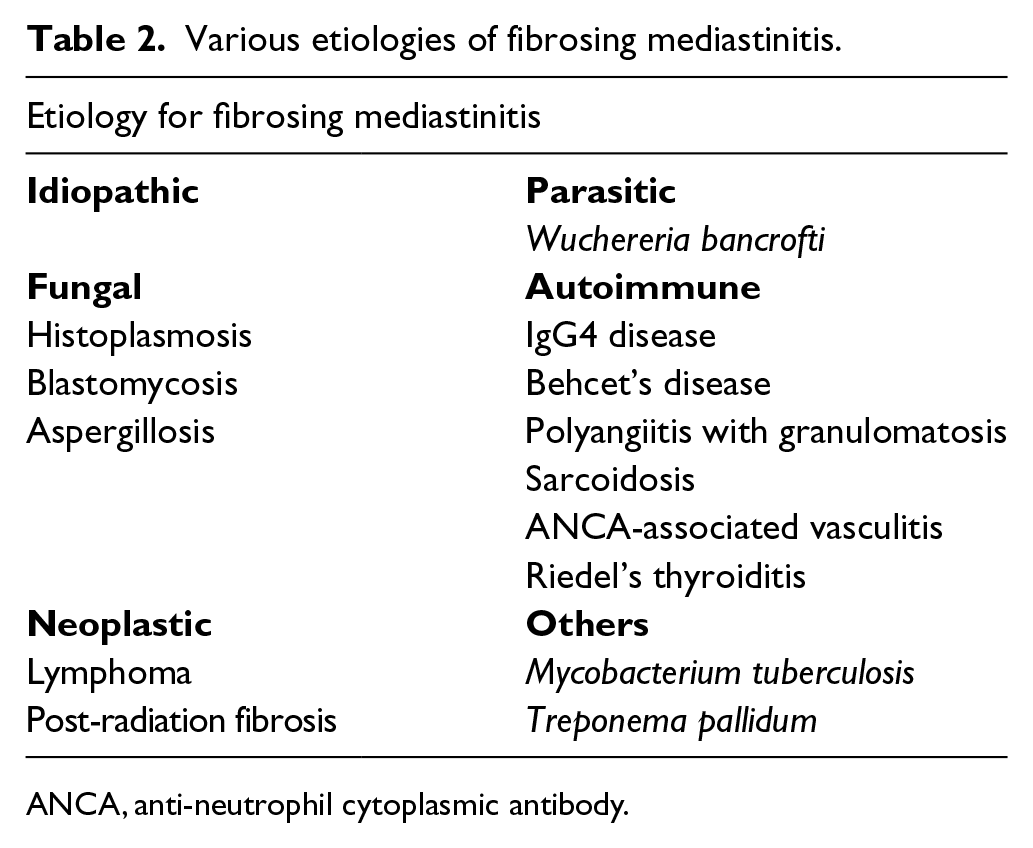
ANCA, anti-neutrophil cytoplasmic antibody.
Pharmacologic treatment of fibrosing mediastinitis (FM)
Treatment of FM is extremely challenging as the tissue is metabolically active. Several medical therapies have been attempted without clear benefit. One case report describes successful treatment with tamoxifen and steroids. 6 Tamoxifen has been used in the treatment of other fibroinflammatory disorders, such as desmoid tumors and retroperitoneal fibrosis.15,16 It is hypothesized that fibroblasts contain estrogen receptors and tamoxifen may inhibit pro-fibrotic cytokines, such as protein kinase C, epidermal derived growth factors and calmodulin. 17 The role of B-lymphocytes in triggering the fibrotic response has also been hypothesized. 5 Rituximab may reduce metabolic activity and lead to clinical improvement and a reduction in fibrotic activity. This outcome was reported in a case series of three patients conducted by Westerly et al. 18 A reduction in metabolic activity and mediastinal mass size with rituximab therapy was demonstrated in one patient and a reduction in pleural effusion in another two patients with FM who were concomitantly treated with steroids as well. Another study demonstrated the efficacy of rituximab and steroid in combination in nine patients with FM. 19 However, five out of nine patients had undergone several airway and vascular interventions for compressive symptoms before rituximab treatment was initiated. The utility of rituximab remains unclear due to the lack of prospective randomized controlled trials (RCTs). In cases of significant obstructive symptoms, open surgical or endovascular procedures have been performed.
SVCS is a known complication that requires revascularization to prevent cerebral edema and its hemodynamic sequelae. Central venous occlusion is a common manifestation of FM and may occur in up to 42% of patients. 5 Thoracic central vein obstruction (TCVO) can be classified as partial (stenosis) or complete obstruction (occlusion). 20 Central venous obstruction is defined as involvement of the SVC or brachiocephalic veins or a venous obstruction in close proximity to the right atrium. 20 For the ease of classification, the Society for Interventional Radiology Reporting Standards for Thoracic Central Vein Obstruction classifies TCVO into four patterns based on the location of obstruction and the remaining patent veins (Table 3).
Classification of TCVO by the Society for Interventional Radiology Reporting Standards for TCVO 20 .
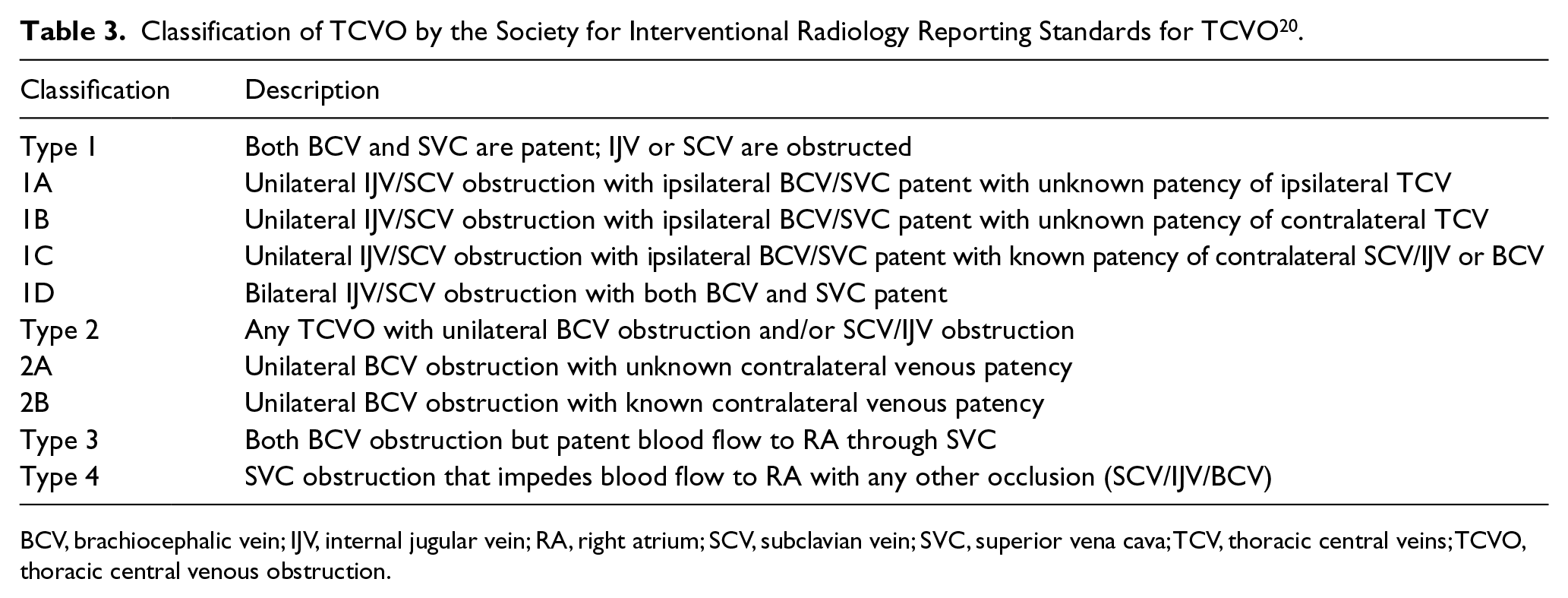
BCV, brachiocephalic vein; IJV, internal jugular vein; RA, right atrium; SCV, subclavian vein; SVC, superior vena cava; TCV, thoracic central veins; TCVO, thoracic central venous obstruction.
Endovascular stenting in superior vena cava syndrome due to FM
Endovascular stent placement has frequently been utilized to treat hemodynamically or clinically significant central venous occlusions in malignant SVCS as it provides significant short-term palliation. 10 This treatment is preferred due to its percutaneous approach and fewer complications, as compared to open surgical interventions such as bypass grafting, and is a reasonable choice in cancer patients with limited life expectancy. Owing to the rarity of SVCS and fibrosing mediastinitis, RCTs to assess the role of endovascular stents have been difficult to conduct. Most data in the available literature have been derived from small retrospective analyses and case reports. Most studies demonstrate significant clinical and hemodynamic improvement after transcutaneous angioplasty and stenting; however, the recurrence of stenosis is common, and frequent stent revisions are required. 21
Majumdar et al. demonstrated that the use of endovascular stenting relieved symptoms within 24 hours in nine out of 10 patients with SVCS secondary to FM. In their study, primary stents lasted a median of 31 months with subsequent revisions lasting a median of 6 months. 22 Albers et al. placed 77 stents for 59 lesions (pulmonary artery – 26, pulmonary vein – 21, and SVC – 12) in 40 patients with central venous obstruction due to FM. 23 They demonstrated a significant reduction in pressure gradients across the stenosis and an increase in the vascular caliber of the pulmonary artery, pulmonary vein, and SVC affected by fibrosing mediastinitis. All patients who underwent SVC stenting reported symptomatic relief. However, seven out of 12 patients with SVC stents needed re-stenting due to recurrence, which was mainly attributed to in-stent tissue growth. The median time to recurrence was 115 months, more commonly occurring in SVC stents compared to pulmonary vein stents. They also reported improved 5-year overall survival in the intervention group (89.5%) compared to the nonintervention group (52.5%). 23 Ferguson et al. also demonstrated clinical and hemodynamic improvement in a case series of six patients requiring 13 percutaneous interventions. 24 Owing to prior reports of frequent restenosis and in-stent thrombosis with self-expanding stents, they utilized larger balloon expanding SVC stents. However, they found no advantage in terms of restenosis between the two. 24
Kalra et al. conducted a retrospective analysis of 32 patients to assess open surgical and percutaneous endovascular interventions for SVCS secondary to benign etiology. 25 Their study cohort had 19 patients with FM. Primary percutaneous transluminal angioplasty (PTA) and endovascular stenting was performed in three patients while eight patients who underwent surgical bypass grafting subsequently required PTA alone (four grafts) or PTA with stenting (five grafts). In early results for the bypass subgroup of 29 patients, five re-interventions of thrombectomy were required for graft thrombosis with a 30-day primary patency of 84% and secondary patency of 100%. Interestingly, for the three patients in the primary PTA/endovascular stenting group, 30-day primary patency was 100%. The bypass subgroup showed complications, such as acute deep vein thrombosis (DVT) of the popliteal vein, mediastinal hematoma, and bilateral vocal cord paralysis. Four patients experienced prolonged ventilator dependence. No in-hospital mortality was observed. Owing to the lower number of patients receiving primary PTA/stenting, complication rates could not be correlated. However, in the long-term follow-ups at 1 year, 3 years, and 5 years, the primary patency rates for the bypass group were 63%, 53%, and 53%, respectively. All three patients in the primary PTA/stenting group required re-stenting, with a mean clinical follow-up for patency of 2 years. 25
Covered versus uncovered stents
Gordon et al. demonstrated that intimal hyperplasia and in-stent stenosis occurred more frequently in the venous system compared to the arterial system when the same stents were used. 26 The venous system is a low flow state and more prone to thrombosis. Another concern is that of intimal/intraluminal hyperplasia that may cause occlusion of the vessel. Whether the stent material interacts with venous endothelium differently than with the arterial endothelium is unclear. Thus, it is important to utilize a stent that provides the minimum risk of thrombosis or intimal reaction. Ideally, it should withstand radial, axial, and shear stresses in the venous system and be flexible in tolerating external compression forces. 27
Balloon expandable stents are utilized when precise deployment is required. They can be adjusted to the size of the native vessel. However, being less flexible, they are externally compressible and prone to kinking. Self-expanding stents are more flexible and less prone to compression. They are useful in locations requiring more movement and flexion of the vessel, such as subclavian and innominate veins. 28
Bare metal stents (BMS), also known as uncovered stents, have been utilized for central venous occlusive disease (CVOD). However, no data suggest it is superior to balloon angioplasty alone. Besides, the higher risk of in-stent stenosis or thrombosis in uncovered stents is a concern. Uncovered stents are sometimes preferred as occlusion of venous branches can be avoided. 27 However, due to the risk of intimal hyperplasia and thrombosis, covered stents are now the preferred stents of choice.
Covered stents have a membranous covering of an inactive material that reduces the risk of intimal hyperplasia while fostering endothelialization to occur. They are designed to act as a safety net in order to trap any unstable plaque or thrombus, thus preventing embolization. 29 This feature makes them favorable for use as the risk of restenosis or thrombosis is lower. The most commonly used compound is polytetrafluoroethylene (PTFE), which is the least thrombogenic and does not evoke a robust cellular reaction compared to other products, such as polyurethane (PU) or polyethylene terephthalate (PET). 29
Haddad et al. compared covered with uncovered stents for SVCS due to benign etiology. Thirty percent (14/47) of patients in their study had FM. They found symptoms recurred in 60% (18/30) patients with uncovered stents compared to 29.4% (5/17) patients with covered stents. They also found a significant difference in the mean percentage of SVC stenosis in uncovered (48%) versus covered stents (17.9%). No clinically significant difference was found in the mean duration of symptom recurrence in both stented groups. Their study suggested that both types of stents are useful for endovascular stenting, with covered stents demonstrating superiority in terms of reduced recurrence and severity of restenosis. 30
Role of anticoagulation after endovascular stenting
No prospective trials have assessed the role of anticoagulation in venous stenting. However, due to the low-flow and low-shear states of the venous system, the risk of thrombosis is high. Anticoagulation has been recommended for iliocaval stenting in chronic venous insufficiency and post-thrombotic stenosis. However, the practice of using anticoagulation after venous stenting is extrapolated from studies assessing the role of anticoagulation for stenting in the arterial system. 31 Patients who received endovascular stents are often anticoagulated, especially if the patient has underlying thrombophilia, malignancy, previous venous thrombosis, or atrial fibrillation. Haddad et al. conducted a retrospective study to assess the role of anticoagulation in SVC stenting for benign causes. In anticoagulated and nonanticoagulated groups, there was no significant difference in the return of symptoms (44.4% vs 50%, respectively), mean percent of stenosis (40.4% ± 34.7% vs 32.1% ± 29.2, respectively) or mean duration of recurrence of symptoms (735.9 ± 1003.1 days vs 478 ± 826.6 days, respectively) in anticoagulated and nonanticoagulated groups. 32 Endo et al. conducted a retrospective study to assess the role of antiplatelet therapy in iliocaval venous stenting. They found that antiplatelet therapy and concomitant anticoagulation was significantly associated with primary stent patency compared to anticoagulation alone. 33 However, in a survey of various specialists regarding the role of antithrombotic therapy in deep vein stenting, the international Delphi consensus found no agreement on the role of antiplatelet therapy, while most agreed on the utility of anticoagulation therapy for 6–12 months after stenting. 34 The role of anticoagulation in SVCS due to external compression such as FM has not been explored. Anticoagulation therapy should be individualized to each patient’s needs and risk factors for thrombosis. The role of anticoagulation or antiplatelet therapy in endovascular stenting for SVCS due to FM may warrant further studies as the risk of bleeding with anticoagulation is always a concern.
Complications of endovascular stenting
Endovascular interventions are usually safe procedures with complications being uncommon, as documented in the abovementioned studies. An insertion site hematoma can occur but is often a minor complication. 35 Pericardial hemorrhage and cardiac tamponade have been reported as life-threatening complications of the endovascular approach.5,30 Rare cases of SVC perforation or aortic laceration have also been documented in the literature and are usually lethal.35,36
Endovascular stenting vs surgical options
Spiral vein graft bypass surgery has long been first-line surgical treatment for SVCS secondary to benign disease, such as FM. Doty et al. demonstrated long-term efficacy of spiral vein grafts in nine patients with benign causes of SVCS (seven patients with FM). They found seven out of nine grafts remained patent during 15 years’ follow-up, and eight patients remained free of SVCS. 7 Composite grafts from femoral, saphenous, innominate or internal jugular veins and expanded PTFE (ePTFE) grafts have been utilized with success. Several studies have demonstrated a long-term patency rate with bypass surgery. However, endovascular procedures have frequently been required to improve the patency of these grafts. 37
Rizvi et al. compared the durability and efficacy of endovascular repair to open surgical reconstruction in SVCS due to benign causes. 38 Their study cohort of 70 patients included 31 patients with mediastinal fibrosis. The endovascular repair included PTA, thrombolysis with PTA, and stenting. They had success in 88% (28/32) of patients requiring endovascular repair, and four patients with failed treatment underwent open surgical intervention. The morbidity in the open surgical group was 19% and was more severe. It included mediastinal hematoma, veno-thromboembolism, symptomatic pericardial effusion, prolonged mechanical ventilation, and subsequent tracheostomy. In contrast, only one patient (4%) in the endovascular repair group developed a minor complication of an arm hematoma at the access site. However, two patients were reported to have cardiac tamponade, but no deaths occurred in patients who received subsequent secondary intervention. 38 Both groups had 93% of patients with symptomatic relief (39/42 in the open surgical group and 26/28 in the endovascular repair group). 38 A systematic review of 13 studies found endovascular intervention provided more symptomatic relief (97.3%) compared to open surgical repair (93.5%), with no 30-day mortality in either group. 39 However, the same review found that patients with mediastinal fibrosis required open surgical intervention more often as they presented with a severe type of SVCS. 39 A multidisciplinary approach has been suggested for management of FM. 21 Patients should be given a trial of rituximab and steroids on presentation. If symptoms do not improve, endovascular stenting can be considered first-line therapy to achieve revascularization, especially in high-risk patients who have evidence of pulmonary hypertension and intricate anatomy. Surgery should be reserved for patients for whom both medical and endovascular treatments have failed. 21
Limitations of this review
The purpose of the article was to help guide strategies and decision making for SVCS in patients with FM. This review has several limitations. It is a focused, nonsystematic literature review, as compared to a systematic review and, as a result, does not encompass all the literature pertinent to this topic. We chose to do a focused review as we intended to cover several aspects of endovascular stenting in SVCS resulting from FM. Another reason for utilizing a nonsystematic approach, rather than a meta-analysis or systematic review, is that FM is a rare disease and data are derived mainly from case series and retrospective studies. An additional limitation to the review is the heterogeneity in the data. Most studies encompassed patients with SVCS from benign causes that included disease entities other than FM and included very few patients with FM. We presented two cases of SVCS from FM treated with endovascular stenting at our institution, in accordance with the reviewed literature and recommendations from clinical experts in the field. The goal was to highlight current practices, review different types of stents being used (covered versus uncovered), discuss the role of anticoagulation in venous stenting, and provide a brief overview of surgical treatment options. Only English language articles were included in the review, which may have resulted in the exclusion of potential studies reported in other languages.
Summary
Although most data come from retrospective studies, the current trend is towards using endovascular repair as first-line therapy and reserving open surgical repair for patients who fail endovascular repair or have more severe disease (Type III or Type IV SVCS). Endovascular repair offers acceptable short-term benefit with symptom relief and minimal complications. Open surgical repair offers long-term patency and symptom relief, but with the cost of an increased rate and severity of complications. Both interventions require subsequent secondary repair, due to either stent or graft thrombosis. Local experience and expertise must inform the selection of the percutaneous versus the surgical approach. The role of anticoagulation therapy following endovascular stenting or surgical repair of nonthrombotic SVC occlusion remains unclear but can be considered if additional indication for anticoagulation exists.
Footnotes
Acknowledgements
The authors thank Linda Conry, MA, for editorial assistance in the preparation of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
