Abstract
Vascular surgery is the largest (non-cardiac) user of blood transfusion which is associated with increased risk to patients. Patient Blood Management (PBM) is a quality improvement programme in transfusion medicine involving educational change and recommendations through preoperative, operative, and postoperative surgery. We wished to assess the feasibility to implement a PBM programme in vascular surgery. A multidisciplinary programme was developed at a vascular unit by PBM experts. The PBM programme involved a series of educational lectures, consultations, and discussions with doctors, nurses, and theatre staff. A one-page PBM checklist of recommendations was developed for all patients undergoing vascular surgery. Prospective audits were conducted before (October 2014 to March 2015) and after (November 2015 to February 2016) PBM implementation. Outcomes were blood transfusion and haemoglobin concentration (Hb) trigger threshold. A total of 211 patients were admitted under vascular surgery: 127 for Audit 1 and 84 for Audit 2. Overall, 30% of patients were transfused, with 193 units transfused in Audit 1 and 85 in Audit 2. PBM implementation was associated with a reduction in patients receiving a blood transfusion (37% to 20%; p = 0.01). However, there was no difference in Hb trigger threshold (76 g/L vs 72 g/L, p = 0.051). A PBM programme is feasible and can be implemented in vascular surgery. PBM was associated with an improvement in transfusion use and length of patient stay that merits further investigation.
Introduction
Blood transfusion is a common and accepted method to treat anaemia, particularly in surgical patients. However, current practices may reflect an over-reliance on the use of red blood cell (RBC) transfusions.1–3 Blood transfusion, as with any intervention, exposes patients to risks – specifically, alloimmunisation and increased infection, but, more commonly, pulmonary injury or transfusion-associated circulatory overload (TACO).4,5 In the setting of surgery, blood transfusion is associated with an increase in morbidity and mortality.6–8
Vascular patients often have multiple comorbidities that increase the risk of anaemia; are on anticoagulant or antiplatelet therapies that increase the risk of bleeding; and undergo interventions that often involve blood loss. These factors may contribute to why vascular surgery is the second largest user of blood transfusion (per patient) after cardiac surgery, although, overall, due to the volume of patients, orthopaedic surgery uses the most blood in the operative period.3,9,10
Patient Blood Management (PBM) is a patient-centred, multidisciplinary, evidence-based approach that aims to optimise the use of blood products to ultimately enhance patient outcomes. PBM targets three areas: optimisation of RBC mass, minimisation of blood loss, and tolerance of anaemia. These translate well into surgical practice where the three independent risk factors that contribute to patient outcome in the surgical setting are anaemia, blood loss, and transfusion. 11
PBM has been endorsed by National Health Service (NHS) Blood and Transplant guidelines, National Institute for Health and Care Excellence (NICE) quality standards, the British Committee for Standards in Haematology, as well as a variety of international consensus statements.12–17 However, implementing guidelines can be a challenging task – often due to poor adherence, resistance from healthcare professionals, and existing culture. 18 Local and national audits comparing local practice against recommended standards continue to show inappropriate use of blood outside of the recommended guidelines.1,2 Consequently, there is a need to address the current practice of PBM in vascular surgery, focusing on anaemia management, reduction of bleeding, and appropriate transfusion practice.
The aim of this study was to assess the feasibility to implement a PBM programme in vascular surgery.
Methods
Audit
A prospective audit was undertaken at a regional vascular unit before and after a PBM intervention with approval from the hospital transfusion committee and audit committee. The first audit (Audit 1) was conducted between October 2014 and March 2015. Data were reviewed and an educational programme combined with a one-page PBM checklist for the preoperative, intraoperative, and postoperative management of patients undergoing vascular intervention was implemented. Following this, a second audit (Audit 2) was conducted between November 2015 and February 2016.
Both audits were observational, and data were collected in a prospective manner from daily ward rounds from sequential patients who underwent vascular surgery at one hospital. There was no input to the surgical management of vascular patients. Patient demographics, medication (i.e. antiplatelet or anticoagulant use), presenting complaint (i.e. acute or chronic limb ischaemia), operative procedure (i.e. bypass, endarterectomy, angioplasty or amputation), risk factors, and laboratory variables were collected. Anaemia was defined according to the World Health Organization (WHO) classification: men < 130 g/L and women < 120 g/L. A single-unit blood transfusion was documented as all, or part of, a single unit transfused within a 24-hour period. As some surgeries do require large transfusions (we defined it as 4 units or more), we looked at whether excluding this group would make a difference in units used after the programme.
The primary outcome was feasibility of the PBM as an intervention in vascular surgery. To assess for feasibility, we looked at whether the intervention reduced transfusion usage, lowered haemoglobin concentration (Hb) trigger threshold, and reduced hospital LOS. Secondary outcomes for comparison between the two audits were: the proportion of patients receiving a RBC transfusion, the Hb before blood transfusion (trigger Hb) and Hb on discharge from hospital, and hospital LOS. The direct cost of blood transfusion was estimated where each unit of RBC cost £121.85 (US$152). 19
Patient Blood Management
PBM is a multimodal, multidisciplinary approach to patient care. The process of PBM intervention was based on the Western Australia PBM project. 9 In brief, this involved benchmarking of existing practice, consultation with all staff to assess current practices, identification of a programme ‘champion’, a programme of education and change management, and implementation of PBM guidelines followed by a repeat audit of practice.
Following an initial benchmarking audit (Audit 1), results were reviewed at the multidisciplinary meeting with engagement of all staff involved in vascular patient care. The programme was led by a consultant surgeon (TR) and a consultant anaesthetist (NS), both familiar with PBM practice and guidelines. Engagement with other departments/staff included: pharmacy to discuss patient medications and where appropriate iron therapy; nursing staff for transfusion practice, medication administration, and patient care; and ward doctors, surgeons, and anaesthetists to review established practices.
A simplified PBM checklist (online Supplementary File 1), condensed to a single page for ease of use, was developed from published guidelines, recommendations, and directives. These included a review of anaemia management and investigations, alternatives to transfusion, restrictive transfusion strategy, and a change of policy to single-unit transfusions.17,20,21 This PBM checklist was reviewed and approved by the hospital transfusion committee.
A biomedical research student was identified and trained to ‘champion’ the PBM programme. The student was supported by the lead consultants, and a range of strategies was used to educate staff at the hospital. These strategies included presentations to foundation doctors, nurses, and pharmacists to raise awareness of PBM and one-on-one meetings with house officers and ward staff to introduce the PBM checklist. Handouts and checklists were distributed following the presentations as supporting materials.
Patient Blood Management checklist
A checklist was developed based on best practice in PBM in agreement with all staff following consultation. For instance, CosmoFer® (Pharmacosmos A/S, Denmark) was identified as the preferred intravenous iron of choice by pharmacy (cost) and nursing staff (one infusion). All surgeons agreed to use cell salvage for open abdominal aortic aneurysm and lower limb bypass (variable). Tranexamic acid was introduced and promoted at operation following education and reassurances that it did not cause clots or deep vein thrombosis. Heparin usage was encouraged, with point of care testing during vascular procedures, and all staff were supported to question the need for more than one unit of blood to be transfused at a time. The checklist was reviewed and approved by the local hospital transfusion committee and by the vascular and anaesthetic governance (see online Supplementary File 1).
Data analysis
Statistical analysis was performed using GraphPad InStat 3.0 (GraphPad Software, Inc., San Diego, CA, USA) and SPSS 25.0 (IBM Corp., Armonk, NY, USA). Continuous variables are expressed as means with SD, or medians with IQR. Audit 1 was compared to Audit 2 using the independent samples t-test for parametric data and Mann–Whitney U-test for nonparametric data. Categorical variables are expressed as counts with corresponding percentages. Similarly, when comparing Audit 1 to Audit 2, the z-test score and chi-squared test were used for parametric and nonparametric data, respectively. Where expected numbers were small, Fisher’s exact test was used instead of the chi-squared test. As PBM is a multifaceted intervention with multiple aspects from educational support, engagement, and recommendations, individual components or ‘tick boxes’ were not compared; rather, the programme as a whole was regarded as ‘the intervention’.
Results
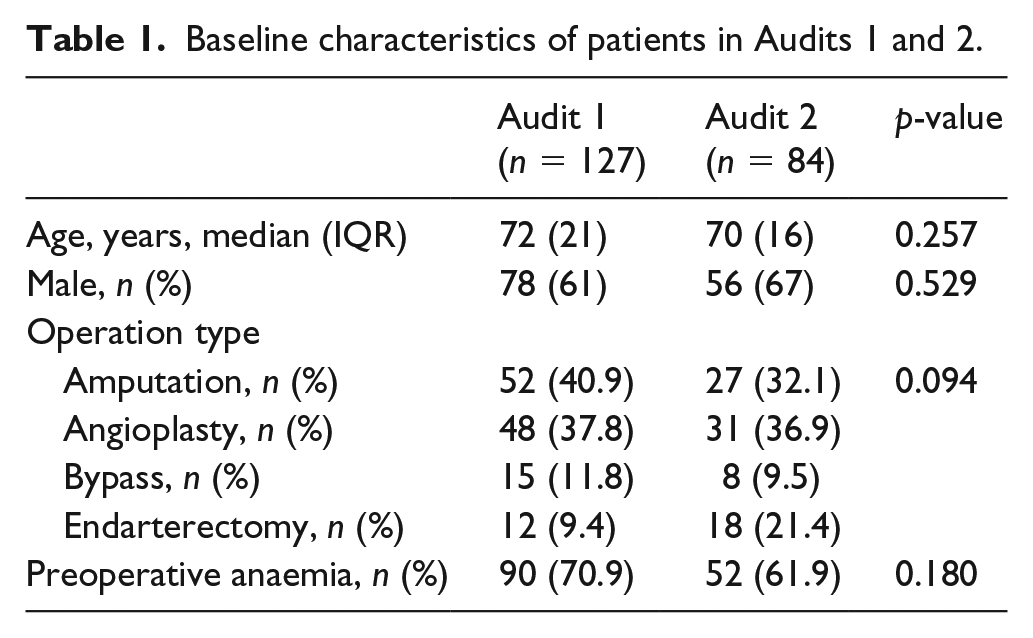
Overall, a total of 211 patients were admitted under vascular surgery and underwent an operation: 127 for Audit 1 and 84 for Audit 2. The average age was 70 years (SD 12) and two-thirds of patients were male. Anaemia at admission was common (average Hb was 116 g/L (SD 23)). Most patients were on antiplatelet therapy (58%), dual antiplatelets (18%) or anticoagulants (16%). A total of 36% of patients were elective surgery, and 25% underwent more than one procedure. There was no significant difference between both audits for age, sex or operation type (Table 1).
Baseline characteristics of patients in Audits 1 and 2.
RBC transfusion
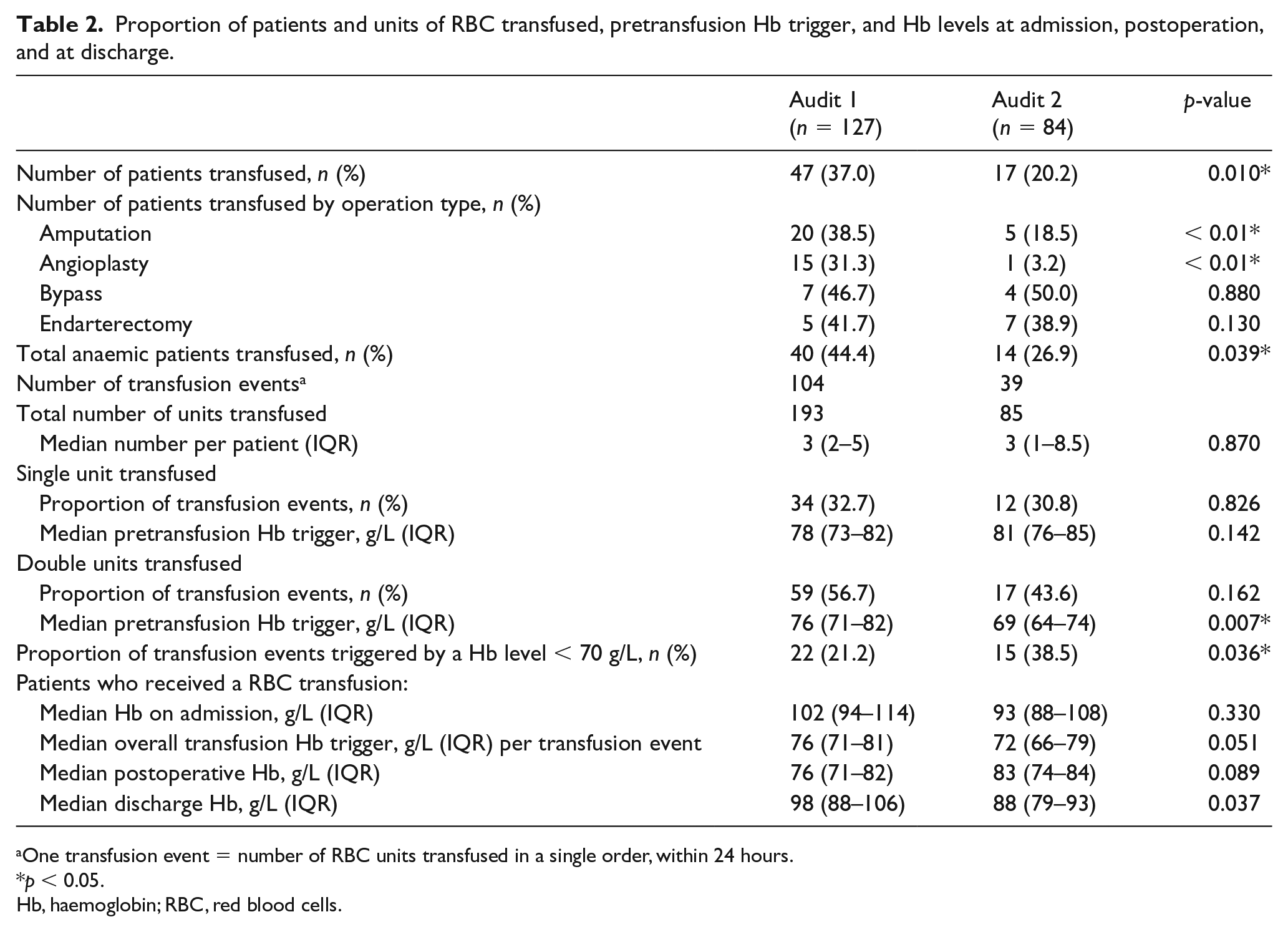
Overall, 64 (30%) patients were transfused in the study (Table 2). Audit 1 revealed a high number (n = 47, 37%) received a blood transfusion; following the PBM intervention, the proportion used was reduced (n = 17, 20.2%; p = 0.010). In terms of volume of blood received, 193 units of RBC were transfused in Audit 1 compared to 85 in Audit 2. There was no difference in the median number of RBC units transfused per patient. A large blood transfusion (4 or more units) occurred in 23 patients: 16 patients in Audit 1 and seven patients in Audit 2. If we exclude these patients, the median number of RBC units transfused was 2 (IQR 2–3) in Audit 1; following the PBM implementation intervention, this was reduced to 1.5 RBC units (IQR 1–2.25) in Audit 2 (p = 0.78).
Proportion of patients and units of RBC transfused, pretransfusion Hb trigger, and Hb levels at admission, postoperation, and at discharge.
One transfusion event = number of RBC units transfused in a single order, within 24 hours.
p < 0.05.
Hb, haemoglobin; RBC, red blood cells.
Transfusion trigger
Overall, the Hb at which transfusion was initiated was 75 g/L (SD 10). There was no difference following PBM intervention (Table 2).
The proportion of transfusions triggered by a Hb level above 70 g/L reduced from 79% in Audit 1 to 61% in Audit 2 (p = 0.036); for a Hb level above 80 g/L, this also reduced from 31% in Audit 1 to 23% in Audit 2 (p = 0.240).
In Audit 1, a third (33%) of transfusions were administered as a single unit, 57% as double, and the remaining transfusions (11%) were 3 or more units of RBC. In Audit 2, 31% of all transfusion events were administered as a single unit, 44% were double, and the remaining transfusions were 3 or more units of RBC (26%).
Hospital LOS in different operation types and with or without anaemia
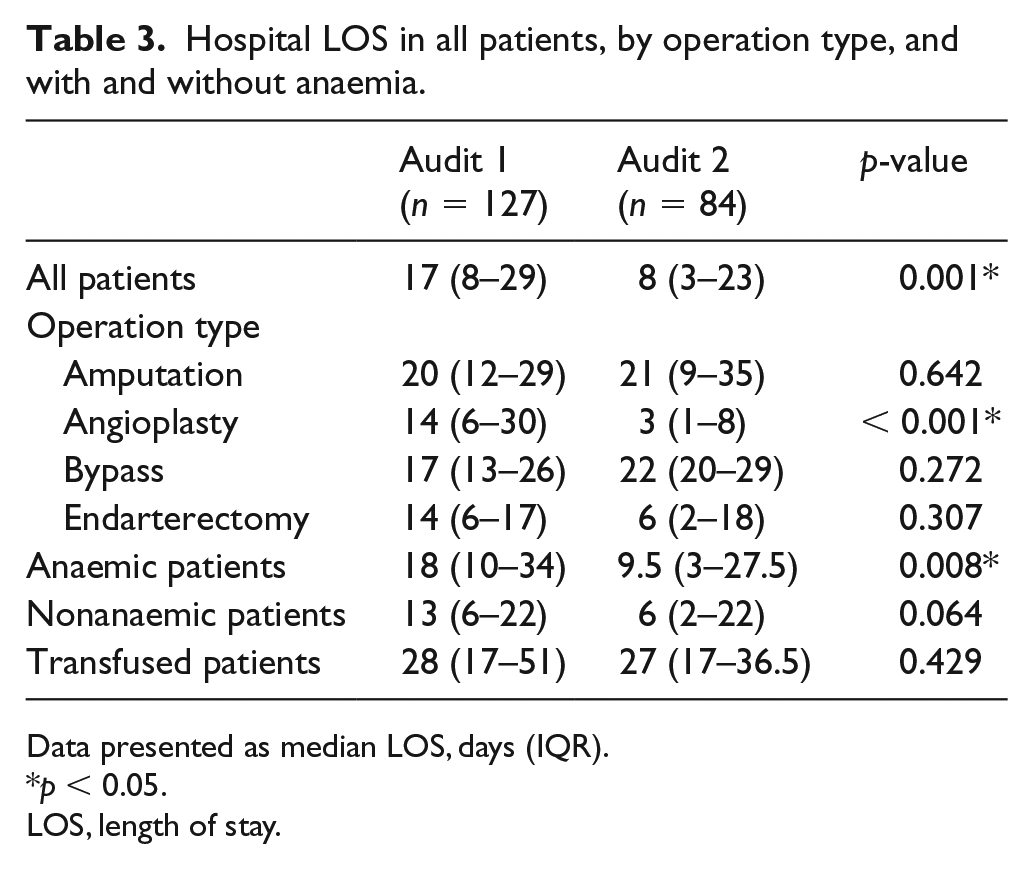
The hospital LOS was examined by operation types, anaemia, and if the patient was transfused (Table 3). The overall median LOS reduced from 17 days to 8 days (p = 0.001) following the introduction of the PBM programme. This was predominantly explained by those undergoing angioplasty (14 days vs 3 days, p < 0.001), which was also associated with a significant reduction in transfusion use (31% vs 3%, p < 0.01).
Hospital LOS in all patients, by operation type, and with and without anaemia.
Data presented as median LOS, days (IQR).
p < 0.05.
LOS, length of stay.
Patients presenting with anaemia had a significant reduction in median LOS following the PBM intervention (18 days vs 9.5 days, p = 0.008).
Transfusion cost
A rough estimate of the cost of transfusion was calculated for this study. In Audit 1, 47 of the 127 patients in the study were transfused and a total of 193 units of RBC were used. This attributes to a direct cost of £23,517.05 (US$29,517). In comparison, 17 out of 84 patients were transfused in Audit 2 and a total of 85 units of RBC were used. This results in a direct cost of £10,357.25 (US$13,000). On a per month basis, 32 RBC units were used on average in Audit 1, whereas 22 RBC units were used in Audit 2. Cost per patient averages to approximately £523.36 (US$656).
Discussion
A programme of PBM can be successfully implemented in a regional vascular surgery unit with associated improvement in blood transfusion use. The intervention of PBM was associated with a reduction of blood transfusions (37% to 20%). There was an associated reduction in hospital LOS, particularly for those patients with preoperative anaemia (18 vs 10 days).
In the UK, PBM is currently under-represented and a change in paradigm from conventional transfusion practice to PBM is necessary. Our study examines the influence of a small-scale educational programme (pilot) on transfusion usage, the Hb trigger threshold for transfusion, and LOS. This PBM implementation intervention has led to an overall reduction in transfusion rates, but no significant change to the proportion of patients who received single and double-unit transfusions was observed. This suggests an opposition to the recommended use of a single-unit policy, an issue which has historically been surrounded by controversy and has faced resistance due to the traditional practice of administering 2 units. 22
Patients with a history of cardiac disease generally have a trigger threshold cut-off of 80 g/L, which is slightly higher than the NICE 17 recommended threshold. The results for this study suggest a shift in transfusion practice towards using restrictive transfusion strategies; notably, the proportion of patients transfused below 70 g/L in line with NICE guidelines for patients without a history of cardiac disease. However, the proportion of anaemic patients receiving a RBC transfusion markedly decreased from 44% to 27% (p = 0.039). This may be due to practice patterns shifting towards the use of alternatives to transfusions.
We observed no significant differences in the pretransfusion triggers for single-unit transfusion, but a significant decrease was observed for double-unit transfusions from 76 g/L to 69 g/L between the two audits (p = 0.007). Interestingly, the median discharge Hb concentrations exceeded the recommended targets of between 70 g/L and 90 g/L among patients transfused in Audit 1, but this decreased to 88 g/L in Audit 2. This encouraging result suggests that clinicians are evaluating the effect of each transfused unit to avoid going beyond the recommended targets.
In terms of cost, 32 RBC units were used on average per month in Audit 1, costing £3899.20 (US$4893), compared to Audit 2, where 22 RBC units were used on average per month costing £2680.70 (US$3363). This represents a considerable direct saving of £1218.50 (US$1528) per month. In addition to this, there would also be indirect savings (e.g. processing, labour, disposables, staff hours) incurred as a result of reduced RBC usage, although we have not quantified the cost for PBM implementation.
Although this study has shown a trend in reducing inappropriate blood transfusion, this change needs to be sustained. From our experience, previous quality improvement projects at the Royal Free Hospital, London, UK have shown effective results post-intervention; however, in the long term, these results are not sustained. Key factors in sustaining change involve support from leaders, access to facilities and human resources, and having the right staff with the right knowledge and skills. 23 When implementing any change in any clinical setting, plans need to be long term, strategic, and practical to ensure success and sustainability.
Limitations
There are several limitations to this study. First, we were unable to evaluate the efficacy for each of the individual recommendations outlined in the PBM checklist, as the numbers involved would not support subgroup analysis and consequently certain data (e.g. tranexamic acid and cell salvage) were not collected. As a result, we were unable to determine the level of adherence to the PBM checklist by clinicians at the hospital. Second, intravenous iron was only used in seven patients in Audit 2. We suspect this may be due to the lack of awareness and knowledge of the protocol as doctors were rotating each week; however, due to the lack of resources, we were unable to confirm this. Third, results may be subject to the Hawthorne effect, where clinical staff behaviour may be altered due to awareness of being observed by researchers. Last, the sample size and audit duration were different in Audit 1 compared to Audit 2, and so results needs to be interpreted with caution. Specifically, there was an apparent difference in the numbers of patients undergoing angioplasty and it is logical that minimally invasive revascularisation had a lower risk of blood transfusion compared to open surgery.
Data were collected prospectively; there is no specific reason why this occurred and most likely it is just a reflection in the variability of workload in vascular surgery. Nevertheless, results of this before and after audit must be interpreted with caution with regards to secondary outcomes such as LOS. These differences in interventions may confound the secondary outcomes, which should be regarded as associative and hypothesis-generating for future larger-scale implementation strategies or clinical trials.
Conclusion
PBM can be implemented in vascular surgery through a programme of education and audit. Despite being a single-centre study, these results could be generalised to other hospitals looking to use a similar approach to introduce PBM algorithms to address blood transfusion practice and patient outcomes.
Supplemental Material
10.1177_1358863X19883940_Supplementary_file – Supplemental material for Development and feasibility of a Patient Blood Management implementation programme in vascular surgery
Supplemental material, 10.1177_1358863X19883940_Supplementary_file for Development and feasibility of a Patient Blood Management implementation programme in vascular surgery by Marisa Chau, Divisha Gohil, Nick Schofield, Sandaruwani Abeysiri, Andrew A Klein and Toby Richards in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: there is no external or industry involvement in this project. MC: salary – supported by Pharmacosmos A/S. DG: no conflict of interests declared. NS: no conflict of interests declared. SA: no conflict of interests declared. AK: AK or his institution has received educational grant funding, honoraria or travel expenses from Fisher and Paykel, Pharmacosmos, Vifor Pharma, Masimo, Hemonetics, and Hemosonics. TR: grants – NIHR HTA (UK), NHMRC (Australia), NIAA / BJA / ACTA / VASGBI (UK), Mason Medical Research Foundation, UCH League of Friends; grants, personal fees, and nonfinancial support – Pharmacosmos, Vifor Pharma, Acelity, Stroke Association; grants and nonfinancial support – Libresse / Bodyform; all outside the submitted work. TR is a regular speaker at national and international conferences on anaemia, blood transfusion, wound healing, and vascular diseases, for which he has received expenses for travel, accommodation, and sundries. TR is the Director of The Iron Clinic Ltd, Director of Veincare London Ltd, and is also the vascular lead for 18-week wait Ltd.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
