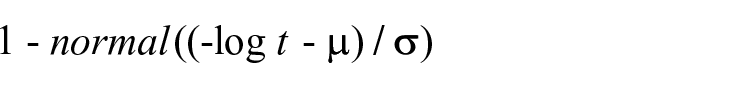Abstract
Following an anticoagulation-associated intracerebral hemorrhage (ICH), whether and when to resume anticoagulation is controversial. Patient-level recurrence risk is difficult to predict with accuracy, but time-based recurrence risk may be more predictable. To better inform clinical decisions, we set out to estimate the net clinical benefit of anticoagulation over time among patients with atrial fibrillation. Using a large administrative dataset with 5339 index ICH hospitalizations and 132 readmissions for ICH, we created a two-stage prediction model, first predicting patient-level risk of recurrence and then predicting timing, conditional on recurrence. A log-normal survival function best explained the declining risk of recurrent ICH over time. We then compared risk of recurrent ICH over time against ischemic stroke risk, weighting the two outcomes to compute the net clinical benefit on each day following an index discharge. Using a bootstrapping approach, we identified the first day following discharge on which anticoagulation would lead to net benefit rather than net harm. Anticoagulation remains harmful for at least 11 days following index discharge and, depending on desired confidence level and assumptions, may remain harmful for as long as 62 days after discharge. Results were sensitive to the overall ICH recurrence risk. Although patient-level risk of recurrent ICH is difficult to predict accurately, recurrence risk declines rapidly over time. The survival function presented herein can inform decision-analytic models regarding when patients should resume anticoagulation following ICH.
Introduction
Following an anticoagulation-associated intracerebral hemorrhage (ICH), whether and when to resume anticoagulation is unclear, as the harms of recurrent hemorrhage relative to the harms of subsequent ischemic stroke are difficult to quantify.
However, the balance of benefits and harms likely changes over time. Theory and data suggest that the risk of recurrent ICH decreases over time following an initial event. 1 Meanwhile, among patients with atrial fibrillation, the risk of ischemic stroke remains roughly constant. 1 At some point in time following an initial ICH, the expected benefits of anticoagulation may exceed the expected harms. Estimating that time point first requires estimating the rate of recurrent ICH over time; however, experimental data on this question are lacking.
A decision-analytic model incorporating change over time would require estimates of both overall recurrence rate in the cohort and time period under consideration (e.g. 1-year recurrence among patients with atrial fibrillation) and an estimate of how risk evolves over that period. Overall recurrence rates can come from cohort studies, but because recurrent ICH is rare, the latter parameter requires very large numbers of patients. Using a large administrative dataset, we set out to estimate the earliest time at which anticoagulation can be safely resumed following an initial ICH.
Methods
Risk of recurrent ICH over time
The Nationwide Readmissions Database (NRD) is a large database designed to support analyses of hospital readmissions in the United States, including uncommon conditions. 2 Using years 2011–2014 of the NRD, we first limited our analysis to admissions with all of the following criteria:
A diagnosis of atrial fibrillation or flutter (International Classification of Diseases, Ninth Revision (ICD-9) codes 427.31 or 427.32)
Use of long-term anticoagulation (ICD-9 code V58.61)
A diagnosis of ICH (ICD-9 code 431)
A diagnosis-related group (DRG) of 64, 65, or 66 (‘Intracranial hemorrhage or cerebral infarction’)
Patient survival to discharge.
For patients with multiple admissions meeting these inclusion criteria, we included only the first admission. Among this cohort, we identified as our outcome of interest the next admission that met the following criteria:
A diagnosis of ICH (ICD-9 code 431)
A DRG of 64, 65, or 66.
We created a two-stage prediction model, with the first stage predicting our outcome of interest (hereafter, ‘readmission for ICH’) and the second stage predicting timing of readmission, conditional on readmission for ICH. A patient selection diagram is shown in the online Supplemental Figure.
We used logistic regression to predict risk of readmission for ICH as a complete-case analysis. To avoid overfitting, we tested no more than one predictor per 10 events. We selected predictor variables that have been previously associated with risk of ICH (primary or recurrent) and which were available in this dataset: age, history of hypertension, tobacco use, renal insufficiency, and craniotomy during the index admission.3–5 (Many predictors, such as anticoagulation intensity, deep vs lobar location of ICH, and presence of microbleeds, are not captured in administrative data.) We added index length of stay and number of chronic conditions, which are predictors of overall readmission risk, as possible predictors of readmission for ICH. 6 We hypothesized that age and length of stay could have nonlinear relationships with risk of readmission for ICH.
For the second stage of our model, we limited our dataset to patients readmitted for ICH. We compared standard survival functions (exponential, Gompertz, Weibull, gamma, log-normal, and log-logistic), selecting for model fit according to the lowest Akaike’s and Bayesian information criteria (AIC and BIC). After selecting the model that best described the data overall, we tested for interactions using the same predictor variables described above. We also tested whether resumption of anticoagulation (determined by ICD-9 code during the second hospitalization) modified the survival function.
Risk of ischemic stroke over time
Among the same cohort described above, we identified as our outcome of interest the next admission that met the following criteria:
A diagnosis of ischemic stroke (ICD-9 code 433.01, 433.11, 433.21, 433.31, 433.81, 433.91, 434.00 434.01, 434.10, 434.11, 434.90, 434.91, or 436)
A DRG of 61, 62, 63, 64, 65, 66, 67, or 68 (which includes the ‘Acute ischemic stroke’, ‘Intracranial hemorrhage or cerebral infarction’, and ‘Nonspecific stroke and precerebral occlusion’ DRGs).
We used logistic regression to predict risk of readmission for ischemic stroke as a complete case analysis, testing the same patient-level variables described above. We also tested CHA2DS2-VASc score, a composite score predicting risk of ischemic stroke, as computed from the index admission. However, because the first-stage model had poor discrimination and calibration, the second-stage model showed no clear temporal relationship of risk of ischemic stroke over time, and well-validated alternate risk-prediction schemes exist for ischemic stroke risk, we abandoned this prediction model. Instead, we calculated the weighted average 1-year risk of ischemic stroke, as per the patient-specific CHA2DS2-VASc score, according to data published from four large cohorts.7–11
Net clinical benefit over time
The outcomes of intracranial hemorrhage while receiving warfarin are generally worse than the outcomes of thromboembolic stroke while not receiving warfarin.12,13 To account for the different consequences of the two clinical conditions, many authors report ‘net clinical benefit’ of anticoagulation, a weighted composite of ICH and stroke. We computed the net clinical benefit as follows:
where the weighting ratio accounts for the generally worse outcomes of ICH compared with ischemic stroke.12,14 We used a previously published method to compute the weighting ratio for each patient, assuming a discount rate of 3%. 15 Because our cohort likely underestimates the overall proportion of patients with recurrence of ICH, we performed sensitivity analyses at recurrence rates from other ICH cohorts. 16 We also analyzed by CHA2DS2-VASc score to test for sensitivity to underlying risk of ischemic stroke.
We used previously published data to estimate the treatment effect of warfarin for ischemic stroke and recurrent ICH. Specifically, we assumed that warfarin reduced the risk of ischemic stroke by a rate ratio centered at 0.27 (95% CI 0.20–0.36) and increased the risk of ICH by a rate ratio centered at 5.44 (95% CI 1.88–11.65). 17 We did not consider time lag in anticoagulation treatment effects, such as the time to therapeutic range or time to a supratherapeutic range.
After estimating the net clinical benefit of anticoagulation for each patient on each day following index discharge, we bootstrapped the mean net clinical benefit and 95% CIs for anticoagulation on each day. Our primary endpoint was the first day on which the net clinical benefit of anticoagulation was greater than zero (the earliest day on which anticoagulation would be expected to offer some benefit).
All analyses were performed in Stata statistical software, release 14 (StataCorp LP, College Station, TX, USA). The analysis code is available from the corresponding author on request. Data used are available as part of the Agency for Healthcare Research and Quality (AHRQ)’s Healthcare Cost and Utilization Project (HCUP).
Results
We identified 5339 index hospitalizations, of whom 1631 were readmitted (30.5%) by the end of the calendar year. The overall 30-day readmission rate was 14.9%. Patients included were 52% male, with a mean age of 78.0 years (SD 9.4). The median CHA2DS2-VASc score was 4 (IQR 3–5; range 0–9). We identified 132 ICH readmissions and 198 stroke readmissions for analysis.
Risk of recurrent ICH over time
In our first-stage model, tobacco use and index length of stay were statistically significant predictors of ICH readmission risk. Too few craniotomies were identified to test, owing to collinearity. No other tested predictors were statistically significant. Length of stay demonstrated a nonlinear relationship with predicted probability of readmission. This model displayed modest discrimination (c-statistic: 0.61) and adequate calibration across nine groups (a roughly fivefold gradient of risk, from 1.3% to 6.3%) based on Hosmer–Lemeshow goodness-of-fit testing. Although this model had reasonable discrimination and calibration, it explained little patient-level variance (pseudo R2 ≅ 0.02). The mean predicted risk of ICH recurrence within 1 year was 2.46%. The receiver-operator curve for this model is shown in Figure 1.
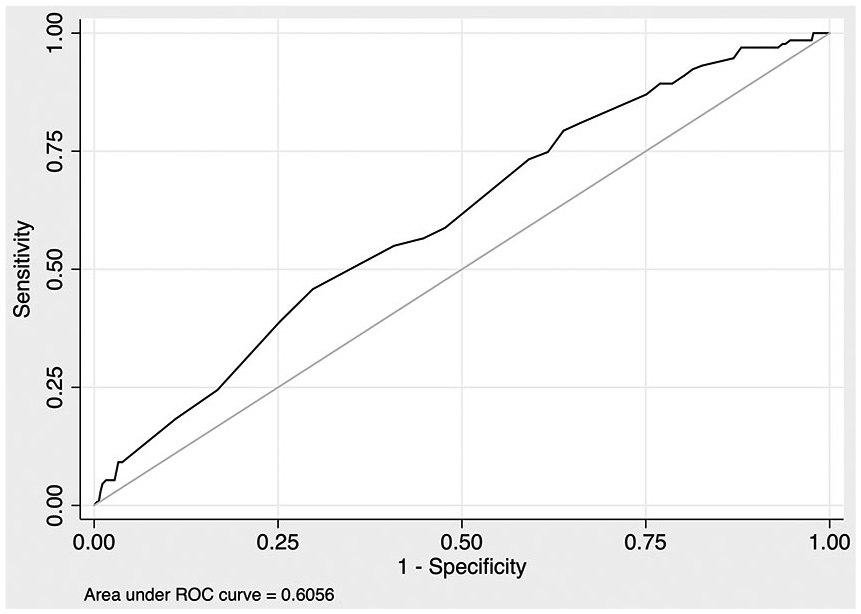
Receiver-operator curve for first-stage prediction model.
In our second-stage model, a generalized gamma model best fit the risk of ICH readmission over time. Of parameterized models, a log-normal model demonstrated the best fit. No tested predictor variables added explanatory power to a model based only on time. Our parameterized second-stage survival model can be expressed as:
where t = days after index discharge,
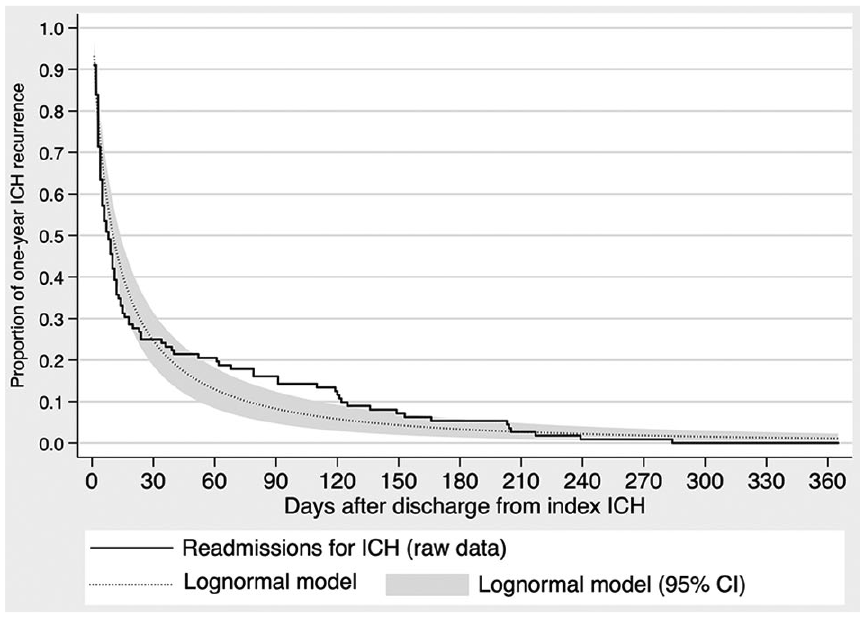
Risk of ICH recurrence over time.
Risk of ischemic stroke over time
In our first-stage model, only female sex was a statistically significant predictor of ischemic stroke following index ICH. This model displayed poor discrimination (c-statistic: 0.52) and explained little variance (pseudo R2 ≅ 0.02). Rather than use an inferior model to predict risk of ischemic stroke, we calculated the weighted average 1-year risk of ischemic stroke, according to patient-specific CHA2DS2-VASc score, using data published from four large cohorts (described above). We then computed daily risks from those annual risks, assuming a constant daily risk over the year.
Net clinical benefit over time
As risk of recurrent ICH decreases, net clinical benefit increases, asymptotically approaching the net clinical benefit that would be expected at a patient’s baseline risk of ICH. In our base-case analysis, the mean net clinical benefit exceeded zero on day 11 following the index discharge, while the 95% CI exceeded zero at 42 days after the index discharge. In our sensitivity analysis, where we assumed a higher risk of recurrent ICH (7.2%, from a previous cohort), the mean net clinical benefit exceeded zero on day 19 following the index discharge, and the lower bound of the 95% CI exceeded zero at 62 days after the index discharge. Net clinical benefit over time for our base-case and key sensitivity analysis is shown in Figure 3. At a 1% risk of ICH recurrence (a hypothetical low-risk sensitivity analysis), the mean net clinical benefit exceeded zero 7 days following index discharge, and the 95% CI was greater than zero 28 days after the index discharge.
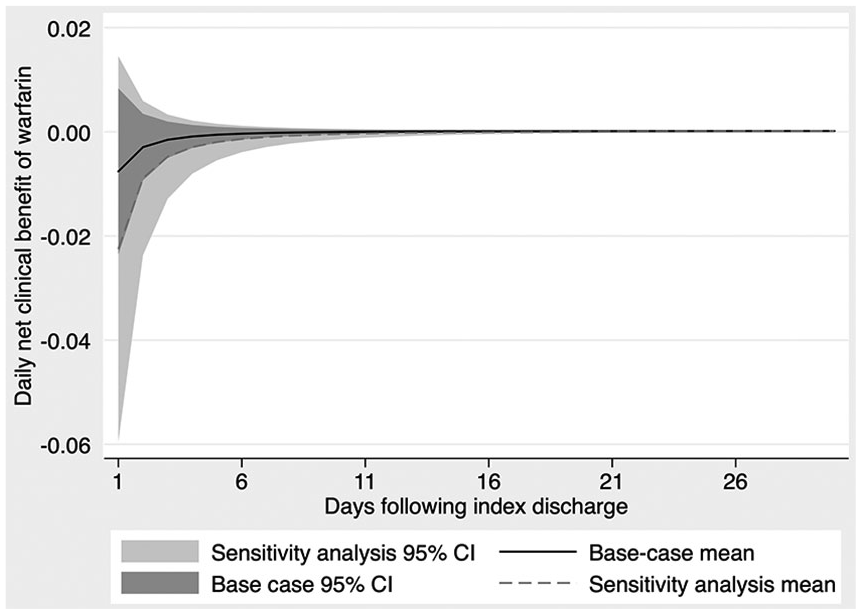
Net clinical benefit of resuming anticoagulation over time.
In other sensitivity analyses, stroke risk (estimated by CHA2DS2-VASc score) changed decision-making to a small extent: mean net clinical benefit exceeded zero on day 9 for patients whose CHA2DS2-VASc score is 6, compared to day 16 for patients whose CHA2DS2-VASc score is 3.
Discussion
Using a large, nationally representative administrative sample of patient readmission, we have estimated the risk of ICH recurrence and net clinical benefit of anticoagulation, across patients and over time. Previous cohort studies have estimated 1-year ICH recurrence between 1.8% and 7.2%. 16 Our predicted rate of ICH recurrence by 1 year (2.5%) is at the low end of this range. Some of this difference is attributable to the time periods captured: because the NRD is censored at the end of each year and not linked between years, patients whose index event is late in the year have short periods of observation. Also, some fraction of patients with recurrent ICH are not admitted at all, either because the recurrence was mild or because the recurrence led to death without readmission. In any case, it is likely that our estimated overall recurrence rate is lower than what would be expected in clinical practice.
Our study, like many before it, has shown the difficulty of predicting patient-level risk of ICH. 9 Although our first-stage model predicting patient-level risk of recurrent ICH demonstrated reasonable calibration and discrimination, it explained little variance in patient-level risk. Further, with an overall recurrence rate of 2.5%, a null model predicting that no patients would sustain recurrent ICH would be accurate 97.5% of the time. Simply put, rare events are difficult to predict.
Time-based recurrence risk is more predictable. The greatest strength of this study lies in the second-stage model, which suggests how rapidly recurrence risk decreases over time. While this finding is difficult to use in isolation, it can inform clinical practice and decision analyses. For example, of patients who sustain recurrent ICH, around 50% will have done so by day 38 after discharge, 80% will have done so by day 124 after discharge, and 95% will have done so by day 263 after discharge.
Using that decay function, estimates of ischemic stroke risk, and multiple estimates of ICH recurrence risk (both our first-stage prediction and higher rates from previous cohorts), we have estimated the net clinical benefit of anticoagulation over time. In our base-case analysis, anticoagulation is no longer harmful between 11 and 42 days following discharge (depending on the confidence desired) for patients who survive an index ICH. In our sensitivity analysis with a high ICH recurrence risk (7.2% in the first year), anticoagulation remains harmful for between 19 and 62 days, again depending on the confidence desired. More precise estimates of recurrence risk could serve to further personalize resumption decisions, and more precise estimates of anticoagulation’s treatment effects could narrow CIs.
Other information would be helpful in personalizing anticoagulation resumption. We have estimated the day on which net clinical benefit exceeds zero, but that is the absolute earliest that anticoagulation should be restarted: clinicians should expect meaningful and cost-effective benefit, not simply the absence of harm. Net clinical benefit does not account for the disutilities of anticoagulation (e.g. cost, inconvenience) or other harms that can result (e.g. extracranial hemorrhage, hospitalization for the same). Considering that information would lead clinicians to delay anticoagulation beyond the times presented here. Other studies of anticoagulation resumption have suggested that the optimal time to resume anticoagulation is between 7 and 30 weeks; the time difference may be at least partly due to factors not captured in a measure of net clinical benefit.18,19 Still, this analysis offers the earliest time at which anticoagulation may be considered: it would be difficult to justify anticoagulation earlier than 11 days after discharge, or 19 days in a high-risk cohort. If clinicians expect a high degree of confidence that anticoagulation is not actively harmful, these data suggest that anticoagulation should be delayed for 6 weeks (our base-case) or longer (nearly 9 weeks in our high-risk sensitivity analysis).
Limitations
The limitations of this analysis warrant discussion. First, some fraction of readmissions (particularly shortly after the index admission) are likely related to the initial ICH, not to a recurrent hemorrhage. Such readmissions may actually capture an inability to function at home or near-term sequela of the initial ICH. Counting these as recurrent events may inflate our recurrence estimates shortly after index discharge. Similarly, anticoagulation did not add explanatory power to a log-normal model, but clinicians’ decision to resume anticoagulation is likely based in part on time. That is, we cannot be certain that resumption of anticoagulation is not endogenous to our estimate of risk over time. Second, as highlighted above, our first-stage model had only modest discrimination, perhaps due to clinical features unavailable in this dataset (lobar location, number of microbleeds, etc.). Overall recurrence rates and patient-specific recurrence risk would likely be better estimated using well-designed cohort studies.4,20 Future applications may therefore be better-served by replacing the first stage of our model with an appropriate estimate for the cohort or patient under consideration, while using our second-stage model to estimate the change in risk over time. The key assumption underlying our second-stage model is that timing of recurrent ICH is not systematically different between patients hospitalized for recurrent ICH and patients who suffer recurrence but are not readmitted. If recurrent ICH events that do not result in admission (either due to death without admission or because the bleed was very minor) occur at different times, that would represent a threat to the validity of this model.
Otherwise, the results we present herein should be valid and useful for clinical practice and decision-analytic models, when used in the right context. First, these estimates should be used among patients who have survived an index ICH to discharge. Second, they should be used in conjunction with patient-specific estimates of the overall 1-year recurrence rate, as the 1-year recurrence risk captured in our dataset may be an underestimate. Third, they should be used with recognition that we have estimated the earliest safe time to resume anticoagulation, not the optimal time to resume. Clinicians can and should consider other harms (e.g. extracranial hemorrhage or financial toxicity) that would delay resumption of anticoagulation beyond the times presented here.
Conclusions
Following an initial ICH, the risk of recurrence decreases rapidly over time, and can be represented by a log-normal survival function. Future studies may be able to use our estimates of recurrence risk over time to arrive at optimal times to resume anticoagulation. Anticoagulation is likely harmful for at least 2 weeks following discharge from an initial ICH and may remain harmful for a number of weeks thereafter.
Supplemental Material
10.1177_1358863X19883027_Supplementary_figure – Supplemental material for Net clinical benefit of anticoagulation for atrial fibrillation following intracerebral hemorrhage
Supplemental material, 10.1177_1358863X19883027_Supplementary_figure for Net clinical benefit of anticoagulation for atrial fibrillation following intracerebral hemorrhage by Matthew A Pappas and James F Burke in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.