Abstract

The overall prevalence of severe internal carotid artery (ICA) disease in patients undergoing cardiac surgery ranges from 5% to 13%.1,2 The presence of carotid artery stenosis constitutes a contributing risk factor for perioperative stroke, with a stroke risk < 2% in patients without significant stenosis, 3% with unilateral stenosis, 5% with bilateral stenosis, and 7–11% with occlusive disease. 3 Carotid endarterectomy (CEA) and coronary artery bypass grafting (CABG) in a staged or concomitant fashion has been performed since the 1970s. However, staged CEA and open heart surgery (OHS) carries with it an increased risk of perioperative myocardial infarction (MI), and both staged and combined CEA-OHS carry an increased risk of late-phase death compared to staged carotid artery stent (CAS) with OHS. 1 Nonetheless, despite the lowered risk of interstage MI following the index CAS compared to CEA, it is not nil. 1 Thus, some have evaluated the possibility of performing one-day sequential CAS-OHS to obviate the risk of interstage adverse events with favorable results.
The study by Dzierwa and colleagues adds to the previous literature to support the feasibility of CAS-OHS in a sequential fashion. 4 The authors retrospectively reviewed data from patients undergoing CAS followed by either CABG or other cardiac surgery in their institution. Specifically, these were patients with an elevated Canadian Cardiovascular Society (CCS) angina grade, a New York Heart Association (NYHA) heart failure class, or unstable angina/non-ST segment elevation MI. In-hospital and 30-day clinical follow-up showed no major adverse cardiac and cerebrovascular events (MACCE) related to the carotid intervention. Of the total 70 patients who were treated with this strategy, only one patient had perioperative MI due to incomplete revascularization and two deaths were deemed related to high-risk operative profiles, constituting a 4.3% risk of 30-day MACCE. The authors should be applauded for a well-executed study with identification of patients most appropriate for one-day sequential procedures, and their adherence to a consistent, ‘tailored’ CAS strategy for their patients. Nonetheless, two important considerations must be addressed.
First, this is a single-arm, retrospective, observational study. Generalizability must be limited to a patient population similar to the study cohort in terms of cardiac and cerebrovascular profiles and symptoms. Furthermore, given the lack of a control arm, one cannot concede that the one-day sequential CAS-OHS technique is superior to staged CAS-OHS in this patient population. Similar to the preceding studies evaluating sequential CAS-OHS, this study only demonstrates that this method is feasible. This is particularly important given the timing of dual antiplatelet therapy (DAPT) and the need for early initiation of thienopyridine in the postoperative phase for protection of the carotid stent. The rate of reoperation due to bleeding with the DAPT strategy outlined by the authors in this current study was 8.6%, which is higher than historical data (2.4–3.6%) for OHS only. 5 This is not to be understated, as the overall mortality significantly increases with reoperation, with a 3- to 4.5-fold difference of in-hospital mortality. 5 For this reason, a larger randomized controlled trial evaluating the safety of sequential versus staged CAS-OHS is necessary to fully elucidate whether one strategy is superior compared to the other.
Second, the number of patients with symptomatic carotid artery disease represented only 27% of the study population. We understand from historical data that the presence of significant carotid artery disease and a history of prior neurologic events confers the highest risk of perioperative stroke. 6 Conversely, those with asymptomatic unilateral carotid artery stenosis undergoing CABG have a considerably lower risk of stroke, regardless of preventative CEA of the involved carotid vessel. 7 In a meta-analysis evaluating only patients with asymptomatic carotid artery stenosis, the presence of unilateral 50–99% stenosis conveyed a stroke risk of 2.0% in the absence of prophylactic intervention. 8 The risk was comparable in patients with 70–99% and 80–99% stenosis. 8 A baseline low rate of incident stroke in the asymptomatic population may account for the lack of any cerebrovascular signal in this current, relatively small study.
Given this low event rate, many have questioned whether pre-emptive carotid revascularization is truly necessary in those with asymptomatic carotid artery disease prior to OHS. 9 Indeed, the etiology of stroke in a patient undergoing OHS is multifactorial, and may encompass: (1) embolization from an atherosclerotic aorta, intracardiac source, or intracranial vascular lesion; (2) microembolization during cardiopulmonary bypass; or (3) hemodynamic fluctuations during surgery. 2 Aside from carotid artery stenosis, other risk factors that contribute to perioperative stroke risk include increased age, prior cerebrovascular phenomenon, peripheral artery disease, left ventricular dysfunction, atrial fibrillation, and hypertension. 2 Imaging-based evaluation of perioperative strokes have suggested that 50–76% of strokes occur in the absence of significant carotid artery disease, and approximately 60% of strokes are not confined to a single carotid territory. 10 Given these findings, the presence of carotid artery stenosis serves more as a marker of increased vulnerability for stroke, akin to the other risk factors above, rather than a causal relationship for perioperative cerebrovascular events.
In the current era, an individualized approach to screen for and to treat carotid artery stenosis prior to OHS is most appropriate. The most recent guidelines 11 suggest selective screening of patients undergoing cardiac surgery for carotid artery stenosis, with revascularization reserved only for those with severe, symptomatic stenosis (Figure 1). The cornerstone of therapy remains medical optimization and maintenance of hemodynamic stability during the operation. In the patients deemed appropriate for carotid revascularization, a tailored treatment strategy should be decided based on clinical risk factors and anatomical profile. One-day sequential CAS-OHS may be a promising treatment strategy for those who satisfy anatomic criteria, though its superiority over staged CAS-OHS will need further investigation.
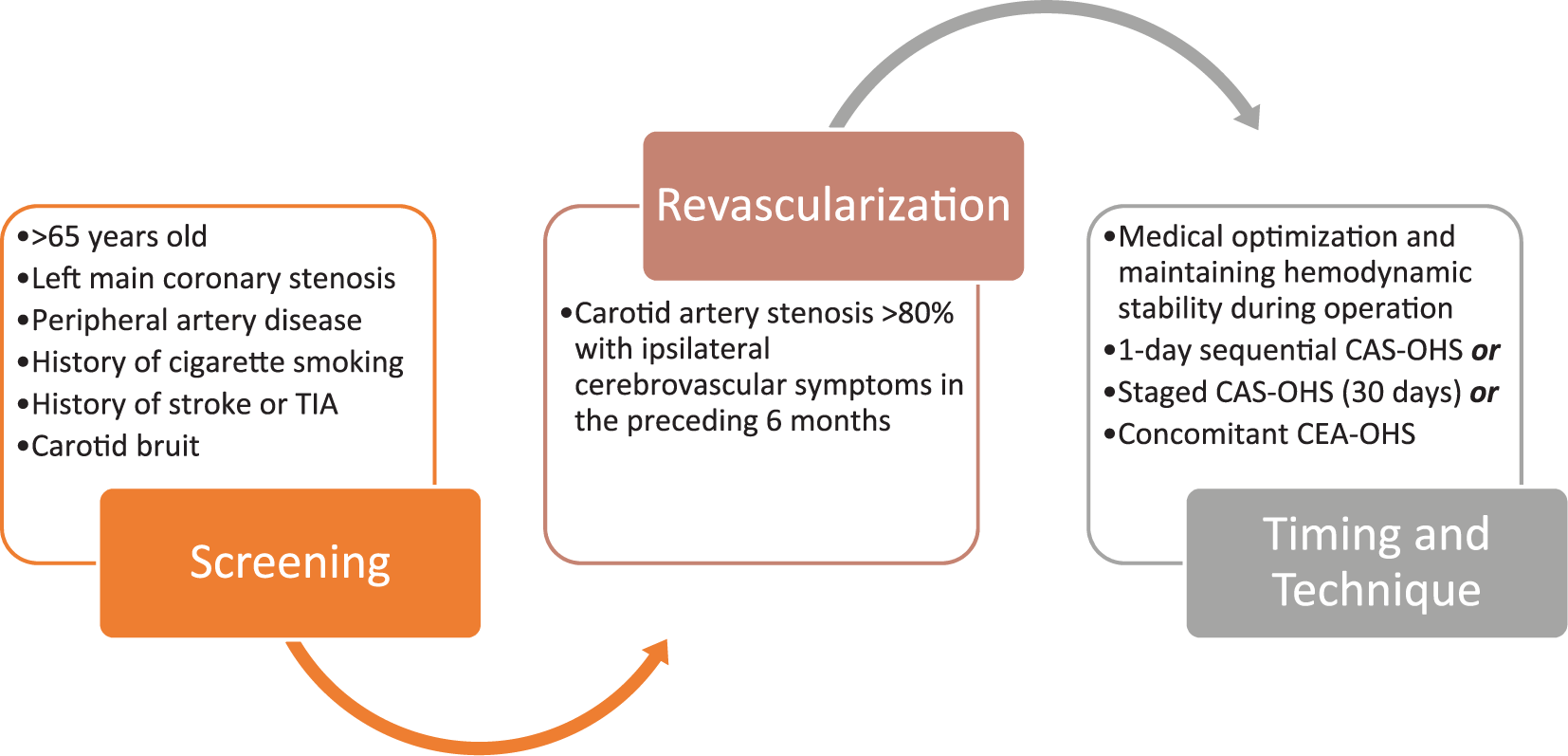
Screening and treatment strategy for carotid artery stenosis prior to open heart surgery based on multisocietal guidelines. 11
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Li serves as a consultant for Abbott Vascular, Medtronic, and Abiomed. Dr Shishehbor serves on the global advisory board for Abbott Vascular, Medtronic, Terumo, Phillips, and Boston Scientific.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
